Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (21): 3407-3412.doi: 10.12307/2024.088
Previous Articles Next Articles
Effects of biomechanics on biological characteristics of osteoblasts
Xiong Wanqi1, Li Zhenhao1, Cui Yan1, Liu Jiahe1, Li Chenzhi1, Wu Mingjian1, Li Yancheng1, Yang Fan1, 2, Liu Baoyi1
- 1Zhongshan Hospital Affiliated to Dalian University, Dalian 116001, Liaoning Province, China; 2Institute of Metal Research, Chinese Academy of Sciences, Shenyang 110000, Liaoning Province, China
-
Received:2023-05-06Accepted:2023-07-04Online:2024-07-28Published:2023-09-28 -
Contact:Liu Baoyi, PhD, Chief physician, Zhongshan Hospital Affiliated to Dalian University, Dalian 116001, Liaoning Province, China Yang Fan, PhD, Associate researcher, Zhongshan Hospital Affiliated to Dalian University, Dalian 116001, Liaoning Province, China; Institute of Metal Research, Chinese Academy of Sciences, Shenyang 110000, Liaoning Province, China -
About author:Xiong Wanqi, Master candidate, Zhongshan Hospital Affiliated to Dalian University, Dalian 116001, Liaoning Province, China -
Supported by:Postdoctoral Science Foundation of Dalian, No. 285395 (to YF); Dalian Medical Science Research Project, No. 2111038 (to YF); Key Research Project of Education Department of Liaoning Province, No. LJKZZ20220148 (to LBY)
CLC Number:
Cite this article
Xiong Wanqi, Li Zhenhao, Cui Yan, Liu Jiahe, Li Chenzhi, Wu Mingjian, Li Yancheng, Yang Fan, Liu Baoyi. Effects of biomechanics on biological characteristics of osteoblasts[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(21): 3407-3412.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
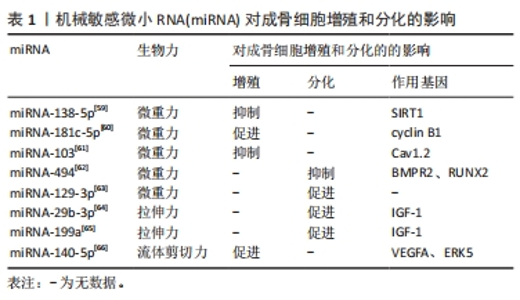
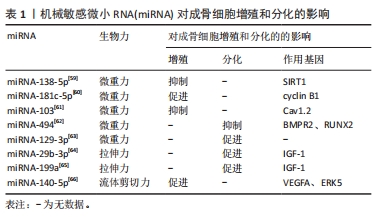
2.1 微重力对成骨细胞的影响 在微重力状态下,成骨细胞受到外界的牵引和压力较小,处于一个相对静止的环境中。相比之下,在地球上的正常1 g重力环境下,成骨细胞会受到更强的压力和牵引,从而促进其增殖和分化。因此,处于微重力环境中的成骨细胞的生物特性受到抑制,包括细胞增殖和分化的能力。长期处于微重力环境中的人,如宇航员,可能会面临骨质疏松的风险,这也是宇航员健康面临的一个重要挑战。为了解决这个问题,研究人员一直在探索利用生物力学刺激等方法来促进骨细胞的生长和发育,从而预防和治疗骨质疏松等骨骼疾病[13]。FAN等[14]发现微重力对成骨细胞的影响是通过下调Wnt/β-catenin信号通路上游基因局部黏附激酶的表达,减少细胞的局部黏附,改变细胞骨架结构来实现的。骨形态发生蛋白2、Ⅰ型胶原和碱性磷酸酶活性均下降,从而导致成骨细胞的增殖和分化受到抑制。同时随着施加在成骨细胞上的重力越小,碱性磷酸酶的活性下降越明显[15]。MORABITO等[16]研究发现,微重力还能通过代谢途径影响细胞,微重力影响成骨细胞的氧化状态,增加了活性氧的产生,从而诱导氧化应激的发生,促进成骨细胞的凋亡,抗氧化剂的使用能延缓该过程。XU等[17]研究发现微重力通过调节细胞骨架的动态变化来调节成骨细胞的功能,微重力使成骨细胞肌动蛋白微丝断裂,进而使肌动蛋白微丝和Smad蛋白的结合增加,从而抑制了骨形态发生蛋白2信号诱导的成骨细胞分化。 以上研究结果表明,微重力环境下成骨细胞的功能受到抑制,这将导致骨量和骨密度的下降。对于长期在太空中工作的宇航员,以及需要长期卧床或不活动的人群,这种骨质丢失的风险更加显著[18]。因此,需要采取相应的预防和治疗措施以维护骨骼健康。 2.2 超重力对成骨细胞的影响 除了微重力外,超重力也是一个不可忽视的因素。微重力和超重力都是不同于地球正常1 g重力的状态,它们对成骨细胞的影响也不同。超重力指高于地球正常1 g重力的状态,如在高速飞行的飞行员或乘坐过山车时所经历的高重力状态。与微重力相比,超重力可能对成骨细胞的影响更为有益。已有研究表明,暴露于适度超重力环境下的成骨细胞可以增加骨量[19],并且有助于预防骨质疏松。因此,了解超重力对成骨细胞的影响,有助于设计更好的骨生长和疾病治疗方案,并提供更好的防护措施。MIWA等[20]的研究表明,超重力在体外通过前列腺素E2介导的机制促进了小鼠成骨细胞系MC3T3-E1的增殖。ZHOU等[21]研究发现,将细胞置于20 g的超重力环境下,超重力可能通过增加细胞的局灶黏附,上调Runt相关转录因子2的表达,从而增加成骨细胞的标志物骨桥蛋白的表达。另外,KAWAO等[22]的研究发现,当小鼠暴露于3 g的超重力环境时,Wnt/β-catenin信号通路中的负向调控因子(如Dickkopf WNT信号通路抑制剂2)的表达受到抑制,随之促进小鼠成骨细胞的碱性磷酸酶活性和矿化作用。这项研究表明,超重力可以通过影响Wnt/β-catenin信号通路来影响成骨细胞的功能。 