Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (33): 5363-5369.doi: 10.12307/2023.498
Previous Articles Next Articles
Tolerogenic dendritic cells can inhibit pyroptosis of spleen cells in collagen-induced arthritis rats
Long Tiaoyu1, Bao Lunmin2, Wan Xiufang1, Li Honghong1, Zhang Yundong2, Jiang Hongmei1
- 1Department of Clinical Microbiology and Immunology, School of Clinical Laboratory Science, Guizhou Medical University, Guiyang 550025, Guizhou Province, China; 2Department of Laboratory Medicine, People’s Hospital of Anshun City, Anshun 561000, Guizhou Province, China
-
Received:2022-06-16Accepted:2022-07-27Online:2023-11-28Published:2023-03-30 -
Contact:Jiang Hongmei, MD, Professor, Doctoral supervisor, Master’s supervisor, Department of Clinical Microbiology and Immunology, School of Clinical Laboratory Science, Guizhou Medical University, Guiyang 550025, Guizhou Province, China -
About author:Long Tiaoyu, Master candidate, Department of Clinical Microbiology and Immunology, School of Clinical Laboratory Science, Guizhou Medical University, Guiyang 550025, Guizhou Province, China -
Supported by:the Regional Science Fund Project of National Natural Science Foundation of China, No. 81960393 (to JHM); the Project of Natural Science Research Innovation Group of General Undergraduate Colleges in Guizhou Province, No. KY[2021]016 (to JHM)
CLC Number:
Cite this article
Long Tiaoyu, Bao Lunmin, Wan Xiufang, Li Honghong, Zhang Yundong, Jiang Hongmei. Tolerogenic dendritic cells can inhibit pyroptosis of spleen cells in collagen-induced arthritis rats[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5363-5369.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
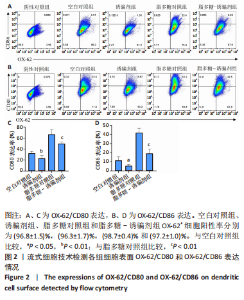
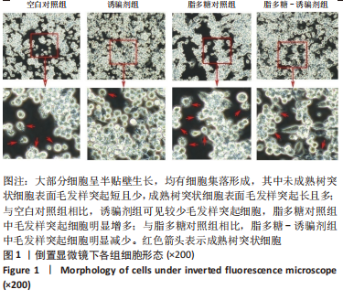
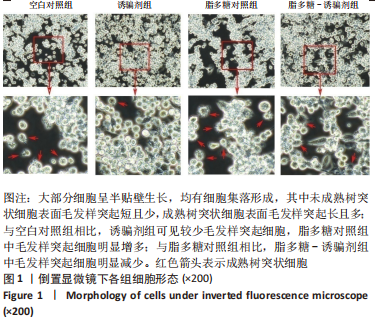
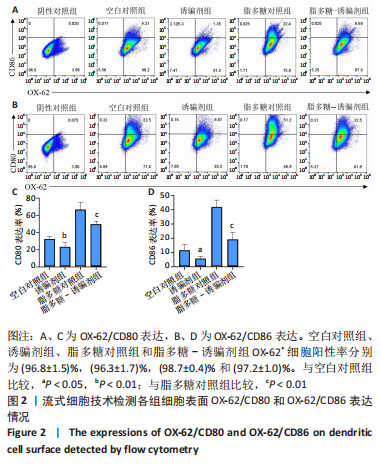
2.2 树突状细胞的鉴定 为鉴定大鼠骨髓源树突状细胞的纯度,采用流式细胞术检测其表面特异性标记物OX-62的表达情况,结果显示:OX-62+细胞在空白对照组、诱骗剂组、脂多糖对照组和脂多糖-诱骗剂组中的表达分别为(96.8±1.5)%,(96.3±1.7)%,(98.7±0.4)%和(97.2±1.0)%;为了评价tolDC的稳定性,实验加入脂多糖刺激细胞成熟,然后采用流式细胞术检测CD80和CD86的表达,评价树突状细胞的成熟度,结果显示:与空白对照组相比,诱骗剂组CD80+、CD86+细胞阳性率明显降低(P < 0.01,P < 0.05);与脂多糖对照组相比,脂多糖-诱骗剂组CD80+、CD86+细胞阳性率明显降低(P < 0.01),见图2,说明制备的tolDC纯度高、稳定性好,符合后续实验要求。"
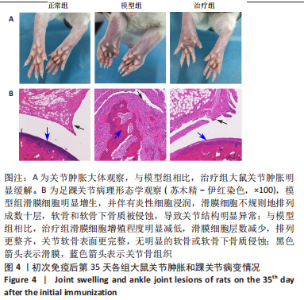
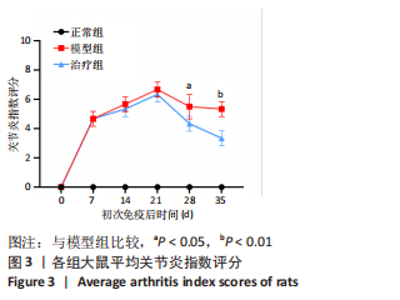
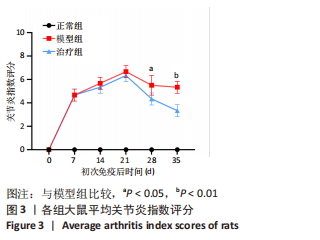
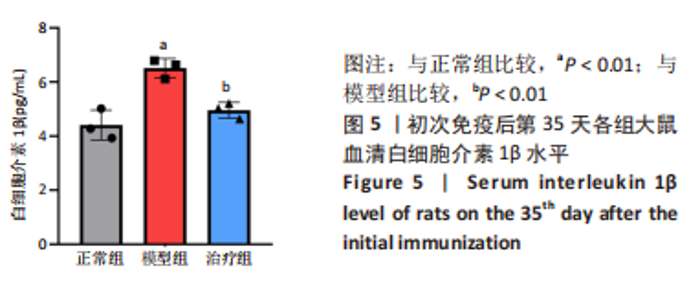
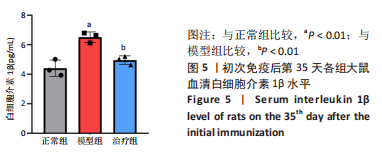
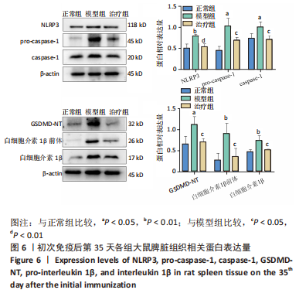
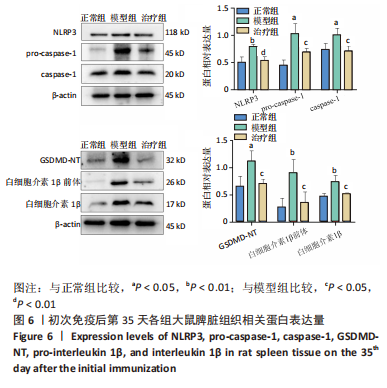
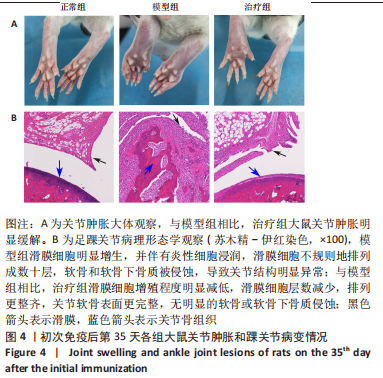
