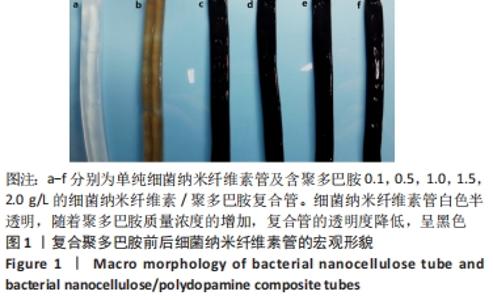
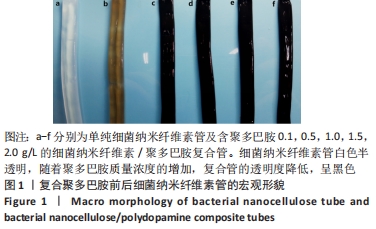
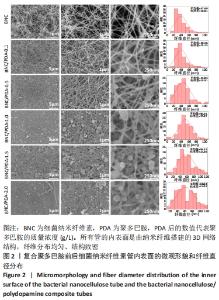
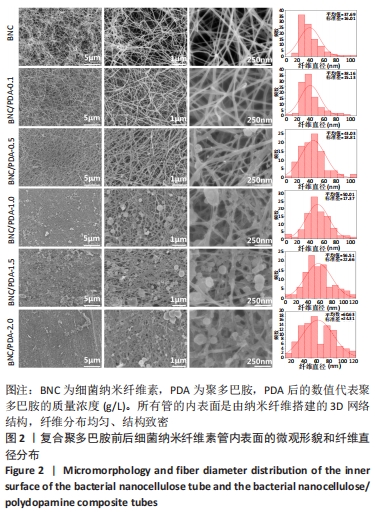
Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (22): 3535-3542.doi: 10.12307/2022.282
Previous Articles Next Articles
Potential of bacterial nanocellulose/polydopamine composite tubes as small-diameter artificial blood vessel
Liu Liang1, 2, 3, Hu Gaoquan1, 2, 3, Wei Zhao1, 2, 3, Chen Lin1, 2, 3, Hong Feng1, 2, 3
- 1College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China; 2Shanghai Engineering Research Center of Nano-Biomaterials and Regenerative Medicine, Donghua University, Shanghai 201620, China; 3Scientific Research Base of Bacterial Nanofiber Manufacturing and Composite Technology, China Textile Engineering Society, Shanghai 201620, China
-
Received:2020-11-20Revised:2021-01-16Accepted:2021-05-23Online:2022-08-08Published:2022-01-12 -
Contact:Hong Feng, Professor, College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China; Shanghai Engineering Research Center of Nano-Biomaterials and Regenerative Medicine, Donghua University, Shanghai 201620, China; Scientific Research Base of Bacterial Nanofiber Manufacturing and Composite Technology, China Textile Engineering Society, Shanghai 201620, China -
About author:Liu Liang, Master, College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China; Shanghai Engineering Research Center of Nano-Biomaterials and Regenerative Medicine, Donghua University, Shanghai 201620, China; Scientific Research Base of Bacterial Nanofiber Manufacturing and Composite Technology, China Textile Engineering Society, Shanghai 201620, China -
Supported by:National Key Research and Development Program of China, No. 2018YFC1105501 (to HF)
CLC Number:
Cite this article
Liu Liang, Hu Gaoquan, Wei Zhao, Chen Lin, Hong Feng. Potential of bacterial nanocellulose/polydopamine composite tubes as small-diameter artificial blood vessel[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3535-3542.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
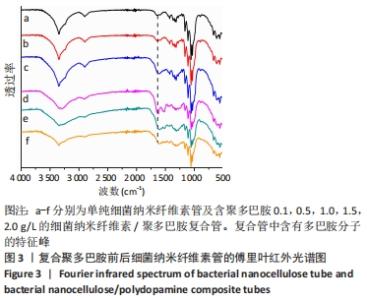
