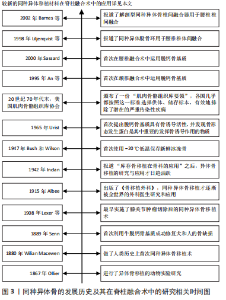
Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (16): 2571-2579.doi: 10.12307/2023.139
Previous Articles Next Articles
Application of allograft bone materials in spinal fusion
Cai Yuanqing, Liu Mozhen, Li Zhonghai
- Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116600, Liaoning Province, China
-
Received:2022-04-16Accepted:2022-05-28Online:2023-06-08Published:2022-11-11 -
Contact:Li Zhonghai, MD, Master’s supervisor, Associate chief physician, Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116600, Liaoning Province, China -
About author:Cai Yuanqing, Master candidate, Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116600, Liaoning Province, China -
Supported by:Science and Technology Benefit Project of Science and Technology Innovation Fund of Dalian, No. 2020JJ27SN070 (to LZH)
CLC Number:
Cite this article
Cai Yuanqing, Liu Mozhen, Li Zhonghai. Application of allograft bone materials in spinal fusion[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2571-2579.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
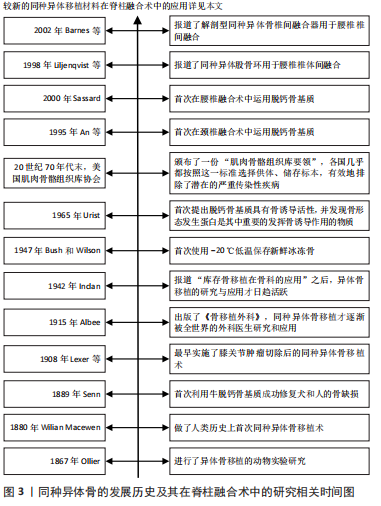
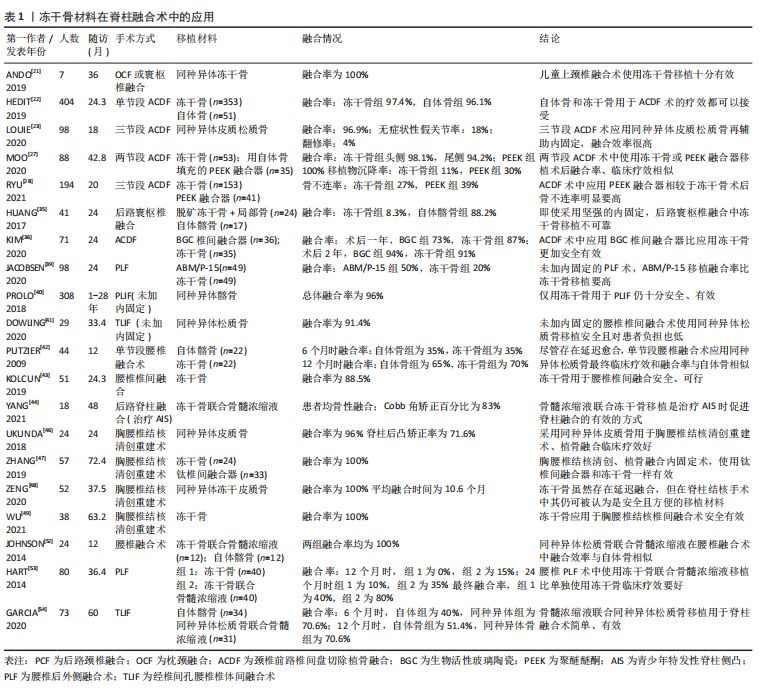
2.2 同种异体骨移植材料的种类 同种异体骨取骨于活体供者(如人工髋关节置换术切除的股骨头和股骨颈等,需供者本人同意)或尸体(需供者生前同意或死者家属同意),通过灭菌、脱水、分离解冻、脱钙等工艺制备出不同的类型,主要包括新鲜冰冻骨、冷冻干燥骨、脱钙骨基质(demineralized bone matrix,DBM)和同种异体骨椎间融合器。 2.2.1 新鲜冰冻骨(下文简称冰冻骨) 是将同种异体骨表面软组织、骨膜和骨端软骨彻底剔除,根据需要制成不同形状、大小的移植骨材料[8]。保存技术方面,Bush和Wilson于1947年首次使用-20 ℃低温保存冰冻骨,但此温度下骨内一些酶活性仍存在,可导致骨结构的破坏[9]。目前常用的保存方法为深低温(-80 ℃)保存,具体冰冻步骤为处理好的异体骨在10%的二甲基亚砜或甘油中浸泡30 min后逐渐降温,再放入-4 ℃的冷库中12 h,最后转入-80 ℃中保存。在-80 ℃的条件下,冰冻骨中酶的活性被破坏从而使骨的完整性得到保存,且由于胶原酶的作用消失,冰冻骨的免疫原性得到一定的降低,冰冻骨可以保存数年[10]。 2.2.2 冷冻干燥骨(下文简称冻干骨) 包括松质骨和皮质骨,冻干骨是通过分离解冻后冷冻骨内的水分,充分脱水,将骨组织内的水分控制在5%以下,在其制备过程中,水分的丢失和辐射的电离破坏了其力学性能[11]。冻干骨的制备过程可有效降低移植物的免疫排斥反应,且由于成骨活性蛋白的保留使其有较强的成骨能力,还兼顾很强的骨传导能力和一定的骨诱导能力,因此,冻干骨是临床应用最为广泛的同种骨移植材料[12]。 2.2.3 脱钙骨基质 1965年,URIST等[13]首次提出脱钙骨基质具有骨诱导活性,并发现骨形态发生蛋白(bone morphogenetic protein,BMP)是其中重要的发挥骨诱导作用的物质。脱钙骨基质是通过一系列化学方法对同种异体骨进行脱钙、去脂等处理,去除了骨的主要矿化成分[14],其主要由胶原蛋白(主要为Ⅰ型、Ⅳ型)、非胶原蛋白、生长因子(如BMP)、少量磷酸钙和细胞碎片等组成[15],具有良好的骨诱导性及一定的骨传导性,且呈低抗原性,在各种骨修复手术中应用广泛[16]。 2.2.4 同种异体骨椎间融合器 Badgy和Kuslich于1983年设计出适用于人体的椎间融合器,即BAK(Bagby and Kuslich)系统[17],用于下腰痛患者的腰椎融合术。同种异体骨所制成的椎间融合器具有三维网状结构,易被新生骨爬行替代,同时同种异体骨有较强的骨传导和骨诱导作用且其弹性模量更接近于人骨,所以临床上发展出了采用同种异体腓骨、股骨等长骨简单加工后形成的早期环状融合器。由于其形状和大小所限,只能用于前路椎间融合术,且多用于腰椎[18]。针对简单腓骨环、股骨环的不足,符合椎间隙解剖学特点的融合器逐渐应用于椎间融合术。 2.3 冰冻骨在脊柱融合术中的应用 冰冻骨具有最高的机械稳定性,但由于存在免疫排斥反应和传播HIV、肝炎病毒等危险[19],在临床中较少运用。而KASIS等[20]通过将冰冻骨和自体髂骨混合填充入椎间融合器中用于前路腰椎椎间融合术,发现术后融合率为94%,患者预后良好,且使用冰冻骨可避免大量取骨引发的取骨区疼痛等症状。 2.4 冻干骨在脊柱融合术中的应用 2.4.1 冻干骨在颈椎融合术中的应用 冻干骨材料在颈椎融合术中已得到广泛应用,大多数研究已证实,颈椎融合术中使用冻干骨材料融合率高、假关节率和翻修率低,可避免自体骨取骨引发的取骨区疼痛等并发症,是颈椎融合术中良好的骨移植材料。ANDO等[21]研究发现,用冻干骨材料对7例儿童行枕颈融合或寰枢椎融合,患儿术后均骨性融合。HEIDT等[22]研究显示单节段颈椎前路椎间盘切除植骨融合术(anterior cervical discectomy and fusion,ACDF)中应用冻干骨移植术后融合率超过96%。LOUIE等[23]3个节段ACDF术的研究中,患者术后融合率为96.9%,无症状性假关节率和翻修率分别为18%和4%。 聚醚醚酮椎间融合器因其良好的生物相容性已被广泛应用于ACDF术中[24],ACDF术中使用冻干骨移植相较于聚醚醚酮融合器移植,术后骨不连率低,患者预后更好。并且在单节段和多节段ACDF术中,冻干骨移植术后假关节发生率都比聚醚醚酮融合器要低[25-26]。MOO等[27]一项平均随访42个月的研究显示,2个节段ACDF术使用冻干骨或聚醚醚酮融合器术后融合效率相似,但使用聚醚醚酮融合器术后移植物沉降率更高(30%比11%)。RYU等[28]一项研究中纳入194例行ACDF术患者(接受冻干骨移植为79%,聚醚醚酮融合器为21%),与聚醚醚酮融合器相比,使用冻干骨移植患者术后骨不连率低(37.7%比62.5%)。然而,融合效率不仅和移植物的选择有关,还和融合的节段数目、患者年龄、吸烟状态相关[29-32],因为脊柱融合术后患者出现骨不连可导致复发性疼痛、神经根病和后凸畸形,严重影响预后,所以临床医生应综合考虑上述因素,为患者做出最佳的移植选择。 存在部分研究表明,颈椎融合术中冻干骨移植存在融合率低、延迟愈合、移植物沉降率高等问题,所以更推荐自体骨移植[33-34]。HUANG等[35]通过前瞻性研究分析41例行后路寰枢椎融合术患者的资料,发现自体骨移植融合率为88.2%,冻干骨融合率仅为8.3%,他们认为冻干骨不适用于寰枢椎融合术。KIM等[36]回顾了36例用生物活性玻璃陶瓷和35例用冻干骨行ACDF术患者的临床资料,发现术后2年两组融合率相似(陶瓷组94%,冻干骨组91%),但冻干骨组移植物沉降率更高(43%和19%)。 2.4.2 冻干骨在腰椎融合术中的应用 部分研究认为,腰椎融合术中应用冻干骨材料存在着费用较高、在未辅助内固定的腰椎融合术中融合率较低等问题[37-38]。JACOBSEN等[39]研究发现冻干骨移植在60岁以上行腰椎后外侧融合术(posterolateral lumbar fusion,PLF)的患者中术后融合率仅有20%。而部分学者与之前研究结果不同,肯定了冻干骨材料在未加内固定的腰椎融合术中的融合效果。在两项未加内固定的研究中,PROLO等[40]对1981-2006年间308例将同种异体髂骨用于后路腰椎椎间融合患者的回顾性分析发现,患者术后总体融合率为96%。DOWLING等[41]将同种异体松质冻干骨用于经椎间孔腰椎椎间融合术(transforaminal lumbar interbody fusion,TLIF),患者术后融合率为91.4%。 冻干骨在未加内固定的腰椎融合术中的融合效率各学者意见存在分歧,但是其融合效率在辅助内固定的一些研究中得到一致肯定。PUTZIER等[42]一项研究发现,单节段腰椎融合术使用冻干松质骨在6个月时相较自体髂骨组融合率明显偏低(15%和35%),但是12个月时两组融合率相当(65%和70%),即使存在着融合延迟,作者仍推荐冻干骨用于单节段腰椎融合术。