2.3.3 各组大鼠关节肿胀情况及足踝关节病理形态学观察 从初次免疫的第7天起,与正常组相比,模型组和治疗组大鼠关节明显肿胀,第21天到达高峰;与模型组相比,治疗组于初次免疫第28,35天关节肿胀明显缓解,见图4A。 踝关节苏木精-伊红染色显示,正常组滑膜组织光滑,滑膜细胞排列整齐有序,由1-3层细胞组成,滑膜下层由脂肪细胞和成纤维细胞组成,无血管增生或炎性细胞浸润;模型组滑膜细胞明显增生,并伴有炎性细胞浸润,滑膜细胞不规则地排列成数十层,滑膜组织几乎占据整个关节腔,软骨和软骨下骨质被侵蚀,导致关节结构明显异常;与模型组相比,治疗组滑膜细胞增殖程度明显减低,滑膜细胞层数减少,排列更整齐,关节软骨表面更完整,无明显的软骨或软骨下骨质侵蚀,见图4B。说明tolDC可缓解胶原诱导性关节炎大鼠滑膜增生与骨损伤。"
| [1] ZHAI Z, YANG F, XU W, et al. Attenuation of rheumatoid arthritis through the inhibition of tumor necrosis factor-induced caspase 3/gasdermin e-mediated pyroptosis. Arthritis Rheumatol. 2022;74(3):427-440. [2] OTON T, CARMONA L. The epidemiology of established rheumatoid arthritis. Best Pract Res Clin Rheumatol. 2019;33(5):101477. [3] WANG W, ZHOU H, LIU L. Side effects of methotrexate therapy for rheumatoid arthritis: A systematic review. Eur J Med Chem. 2018;158:502-516. [4] MOSANYA CH, ISAACS JD. Tolerising cellular therapies: What is their promise for autoimmune disease? Ann Rheum Dis. 2019;78(3):297-310. [5] JOHNSON DJ, OHASHI PS. Molecular programming of steady-state dendritic cells: Impact on autoimmunity and tumor immune surveillance. Ann N Y Acad Sci. 2013;1284:46-51. [6] ORNING P, LIEN E, FITZGERALD KA. Gasdermins and their role in immunity and inflammation. J Exp Med. 2019;216(11):2453-2465. [7] CHOULAKI C, PAPADAKI G, REPA A, et al. Enhanced activity of NLRP3 inflammasome in peripheral blood cells of patients with active rheumatoid arthritis. Arthritis Res Ther. 2015;17:257. [8] XIA S, ZHANG Z, MAGUPALLI VG, et al. Gasdermin D pore structure reveals preferential release of mature interleukin-1. Nature. 2021;593(7860):607-611. [9] DING J, WANG K, LIU W, et al. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature. 2016;535(7610):111-116. [10] FINK SL, COOKSON BT. Caspase-1-dependent pore formation during pyroptosis leads to osmotic lysis of infected host macrophages. Cell Microbiol. 2006;8(11): 1812-1825. [11] XU L, WANG H, YU QQ, et al. The monomer derivative of paeoniflorin inhibits macrophage pyroptosis via regulating TLR4/ NLRP3/ GSDMD signaling pathway in adjuvant arthritis rats. Int Immunopharmacol. 2021;101(Pt A):108169. [12] TIAN Y, SHI P, ZHOU Y, et al. Dir-labeled tolerogenic dendritic cells for targeted imaging in collagen- induced arthritis rats. Int Immunopharmacol. 2021;91: 107273. [13] 马贵兰,谭艳,田娅玲,等.耐受性树突状细胞通过降低Th1细胞和Th17细胞比例减轻CIA大鼠的关节炎症和病变[J].细胞与分子免疫学杂志,2021, 37(3):193-198. [14] 谭艳,田娅玲,宁丽常,等.胶原蛋白诱导性关节炎(CIA)大鼠耐受性树突状细胞制备技术的优化[J]. 细胞与分子免疫学杂志,2019,35(12):1076-1081. [15] SPARKS JA. Rheumatoid arthritis. Ann Intern Med. 2019;170(1):ITC1-ITC16. [16] LIN YJ, ANZAGHE M, SCHULKE S. Update on the pathomechanism, diagnosis, and treatment options for rheumatoid arthritis. Cells. 