Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (2): 216-223.doi: 10.12307/2023.991
Previous Articles Next Articles
Mechanism of beta-sitosterol on hypertrophic scar fibroblasts: an analysis based on network pharmacology
Zuo Jun, Ma Shaolin
- Department of Plastic Surgery, The First Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, Xinjiang Uygur Autonomous Region, China
-
Received:2022-11-02Accepted:2022-12-12Online:2024-01-18Published:2023-06-30 -
Contact:Ma Shaolin, Master, Chief physician, Professor, Doctoral supervisor, Department of Plastic Surgery, The First Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, Xinjiang Uygur Autonomous Region, China -
About author:Zuo Jun, MD candidate, Attending physician, Department of Plastic Surgery, The First Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, Xinjiang Uygur Autonomous Region, China -
Supported by:Regional Foundation of National Natural Science Foundation of China, No. 81760345 (to MSL); Youth Program of Natural Science Foundation of Hunan Province, No. 2021JJ40487 (to ZJ)
CLC Number:
Cite this article
Zuo Jun, Ma Shaolin. Mechanism of beta-sitosterol on hypertrophic scar fibroblasts: an analysis based on network pharmacology[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 216-223.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

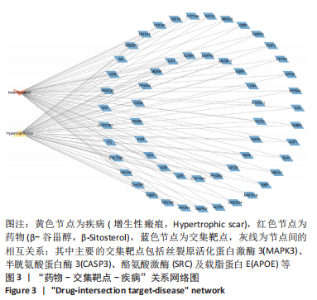
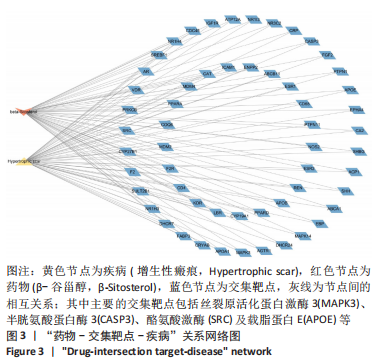
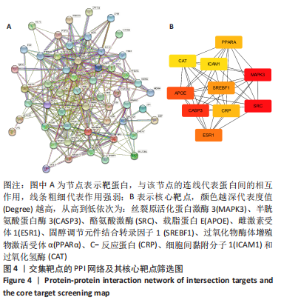
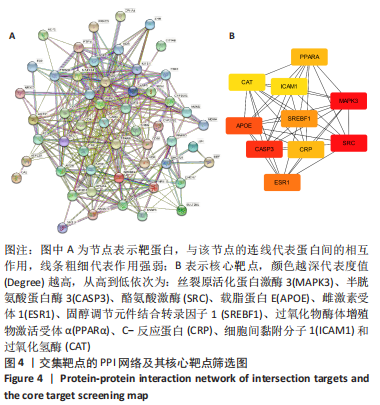
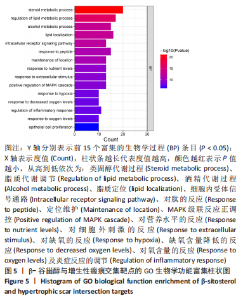
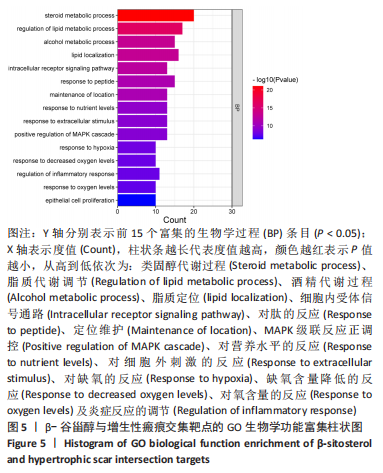
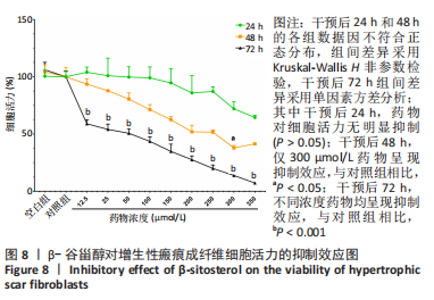
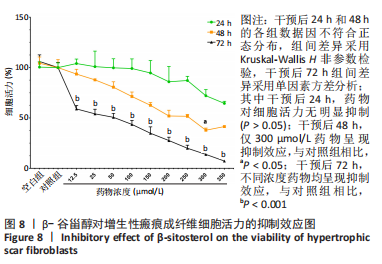
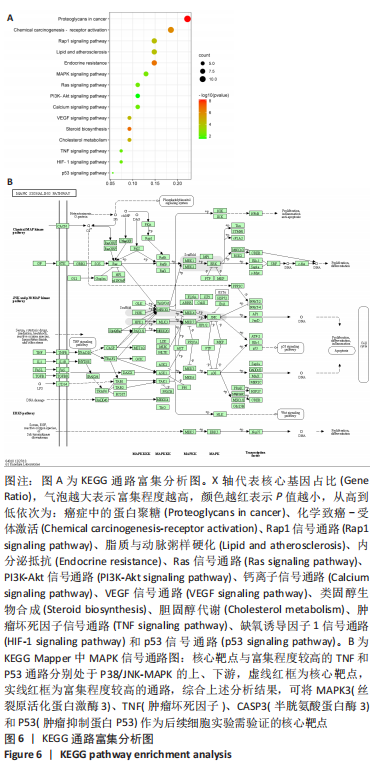
2.7 KEGG通路富集分析结果 KEGG富集分析共获得83条信号通路(P < 0.05),富集程度排列前15位的通路见图6A,包括脂质和动脉粥样硬化通路、MAPK信号通路和PI3K-AKt信号通路等。根据增生性瘢痕相关文献[18-19]、GO生物学功能富集分析结果和富集通路中核心靶点基因占比(Gene Ratio),作者预测MAPK信号通路与交集靶点关系密切。在KEGG Mapper功能模块中搜索“MAPK信号通路”进一步发现,MAPK信号通路中包含2个核心靶点:MAPK3(ERK1/2)和CASP3,富集程度较高的肿瘤坏死因子和P53信号通路也分别处于P38/JNK-MAPK的上、下游,见图6B。"
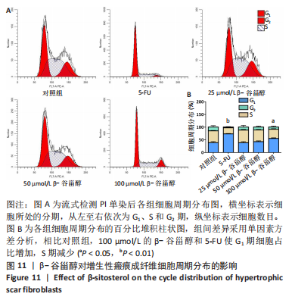
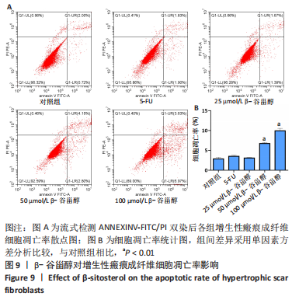
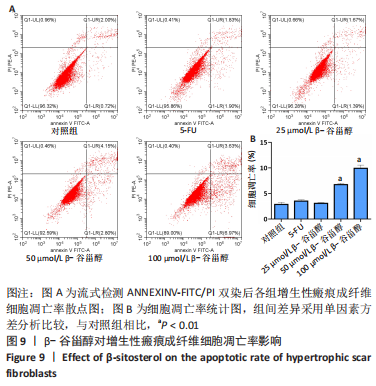
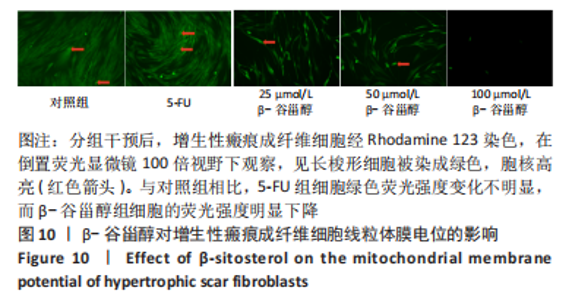
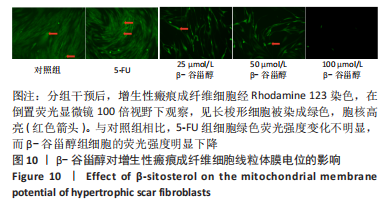
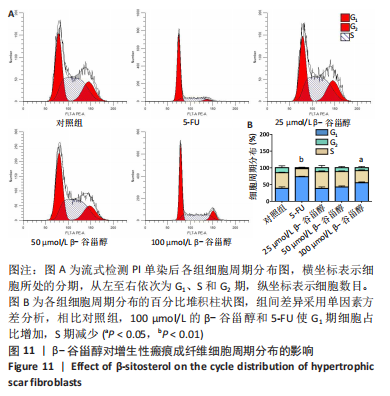