KOLCUN等[43]回顾了51例将冻干骨用于腰椎椎间融合术患者的资料,平均随访时间为24.3个月,术后融合率为88.5%。YANG等[44]将冻干骨用于青少年特发性脊柱侧凸(adolescent idiopathic scoliosis,AIS)后路融合术移植,患者术后均骨性融合,且Cobb角矫正百分比为83%。 脊柱结核通常累及前柱和中柱,造成骨质破坏和脊柱不稳,除了抗结核治疗外,治疗方式包括前柱的清创重建、植骨融合内固定等,目的是重建因结核而破坏的脊柱正常的生理曲度,恢复脊柱的稳定性,而胸腰椎结核植骨融合过程中使用同种异体冻干骨移植材料,患者术后融合率高,且能有效矫正胸腰椎结核所致的后凸畸形[45]。UKNUDA等[46]通过对24例胸腰椎结核术中使用冻干骨材料患者的随访,发现术后融合率为96%,脊柱后凸矫正率为71.6%。ZHANG等[47]和ZENG等[48]分别在后路和前路的胸腰椎结核手术中使用冻干骨材料,两组患者术后均骨性融合。WU等[49]在钛金属椎间融合器中填充冻干骨用于胸腰椎结核后路腰椎椎间融合术,患者术后也全部融合。当然,脊柱结核治疗的基本原则是个体化综合治疗,包括休息制动、抗结核药物、手术等,脊柱结核是否病灶清除、一期植骨融合目前还存在争议,应该严格按照手术指征,谨慎选择手术治疗[50]。 骨髓浓缩液是由骨髓穿刺液浓缩而来,因其富含间充质干细胞、血小板和其他生长因子,已被证实能促进骨骼和肌肉组织再生[51],具有良好的骨诱导活性,多位学者研究证明骨髓浓缩液能显著提高冻干骨材料在腰椎融合术中的融合效率。JOHNSON等[52]一项前瞻性研究显示,腰椎融合术中同种异体松质骨与骨髓浓缩液混合移植和自体髂骨移植两组患者术后均骨性融合。HART等[53]发现PLF术中应用自体骨髓浓缩液联合冻干骨移植融合率能达到80%。GARCíA等[54]一项关于自体骨髓浓缩液联合冻干骨或自体骨用于腰椎TLIF和前路腰椎融合术移植的研究结果显示,自体骨组在术后6,12个月的融合率是40%、51.4%,而冻干骨组术后6,12个月的融合率均可达70.6%,因此他们认为腰椎融合术中骨髓浓缩液联合冻干骨移植是可行且有效的。 综上,颈椎和腰椎融合术中应用冻干骨总体融合率都很高,但是未辅助内固定的腰椎融合术中应用冻干骨临床疗效存在一定分歧;腰椎融合术中,冻干骨材料与骨髓浓缩液的混合移植能极大地促进其融合效率,使其在治疗腰椎结核、创伤和纠正脊柱畸形方面发挥中越来越重要的作用。冻干骨材料在脊柱融合术中的应用详见表1。"
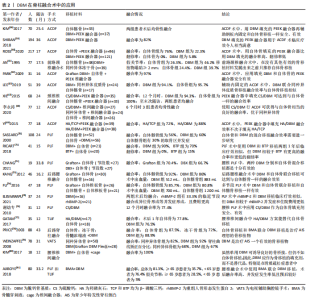
2.5 脱钙骨基质在脊柱融合术中的应用 1889年,SENN等[55]首次报道了脱钙骨基质在治疗骨缺损中的成功应用。脱钙骨基质是一种多孔、可生物降解的骨移植替代材料,其中富含BMP等生长因子,使其具有良好的骨诱导和骨传导能力。但不同形式的脱钙骨基质产品不易于临床医师运用,其原因在于脱钙骨基质常呈颗粒状或粉末状,术中会因为冲洗或血流因素而弥散;且其形状常与融合部位不匹配,会导致延迟融合。所以,临床上脱钙骨基质常常是与其他载体联用,最终产品外形可塑,可与融合部位牢固结合[15]。与脱钙骨基质联用的载体通常包括甘油、泊洛沙姆、硫酸钙、羟磷灰石、透明质酸等,其最终的脱钙骨基质产品名称包括Grafton DBM、OrthoblastⅡ、llomatrix、DBX等[56-57]。 2.5.1 脱钙骨基质在颈椎融合术中的应用 颈椎融合术中,临床研究部分集中于脱钙骨基质填充于聚醚醚酮融合器在ACDF术中的临床疗效。KIM等[58]研究发现,单节段ACDF术中应用脱钙骨基质填充的聚醚醚酮融合器或应用自体骨移植患者术后100%融合,且脱钙骨基质组患者术后椎间高度、颈椎节段角保持的均比自体骨组要好,他们认为ACDF术中应用脱钙骨基质填充的聚醚醚酮融合器安全有效。这与SHIBAN等[59]研究结果一致,该项研究中患者术后融合率为82%。而ROSS等[60]比较了ACDF术中分别用脱钙骨基质和自体骨填充聚醚醚酮融合器移植的融合效率,结果显示脱钙骨基质组术后融合率仅为22.3%,翻修率为5.8%;自体骨组融合率为76%,无患者需翻修手术,他们分析脱钙骨基质组患者低融合率可能与患者年龄偏大、吸烟占比多有关。 脱钙骨基质常作为移植物填充剂应用于颈椎融合术中,不仅能增加移植物的容积,再加上其良好的骨诱导和骨传导能力,还能潜在的诱导成骨,有着不错的运用。甘油具有良好的生物相容性和黏性,常作为脱钙骨基质的载体,其最终产品为Grafton DBM。1995年,AN等[61]首次报道了Grafton DBM在颈椎中的应用,该研究发现颈椎融合术中与自体骨移植相比,脱钙骨基质联合冻干骨移植术后假关节发生率高(脱钙骨基质组46%,自体骨组26%),且脱钙骨基质组移植物塌陷率也很高(脱钙骨基质组39.7%,自体骨组24.4%)。而使用钢板或螺钉固定可有效改善患者的预后,提高脱钙骨基质作为移植物填充剂运用时的融合效率。在两项辅助内固定的研究中,PARK等[62]在31例患者中使用填充自体骨和Grafton DBM的聚醚醚酮融合器,术后1年患者融合率为97%,且目测类比评分和JOA评分有着显著改善。LEE等[63]研究发现,ACDF术中脱钙骨基质(Orthoblast Ⅱ)联合同种异体皮质骨移植术后融合效率与自体髂骨相似(分别为94.1%和95.7%)。 与脱钙骨基质联用的载体还包括羟基磷灰石(hydroxyapatite,HA)和硫酸钙(calcium sulphate,CS)等,硫酸钙可煅烧成半水硫酸钙,具有塑形作用,非常适用于填充不规则的融合部位,且融合部位局部钙离子浓度的升高可促进成骨细胞的分化,从而有效地促进融合[64-65]。羟基磷灰石具有良好的骨传导能力和一定的骨诱导能力,常作为移植物的填充剂或替代品[66]。XIE等[67]比较了ACDF术中分别运用羟基磷灰石/脱钙骨基质或自体骨填充于聚醚醚酮融合器的融合效率,结果显示术后1年脱钙骨基质组融合率较自体骨组低(94.3%和100%),但是末次随访两组患者均骨性融合。李永涛等[68]在ACDF术中运用填充硫酸钙/脱钙骨基质的融合器移植,患者术后6个月均融合。YI等[69]研究了羟基磷灰石/β-磷酸三钙(β-tricalcium phosphate, TCP)和羟基磷灰石/脱钙骨基质填充于聚醚醚酮融合器在ACDF术中的融合效果,术后1年羟基磷灰石/β-磷酸三钙组患者融合率为72%,羟基磷灰石/脱钙骨基质组为88%。 2.5.2 脱钙骨基质在腰椎融合术中的应用 腰椎融合术中,脱钙骨基质常作为自体骨填充剂,其融合效率同样值得肯定。SASSARD等[70]首次报道了脱钙骨基质在腰椎中的应用,该研究比较了Grafton DBM联合自体骨移植和自体髂骨移植在辅助内固定的PLF术中的融合效率,最终两组融合效率相似,脱钙骨基质组融合率为60%,自体髂骨组为56%,且自体髂骨组有着30%的取骨区并发症。RICART等[71]一项研究对比了自体骨分别联合Grafton DBM或磷酸三钙在PLF术中的融合效率,结果显示脱钙骨基质组有较高的融合效率(90%比70%)和较低的翻修率(0%比15%)。CHANG等[72]比较了Grafton和DBX这两种脱钙骨基质作为自体骨填充剂时在PLF术中的融合效率,最终结果显示两组融合率相当(Grafton组70.4%,DBX组66.7%),因此他们认为这两种脱钙骨基质作为自体骨移植填充材料用于PLF十分有效。 多项研究也表明,与其他移植物相比,腰椎融合术中应用脱钙骨基质联合自体骨移植,在达到满意的融合效率的同时,还能减少手术时间、降低术中出血量、降低患者住院时间,且治疗费用也较低。KANG等[73]一项前瞻性随机对照试验显示,单节段腰椎PLF术中应用自体骨联合脱钙骨基质术后融合率和应用自体髂骨相似,但是脱钙骨基质组有较低的术中出血量(脱钙骨基质组512 mL,自体髂骨组883 mL)和较短的手术时间(脱钙骨基质组200 min,自体髂骨组226 min)。FU等[74]多节段腰椎PLF的研究得到同样的结果,即应用脱钙骨基质能较少术中出血量(脱钙骨基质组700 mL,自体髂骨组1 200 mL)。ELESWARAPU等[75]一项队列研究结果显示,腰椎融合术中脱钙骨基质联合局部自体骨移植与人重组骨形态发生蛋白2移植两组术后均全部融合,且脱钙骨基质组患者治疗费用明显更低(脱钙骨基质组平均费用为1 522美元,人重组骨形态发生蛋白2组为3 505美元)。 硫酸钙和羟基磷灰石作为脱钙骨基质载体在腰椎融合术中运用时,可达到与自体骨移植相似的融合效率,临床疗效令人满意。谢幼专等[76]将硫酸钙/脱钙骨基质作为自体骨填充剂应用于腰椎PLF术中,末次随访时31例患者融合率为77.4%,无感染、渗出等并发症发生,他们认为硫酸钙/脱钙骨基质作为自体骨填充剂应用于腰椎PLF术安全、有效。GATAM等[77]分别将羟基磷灰石/脱钙骨基质和自体骨用于腰椎椎间融合术中,术后1年患者融合率脱钙骨基质组为76.5%,自体骨组为77.8%,作者认为腰椎椎间融合中羟基磷灰石/脱钙骨基质方案能替代自体骨移植。 2.5.3 脱钙骨基质在治疗AIS中的应用 由于脊柱侧凸的矫正需要广泛的融合,脱钙骨基质可以作为自体骨的替代骨移植选择,在AIS治疗中也有着不错的应用效果。PRICE等[78]的一项回顾性研究比较了AIS的3种移植选择:自体髂骨、冻干骨、脱钙骨基质与骨髓穿刺液联合移植,结果显示自体骨组和脱钙骨基质组融合率相似(分别为87.5%和88.9%),而冻干骨组融合较低(72%),所以AIS治疗中作者推荐使用骨髓穿刺液联合脱钙骨基质移植。WEINZAPFEL等[79]研究了40例后路脊柱融合治疗AIS病例,脊柱曲线矫正百分比方面,同种异体骨组为68%,脱钙骨基质组为67%;融合率方面,同种异体骨组82%,脱钙骨基质组92%。作者认为,脱钙骨基质用于AIS的融合术安全有效。 但是,也有研究发现脱钙骨基质在骨质疏松患者和老年患者中使用时,存在着术后融合率低、假关节等并发症发生率高、骨形成较慢等各种问题。KIM等[80]研究发现,骨质疏松患者PLF术中应用脱钙骨基质移植,尽管术后患者均有桥接骨形成,但融合部位骨生长较少。AJIBOYE等[81]研究结果证实,65岁以上患者在PLF术中应用脱钙骨基质联合骨髓穿刺液移植,术后融合率仅35.7%,假关节率达28.6%,而在65岁以下患者中,术后融合率为76.4%,假关节率为5%。因此需要进一步的研究来阐明脱钙骨基质在这些更具体的情况下的使用。脱钙骨基质在脊柱融合术中的应用详见表2。"