2020;9(4):880. [17] CHAICHIAN Y, GENOVESE MC, WEISMAN MH. The road to rheumatoid arthritis prevention: Challenges and opportunities. Clin Rheumatol. 2020;39(5):1379-1381. [18] PUT S, WESTHOVENS R, LAHOUTTE T, et al. Molecular imaging of rheumatoidarthritis: emerging markers, tools, and techniques. Arthritis Res Ther. 2014;16(2):208. [19] RODRÍGUEZ-FERNÁNDEZ JL. Antigen presentation by dendritic cells in rheumatoid arthritis. Curr Top Med Chem. 2013;13(6):712-719. [20] MATSUE H, YANG C, MATSUE K, et al. Contrasting impacts of immunosuppressive agents (rapamycin, FK506, cyclosporin A, and dexamethasone) on bidirectional dendritic cell-T cell interaction during antigen presentation. J Immunol. 2002; 169(7):3555-3564. [21] HUBO M, TRINSCHEK B, KRYCZANOWSKY F, et al. Costimulatory molecules on immunogenic versus tolerogenic human dendritic cells. Front Immunol. 2013; 4:82. [22] FLÓREZ-GRAU G, ZUBIZARRETA I, CABEZÓN R, et al. Tolerogenic Dendritic Cells as a Promising Antigen-Specific Therapy in the Treatment of Multiple Sclerosis and Neuromyelitis Optica From Preclinical to Clinical Trials. Front Immunol. 2018;9:1169. [23] SONG HY, KIM WS, HAN JM, et al. Galangin treatment during dendritic cell differentiation confers tolerogenic properties in response to lipopolysaccharide stimulation. J Nutr Biochem. 2021;87:108524. [24] ONAITIS M, KALADY MF, PRUITT S, et al. Dendritic cell gene therapy. Surg Oncol Clin N Am. 2002;11:645-660. [25] LI X, HAN Y, ZHOU Q, et al. Apigenin, a potent suppressor of dendritic cell maturation and migration, protects against collagen-induced arthritis. J Cell Mol Med. 2016;20(1):170-180. [26] MIYOSHI M, LIU S. Collagen-induced arthritis models. Methods Mol Biol. 2018; 1868:3-7. [27] BURDETTE BE, ESPARZA AN, ZHU H, et al. Gasdermin D in pyroptosis. Acta Pharm Sin B. 2021;11(9):2768-2782. [28] LI Z, GUO J, BI L. Role of the NLRP3 inflammasome in autoimmune diseases. Biomed Pharmacother. 2020;130:110542. [29] TENSHIN H, TERAMACHI J, ASHTAR M, et al. TGF-β-activated kinase-1 inhibitor IL-z1640-2 reduces joint inflammation and bone destruction in mouse models of rheumatoid arthritis by inhibiting NLRP3 inflammasome, TACE, TNF-α and RANKL expression. Clin Transl Immunology. 2022;11(1):e1371. [30] KONG R, SUN L, LI H, et al. The role of NLRP3 inflammasome in the pathogenesis of rheumatic disease. Autoimmunity. 2022;55(1):1-7. [31] YANG P, FENG W, LI C, et al. LPS induces fibroblast-like synoviocytes RSC-364 cells to pyroptosis through NF-κB mediated dual signalling pathway. J Mol Histol. 