2.12 β-谷甾醇对增生性瘢痕成纤维细胞周期分布的影响 如图11所示,相比对照组[G1期(38.5±4.4)%、S期(46.7±1.4)%、G2期(14.7±5.8)%],5-FU能显著阻滞增生性瘢痕成纤维细胞周期进展,使G1期细胞占比[(73.4±1.1)%]增加、S期[(23.8±0.3)%]减少(P < 0.001);而增生性瘢痕成纤维细胞在暴露于25 μmol/L [G1期(38.1±4.5)%、S期(49.9±2.9)%、G2期(12.0±6.9)%]和50 μmol/L [G1期(42.5±2.8)%、S期(46.2±1.8)%、G2期(11.2±3.8)%] β-谷甾醇后细胞周期谱无显著变化(P > 0.05);但100 μmol/L β-谷甾醇能使G1期阻滞[(55.2±2.7)%],S期细胞相应减少[(36.5±2.0)%],差异有显著性意义(P < 0.05);各组细胞G2期占比均无显著差异(P > 0.05)。"
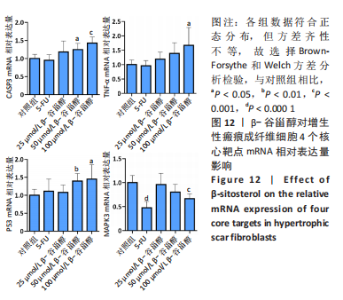
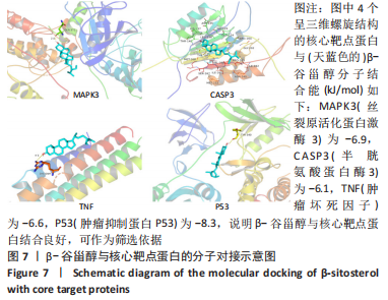
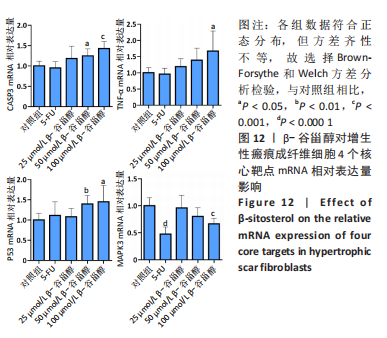
2.13 β-谷甾醇对增生性瘢痕成纤维细胞核心靶点mRNA表达的影响 如图12所示,与对照组相比,100 μmol/L β-谷甾醇使肿瘤坏死因子、Casp3和P53 mRNA表达显著上调(P < 0.05),并降低MAPK3 mRNA表达水平(P < 0.001);50 μmol/L β-谷甾醇也能显著下调Casp3和P53 mRNA表达(P < 0.05);25 μmol/L β-谷甾醇对4个核心靶点mRNA的表达均无显著影响(P > 0.05),与上述表型基本一致;而5-FU除能降低MAPK3 mRNA表达外(P < 0.05),对其他3个核心靶点的mRNA表达无显著影响(P > 0.05)。"
| [1] COENTRO JQ, PUGLIESE E, HANLEY G, et al. Current and upcoming therapies to modulate skin scarring and fibrosis. Adv Drug Deliv Rev. 2019;146:37-59. [2] ZHANG H, WANG HY, WANG DL, et al. Effect of pressure therapy for treatment of hypertrophic scar. Med Baltim. 2019;98(26):e162-e163. [3] TAN J, ZHOU J, HUANG L, et al. Hypertrophic scar improvement by early intervention with ablative fractional carbon dioxide laser treatment. Lasers Surg Med. 2021;53(4):450-457. [4] TUNCA M, GAMSIZKAN M, YÜREKLI A, et al. Cryosurgery to remove perichondrium for the rabbit ear hypertrophic scar model: a simplified method. Acta Dermatovenerol. 2019;28(2):57-59. [5] BI M, SUN P, LI D, et al. Intralesional injection of botulinum toxin type A compared with intralesional injection of corticosteroid for the treatment of hypertrophic scar and keloid: a systematic review and meta-analysis. Med Sci Monit. 2019;25:2950-2958. [6] ELSAIE ML. Update on management of keloid and hypertrophic scars: a systemic review. J Cosmet Dermatol. 2021;20(9):2729-2738. [7] CHEN D, LI Q, ZHANG H, et al. Traditional Chinese medicine for hypertrophic scars-A review of the therapeutic methods and potential effects. Front Pharmacol. 2022;13:1025602. [8] 王琳奇,郑明明,薛婷丽,等.α-亚麻酸植物甾醇酯调控TGF-β1/Smad信号通路改善小鼠肝纤维化[J].营养学报,2021,43(3):236-241. [9] PARK YJ, BANG IJ, JEONG MH, et al. Effects of β-sitosterol from corn silk on TGF-β1-induced epithelial-mesenchymal transition in lung alveolar epithelial cells. J Agric Food Chem. 2019;67(35):9789-9795. [10] BAE H, PARK S, HAM J, et al. ER-mitochondria calcium flux by β-sitosterol promotes cell death in ovarian cancer. Antioxidants (Basel). 