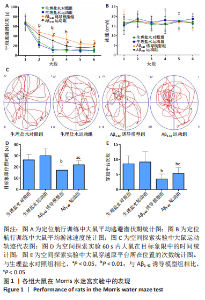
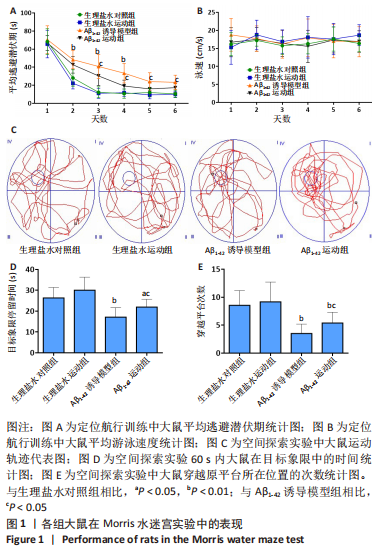
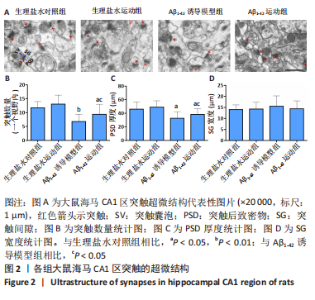
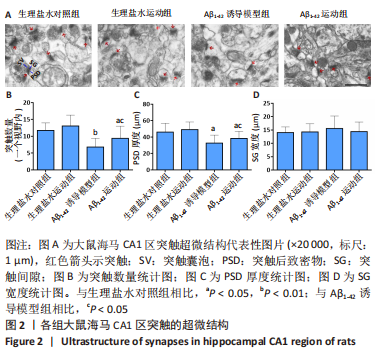
[1] FLORIDDIA ELISA,Glia heterogeneity in AD. Nat Neurosci. 2022;25: 531.
[2] GUARINO A, FAVIERI F, BONCOMPAGNI I, et al. Executive functions in Alzheimer disease: A systematic review. Front Aging Neurosci. 2019;10: 437.
[3] ETNIER JL, KARPER WB, LABBAN JD, et al. The physical activity and Alzheimer’s disease (PAAD) Study: Cognitive outcomes. Ann Behav Med. 2018;52(2):175-185.
[4] 李垂坤,张业廷,魏翠兰,等.不同强度有氧运动对APP/PS1双转基因小鼠海马双皮质素及β-淀粉样蛋白的影响[J].西安体育学院学报,2022,39(2):214-223.
[5] JIANG L, MA J, ZHANG Y, et al. Effect of running exercise on the number of the neurons in the hippocampus of young transgenic APP/PS1 mice.Brain Res. 2018;1692:56-65.
[6] 张业廷,付燕,胡小勇,等.身体活动对阿尔兹海默症影响的CiteSpace知识图谱可视化研究[J].中国康复医学杂志,2020,35(6):716-722.
[7] PANZA GA, TAYLOR BA, MACDONALD HV, et al. Can exercise improve cognitive symptoms of Alzheimer’s disease? A Meta-Analysis. J Am Geriatr Soc. 2018;10:1111.
[8] LEE JH,YANG DS, GOULBOURNE CN, et al. Faulty autolysosome acidification in Alzheimer’s disease mouse models induces autophagic build-up of Aβ in neurons, yielding senile plaques. Nat Neurosci. 2022; 16(3):220-233.
[9] MANSVELDER HD, VERHOOG MB, GORIOUNOVA NA. Synaptic plasticity in human cortical circuits: cellular mechanisms of learning and memory in the human brain? Curr Opin Neurobiol. 2018;18:30069.
[10] MÜLLER-SCHIFFMANN A, HERRING A, ABDEL-HAFIZ L, et al. Amyloid-β dimers in the absence of plaque pathology impair learning and synaptic plasticity. Brain. 2016;139(Pt 2):509-525.
[11] 付燕,张业廷,罗笑,等. 有氧运动对Aβ1-42诱导阿尔茨海默病大鼠学习记忆能力及海马炎症状态的影响[J]. 中国运动医学杂志, 2018,37(8):676-684.
[12] GÜLDNER FH, INGHAM CA. Increase in postsynaptic density material in optic target neurons of the rat suprachiasmatic nucleus after bilateral enucleation. Neurosci Lett. 1980;17:27-31.
[13] JONES DG, DEVON RM. An ultrastructural study into the effects of pentobarbitone on synaptic organization. Brain Res. 1978;147:47-63.
[14] RAGHURAMAN R, KARTHIKEYAN A, WEI WL, et al. Activation of microglia in acute hippocampal slices affects activity-dependent long-term potentiation and synaptic tagging and capture in area CA1.Neurobiol Learn Mem. 2019;163:107039.