2021;52(4):661-669. [32] GUO C, FU R, WANG S, et al. NLRP3 inflammasome activation contributes to the pathogenesis of rheumatoid arthritis. Clin Exp Immunol. 2018;194(2):231-243. [33] PANDEYA A, LI L, LI Z, et al. Gasdermin D (GSDMD) as a new target for the treatment of infection. Medchemcomm. 2019;10(5):660-667. [34] EVAVOLD CL, HAFNER-BRATKOVIC I, DEVANT P, et al. Control of gasdermin d oligomerization and pyroptosis by the ragulator-rag-mtorc1 pathway. Cell. 2021; 184(17):4495-4511 e4419. [35] HUANG Y, XU W, ZHOU R. NLRP3 inflammasome activation and cell death. Cell Mol Immunol. 2021;18(9):2114-2127. [36] 余明芳,陈白露,何欣玲,等.时辰艾灸干预类风湿关节炎模型大鼠核心钟基因Clock、Bmal1表达与细胞焦亡的关系[J].中国组织工程研究,2022, 26(35):5700-5706. [37] EL-WASEEF D. A highlight on CD4+ T-cells in the spleen in a rat model of rheumatoid arthritis and possible therapeutic effect of omega-3. Histological and immunofluorescence study. Int Immunopharmacol. 2020;81:106283. [38] WANG Z, ZHUO F, CHU P, et al. Germacrone alleviates collagen-induced arthritis via regulating Th1/Th2 balance and nf-kappab activation. Biochem Biophys Res Commun. 2019;518(3):560-564. [39] RANA AK, LI Y, DANG Q, et al. Monocytes in rheumatoid arthritis: Circulating precursors of macrophages and osteoclasts and, their heterogeneity and plasticity role in RA pathogenesis. Int Immunopharmacol. 2018;65:348-359. [40] YU P, ZHANG X, LIU N, et al. Pyroptosis: Mechanisms and diseases. Signal Transduct Target Ther. 2021;6(1):128. [41] NEGREI C, BOJINCA V, BALANESCU A, et al. Management of rheumatoid arthritis: Impact and risks of various therapeutic approaches. Exp Ther Med. 2016;11(4): 1177-1183. [42] ACOSTA-RODRIGUEZ EV, NAPOLITANI G, LANZAVECCHIA A, et al. Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human t helper cells. Nat Immunol. 2007;8(9):942-949. |
| [1] | Wang Qiuyuan, Liu Youwen, Yue Chen, Hou Hongli, Zhang Xue. Expression and significance of pyroptosis-related factors in patients with rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(32): 5209-5213. |
| [2] | Wang Hao, Ma Chenghao, Qin Zuohai, Zhou Haibo, Ding Haoyuan, Han Dapeng, Nie Zhixing, Pan Peijun, Gao Chenxin, Ouyang Guilin. Effect of tourniquet use on perioperative blood loss in total knee arthroplasty in patients with rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(31): 4943-4948. |
| [3] | Liu Guanjuan, Song Na, Huo Hua, Luo Shanshan, Cheng Yuting, Xiong Yue, Hong Wei, Liao Jian. Zoledronic acid inhibits lipopolysaccharide-induced osteoclast differentiation by regulating NLRP3 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(29): 4677-4683. |