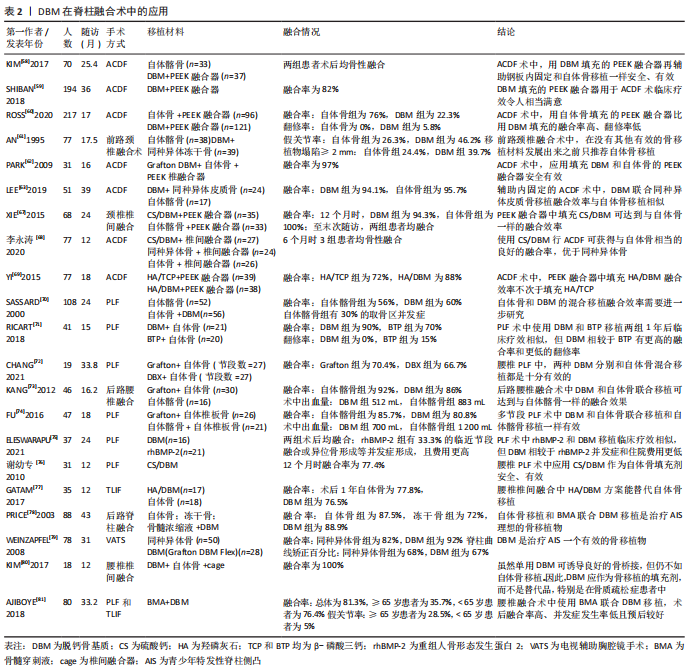
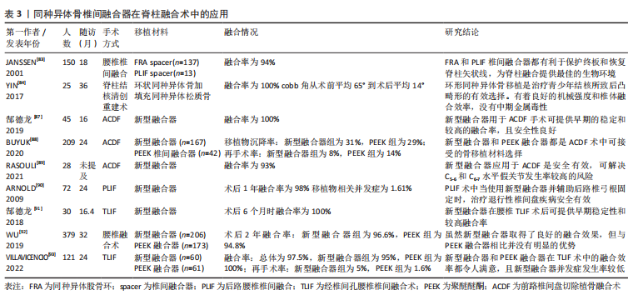
综上,脱钙骨基质具有良好的骨诱导、骨传导活性,使其在颈椎和腰椎融合术中应用十分广泛。脱钙骨基质制备过程中,骨的无机成分的部分丧失和生长因子的活性受影响,其力学性能及成骨能力下降,且脱钙骨基质常与融合部位形状不相匹配,为达到在融合部位的充分固定和提高融合效果,其常与其它材料联合使用,如甘油、羟基磷灰石、硫酸钙等,也常作为移植物填充剂以增加移植物容积,使其在脊柱融合中的融合效率得到进一步提高。同时,脱钙骨基质的运用还有着减少手术时间、降低术中出血量、降低患者住院时间等优点。但是脱钙骨基质还是存在着一些缺点,例如由于制备工艺还不够标准化,生产出的脱钙骨基质产品之间骨诱导活性差异大、在老年人和骨质疏松患者中运用时融合率低且并发症发生率高等,所以脱钙骨基质产品的制作工艺需要进一步发展,也需要更多的临床与基础研究来证明脱钙骨基质的安全有效性[57, 82]。 2.6 同种异体骨椎间融合器在脊柱融合术中的应用 2.6.1 早期环状同种异体骨椎间融合器在脊柱融合术中的应用 早期融合器制作工艺简单,但其仍具有良好的骨诱导和骨传导能力,是早期脊柱融合术中良好的骨移植材料。JANSSEN等[83]对137例行后路腰椎融合术的患者采用同种异体股骨环移植,术后融合率为94%。YIN等[84]回顾了25例脊柱结核伴后凸畸形的患者使用同种异体骨环治疗的临床资料,术后3个月患者均骨性融合,且Cobb角从术前的平均65°矫正到术后的平均14°。尽管同种异体骨环有着不错的临床运用效果,但因其规格不规范、尺寸过大或过小、未加工的天然外形使得医生难以操作、容易出现移植物移碎裂、沉降等缺点,难以适应现代微创脊柱外科的发展需求[85-86]。 2.6.2 同种异体骨椎间融合器在颈椎融合术中的应用 针对以上早期融合器的不足,新型解剖型同种异体骨椎间融合器逐渐发展并运用于脊柱融合术,因其取材不受限制、外形可加工、符合解剖学特点,在颈椎融合术中运用融合率高、并发症发生率低。尤其是在ACDF术中,郜德龙等[87]对45例使用新型融合器行ACDF术的患者进行了平均16个月的随访,发现患者术后全部融合。BUYUK等[88]对比了167例使用新型融合器和42例使用聚醚醚酮融合器用于ACDF术患者的术后再手术率,结果显示,新型融合器组和聚醚醚酮组移植物沉降率分别为31%和29%,因假关节而需的翻修率分别为8%和14%,他们认为这两种融合器都是ACDF术中可接受的骨移植材料选择。RASOULI等[89]对28例行ACDF术患者使用新型融合器移植,患者术后融合率为93%。 2.6.3 新型融合器在腰椎融合术中的应用 腰椎融合术中,使用新型融合器早期融合率高,移植物移位、因假关节而需翻修手术等并发症发生率较低,能改善患者的脊柱功能,可获得较好的临床预后结果。ARNOLD等[90]报道了72例患者使用新型融合器进行PLIF术的结果,发现术后1年的融合率为98%,移植物并发症仅为1.61%,证明了其安全有效性。郜德龙等[91]对30例用新型融合器行TLIF的患者平均16个月的随访发现,患者术后均融合,仅1例术后6个月出现移植物下缘碎裂,不伴移植物移位等其他并发症。WU等[92]研究发现,新型融合器相较PEEK融合器在腰椎融合术术后融合率相似(97.57%和96.53%),但融合所需时间更短。VILLAVICENCIO等[93]对121例TLIF术中分别使用新型融合器和聚醚醚酮融合器的患者进行了平均2年的随访,发现术后聚醚醚酮组全部融合,翻修率为1.6%;新型融合器组融合率为95%,翻修率为5%,他们认为两种融合器对患者预后提升都很明显。新型融合器在脊柱融合术中的应用详见表3。"


综上,早期环状融合器和新型融合器都有着不错的融合效率,且由于新型融合器有着外形可加工、符合解剖结构等特点,临床融合效率更好,在脊柱融合术后维持椎间隙高度和椎间孔正常形状也更好,是脊柱融合术中良好的选择。但是,股骨和腓骨的皮质骨所制成的解剖型椎间融合器都有着不同程度的移植物碎裂等并发症发生,这与其刚性较低有关;并且在重度骨质疏松患者中运用和聚醚醚酮等融合器一样容易发生移植物沉降,这些都是限制其发展的关键因素[94-95]。当与聚醚醚酮融合器相比时,新型融合器弹性模量更接近于人骨,且生物力学性能更好,但是脊柱融合术中运用时,其在融合效率、并发症等方面并没有明显的优势,所以临床上新型融合器不能完全替代聚醚醚酮融合器[92]。新型融合器成骨活性好,但是还需更多的大样本、多中心临床随机对照研究以充分验证其运用效率。"

| [1] RAVINDRA VM, SENGLAUB SS, RATTANI A, et al. Degenerative Lumbar Spine Disease: Estimating Global Incidence and Worldwide Volume. Global Spine J. 2018;8(8):784-794. [2] LOMAS R, CHANDRASEKAR A, BOARD TN. Bone allograft in the U.K.: perceptions and realities. Hip Int. 2013;23(5):427-433. [3] ITO H, KOEFOED M, TIYAPATANAPUTI P, et al. Remodeling of cortical bone allografts mediated by adherent rAAV-RANKL and VEGF gene therapy. Nat Med. 2005;11(3):291-297. [4] URABE K, ITOMAN M, TOYAMA Y, et al. Current trends in bone grafting and the issue of banked bone allografts based on the fourth nationwide survey of bone grafting status from 2000 to 2004. J Orthop Sci. 2007;12(6):520-525. [5] STERNHEIM A, DREXLER M, KUZYK PR, et al. Treatment of failed allograft prosthesis composites used for hip arthroplasty in the setting of severe proximal femoral bone defects. J Arthroplasty. 2014;29(5):1058-1062. [6] BUTTERMANN GR, GLAZER PA, BRADFORD DS. The use of bone allografts in the spine. Clin Orthop Relat Res. 1996;(324):75-85. [7] 李丹,毕龙,胡蕴玉.综合骨库在现代矫形外科中的作用[J].中国矫形外科杂志,2009,17(21):1639-1642 [8] SUAREZ LS, RICHMOND JC. Overview of procurement, processing, and sterilization of soft tissue allografts for sports medicine. Sports Med Arthrosc Rev. 2007;15(3):106-113. [9] CURTISS PH Jr, HERNDON CH. Immunological factors in homogenous-bone transplantation. I. Serological studies. J Bone Joint Surg Am. 1956;38-a(1):103-110. [10] BOYCE T, EDWARDS J, SCARBOROUGH N. Allograft bone. The influence of processing on safety and performance. Orthop Clin North Am. 1999;30(4): 571-581. [11] APONTE-TINAO LA, RITACCO LE, ALBERGO JI, et al. The principles and applications of fresh frozen allografts to bone and joint reconstruction. Orthop Clin North Am. 2014;45(2):257-269. [12] STEVENSON S. Enhancement of fracture healing with autogenous and allogeneic bone grafts. Clin Orthop Relat Res. 1998;(355 Suppl):S239-246. [13] URIST MR. Bone: formation by autoinduction. Science. 