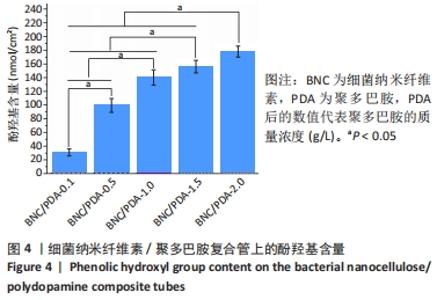
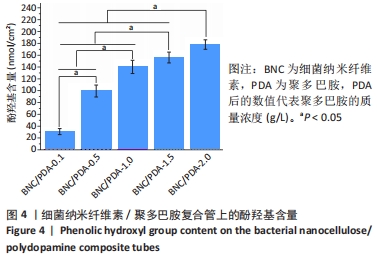
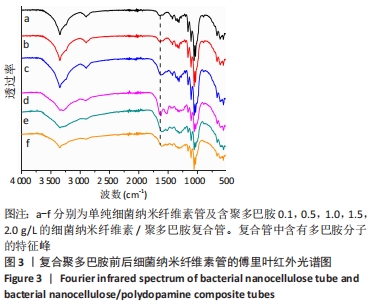
根据Image J软件统计纤维直径的数据可知,与细菌纳米纤维素管相比,BNC/PDA复合管的纤维直径增大,且随多巴胺溶液质量浓度的增加纤维直径增大。其中细菌纳米纤维素管的直径为37.7 nm,而BNC/PDA-2.0复合管的直径增大至60.6 nm。 2.3 细菌纳米纤维素管及BNC/PDA复合管的傅里叶红外光谱图分析 由图3a可以看出细菌纳米纤维素管的特征吸收峰,红外谱图中 3 600-3 100,1 330,1 250-850 cm-1处的吸收峰分别对应细菌纤维素上O-H、C-H和C-O 的伸缩振动。在BNC/PDA复合管上仍有细菌纳米纤维素管的特征吸收峰,但是在3 000-3 500 cm-1出现了宽带,见图3b-f,那是由多巴胺分子中存在分子间氢键O-H伸缩振动或是由芳香族仲胺 N-H 伸缩振动引起的[27]。而在1 640 cm-1处的吸收峰对应的是酰胺Ⅰ键C=O的伸缩振动[10]。这些结果从官能团角度证明了聚多巴胺的成功涂覆。"
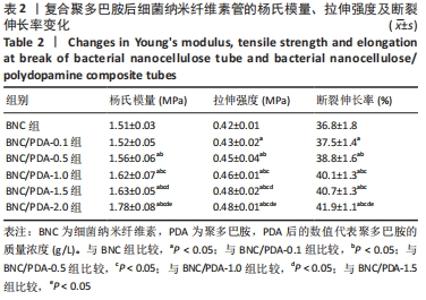
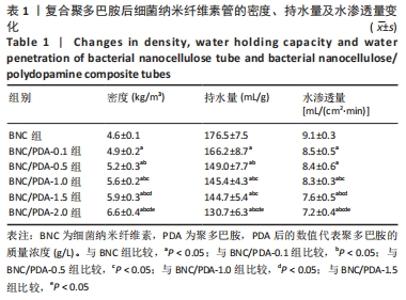
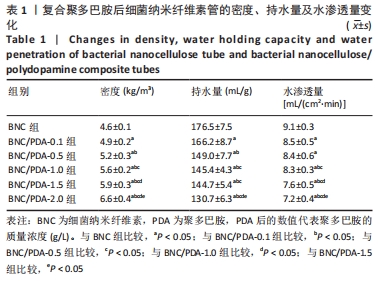
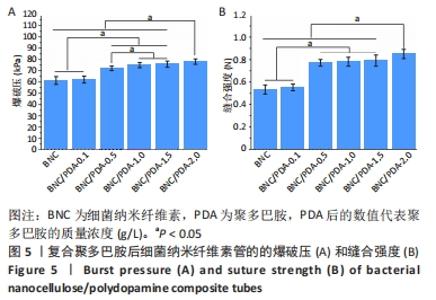
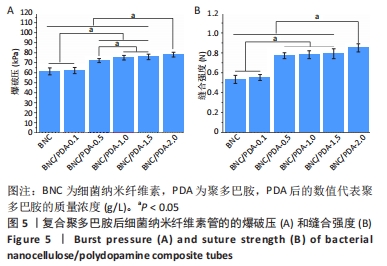
细菌纳米纤维素管、BNC/PDA-0.1、BNC/PDA-0.5、BNC/PDA-1.0、BNC/PDA-1.5和BNC/PDA-2.0复合管的爆破压分别为(61.2±3.5),(62.0±3.1),(72.0±2.0),(74.8±2.3),(75.7±2.7)和(78.0±2.4 ) kPa,缝合强度分别为(0.53±0.04),(0.55±0.03),(0.77±0.03),(0.78±0.04),(0.79±0.05)和(0.85±0.04) N。可以看出,与细菌纳米纤维素管相比,BNC/PDA复合管的爆破压增大,但是细菌纳米纤维素管和BNC/PDA-0.1复合管间的爆破压比较差异无显著性意义(P > 0.05);聚多巴胺复合量越大管的爆破压也越大,但是BNC/PDA-1.0和BNC/PDA-1.5复合管间的爆破压比较差异无显著性意义(P > 0.05)。细菌纳米纤维素管与BNC/PDA-0.1复合管的缝合强度比较差异无显著性意义(P > 0.05);BNC/PDA-0.5、BNC/PDA-1.0、BNC/PDA-1.5和BNC/PDA-2.0复合管的缝合强度均明显增加,但是BNC/PDA-0.5、BNC/PDA-1.0、BNC/PDA-1.5复合管之间比较差异无显著性意义(P > 0.05)。 2.7 细菌纳米纤维素管及BNC/PDA复合管的轴向拉伸力学测试结果 表2列出了细菌纳米纤维素管和BNC/PDA复合管的杨氏模量、拉伸强度及断裂伸长率,可以看出随着聚多巴胺复合量的增加,管的杨氏模量、拉伸强度和断裂伸长率均增加,这表明聚多巴胺的复合可以提升细菌纳米纤维素管的轴向力学性质。但是细菌纳米纤维素管与BNC/PDA-0.1复合管之间的杨氏模量比较差异无显著性意义(P > 0.05),BNC/PDA-1.0与BNC/PDA-1.5复合管之间的断裂伸长率比较差异无显著性意义(P > 0.05)。"
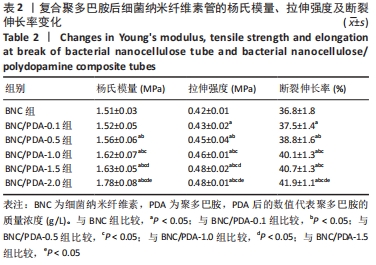
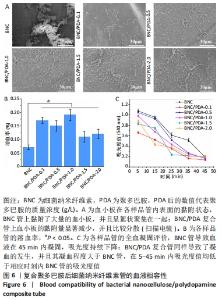
2.8 细菌纳米纤维素管及BNC/PDA复合管的血液相容性 图6A显示了细菌纳米纤维素管和BNC/PDA复合管的血小板黏附状况,可以看出纯细菌纳米纤维素管上黏附了大量的血小板,并且呈絮状聚集在一起,唐敬玉等[28]的研究也表明,血小板容易黏附在细菌纳米纤维素上且相互聚集形成血小板栓子。然而BNC/PDA复合管上血小板的黏附量显著减少,并且比较分散,激活程度较小。 图6B显示了细菌纳米纤维素管和BNC/PDA复合管的溶血率,结果显示细菌纳米纤维素管的溶血率为0.07%,复合聚多巴胺后的溶血率略微提高,BNC/PDA-0.1、BNC/PDA-0.5、BNC/PDA-1.0、BNC/PDA-1.5和BNC/PDA-2.0复合管的溶血率分别为(0.17±0.01)%,(0.15±0.01)%,(0.19±0.02)%,(0.11±0.02)%和(0.12±0.02)%。 全血凝固实验可以直观地评价材料的致凝血性能。图6C展示了兔全血与细菌纳米纤维素管和BNC/PDA复合管接触后的凝血过程,细菌纳米纤维素管导致血液在45 min内凝固,吸光度持续下降;BNC/PDA复合管同样导致了凝血的发生,并且其凝血程度大于纯细菌纳米纤维素管,在5-45 min内吸光度值均低于相应时刻内细菌纳米纤维素管的吸光度值,但是不同聚多巴胺复合量的BNC/PDA复合管之间的全血凝固结果无明显差异。"