2021;10(10):1583. [11] KHAN Z, NATH N, RAUF A, et al. Multifunctional roles and pharmacological potential of β-sitosterol: emerging evidence toward clinical applications. Chem Biol Interact. 2022;365:110117. [12] GUO Q, LI Y, CHEN Y, et al. β-Elemene induces apoptosis by activating the P53 pathway in human hypertrophic scar fibroblasts. IUBMB Life. 2022; 74(6):508-518. [13] 郭惠中.中草药独角莲根茎提取物β-谷甾醇(β-sitosterol)对瘢痕疙瘩成纤维细胞作用的实验研究[D].大连:大连医科大学,2015. [14] 世界中医药学会联合会.网络药理学评价方法指南[J].世界中医药, 2021,16(4):527-532. [15] 张青,徐月,彭伟,等.分子对接结合网络药理学研究桂枝芍药知母汤治疗类风湿关节炎的分子作用机制[J].中草药,2020,51(18):4673-4684. [16] HAN T, LIN DF, JIANG H. Wound natural healing in treatment of tumor-like hypertrophic scar. An Bras Dermatol. 2017;92(4):474-477. [17] 刘文洁,尹雪云,王红,等.M1型小胶质细胞源性外泌体携载miR-20a-5p对氧糖剥夺/复氧后神经元损伤的影响[J].中华危重病急救医学, 2022,34(8):842-847. [18] MA F, SHEN J, ZHANG H, et al. A novel lncRNA FPASL regulates fibroblast proliferation via the PI3K/AKT and MAPK signaling pathways in hypertrophic scar. Acta Biochim Biophys Sin (Shanghai). 2022. doi: 10.3724/abbs.2022122. [19] CHAI CY, SONG J, TAN Z, et al. Adipose tissue-derived stem cells inhibit hypertrophic scar(HS) fibrosis via p38/MAPK pathway. J Cell Biochem. 2019; 120(3):4057-4064. [20] KASIRZADEH S, GHAHREMANI MH, SETAYESH N, et al. β-sitosterol alters the inflammatory response in CLP rat model of sepsis by modulation of NFκB signaling. Biomed Res Int. 2021;2021:5535562. [21] DEVARAJ E, ROY A, ROYAPURAM VEERARAGAVAN G, et al. β-Sitosterol attenuates carbon tetrachloride-induced oxidative stress and chronic liver injury in rats. Naunyn Schmiedebergs Arch Pharmacol. 2020;393(6):1067-1075. [22] PARK YJ, BANG IJ, JEONG MH, et al. Effects of β-sitosterol from corn silk on TGF-β1-induced epithelial-mesenchymal transition in lung alveolar epithelial cells. J Agric Food Chem. 2019;67(35):9789-9795. [23] 张琳竺,曾睿,黄楚渝,等.基于网络药理学探讨苦参治疗增生性瘢痕的作用机制[J].中医学报,2022,37(9):1954-1961. [24] 许小琪,赖建辉,时军,等.丹参酮ⅡA治疗增生性瘢痕的网络药理学研究[J].中药新药与临床药理,2020,31(1):64-71. [25] KESAVARDHANA S, MALIREDDI RKS, KANNEGANTI TD. Caspases in cell death, inflammation, and pyroptosis. Annu Rev Immunol. 