1965;150(3698): 893-899. [14] SCHUBERT T, LAFONT S, BEAURIN G, et al. Critical size bone defect reconstruction by an autologous 3D osteogenic-like tissue derived from differentiated adipose MSCs. Biomaterials. 2013;34(18):4428-4438. [15] GRUSKIN E, DOLL BA, FUTRELL FW, et al. Demineralized bone matrix in bone repair: history and use. Adv Drug Deliv Rev. 2012;64(12):1063-1077. [16] HOLT DJ, GRAINGER DW. Demineralized bone matrix as a vehicle for delivering endogenous and exogenous therapeutics in bone repair. Adv Drug Deliv Rev. 2012;64(12):1123-1128. [17] BAGBY GW. Arthrodesis by the distraction-compression method using a stainless steel implant. Orthopedics. 1988;11(6):931-934. [18] LILJENQVIST U, O’BRIEN JP, RENTON P. Simultaneous combined anterior and posterior lumbar fusion with femoral cortical allograft. Eur Spine J. 1998;7(2): 125-131. [19] EHRLER DM, VACCARO AR. The use of allograft bone in lumbar spine surgery. Clin Orthop Relat Res. 2000;(371):38-45. [20] KASIS AG, JENSEN C, DHARMADHIKARI R, et al. Novel bone grafting technique in stand-alone ALIF procedure combining allograft and autograft (‘Northumbria Technique’)-Fusion rate and functional outcomes in 100 consecutive patients. Eur Spine J. 2021;30(5):1296-1302. [21] ANDO K, KOBAYASHI K, ITO K, et al. Occipitocervical or C1-C2 fusion using allograft bone in pediatric patients with Down syndrome 8 years of age or younger. J Pediatr Orthop B. 2019;28(4):405-410. [22] HEIDT ST, LOUIE PK, KHAN JM, et al. Comparing Allografts to Autografts for Maintenance of Cervical Sagittal Parameters and Clinical Outcomes Following Anterior Cervical Discectomy and Fusion With Anterior Cervical Plating. Neurospine. 2019;16(3):618-625. [23] LOUIE PK, SEXTON AC, BOHL DD, et al. Rigid-Plating and Cortico-Cancellous Allograft Are Effective for 3-Level Anterior Cervical Discectomy and Fusion: Radiographic and Clinical Outcomes. Neurospine. 2020;17(1):146-155. [24] SEAMAN S, KEREZOUDIS P, BYDON M, et al. Titanium vs. polyetheretherketone (PEEK) interbody fusion: Meta-analysis and review of the literature. J Clin Neurosci. 2017;44: 23-9. [25] KRAUSE KL, OBAYASHI JT, BRIDGES KJ, et al. Fivefold higher rate of pseudarthrosis with polyetheretherketone interbody device than with structural allograft used for 1-level anterior cervical discectomy and fusion. J Neurosurg Spine. 2018;30(1):46-51. [26] TETON ZE, CHEANEY B, OBAYASHI JT, et al. PEEK interbody devices for multilevel anterior cervical discectomy and fusion: association with more than 6-fold higher rates of pseudarthrosis compared to structural allograft. J Neurosurg Spine. 2020:1-7. doi: 10.3171/2019.11.SPINE19788. [27] MOO IH, KAM CJW, LAI MWS, et al. A comparison of contiguous two-level anterior cervical discectomy and fusion using a structural allograft versus a Polyetheretherketone (PEEK) cage: the results of a three-year follow-up. BMC Musculoskelet Disord. 2020;21(1):331. [28] RYU WHA, RICHARDS D, KEROLUS MG, et al. Nonunion Rates After Anterior Cervical Discectomy and Fusion: Comparison of Polyetheretherketone vs Structural Allograft Implants. Neurosurgery. 2021;89(1):94-101. [29] LI Y, ZHENG LM, ZHANG ZW, et al. The Effect of Smoking on the Fusion Rate of Spinal Fusion Surgery: A Systematic Review and Meta-Analysis. World Neurosurg. 2021;154:e222-e235. [30] PIRKLE S, KASKOVICH S, COOK DJ, et al. Cages in ACDF are Associated With a Higher Nonunion Rate Than Allograft: A Stratified Comparative Analysis of 6130 Patients. Spine (Phila Pa 1976). 2019;44(6):384-388. [31] BOLESTA MJ, RECHTINE GR 2nd, CHRIN AM. Three- and four-level anterior cervical discectomy and fusion with plate fixation: a prospective study. Spine (Phila Pa 1976). 