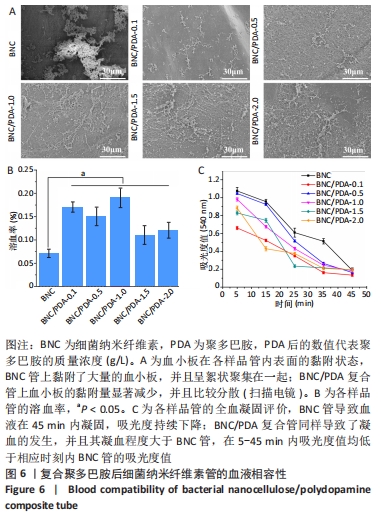
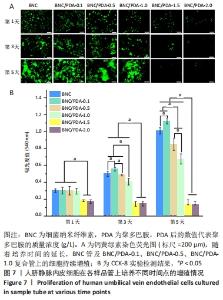
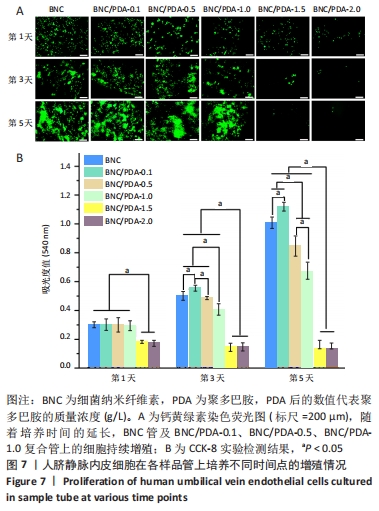
2.9 细菌纳米纤维素管及BNC/PDA复合管的细胞相容性 图7A是人脐静脉内皮细胞在各样品管上培养1,3,5 d后的钙黄绿素荧光染色结果,可以看出基本趋势与CCK-8实验结果一致。在整个培养周期内,细菌纳米纤维素管、BNC/PDA-0.1、BNC/PDA-0.5以及BNC/PDA-1.0复合管上的细胞持续增殖,到第5天时细胞在这些材料上已经成簇铺展开,与细菌纳米纤维素管上的细胞相比,BNC/PDA-0.1复合管上细胞更多,而BNC/PDA-0.5及BNC/PDA-1.0复合管上的细胞较少。在整个培养周期,BNC/PDA-1.5和BNC/PDA-2.0复合管上的细胞没有增殖,荧光最弱,尤其到第3天和第5天基本看不到荧光。 图7B显示人脐静脉内皮细胞在样品管上培养1,3,5 d后的CCK-8实验结果,可以看出在第1天时,细菌纳米纤维素管与BNC/PDA-0.1、BNC/PDA-0.5及BNC/PDA-1.0复合管之间细胞数量无明显差异,但是BNC/PDA-1.5和BNC/PDA-2.0复合管上的细胞数量较少;随着培养时间的延长,细菌纳米纤维素管、BNC/PDA-0.1、BNC/PDA-0.5和BNC/PDA-1.0复合管上的细胞持续增殖,但增殖趋势不同,与细菌纳米纤维素管相比,BNC/PDA-0.1复合管上第3,5天的细胞数量较多,而BNC/PDA-0.5和BNC/PDA-1.0复合管上的细胞数量较少,第3天时细菌纳米纤维素管与BNC/PDA-0.5复合管上的细胞数量无明显差异。在整个5 d的培养过程中,BNC/PDA-1.5和BNC/PDA-2.0复合管上的细胞没有表现出增殖结果。"
| [1] VIRANI SS, ALONSO A, BENJAMIN EJ, et al. Heart disease and stroke statistics-2020 update: a report from the American Heart Association. Circulation. 2020;141(9): E139-E596. [2] JEREMIAS A, KAUL S, ROSENGART TK, et al. The impact of revascularization on mortality in patients with nonacute coronary artery disease. Am J Med. 2009; 122(2):152-161. [3] KIESER TM, LEWIN AM, GRAHAM MM, et al. Outcomes associated with bilateral internal thoracic artery grafting: the importance of age. Ann Thorac Sugr. 2011; 92(4):1269-1275. [4] ZHU AP, MING Z, JIAN S. Blood compatibility of chitosan/heparin complex surface modified ePTFE vascular graft. Appl Surf Sci. 2005;241(3-4):485-492. [5] LI J, LIN F, LI LD, et al. Surface engineering of poly(ethylene terephthalate) for durable hemocompatibility via a surface interpenetrating network technique. Macromol Chem Phys. 2012; 213(20):2120-2129. [6] SHIN YM, LEE YB, KIM SJ, et al. Mussel-inspired immobilization of vascular endothelial growth factor (VEGF) for enhanced endothelialization of vascular grafts. Biomacromolecules. 