所以,超重力环境下,由于成骨细胞接受到的机械刺激明显增加,因此可以促进成骨细胞的增殖和分化,有着积极作用。这些研究成果为骨组织工程和骨疾病的治疗提供了新的思路和方法,比如可以利用超重力技术促进骨组织的再生和修复。但是,目前超重力技术的应用仍受到技术和设备限制,需要进一步的研究和改进,以提高技术的可行性和安全性,以便更好地应用于骨组织工程和骨疾病的治疗。 2.3 压应力对成骨细胞的影响 压应力是指施加在骨骼系统上的机械压力。压应力不仅能够促进骨骼生长和增强骨密度,还能够影响成骨细胞的生物学特性。研究表明,压应力对成骨细胞的影响受到力度和持续时间的影响,较小的压力可以刺激成骨细胞增殖和分化,但较高的压力则会抑制成骨细胞的增殖和分化[23-24]。此外,压应力的时间依赖性也是影响成骨细胞生物学行为的重要因素。短期的压应力可以促进成骨细胞的增殖和分化,而长时间的压应力则可能导致成骨细胞的凋亡和功能下降。因此,对于不同程度和时间的压力,成骨细胞的生物学反应可能会有所不同,这也为骨组织工程和骨骼疾病的治疗提供了重要的理论和实践基础[25]。SHEN等[26]进行了一项针对MC3T3-E1细胞的3D培养实验,将细胞置于0,1,2,3,4和5 g/cm2的压应力下,并探究了不同压应力下对细胞活力和分化的影响,结果表明,压应力在5 g/cm2以内对细胞活力没有明显的影响;当应力为2 g/cm2时,成骨细胞分化的水平有所增强,然而当压应力超过2 g/cm2时,成骨细胞分化的水平没有进一步的增强。这些结果提示了压应力对成骨细胞分化的影响具有一定的阈值效应。YONG等[27]研究发现压应力可以激活β-catenin和丝裂原活化蛋白激酶信号通路,而抑制丝裂原活化蛋白激酶则可改变压力诱导的β-catenin激活,因此在压力作用下,丝裂原活化蛋白激酶与β-catenin信号通路是存在相互作用的。CHEN等[28]的研究表明,循环压应力可以透过Wnt/β-catenin信号通路促进成骨分化,并且施加的压力时间和强度对于成骨分化也有显著影响。此外,细胞在0.5 MPa压力下作用6 h后的成骨分化相关基因表达达到最高峰,然而,1 MPa的压力对于成骨细胞的分化没有明显影响,较长时间的压力则对成骨细胞分化产生负面影响。同时,SOMEMURA等[29]的研究发现,在循环压应力的作用下,应力诱导了成骨细胞葡萄糖转运蛋白1(glucose transporters type 1,Glut1)的激活并抑制了NAD-依赖性去乙酰化酶Sirtuin-1(NAD-dependent deacetylase sirtuin-1,SIRT1)的活性,从而导致成骨细胞中Runt相关转录因子2的上调,这提示在压应力作用下,成骨细胞的分化与Glut1/SIRT1/Runt相关转录因子2信号通路有关。INOUE等[30]的研究报道了在成骨细胞中表达的囊状核苷酸转运蛋白,在压力作用下通过诱导ATP释放和上调嘌呤能受体P2X7的表达,从而抑制成骨细胞的分化。XU等[31]使用循环单轴压应力(0.5 Hz,4 000 μstrain)来模拟咬合创伤力,对成骨细胞进行创伤性压力刺激实验,发现该创伤性压力可以通过激活核因子κB信号通路促进β-catenin的降解,最终抑制体外成骨分化。LU等[32]采用与XU等[31]相同的加载方式对成骨细胞进行刺激,并发现压应力可以下调分化抑制性长链非编码RNA的表达,从而间接激活核因子κB信号通路,抑制成骨分化。另外,SEN等[33]的研究发现,在正畸治疗中,牙槽骨成骨细胞在压应力的刺激下通过增强轴突导向因子3A的表达来促进成骨分化。 总的来说,在骨骼生物力学研究中,压应力是一个非常重要的因素。一定程度的压应力可以刺激成骨细胞增殖、分化以及骨组织形成,从而对骨骼健康起到积极的影响。但是,过度的压力则会对骨骼健康产生负面影响。因此,在骨骼相关疾病的研究和治疗中,需要综合考虑压应力的强度和时间等因素,以达到最佳治疗效果。同时,控制压应力的程度和时间也是骨组织工程和骨疾病治疗中的重要研究方向之一。未来需要进一步深入研究压应力对骨骼健康的影响机制,以及开发更为先进的技术手段来实现精准的压应力控制,从而更好地维护人类骨骼健康。 2.4 拉伸应力对成骨细胞的影响 拉伸应力是指细胞所承受的外力拉伸作用。拉伸应力对成骨细胞的影响取决于应力的大小,适当的拉伸应力可以促进成骨细胞的增殖、分化以及骨基质合成,但是过度的拉伸刺激会抑制成骨细胞成骨分化[34-35]。YU等[36]的研究发现,高强度机械拉伸可以诱导成骨细胞凋亡。此外,研究表明骨膜蛋白对机械拉伸诱导的成骨细胞凋亡具有保护作用。值得注意的是,在生理负荷条件下,机械拉伸的强度通常不足以导致细胞凋亡。近年来,Flexcell拉伸系统广泛应用于研究当中。利用该系统对成骨细胞进行加载后发现,不同的伸长率对于细胞的存活、细胞因子的产生、成熟度以及分化程度等方面都产生了显著影响。研究结果表明,成骨分化因子的上调程度取决于应变大小小于18%的伸长率。此外,研究还发现,成骨细胞的增殖在0-12%的伸长率下受到刺激,而更高的伸长率则会对其产生不利影响[37-38]。XIAO等[39]对成骨细胞施加了循环拉伸载荷(10%伸长率,0.5 Hz)后,发现细胞外信号调节激酶1/2和信号转导和转录激活因子3被依次激活,这显著促进了成骨细胞成骨相关基因的表达以及矿化结节的形成。同时,GONG等[40]研究发现拉伸应力作用于成骨细胞时,可以促进信号转导和转录激活因子3的表达升高,进而促进成骨细胞的分化。DANCIU等[41]的研究探索了机械拉伸促进成骨细胞分化和凋亡的早期通路,他们发现,拉伸力可导致成骨细胞内Ca2+的快速升高,并促进磷脂酰肌醇激酶通路的磷酸化,从而调节成骨细胞的分化和凋亡。在ZENG等[42]的研究中发现,周期性拉伸刺激能够激活蛋白激酶B/哺乳动物雷帕霉素靶蛋白/核糖体S6激酶信号通路,从而增强成骨细胞样MG-63细胞的能量代谢。具体而言,这种刺激可增加葡萄糖消耗、提高乳酸水平以及增强ATP水平等代谢方面的表现。WANG等[43]研究发现,拉伸力可显著促进MG-63细胞中成骨细胞分化相关标志物的表达,包括碱性磷酸酶、骨形态发生蛋白2、Ⅰ型胶原蛋白、骨钙素和Runt相关转录因子2。此外,机械拉伸还可引起哺乳动物雷帕霉素蛋白磷酸化和核因子κB磷酸化及其核转位的增加。 综上所述,可以得出周期性和静态的拉伸力、单向和多向的拉伸方向对成骨细胞的响应具有不同的影响。因此,在研究和治疗骨骼相关疾病时,需要综合考虑拉伸力的强度、周期、方向等因素,以便更好地了解骨细胞的反应,从而制定出更为科学合理的治疗方案和预防措施。 2.5 流体剪切力对成骨细胞的影响 流体剪切力是一种机械刺激,由细胞外液体流过细胞膜表面引起,通过对骨骼施加负荷(包括机械负荷、肌肉收缩、血压和淋巴引流)导致组织间液体流动,从而压缩腔隙-泪小管系统,诱发包括流体剪切力在内的各种机械刺激[44-45]。流体剪切力可进一步诱导成骨细胞生物力学性能发生改变。LEI等[46]在探讨流体剪切力对钛表面MG-63细胞中细胞增殖和黏着斑激酶表达影响的研究中发现,在1.6 Pa的流体剪切力下,喷砂/酸蚀表面对局部黏着斑激酶的表达有积极影响。