2000;25(16):2040-2044; discussion 5-6. [32] PHILLIPS FM, CARLSON G, EMERY SE, et al. Anterior cervical pseudarthrosis. Natural history and treatment. Spine (Phila Pa 1976). 1997;22(14):1585-1589. [33] PARK JH, BAE YK, SUH SW, et al. Efficacy of cortico/cancellous composite allograft in treatment of cervical spondylosis. Medicine (Baltimore). 2017; 96(33):e7803. [34] WOO JB, SON DW, LEE SH, et al. Risk Factors of Allogenous Bone Graft Collapse in Two-Level Anterior Cervical Discectomy and Fusion. J Korean Neurosurg Soc. 2019;62(4):450-457. [35] HUANG DG, ZHANG XL, HAO DJ, et al. Posterior atlantoaxial fusion with a screw-rod system: Allograft versus iliac crest autograft. Clin Neurol Neurosurg. 2017;162:95-100. [36] KIM HC, OH JK, KIM DS, et al. Comparison of the effectiveness and safety of bioactive glass ceramic to allograft bone for anterior cervical discectomy and fusion with anterior plate fixation. Neurosurg Rev. 2020;43(5):1423-1430. [37] THEOLOGIS AA, TABARAEE E, LIN T, et al. Type of bone graft or substitute does not affect outcome of spine fusion with instrumentation for adolescent idiopathic scoliosis. Spine (Phila Pa 1976). 2015;40(17):1345-1351. [38] BUSER Z, BRODKE DS, YOUSSEF JA, et al. Allograft Versus Demineralized Bone Matrix in Instrumented and Noninstrumented Lumbar Fusion: A Systematic Review. Global Spine J. 2018;8(4):396-412. [39] JACOBSEN MK, ANDRESEN AK, JESPERSEN AB, et al. Randomized double blind clinical trial of ABM/P-15 versus allograft in noninstrumented lumbar fusion surgery. Spine J. 2020;20(5):677-684. [40] PROLO LM, OKLUND SA, ZAWADZKI N, et al. Uninstrumented Posterior Lumbar Interbody Fusion: Have Technological Advances in Stabilizing the Lumbar Spine Truly Improved Outcomes? World Neurosurg. 2018;115:490-502. [41] DOWLING Á, BáRCENAS JGH, LEWANDROWSKI KU. Transforaminal endoscopic decompression and uninstrumented allograft lumbar interbody fusion: A feasibility study in patients with end-stage vacuum degenerative disc disease. Clin Neurol Neurosurg. 2020;196:106002. [42] PUTZIER M, STRUBE P, FUNK JF, et al. Allogenic versus autologous cancellous bone in lumbar segmental spondylodesis: a randomized prospective study. Eur Spine J. 2009;18(5):687-695. [43] KOLCUN JPG, GHOBRIAL GM, CRANDALL KM, et al. Minimally Invasive Lumbar Interbody Fusion With an Expandable Meshed Allograft Containment Device: Analysis of Subsidence With 12-Month Minimum Follow-Up. Int J Spine Surg. 2019;13(4):321-328. [44] YANG P, XING J, CHEN B, et al. The clinical use of the enriched bone marrow obtained by selective cell retention technology in treating adolescent idiopathic scoliosis. J Orthop Translat. 2021;27:146-152. [45] MEENA S, MITTAL S, CHOWDHARY B. Spinal tuberculosis: which is the best surgical approach? Med Princ Pract. 2014;23(1):96. [46] UKUNDA UNF, LUKHELE MM. The posterior-only surgical approach in the treatment of tuberculosis of the spine: outcomes using cortical bone allografts. Bone Joint J. 2018;100-b(9):1208-1213. [47] ZHANG HQ, LI M, WANG YX, et al. Minimum 5-Year Follow-Up Outcomes for Comparison Between Titanium Mesh Cage and Allogeneic Bone Graft to Reconstruct Anterior Column Through Posterior Approach for the Surgical treatment of Thoracolumbar Spinal Tuberculosis with Kyphosis. World Neurosurg. 2019;127:e407-e415. [48] ZENG Y, FAN Y, LUO F, et al. Tricortical iliac crest allograft with anterolateral single rod screw instrumentation in the treatment of thoracic and lumbar spinal tuberculosis. Sci Rep. 2020;10(1):13037. [49] WU W, LI Z, LIN R, et al. Single-stage posterior-only debridement, decompression and interbody fusion for the treatment of thoracolumbar spinal tuberculosis complicated with psoas abscesses. BMC Surg. 2021;21(1):84. [50] 王自立,王骞.脊柱结核的手术策略[J].中华骨科杂志,2010,30(7):717-723. [51] CHEN FH, TUAN RS. Mesenchymal stem cells in arthritic diseases. Arthritis Res Ther. 2008;10(5):223. [52] JOHNSON RG. Bone marrow concentrate with allograft equivalent to autograft in lumbar fusions. Spine (Phila Pa 1976). 