2012;13(7):2020-2028. [7] FINK H, HONG J, DROTZ K, et al. An in vitro study of blood compatibility of vascular grafts made of bacterial cellulose in comparison with conventionally-used graft materials. J Biomed Mater Res Part A. 2011;97A(1):52-58. [8] LI X, TANG JY, BAO LH, et al. Performance improvements of the BNC tubes from unique double-silicone-tube bioreactors by introducing chitosan and heparin for application as small-diameter artificial blood vessels. Carbohydr Polym. 2017; 178:394-405. [9] FINK H, FAXALV L, MOLNAR GF, et al. Real-time measurements of coagulation on bacterial cellulose and conventional vascular graft materials. Acta Biomater. 2010;6(3):1125-1130. [10] LAI C, ZHANG SJ, SHENG LY, et al. Comparative evaluation of the biocompatible and physical-chemical properties of poly(lactide-co-glycolide) and polydopamine as coating materials for bacterial cellulose. J Mat Chem B. 2019;7(4):630-639. [11] PERTILE R, MOREIRA S, ANDRADE F, et al. Bacterial cellulose modified using recombinant proteins to improve neuronal and mesenchymal cell adhesion. Biotechnol Progr. 2012;28(2):526-532. [12] FINK H, AHRENSTEDT L, BODIN A, et al. Bacterial cellulose modified with xyloglucan bearing the adhesion peptide RGD promotes endothelial cell adhesion and metabolism - a promising modification for vascular grafts. J Tissue Eng Regen Med. 2011;5(6):454-463. [13] LEE H, DELLATORE SM, MILLER WM, et al. Mussel-inspired surface chemistry for multifunctional coatings. Science (New York, NY). 2007;318(5849):426-430. [14] LUO RF, TANG LL, WANG J, et al. Improved immobilization of biomolecules to quinone-rich polydopamine for efficient surface functionalization. Colloid Suraface B. 2013;106:66-73. [15] KU SH, PARK CB. Human endothelial cell growth on mussel-inspired nanofiber scaffold for vascular tissue engineering. Biomaterials. 2010;31(36):9431-9437. [16] DAVOUDI P, ASSADPOUR S, DERAKHSHAN MA, et al. Biomimetic modification of polyurethane-based nanofibrous vascular grafts: A promising approach towards stable endothelial lining. Mat Sci Eng C-Mater. 2017;80:213-221. [17] YANG ZL, TU QF, ZHU Y, et al. Mussel-inspired coating of polydopamine directs endothelial and smooth muscle cell fate for re-endothelialization of vascular devices. Adv Healthc Mater. 