流体剪切力刺激细胞增殖和局部黏着斑激酶的表达,并受表面形貌和流体剪切力水平以及持续时间的显著协同效应的调节。JIN等[47]通过平行板流室的流动装置生成节律性液体流(pulsating fluid flow)并作用于成骨细胞,观察到细胞骨架、细胞核形态和体积发生明显变化,并伴有黏附蛋白和结构蛋白基因和蛋白表达升高。 除此以外,流体剪切力诱导不同幅度的钙瞬变,导致能量代谢途径发生改变,从而影响细胞的增殖和分化[48]。流体剪切力通过调节细胞外信号调节激酶 5信号通路诱导成骨细胞增殖。DING等[49]研究发现,活化的T-细胞核因子1作为中间产物促进了流体剪切力诱导的细胞外信号调节激酶5磷酸化,从而促进成骨细胞增殖。另外,ZHANG等[50]的研究表明,流体剪切力作用于成骨细胞后细胞外信号调节激酶5能够负调控Kruppel样因子4的表达,进而促进成骨细胞增殖。在WANG等[51]的研究中,他们发现将MC3T3-E1细胞暴露于1.2 Pa的流体剪切力环境中,分别处理30,60,90 min时,长链非编码RNA牛磺酸上调基因1的表达水平具有时间依赖性。长链非编码RNA牛磺酸上调基因1可以结合微小RNA34a来上调成纤维细胞生长因子受体1的表达,促进成骨细胞增殖,并同时对成骨细胞的凋亡产生抑制作用。近年来,针对骨折和骨缺损治疗,研究人员创造了大量的合成材料支架。同时,生物材料在流体剪切力诱导的成骨分化中发挥着重要作用[52]。SALERNO等[53]制作了一种羟基磷灰石支架,它是一种基于羟基磷灰石的3D支架,具有与自体骨高度相似的优势,他们证明了羟基磷灰石支架可以有效地与流体设备耦合,并研究了在动态流动灌注条件下成骨细胞形态和生物学的变化;结果表明,羟基磷灰石支架结合流体剪切力机械刺激可以有效地促进成骨诱导。 总之,流体剪切力作为一种重要的生物力学刺激类型,在成骨细胞研究中具有重要作用。研究表明,流体剪切力可以促进成骨细胞的增殖和分化,这与骨组织的形成、修复和维护密切相关。不同的流体剪切力刺激方式和强度也会对成骨细胞的响应产生不同的影响,因此在研究流体剪切力对成骨细胞的影响时,需要考虑多种因素,如剪切应力大小、方向、时间等。深入探究成骨细胞在不同流体剪切力刺激下的信号传导机制,有助于揭示骨骼生长和疾病的机制,为骨组织工程和骨疾病的治疗提供理论基础。 2.6 静水压力对成骨细胞的影响 静水压是指在液体中,由于液体的静压力而产生的压力。在骨骼系统中,静水压是一种常见的力学刺激形式,HENSTOCK等[54]发现循环静水力可以增强鸡胚胎股骨的体外成骨能力,能够刺激致密骨的形成同时观察到骨成熟标志物均有上升。TAKAI等[55]在探究动态静水压对骨细胞的存活能力和成骨细胞功能的影响时发现,静水压使骨细胞保持活力,并通过骨细胞和成骨细胞之间的相互作用增强成骨细胞的功能。而过高的静水压能导致细胞失活,施加200 MPa静水压可导致细胞凋亡,而高于300 MPa导致细胞坏死。JIN等[47]用不同的高静水压(100-150 MPa,250-300 MPa,450-500 MPa)处理人类成骨细胞和软骨细胞时发现,经200-350 MPa处理的成骨细胞表面变成了带小气孔的海绵状结构,细胞内结构被破坏,细胞直径较其他组明显缩小。在骨移植中处理移植物传统方法可用γ辐照、冷冻干燥、热和化学处理或组合使用,其目的是使移植物不存在任何可能诱导免疫反应的细胞残留物,然而,在加工过程中往往会失去生物特性[56]。例如,冷冻干燥导致缺乏成骨特性、血管形成和即刻强度[57]。而在近期研究中发现,静水压250 MPa作用20 min处理移植物具有灭活作用,与其他方法相比是一种温和的替代方案[58]。 可见,静水压是一种重要的生物力学刺激类型,可以对成骨细胞的增殖、分化和凋亡等方面产生影响。研究表明,适当的静水压刺激可以促进成骨细胞的增殖和分化,从而增强骨组织的形成和修复能力。基于静水压对成骨细胞生物特性的积极影响,静水压可被应用于体外培养成骨细胞的研究,也可用于体内骨生长的研究。然而,静水压的作用机制还需要进一步的研究,以更好地发掘其在骨生物学研究和骨组织工程方面的潜在应用。 2.7 机械敏感微小RNA(microRNA,miRNA) 随着不同的生物力学作用对成骨细胞影响研究的不断深入,机械敏感miRNA逐渐被研究人员发现(表1)。机械敏感miRNA是指在机械刺激下表达量发生变化的miRNA分子。在微重力环境下,miRNA-138-5p靶向SIRT1基因,抑制成骨细胞增殖并诱导成骨细胞凋亡[59]。此外,微重力还通过影响细胞周期抑制成骨细胞增殖。施加微重力后,成骨细胞细胞周期蛋白B1的表达下降,导致细胞周期停滞在G2期。转染miRNA-181c-5p抑制剂可以抵消微重力对成骨细胞的影响[60]。此外,在微重力环境下,miRNA-103抑制成骨细胞增殖,通过抑制L型电压敏感钙通道的主要亚基Cav1.2的表达[61]。同时,miRNA-494直接靶向骨形态发生蛋白Ⅱ型受体和Runt相关转录因子2来抑制骨形态发生蛋白信号诱导的成骨细胞分化[62]。初级纤毛是突出于细胞表面的微管结构,被认为是微重力的传感器[63]。LIU等[63]的研究发现,在微重力环境下,成骨细胞的初级纤毛会迅速缩短或消失;而过表达miRNA-129-3p可以抑制初级纤毛的吸收,从而保护成骨细胞免受微重力的不利影响。ZENG等[64]的研究表明,成骨细胞中的miRNA-29b-3p对拉伸应变做出反应,并通过调节机械拉伸作用下胰岛素样生长因子的分泌来调节成骨细胞分化。同时,林维龙等[65]发现拉伸力能够下调miRNA-199a的表达,并导致胰岛素样生长因子的表达上调,从而诱导成骨细胞分化。除此以外,WANG等[66]发现流体剪切力通过激活血管内皮生长因子a/细胞外调节蛋白激酶5信号通路,进而下调miRNA-140-5p并促进成骨细胞的增殖。研究表明,机械敏感miRNA对成骨细胞的增殖和分化发挥着重要的作用。深入了解这些机械敏感miRNA的调控机制和作用途径,有助于深化对成骨细胞生物学行为的理解,并为相关骨骼疾病的预防和治疗提供新思路。"