2014;39(9):695-700. [53] HART R, KOMZáK M, OKáL F, et al. Allograft alone versus allograft with bone marrow concentrate for the healing of the instrumented posterolateral lumbar fusion. Spine J. 2014;14(7):1318-1324. [54] GARCíA DE FRUTOS A, GONZáLEZ-TARTIèRE P, COLL BONET R, et al. Randomized clinical trial: expanded autologous bone marrow mesenchymal cells combined with allogeneic bone tissue, compared with autologous iliac crest graft in lumbar fusion surgery. Spine J. 2020;20(12):1899-1910. [55] No authors listed. Senn on the Healing of Aseptic Bone Cavities by Implantation of Antiseptic Decalcified Bone. Ann Surg. 1889;10(5):352-368. [56] VAN DER STOK J, HARTHOLT KA, SCHOENMAKERS DAL, et al. The available evidence on demineralised bone matrix in trauma and orthopaedic surgery: A systematic review. Bone Joint Res. 2017;6(7):423-432. [57] ZHANG H, YANG L, YANG XG, et al. Demineralized Bone Matrix Carriers and their Clinical Applications: An Overview. Orthop Surg. 2019;11(5):725-737. [58] KIM SH, LEE JK, JANG JW, et al. Polyetheretherketone Cage with Demineralized Bone Matrix Can Replace Iliac Crest Autografts for Anterior Cervical Discectomy and Fusion in Subaxial Cervical Spine Injuries. J Korean Neurosurg Soc. 2017;60(2):211-219. [59] SHIBAN E, NIES M, KOGLER J, et al. No correlation between radiological and clinical outcome 1 year following cervical arthrodesis. Acta Neurochir (Wien). 2018;160(4):845-853. [60] ROSS DA, POLLOCK JM, LI NP, et al. Anterior Cervical Arthrodesis With Polyetheretherketone Spacers: What is the Role of the Grafting Material? Clin Spine Surg. 2020;33(10):E539-e544. [61] AN HS, SIMPSON JM, GLOVER JM, et al. Comparison between allograft plus demineralized bone matrix versus autograft in anterior cervical fusion. A prospective multicenter study. Spine (Phila Pa 1976). 1995;20(20):2211-2216. [62] PARK HW, LEE JK, MOON SJ, et al. The efficacy of the synthetic interbody cage and Grafton for anterior cervical fusion. Spine (Phila Pa 1976). 2009;34(17): E591-595. [63] LEE JC, JANG HD, AHN J, et al. Comparison of Cortical Ring Allograft and Plate Fixation with Autologous Iliac Bone Graft for Anterior Cervical Discectomy and Fusion. Asian Spine J. 2019;13(2):258-264. [64] GAO C, HUO S, LI X, et al. Characteristics of calcium sulfate/gelatin composite biomaterials for bone repair. J Biomater Sci Polym Ed. 2007; 18(7):799-824. [65] THOMAS MV, PULEO DA, AL-SABBAGH M. Calcium sulfate: a review. J Long Term Eff Med Implants. 2005;15(6):599-607. [66] HUANG H, JIANG C, FENG Z, et al. Comparing the process of creeping substitution between allograft bone and local bone grafting in lumbar interbody fusion. Eur Spine J. 2014;23(10):2068-2074. [67] XIE Y, LI H, YUAN J, et al. A prospective randomized comparison of PEEK cage containing calcium sulphate or demineralized bone matrix with autograft in anterior cervical interbody fusion. Int Orthop. 2015;39(6):1129-1136. [68] 李永涛,宋文慧,刘昌文.硫酸钙/脱钙骨基质和同种异体骨在颈椎前路椎间盘切除减压融合中的疗效对比[J].中国组织工程研究,2020,24(34): 5478-5485. [69] YI J, LEE GW, NAM WD, et al. A Prospective Randomized Clinical Trial Comparing Bone Union Rate Following Anterior Cervical Discectomy and Fusion Using a Polyetheretherketone Cage: Hydroxyapatite/B-Tricalcium Phosphate Mixture versus Hydroxyapatite/Demineralized Bone Matrix Mixture. Asian Spine J. 2015;9(1):30-38. [70] SASSARD WR, EIDMAN DK, GRAY PM, et al. Augmenting local bone with Grafton demineralized bone matrix for posterolateral lumbar spine fusion: avoiding second site autologous bone harvest. Orthopedics. 2000; 23(10):1059-1064; discussion 64-5. [71] RICART PH, GANDHI SD, GEISINGER J, et al. Clinical and CT Analysis of Lumbar Spine Arthrodesis: β-Tricalcium Phosphate Versus Demineralized Bone Matrix. J Am Acad Orthop Surg Glob Res Rev. 2018;2(9):e024. [72] CHANG DG, PARK JB, HAN Y. Surgical outcomes of two kinds of demineralized bone matrix putties/local autograft composites in instrumented posterolateral lumbar fusion. BMC Musculoskelet Disord. 2021;22(1):200. [73] KANG J, AN H, HILIBRAND A, et al. Grafton and local bone have comparable outcomes to iliac crest bone in instrumented single-level lumbar fusions. Spine (Phila Pa 1976). 