2012;1(5):548-559. [18] OBIWELUOZOR FO, TIWARI AP, LEE JH, et al. Thromboresistant semi-IPN hydrogel coating: towards improvement of the hemocompatibility/biocompatibility of metallic stent implants. Mater Sci Eng C-Mater Biol Appl. 2019;99:1274-1288. [19] HUANG Y, ZHAO X, ZHANG Z, et al. Degradable gelatin-based IPN cryogel hemostat for rapidly stopping deep noncompressible hemorrhage and simultaneously improving wound healing. Chem Mater. 2020;32(15):6595-6610. [20] 唐文婷.基于聚多巴胺纳米材料的制备及在肿瘤微波热疗中的应用[D].北京:北京交通大学,2016. [21] JIANG QS, DERAMI HG, GHIM D, et al. Polydopamine-filled bacterial nanocellulose as a biodegradable interfacial photothermal evaporator for highly efficient solar steam generation. J Mater Chem A. 2017;5(35):18397-18402. [22] YANG LY, CHEN CT, HU Y, et al. Three-dimensional bacterial cellulose/polydopamine/TiO2 nanocomposite membrane with enhanced adsorption and photocatalytic degradation for dyes under ultraviolet-visible irradiation. J Colloid Interface Sci. 2020;562:21-28. [23] XIE YJ, YUE LN, ZHENG YD, et al. The antibacterial stability of poly(dopamine) in-situ reduction and chelation nano-Ag based on bacterial cellulose network template . Appl Surf Sci. 2019;491;383-394. [24] BAO LH, TANG JY, HONG FF, et al. Physicochemical properties and in vitro biocompatibility of three bacterial nanocellulose conduits for blood vessel applications. Carbohydr Polym. 2020;239:13. [25] 申芳瑜.用于血管介入器械表面改性的铜介导原位催化产生一氧化氮涂层的构建[D].成都:西南交通大学,2016. [26] 段传双,包露涵,陈琳,等.细菌纳米纤维素/聚乙烯醇小径人工血管的制备及其表征[J].纤维素科学与技术,2020,28(2):1-10. [27] 姚艳.应用于组织工程血管支架的纯镁的表面改性及性能研究[D].重庆:重庆大学,2018. [28] 唐敬玉,包露涵,李雪,等.细菌纳米纤维素与聚乙烯醇复合水凝胶管的生物相容性表征[J].中国组织工程研究,2017,21(34): 5474-5480. [29] 曹海建,钱坤.人造血管的现状及其研究进展[J].四川纺织科技,2004(2):31-33. [30] KONIG G, MCALLISTER TN, DUSSERRE N, et al. Mechanical properties of completely autologous human tissue engineered blood vessels compared to human saphenous vein and mammary artery. Biomaterials. 