| [4] | Song Na, Liu Guanjuan, Cheng Yuting, Xiong Yue, Luo Shanshan, Hong Wei, Liao Jian. Zoledronic acid promotes alveolar bone formation in ovariectomized rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(28): 4494-4501. |
| [5] | Lu Xiaojun, Xiong Bohan, Yang Tengyun, Wang Xu, Zhang Yaozhang, Zhong Ruiying, Li Yanlin. Novel programmed cell death of chondrocytes in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(28): 4571-4576. |
| [6] | Song Jianhui, Mu Jihong, Li Shiwen. miR-23b effects on transforming growth factor beta1 and osteoclast activity in synovial tissue of rats with rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(26): 4175-4180. |
| [7] | Long Zhisheng, Fu Liuxiang, Gong Feipeng, Wen Jiabin, Deng Ying, Min Huan, Deng Zhuan, Chen Gang. Expression and significance of pyroptosis associated protein in peripheral tissues with tantalum cage loosening [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 4057-4062. |
| [8] | Wu Yongli, Li Long, Liu Junwei, Liu Di, Wang Duo. Warm-needling moxibusion inhibits NLRP3 inflammasome activation and improves cartilage injury in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3202-3208. |
| [9] | Wang Chenyu, Yao Jiawei, Xu Xiongfeng, Qiu Bo. Screening and analysis of differentially expressed genes in synovial tissue of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3224-3229. |
| [10] | Zhao Yan, Xia Qiuqiu, Xiang Shaojie, Mao Qiming, Kong Weijun, Du Qian, Liao Wenbo, Xin Zhijun. Exosomal miRNA in the repair mechanism of degenerative diseases of the spine and joints [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(19): 3040-3051. |
| [11] | Zhang Ziyu, Wang Yiming, Li Han, Wang Zhonghan, Lu Jialin, Xu Rui, Jin Hui. Platelet-rich plasma meets the therapeutic needs of different articular cartilage defects [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2772-2779. |
| [12] | Li Fang, Cao Jianmin, Zhai Pengfei, Wang Jianshe, Hong Da. Effects of resveratrol on albumin-induced pyroptosis-related proteins in HK-2 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(14): 2164-2169. |
| [13] | Wang Xuejiao, Shi Wenjuan, Zhang Yan, Xing Dehai, Li Dongxue, Jiao Xiangying. Role of thioredoxin-interacting protein-mediated inflammatory response and apoptosis in myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(11): 1715-1721. |
| [14] | Zhang Haobo, Zhao Yunan, Yang Xuejun. Role and therapeutic implications of pyroptosis in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1445-1451. |
| [15] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||