2020;38:567-595. [26] VO PHT, NGUYEN TDT, TRAN HT, et al. Cytotoxic components from the leaves of Erythrophleum fordii induce human acute leukemia cell apoptosis through caspase 3 activation and PARP cleavage. Bioorg Med Chem Lett. 2021;31:127673. [27] RAJAVEL T, PACKIYARAJ P, SURYANARAYANAN V, et al. β-Sitosterol targets Trx/Trx1 reductase to induce apoptosis in A549 cells via ROS mediated mitochondrial dysregulation and P53 activation. Sci Rep. 2018;8(1):2071. [28] RONKINA N, GAESTEL M. MAPK-activated protein kinases: servant or partner? Annu Rev Biochem. 2022;91:505-540. [29] WANG WD, LI GL, YANG HM, et al. Role of mitogen-activated protein kinases in the formation of hypertrophic scar with model of lipopolysaccharide stimulated skin fibroblast cell. Pakistan J Med Scis. 2018;34(1):215-220. [30] 朱君. ERK1/2MAPK信号通路在表儿茶素抑制增生性瘢痕成纤维细胞增殖中的影响研究[D].温州:温州医科大学,2019. [31] MELOCHE S, POUYSSÉGUR J. The ERK1/2 mitogen-activated protein kinase pathway as a master regulator of the G1- to S-phase transition. Oncogene. 2007;26(22):3227-3239. [32] TASYRIQ M, NAJMULDEEN IA, IN LL, et al. 7α-hydroxy-β-sitosterol from chisocheton tomentosus induces apoptosis via dysregulation of cellular Bax/Bcl-2 ratio and cell cycle arrest by downregulating ERK1/2 activation. Evid Based Complement Alternat Med. 2012;2012:765316. [33] YUAN H, WU X, WANG X, et al. Chinese herbal decoction astragalus and angelica exerts its therapeutic effect on renal interstitial fibrosis through the inhibition of MAPK, PI3K-Akt and TNF signaling pathways. Genes Dis. 2020;9(2):510-521. [34] LIU R, HAO D, XU W, et al. β-sitosterol modulates macrophage polarization and attenuates rheumatoid inflammation in mice. Pharm Biol. 2019;57:161-168. |
| [1] | Liu Baofang, Xu Bin, Chen Lei. Pueraria decoction in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 193-199. |
| [2] | Ran Lei, Han Haihui, Xu Bo, Wang Jianye, Shen Jun, Xiao Lianbo, Shi Qi. Molecular docking analysis of the anti-inflammatory mechanism of Cibotium barometz and Epimedium for rheumatoid arthritis: animal experiment validation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 208-215. |
| [3] | Wang Qian, Lu Ziang, Li Lihe, Lyu Chaoliang, Wang Meng, Zhang Cunxin. Sinomenine effectively inhibits interleukin-1beta-induced apoptosis in nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 224-230. |
| [4] | Chen Simin, Hu Yingjun, Yan Wenrui, Ji Le, Shao Mengli, Sun Ze, Zheng Hongxing, Qi Shanshan. Establishment and evaluation of a streptozotocin-induced diabetic encephalopathy rat model [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 237-241. |