[15] LLORENS-MARTÍN M, BLAZQUEZ-LLORCA L, BENAVIDES-PICCIONE R, et al. Selective alterations of neurons and circuits related to early memory loss in Alzheimer’s disease. Front Neuroanat. 2014;8:1-12.
[16] CHEN C, LI B, CHENG G, et al. Amentoflavone ameliorates Aβ1-42-induced memory deficits and oxidative stress in cellular and rat model. Neurochem Res. 2018;2:10.
[17] POSTU PA, NOUMEDEM JAK, CIOANCA O, et al. Lactuca capensis reverses memory deficits in Aβ1-42-induced an animal model of Alzheimer’s disease. J Cell Mol Med. 2018;22(1):111-122.
[18] HOFFMANN K, SOBOL NA, FREDERIKSEN KS, et al. Moderate-to-high intensity physical exercise in patients with Alzheimer’s disease: a randomized controlled trial. J Alzheimers Dis. 2015;50(2):443-453.
[19] CAMMISULI DM, INNOCENTI A, FUSI J, et al. Aerobic exercise effects upon cognition in Alzheimer’s Disease: A systematic review of randomized controlled trials. Arch Ital Biol. 2018;156(1-2):54-63.
[20] FARZI MA, SADIGH-ETEGHAD S, EBRAHIMI K, et al. Exercise improves recognition memory and acetylcholinesterase activity in the beta amyloid-induced rat model of Alzheimer’s Disease. Ann Neurosci. 2019; 25(3):121-125.
[21] MANCINI A, DE IURE A, PICCONI B. Basic mechanisms of plasticity and learning. Handb Clin Neurol. 2022;184:21-34.
[22] ALONSO-NANCLARES L, MERINO-SERRAIS P, GONZALEZ S, et al. Synaptic Changes in the Dentate Gyrus of APP/PS1 Transgenic Mice Revealed by Electron Microscopy. J Neuropathol Exp Neurol. 2013;72(5):386-395.
[23] SHENG C, XU P, ZHOU K, et al. Icariin attenuates synaptic and cognitive deficits in an Aβ1-42-induced rat model of alzheimer’s disease. Biomed Res Int. 2017;2017:7464872.
[24] YANG X, YAO C, TIAN T, et al. Synaptic mechanism in Alzheimer’s disease: a selective degeneration of an excitatory synaptic pathway in the CA1 hippocampus that controls spatial learning and memory in Alzheimer’s disease. Mol Psychiatry. 2018;23(2):167.
[25] KENNEDY MB. The protein biochemistry of the postsynaptic density in glutamatergic synapses mediates learning in neural networks. Biochemi. 2018;57(27):4005-4009.
[26] BLISS TV, COLLINGRIDGE GL. A synaptic model of memory: long-term potentiation in the hippocampus. Nature. 1993;361(6407):31-39.
[27] TOZZI A, SCLIP A, TANTUCCI M, et al. Region- and age-dependent reductions of hippocampal long-term potentiation and NMDA to AMPA ratio in a genetic model of Alzheimer’s disease. Neurobiol Aging. 2015;36(1):123-133.
[28] KOLOS YA, GRIGORIYEV IP, KORZHEVSKYI DE. A synaptic marker synaptophysin. Morfologiia. 2015;147(1):78-82.
[29] MULLANY PM, LYNCH MA. Evidence for a role for synaptophysin in expression of long-term potentiation in rat dentate gyrus. Neuroreport. 1998;9(11):2489-2494.
[30] WU Q, SUN M, BERNARD LP, et al. Postsynaptic density 95(PSD-95) serine 561 phosphorylation regulates a conformational switch and bidirectional dendritic spine structural plasticity. J Biol Chem. 2017; 292(39):16150-16160.
[31] FRANK RA, GRANT SG.Supramolecular organization of NMDA receptors and the postsynaptic density. Curr Opin Neurobiol. 2017;45:139-147.
[32] EHRLICH I, MALINOW R. Postsynaptic density 95 controls AMPA receptor incorporation during long-term potentiation and experience-driven synaptic plasticity. J Neurosci. 2004;24(4):916-927.
[33] KHAN R, KULASIRI D, SAMARASINGHE S. Functional repertoire of protein kinases and phosphatases in synaptic plasticity and associated neurological disorders. Neural Regen Res. 2021;16(6):1150-1157.
[34] DOZMOROV M, LI R, ABBAS AK, et al. Contribution of AMPA and NMDA receptors to early and late phases of LTP in hippocampal slices. Neurosci Res. 2006;55(2):182-188.
[35] ZHANG Y, MATT L, PATRIARCHI T, et al. Capping of the N-terminus of PSD-95 by calmodulin triggers its postsynaptic release. EMBO J. 2014;33(12):1341-1353.
|