| [1] EL-FARRASH RA, ALI RH, BARAKAT NM. Post-natal bone physiology. Semin Fetal Neonatal Med. 2020;25(1):101077. [2] DATTA HK, NG WF, WALKER JA, et al. The cell biology of bone metabolism. J Clin Pathol. 2008;61(5):577-587. [3] GOULD NR, TORRE OM, LESER JM, et al. The cytoskeleton and connected elements in bone cell mechano-transduction. Bone. 2021;149:115971. [4] UDA Y, AZAB E, SUN N, et al. Osteocyte Mechanobiology. Curr Osteoporos Rep. 2017;15(4):318-325. [5] CHEN X, WANG Z, DUAN N, et al. Osteoblast-osteoclast interactions. Connect Tissue Res. 2018;59(2):99-107. [6] GORI F, SUPERTI-FURGA A, BARON R. Bone Formation and the Wnt Signaling Pathway. N Engl J Med. 2016;375(19):1902-1903. [7] CHEN G, DENG C, LI YP. TGF-β and BMP signaling in osteoblast differentiation and bone formation. Int J Biol Sci. 2012;8(2):272-288. [8] CHAN WCW, TAN Z, TO MKT, et al. Regulation and Role of Transcription Factors in Osteogenesis. Int J Mol Sci. 2021;22(11):5445. [9] ZHANG K, LIU X, WANG L, et al. The mechanosensory and mechanotransductive processes mediated by ion channels and the impact on bone metabolism: A systematic review. Arch Biochem Biophys. 2021;711:109020. [10] HENSLEY AP, MCALINDEN A. The role of microRNAs in bone development. Bone. 2021;143:115760. [11] 姚依村,梁伟国,叶冬平. 细胞骨架与力学信号传导[J]. 中国组织工程研究,2014,18(7):1109-1114. [12] XIONG J, ONAL M, JILKA RL, et al. Matrix-embedded cells control osteoclast formation. Nat Med. 2011;17(10):1235-1241. [13] COLLET P, UEBELHART D, VICO L, et al. Effects of 1- and 6-month spaceflight on bone mass and biochemistry in two humans. Bone. 1997;20(6):547-551. [14] FAN C, WU Z, COOPER DML, et al. Activation of Focal Adhesion Kinase Restores Simulated Microgravity-Induced Inhibition of Osteoblast Differentiation via Wnt/Β-Catenin Pathway. Int J Mol Sci. 2022;23(10):5593. [15] BRAVEBOY-WAGNER J, LELKES PI. Impairment of 7F2 osteoblast function by simulated partial gravity in a Random Positioning Machine. NPJ Microgravity. 2022;8(1):20. [16] MORABITO C, GUARNIERI S, CUCINA A, et al. Antioxidant Strategy to Prevent Simulated Microgravity-Induced Effects on Bone Osteoblasts. Int J Mol Sci. 2020;21(10):3638. [17] XU H, WU F, ZHANG H, et al. Actin cytoskeleton mediates BMP2-Smad signaling via calponin 1 in preosteoblast under simulated microgravity. Biochimie. 2017;138:184-193. [18] ROLVIEN T, AMLING M. Disuse Osteoporosis: Clinical and Mechanistic Insights. Calcif Tissue Int. 2022;110(5):592-604. [19] WOODCOCK EM, GIRVAN P, ECKERT J, et al. Measuring Intracellular Viscosity in Conditions of Hypergravity. Biophys J. 2019;116(10):1984-1993. [20] MIWA M, KOZAWA O, TOKUDA H, et al. Effects of hypergravity on proliferation and differentiation of osteoblast-like cells. Bone Miner. 1991; 14(1):15-25. [21] ZHOU S, YANG X, HU J, et al. Continuous hypergravity alters the cytoplasmic elasticity of MC3T3-E1 osteoblasts via actin filaments. J Biomech. 2018;72: 222-227. [22] KAWAO N, MORITA H, IEMURA S, et al. Roles of Dkk2 in the Linkage from Muscle to Bone during Mechanical Unloading in Mice. Int J Mol Sci. 2020; 1(7):2547. [23] ALFORD AI, KOZLOFF KM, HANKENSON KD. Extracellular matrix networks in bone remodeling. Int J Biochem Cell Biol. 2015;65:20-31. [24] YANG X, JIANG J, ZHOU L, et al. Osteogenic and angiogenic characterization of mandible and femur osteoblasts. J Mol Histol. 