| [5] | Ai Fangfang, Xiao Hongyan, Wang Fang, Zhu Yongzhao, Ma Lijun. Reversal effect of Lycium barbarum polysaccharide in combination with oxaliplatin on drug resistance of colon cancer stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 74-79. |
| [6] | Ma Suilu, He Zhijun, Liu Tao, Li Yan, He Yuanxu, He Bo, Wang Weiwei, Wei Xiaotao. Traditional Chinese medicine monomer in the prevention and treatment of flap necrosis by regulating “autophagy” [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 153-158. |
| [7] | Zheng Rongjiong, Deng Zerun, Han Dan, Sun Lihua. Mechanism underlying rat hepatocyte apoptosis regulated by exosomes derived from bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 44-49. |
| [8] | Shen Feiyan, Yao Jixiang, Su Shanshan, Zhao Zhongmin, Tang Weidong. Knockdown of circRNA WD repeat containing protein 1 inhibits proliferation and induces apoptosis of chondrocytes in knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-6. |
| [9] | He Xi, Wan Yu, Tang Yuting, Yang Anning, Wu Kai, Jiao Yun, Bai Zhigang, Jiang Yideng, Shen Jiangyong. Erastin inhibits proliferation of hypertrophic scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-. |
| [10] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [11] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [12] | Cui Lianxu, Jiang Wenkang, Lu Dahong, Xu Junrong, Liu Xiaocui, Wang Bingyun. Clinical-grade human umbilical cord mesenchymal stem cells affect the improvement of neurological function in rats with traumatic brain injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 835-839. |
| [13] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| [14] | Ke Weiqiang, Chen Xianghui, Chen Xiaoling, Meng Jie, Ma Yanlin. Rituximab combined with autologous peripheral blood stem cell transplantation in the treatment of diffuse large B-cell lymphoma and the expression of related factors [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 915-920. |
| [15] | Zhao Siqi, Du Juan, Qu Haifeng, Li Jianmin, Zhang Yuxin, Liu Junjie. Effects of enriched environment combined with melatonin on learning and memory function and brain neuron apoptosis in SAMP8 mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 701-706. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||