2009;30(8):1542-1550. [31] DOBROVOLSKAIA MA, CLOGSTON JD, NEUN BW, et al. Method for analysis of nanoparticle hemolytic properties in vitro. Nano Lett. 2011;8(8):2180-2187. [32] YANG XL, LIU X, WU QQ, et al. Dopamine-assisted immobilization of peptide arginine-glycine-aspartic acid to enhance the cellular performances of MC3T3-E1 cells of carbon-carbon composites. J Biomater Appl. 2019;34(2):284-296. [33] WEI Q, LI BJ, YI N, et al. Improving the blood compatibility of material surfaces via biomolecule-immobilized mussel-inspired coatings. J Biomed Mater Res Part A. 2011;96A(1):38-45. [34] LEITAO AF, GUPTA S, SILVA JP, et al. Hemocompatibility study of a bacterial cellulose/polyvinyl alcohol nanocomposite. Colloids Surf B. 2013;111:493-502. [35] YANG Y, QI PK, DING YH, et al. A biocompatible and functional adhesive amine-rich coating based on dopamine polymerization. J Mat Chem B. 2015;3(1): 72-81. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | Yang Feng, Zhao Qian, Zhang Shixuan, Zhao Tienan, Feng Bo. Effectiveness and safety of rapamycin combined with CD133 antibody stent in preventing vascular restenosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 579-584. |
| [5] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [6] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [7] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [8] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [9] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [10] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [11] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [12] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [13] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [14] | Liu Junwei, Yang Sixing, Xiong Yunhe, Song Chao, Liao Wenbiao, Meng Lingchao, Li De, Song Qianlin, He Ziqi, Li Bin. Construction of tissue-engineered urine outflow tract using adipose stem cells and expanded polytetrafluoroethylene for canine urinary diversion [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3487-3491. |
| [15] | Guo Yangyan, Yu Zhengwen, Zhang Jian. Research hotspots of magnesium alloy biomaterials in an in vivo animal [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3556-3565. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||