2019; 50(2):105-117. [25] SONG F, WANG Y, JIANG D, et al. Cyclic Compressive Stress Regulates Apoptosis in Rat Osteoblasts: Involvement of PI3K/Akt and JNK MAPK Signaling Pathways. PLoS One. 2016;11(11):e0165845. [26] SHEN XQ, GENG YM, LIU P, et al. Magnitude-dependent response of osteoblasts regulated by compressive stress. Sci Rep. 2017;7:44925. [27] YONG J, VON BREMEN J, RUIZ-HEILAND G, et al. Adiponectin as Well as Compressive Forces Regulate in vitro β-Catenin Expression on Cementoblasts via Mitogen-Activated Protein Kinase Signaling Activation. Front Cell Dev Biol. 2021;9:645005. [28] CHEN X, GUO J, YUAN Y, et al. Cyclic compression stimulates osteoblast differentiation via activation of the Wnt/β-catenin signaling pathway. Mol Med Rep. 2017;15(5):2890-2896. [29] SOMEMURA S, KUMAI T, YATABE K, et al. Physiologic Mechanical Stress Directly Induces Bone Formation by Activating Glucose Transporter 1 (Glut 1) in Osteoblasts, Inducing Signaling via NAD+-Dependent Deacetylase (Sirtuin 1) and Runt-Related Transcription Factor 2 (Runx2). Int J Mol Sci. 2021;22(16):9070. [30] INOUE A, NAKAO-KUROISHI K, KOMETANI-GUNJIGAKE K, et al. VNUT/SLC17A9, a vesicular nucleotide transporter, regulates osteoblast differentiation. FEBS Open Bio. 2020;10(8):1612-1623. [31] XU W, LU Y, YUE J, et al. Occlusal trauma inhibits osteoblast differentiation and bone formation through IKK-NF-κB signaling. J Periodontol. 2020; 91(5):683-692. [32] LU Q, XU W, LIU L, et al. Traumatic compressive stress inhibits osteoblast differentiation through long chain non-coding RNA Dancr. J Periodontol. 2020;91(11):1532-1540. [33] ŞEN S, LUX CJ, ERBER R. A Potential Role of Semaphorin 3A during Orthodontic Tooth Movement. Int J Mol Sci. 2021;22(15):8297. [34] TANG LL, WANG YL, PAN J, et al. The effect of step-wise increased stretching on rat calvarial osteoblast collagen production. J Biomech. 2004;37(1):157-161. [35] SONG CX, LIU SY, ZHU WT, et al. Excessive mechanical stretchmediated osteoblasts promote the catabolism and apoptosis of chondrocytes via the Wnt/βcatenin signaling pathway. Mol Med Rep. 2021;24(2):593. [36] YU KW, YAO CC, JENG JH, et al. Periostin inhibits mechanical stretch-induced apoptosis in osteoblast-like MG-63 cells. J Formos Med Assoc. 2018;117(4):292-300. [37] BHATT KA, CHANG EI, WARREN SM, et al. Uniaxial mechanical strain: an in vitro correlate to distraction osteogenesis. J Surg Res. 2007;143(2):329-336. [38] TANG L, LIN Z, LI YM. Effects of different magnitudes of mechanical strain on Osteoblasts in vitro. Biochem Biophys Res Commun. 2006;344(1):122-128. [39] XIAO X, ZOU S, CHEN J. Cyclic tensile force modifies calvarial osteoblast function via the interplay between ERK1/2 and STAT3. BMC Mol Cell Biol. 2023;24(1):9. [40] GONG X, SUN S, YANG Y, et al. Osteoblastic STAT3 Is Crucial for Orthodontic Force Driving Alveolar Bone Remodeling and Tooth Movement. J Bone Miner Res. 2023;38(1):214-227. [41] DANCIU TE, ADAM RM, NARUSE K, et al. Calcium regulates the PI3K-Akt pathway in stretched osteoblasts. FEBS Lett. 2003;536(1-3):193-197. [42] ZENG Z, JING D, ZHANG X, et al. Cyclic mechanical stretch promotes energy metabolism in osteoblast-like cells through an mTOR signaling-associated mechanism. Int J Mol Med. 2015;36(4):947-956. [43] WANG D, CAI J, ZENG Z, et al. The interactions between mTOR and NF-κB: A novel mechanism mediating mechanical stretch-stimulated osteoblast differentiation. J Cell Physiol. 