2012;37(12):1083-1091. [74] FU TS, WANG IC, LU ML, et al. The fusion rate of demineralized bone matrix compared with autogenous iliac bone graft for long multi-segment posterolateral spinal fusion. BMC Musculoskelet Disord. 2016;17:3. [75] ELESWARAPU A, ROWAN FA, LE H, et al. Efficacy, Cost, and Complications of Demineralized Bone Matrix in Instrumented Lumbar Fusion: Comparison With rhBMP-2. Global Spine J. 2021;11(8):1223-1229. [76] 谢幼专,张蒲,赵杰.应用硫酸钙/脱钙骨基质颗粒促进腰椎后外侧融合的临床研究[J].生物骨科材料与临床研究,2010,7(1):20-23. [77] GATAM AR, GATAM L, LUMBAN TOBING SD. Comparison of Clinical and Radiological Outcomes of Lumbar Interbody Fusion Using a Combination of Hydroxyapatite and Demineralized Bone Matrix and Autografts for Lumbar Degenerative Spondylolisthesis. Asian Spine J. 2017;11(5):706-714. [78] PRICE CT, CONNOLLY JF, CARANTZAS AC, et al. Comparison of bone grafts for posterior spinal fusion in adolescent idiopathic scoliosis. Spine (Phila Pa 1976). 2003;28(8):793-798. [79] WEINZAPFEL B, SON-HING JP, ARMSTRONG DG, et al. Fusion rates after thoracoscopic release and bone graft substitutes in idiopathic scoliosis. Spine (Phila Pa 1976). 2008;33(10):1079-1083. [80] KIM BJ, KIM SH, LEE H, et al. Demineralized Bone Matrix (DBM) as a Bone Void Filler in Lumbar Interbody Fusion: A Prospective Pilot Study of Simultaneous DBM and Autologous Bone Grafts. J Korean Neurosurg Soc. 2017;60(2):225-231. [81] AJIBOYE RM, ECKARDT MA, HAMAMOTO JT, et al. Does Age Influence the Efficacy of Demineralized Bone Matrix Enriched with Concentrated Bone Marrow Aspirate in Lumbar Fusions? Clin Spine Surg. 2018;31(1):E30-e35. [82] SHEPARD NA, RUSH AJ 3rd, SCARBOROUGH NL, et al. Demineralized Bone Matrix in Spine Surgery: A Review of Current Applications and Future Trends. Int J Spine Surg. 2021;15(s1):113-119. [83] JANSSEN ME, LAM C, BECKHAM R. Outcomes of allogenic cages in anterior and posterior lumbar interbody fusion. Eur Spine J. 2001;10 Suppl 2(Suppl 2): S158-168. [84] YIN X, LIU P, LIU YY, et al. Utilization of ring-shaped bone allograft for surgical treatment of adolescent post-tubercular kyphosis: A retrospective study. Medicine (Baltimore). 2017;96(24):e7132. [85] CUTLER AR, SIDDIQUI S, MOHAN AL, et al. Comparison of polyetheretherketone cages with femoral cortical bone allograft as a single-piece interbody spacer in transforaminal lumbar interbody fusion. J Neurosurg Spine. 2006;5(6):534-539. [86] PHILIPP T, RADOSLOVICH SS, YOO JU. Risk Factors Associated With Femoral Ring Allograft Breakage in ALIF. Global Spine J. 2021;11(1):57-62. [87] 郜德龙,方忠,徐勇.同种异体骨椎间融合器在颈椎前路手术中应用的临床效果[J].中国修复重建外科杂志,2019,10(3):231-233 [88] BUYUK AF, ONYEKWELU I, GAFFNEY CJ, et al. Symptomatic pseudarthrosis requiring revision surgery after 1- or 2-level ACDF with plating: peek versus allograft. J Spine Surg. 2020;6(4):670-680. [89] RASOULI J, FIANI B, BELDING J, et al. Anterior Cervical Discectomy and Fusion With “Kissing” Allograft Interbodies. Cureus. 2021;13(11):e19499. [90] ARNOLD PM, ROBBINSS, PAULLUS W, et al. Clinical outcomes of lumbar degenerative disc disease treated with posterior lumbar interbody fusion allograft spacer: a prospective, multicenter trial with 2-year follow-up. Am J Orthop (Belle Mead NJ). 2009;38(7):E115-122. [91] 郜德龙,方忠,孙允龙.同种异体骨Cage在经椎间孔腰椎椎间融合手术中的应用[J].中国修复重建外科杂志,2018,32(7):927-932 [92] WU WJ, LI Y, HOU TY, et al. Application of New Allogeneic Lumbar Fusion Cage (Biocage) in Single-Segment Lumbar Degenerative Disease: A Prospective Controlled Study with Follow-Up for ≥2 Years. World Neurosurg. 2019;126:e1309-e1314. [93] VILLAVICENCIO AT, NELSON EL, RAJPAL S, et al. Prospective, randomized, double-blinded clinical trial comparing PEEK and allograft spacers in patients undergoing transforaminal lumbar interbody fusion surgeries. Spine J. 2022;22(1):84-94. [94] HACKER RJ, CAUTHEN JC, GILBERT TJ, et al. A prospective randomized multicenter clinical evaluation of an anterior cervical fusion cage. Spine (Phila Pa 1976). 2000;25(20):2646-2654; discussion 55. [95] KAYANJA M, ORR RD. Incidence and outcome of graft resorption in anterior lumbar interbody fusion: using femoral ring allografts and recombinant human bone morphogenetic protein-2. Spine (Phila Pa 1976). 2014;39(5):374-380. |