2020. doi: 10.1002/jcp.30184. [44] KUMAR R, TIWARI AK, TRIPATHI D, et al. Anatomical variations in cortical bone surface permeability: Tibia versus femur. J Mech Behav Biomed Mater. 2021;113:104122. [45] PRICE C, ZHOU X, LI W, et al. Real-time measurement of solute transport within the lacunar-canalicular system of mechanically loaded bone: direct evidence for load-induced fluid flow. J Bone Miner Res. 2011;26(2):277-285. [46] LEI X, LIU Q, LI S, et al. Effects of fluid shear stress on expression of focal adhesion kinase in MG-63 human osteoblast-like cells on different surface modification of titanium. Bioengineered. 2021;12(1):4962-4971. [47] JIN J, JASPERS RT, WU G, et al. Shear Stress Modulates Osteoblast Cell and Nucleus Morphology and Volume. Int J Mol Sci. 2020;21(21):8361. [48] YU L, WANG X, GAO X, et al. The calcium transient characteristics induced by fluid shear stress affect the osteoblast proliferation. Exp Cell Res. 2018; 362(1):51-62. [49] DING N, GENG B, LI Z, et al. Fluid shear stress promotes osteoblast proliferation through the NFATc1-ERK5 pathway. Connect Tissue Res. 2019; 60(2):107-116. [50] ZHANG B, AN L, GENG B, et al. ERK5 negatively regulates Kruppel-like factor 4 and promotes osteogenic lineage cell proliferation in response to MEK5 overexpression or fluid shear stress. Connect Tissue Res. 2021;62(2):194-205. [51] WANG X, HE J, WANG H, et al. Fluid shear stress regulates osteoblast proliferation and apoptosis via the lncRNA TUG1/miR-34a/FGFR1 axis. J Cell Mol Med. 2021;25(18):8734-8747. [52] PRODANOV L, SEMEINS CM, VAN LOON JJ, et al. Influence of nanostructural environment and fluid flow on osteoblast-like cell behavior: a model for cell-mechanics studies. Acta Biomater. 2013;9(5):6653-6662. [53] SALERNO E, ORLANDI G, ONGARO C, et al. Liquid flow in scaffold derived from natural source: experimental observations and biological outcome. Regen Biomater. 2022;9:rbac034. [54] HENSTOCK JR, ROTHERHAM M, ROSE JB, et al. Cyclic hydrostatic pressure stimulates enhanced bone development in the foetal chick femur in vitro. Bone. 2013;53(2):468-477. [55] TAKAI E, MAUCK RL, HUNG CT, et al. Osteocyte viability and regulation of osteoblast function in a 3D trabecular bone explant under dynamic hydrostatic pressure. J Bone Miner Res. 2004;19(9):1403-1410. [56] SINGH R, SINGH D, SINGH A. Radiation sterilization of tissue allografts: A review. World J Radiol. 2016;8(4):355-369. [57] TOSOUNIDIS TH, GIANNOUDIS PV. Biological Facet of Segmental Bone Loss Reconstruction. J Orthop Trauma. 2017;31 Suppl 5:S27-S31. [58] WALETZKO-HELLWIG J, POHL C, RIESE J, et al. Effect of High Hydrostatic Pressure on Human Trabecular Bone Regarding Cell Death and Matrix Integrity. Front Bioeng Biotechnol. 