| [1] | Ma Maoxiao, Cui Guofeng, Zhang Xue, Zhang Hong, Liu Youwen, Yue Chen. Complications after spinal fusion in patients with metabolic syndrome: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3602-3608. |
| [2] | Song Kunxiu, Wang Limin, Ma Rongxing, Hu Yongcheng. Application of patent literature in the study of allogenic demineralized bone matrix [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(14): 2229-2233. |
| [3] | Xiang Xinjian, Liu Fang, Wu Liangliang, Jia Daping, Tao Yue, Zhao Zhengnan, Zhao Yu. High-dose vitamin C promotes the survival of autologous fat transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1242-1246. |
| [4] | Li Jian, Bao Zhengqi, Zhou Pinghui, Zhu Ruizhi, Li Zhixiang, Wang Jinzi. Effects of posterior single open-door laminoplasty and anterior cervical corpectomy fusion on cervical sagittal balance parameters in the treatment of multilevel cervical spondylotic myelopathy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 949-953. |
| [5] | Yang Jun, Yang Qun, Zhang Rui, Jiang Chang. A novel slidable pedicle screw-rod system for lumbar tuberculosis: promoting bone graft fusion by producing stress stimulation to fused segment [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 914-918. |
| [6] | Li Ting, Liu Xilin, Wang Fei, Hu Jiang. Robot-assisted minimally invasive transforaminal lumbar interbody fusion in the treatment of lumbar degenerative diseases: accuracy and safety of screw placement [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5812-5818. |
| [7] | Jin Herong, Cui Jingbin, Shao Cang. Materials of interbody fusion cage: advantages and focus of clinical application [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3592-3597. |
| [8] | Zhao Zhenglü, Li Yanlin, Wang Xu, Liu Dejian, Zhang Yan. Characteristics and selection of grafts for posterior cruciate ligament reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1615-1619. |
| [9] | Wang Shuguang, Cai Tongchuan, Feng Xinmin, Nan Liping, Wang Feng, Zhu Lei, Chen Dong, Zhang Liang. Meta-analysis of adverse events between anterior and posterior fusion surgery for multiple-level cervical spondylosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(30): 4907-4914. |
| [10] | Tang Xiaokai, Li Weiming. Role and mechanism of Nel-like molecule-1 in promoting bone fusion after spinal fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3914-3920. |
| [11] | Chen Jiang, Li Jinyu, Zheng Chenying, Bai Chunxiao, Zhang Fan, Liu Chuyin, Zhao Xueqian, Yuan Qiaomei, Di Xueshi, Kang Shengqian, Jia Yusong . Changes in sagittal parameters of cervical spine after double-segment artificial cervical disc replacement and anterior cervical discectomy and fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2341-2346. |
| [12] | Chen Junyi, Wang Ning, Chen Ximiao, Zhu Lunjing, Duan Jiangtao, Zhang Xianping, Bei Chaoyong. Osteogenic differentiation of bone marrow mesenchymal stem cells induced by demineralized bone matrix in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 26-31. |
| [13] | Wu Zhifeng, Luo Min. Biomechanical analysis of chemical acellular nerve allograft combined with bone marrow mesenchymal stem cell transplantation for repairing sciatic nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 991-995. |
| [14] | Xiang Feifan, Tan Xiaoqi, Xiang Yong, Liang Jie, Zhou Wei, Luo Liang, Gu Hao, Yang Yunkang. Effect of anatomic locking plate combined with allograft fibula on proximal humerus fracture: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(36): 5897-5904. |
| [15] | Li Yongtao, Song Wenhui, Liu Changwen, Liang Kaiheng, Zhang Kaixi, Liu Changfeng. Clinical efficacy of calcium sulphate/demineralized bone matrix versus bone allograft in anterior cervical discectomy and fusion [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5478-5485. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||