2021;9:730266. [59] XU L, ZHANG X, LI G, et al. Inhibition of SIRT1 by miR-138-5p provides a mechanism for inhibiting osteoblast proliferation and promoting apoptosis under simulated microgravity. Life Sci Space Res (Amst). 2023;36:59-69. [60] SUN Z, LI Y, WANG H, et al. miR-181c-5p mediates simulated microgravity-induced impaired osteoblast proliferation by promoting cell cycle arrested in the G2 phase. J Cell Mol Med. 2019;23(5):3302-3316. [61] SUN Z, CAO X, HU Z, et al. MiR-103 inhibits osteoblast proliferation mainly through suppressing Cav1.2 expression in simulated microgravity. Bone. 2015;76:121-128. [62] QIN W, LIU L, WANG Y, et al. Mir-494 inhibits osteoblast differentiation by regulating BMP signaling in simulated microgravity. Endocrine. 2019; 65(2):426-439. [63] LIU J, LENG FF, GAO YH, et al. Protection of primary cilia is an effective countermeasure against the impairment of osteoblast function induced by simulated microgravity. J Cell Mol Med. 2023;27(1):36-51. [64] ZENG Q, WANG Y, GAO J, et al. miR-29b-3p regulated osteoblast differentiation via regulating IGF-1 secretion of mechanically stimulated osteocytes. Cell Mol Biol Lett. 2019;24:11. [65] 林维龙,吴晓沛,王晓明,等. miR-199a调控IGF1表达对机械刺激下成骨细胞分化的影响[J]. 上海口腔医学,2022,31(2):132-137. [66] WANG X, GENG B, WANG H, et al. Fluid shear stress-induced down-regulation of microRNA-140-5p promotes osteoblast proliferation by targeting VEGFA via the ERK5 pathway. Connect Tissue Res. 2022;63(2): 156-168. [67] SCHRÖDER M, RESELAND JE, HAUGEN HJ. Osteoblasts in a Perfusion Flow Bioreactor-Tissue Engineered Constructs of TiO2 Scaffolds and Cells for Improved Clinical Performance. Cells. 2022;11(13):1995. [68] ATIF AR, PUJARI-PALMER M, TENJE M, et al. A microfluidics-based method for culturing osteoblasts on biomimetic hydroxyapatite. Acta Biomater. 2021;127:327-337. [69] LUO X, ZHANG S, LUO B, et al. Engineering collagen fiber templates with oriented nanoarchitecture and concerns on osteoblast behaviors. Int J Biol Macromol. 2021;185:77-86 [70] AW YONG KM, HORST E, NEALE D, et al. A Bioreactor for 3D In Vitro Modeling of the Mechanical Stimulation of Osteocytes. Front Bioeng Biotechnol. 2022;10:797542. |
| [1] | Li Zhifei, Yang Yin, Chen Hualong, Liang Qinqiu, Zhong Yuanming, Zhang Yisheng. Finite element analysis of the correlation between tilt angle of titanium cage and postoperative subsidence of titanium cage after anterior subtotal cervical corpectomy, decompression and fusion [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1313-1319. |
| [2] | Chen Mengmeng, Bao Li, Chen Hao, Jia Pu, Feng Fei, Shi Guan, Tang Hai. Biomechanical characteristics of a novel interspinous distraction fusion device BacFuse for the repair of lumbar degenerative disease [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1325-1329. |
| [3] | Liang Cheng, Zhang Linqi, Wang Guan, Li Wen, Duan Ke, Li Zhong, Lu Xiaobo, Zhuo Naiqiang. Finite element and biomechanical analysis of different implants in repair for unilateral unstable pelvic posterior ring injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1336-1341. |
| [4] | Guo Sutong, Feng Dehong, Guo Yu, Wang Ling, Ding Yujian, Liu Yi, Qian Zhengying, Li Mingyang. Construction and finite element analysis of normal and osteoporotic hip models [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1342-1346. |
| [5] | Yang Junliang, Lu Tan, Xu Biao, Jiang Yaqiong, Wang Fucheng. Three-dimensional finite element analysis of effects of partial anterior cruciate ligament rupture on knee joint stress [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1347-1353. |
| [6] | Weng Rui, Lin Dongxin, Guo Haiwei, Zhang Wensheng, Song Yuke, Lin Hongheng, Li Wenchao, Ye Linqiang. Abnormal types of intervertebral disc structure and related mechanical loading with biomechanical factors [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1436-1442. |
| [7] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [8] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [9] | Zhang Xiaoyun, Liu Hua, Chai Yuan, Chen Feng, Zeng Hao, Gao Zhengang, Huang Yourong. Effect of Yishen Gushu Formula on bone metabolic markers and clinical efficacyn in patients with osteoporosis of kidney deficiency and blood stasis type [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1155-1160. |
| [10] | Yue Yun, Wang Peipei, Yuan Zhaohe, He Shengcun, Jia Xusheng, Liu Qian, Li Zhantao, Fu Huiling, Song Fei, Jia Menghui. Effects of croton cream on JNK/p38 MAPK signaling pathway and neuronal apoptosis in cerebral ischemia-reperfusion injury rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1186-1192. |
| [11] | Dai Yuexing, Zheng Liqin, Wu Minhui, Li Zhihong, Li Shaobin, Zheng Desheng, Lin Ziling. Effect of vessel number on computational fluid dynamics in vascular networks [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1206-1210. |
| [12] | Liu Xin, Hu Man, Zhao Wenjie, Zhang Yu, Meng Bo, Yang Sheng, Peng Qing, Zhang Liang, Wang Jingcheng. Cadmium promotes senescence of annulus fibrosus cells via activation of PI3K/Akt signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1217-1222. |
| [13] | Wei Juan, Li Ting, Huan Mengting, Xie Ying, Xie Zhouyu, Wei Qingbo, Wu Yunchuan. Mechanism by which static exercise improves insulin resistance in skeletal muscle of type 2 diabetes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1271-1276. |
| [14] | Sheng Siqi, Xie Lin, Zhao Xiangyu, Jiang Yideng, Wu Kai, Xiong Jiantuan, Yang Anning, Hao Yinju, Jiao Yun. Involvement of miR-144-3p in Cbs+/- mouse hepatocyte autophagy induced by high-methionine diet [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1289-1294. |
| [15] | Wang Wen, Zheng Pengpeng, Meng Haohao, Liu Hao, Yuan Changyong. Overexpression of Sema3A promotes osteogenic differentiation of dental pulp stem cells and MC3T3-E1 [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 993-999. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||