Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (9): 1445-1451.doi: 10.12307/2022.444
Previous Articles Next Articles
Role and therapeutic implications of pyroptosis in intervertebral disc degeneration
Zhang Haobo1, 2, Zhao Yunan1, 2, Yang Xuejun2
- 1Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China; 2Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China
-
Received:2021-06-28Revised:2021-06-30Accepted:2021-07-08Online:2022-03-28Published:2021-12-10 -
Contact:Yang Xuejun, Professor, Chief physician, Master’s supervisor, Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China -
About author:Zhang Haobo, Master candidate, Physician, Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China; Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China
CLC Number:
Cite this article
Zhang Haobo, Zhao Yunan, Yang Xuejun. Role and therapeutic implications of pyroptosis in intervertebral disc degeneration[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1445-1451.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 椎间盘退变的概述 2.1.1 椎间盘的结构 椎间盘是一种无血管组织,主要由中央的髓核、周围的纤维环和软骨终板组成。髓核组织由少量软骨样细胞和大量细胞外基质构成,细胞外基质是由蛋白聚糖和Ⅱ型胶原纤维组成的[16],带负电荷侧链的蛋白聚糖使髓核具有高渗透压,使椎间盘能够抵抗压缩负荷和可逆变形。纤维环是由Ⅰ型和Ⅱ型胶原纤维以及弹性蛋白纤维组成的同心排列的薄片,内环由Ⅰ、Ⅱ型胶原纤维构成组成,而外环主要以Ⅰ型胶原纤维为主,且越靠近外环,Ⅰ型胶原纤维所占比例越高,这使椎间盘能够抵抗拉伸负荷[4]。软骨终板是覆盖在椎体上下缘的透明软骨组织,在椎间盘内外营养和代谢物质的运输中起着重要作用[17],这些结构使椎间盘具有较高的抗压强度和抗拉强度,保持脊柱的灵活性和机械稳定性[18]。椎间盘退变的发展以椎间盘微环境的细胞和生化改变为特征,导致进行性的功能和结构损害,包括蛋白聚糖含量降低、Ⅱ型胶原向Ⅰ型胶原的转变以及髓核细胞数量减少,最终导致椎间盘承受压缩负荷的能力降低并导致椎间盘结构的破坏[19-20]。 2.1.2 椎间盘退变的病理机制 椎间盘退变的病因十分复杂,包括年龄因素、遗传因素(如编码蛋白聚糖和胶原蛋白Ⅰ/Ⅸ/Ⅺ的基因多态性)和生活方式因素(如职业、吸烟、饮酒、缺乏体育活动和夜班工作)等[4,21]。这些因素导致髓核细胞数量和转化减少,造成它们合成细胞外基质的能力下降,同时促使细胞分泌更多促进基质分解的酶(如基质金属蛋白酶和ADAMTS金属蛋白酶),使髓核的水合能力下降,最终导致椎间盘结构遭到破坏。除了细胞外基质分解和细胞丢失以外,炎症也在椎间盘退变中起着重要作用,并且是区分无症状性椎间盘退变和有症状性椎间盘退变的一个重要因素。众所周知,随着椎间盘退变程度的加深和疼痛的进展,髓核细胞不断释放大量促炎细胞因子,其中肿瘤坏死因子α及白细胞介素1β最为突出。这些细胞因子可以破坏细胞外基质分解代谢与合成代谢之间的平衡,并促进细胞死亡。细胞外基质分解、髓核细胞丢失和炎症3者之间相互关联。促炎细胞因子通过上调基质降解酶的表达,导致基质代谢紊乱。基质降解的产物在细胞外积累,进而又刺激髓核细胞的炎症反应。此外,髓核细胞的凋亡和焦亡又与基质的合成和炎症反应有关。 2.2 细胞焦亡概述 "

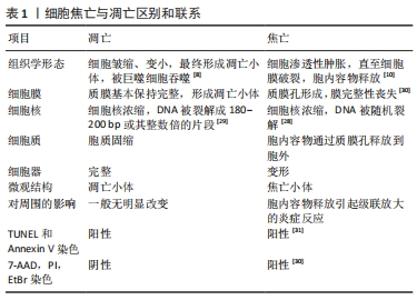
2.2.1 细胞焦亡的概念及形态学特征 近几十年来,对焦亡的定义进行了数次修订。焦亡现象首次发现于1992年,ZYCHLINSKY等[22]在感染了福氏志贺菌的巨噬细胞中发现一种裂解性死亡,其形态学特征有别于凋亡。2001年,COOKSON等[23]提出“焦亡”一词来描述促炎性细胞的程序性坏死。2015年,邵峰院士团队发现,caspase-1和caspase-4/5/11是通过切割一个叫做gasdermin D(GSDMD)的蛋白而诱发焦亡的,GSDMD在被caspase-1和caspase-4/5/11切割后,释放出其N端结构域,该结构域具有成孔活性,这样就导致细胞渗透压发生变化而发生膨胀直至细胞膜破裂[24-25]。随后的研究证实,gasdermin家族的其他蛋白如gasdermin E (GSDME)也被caspase-3激活,从而导致焦亡[26]。细胞焦亡(pyroptosis)这一单词来源于古希腊语,“Pyro”指的是火或发热,表明炎症的性质,而“ptosis”指的是下降,表明其程序性死亡的本质。 2.2.2 细胞凋亡与细胞焦亡的区别 焦亡和凋亡之间有一些相似之处,如DNA损伤、染色质凝结和caspase依赖性,但它与细胞凋亡的区别在于其特殊的形态学特征。首先,在细胞焦亡早期存在一种非常特殊的DNA损伤形式,dUTP末端标记染色阳性,但强度要低于凋亡[27]。其次,焦亡细胞的DNA损伤程度较轻,细胞焦亡的DNA片段是随机的,细胞核保持完整,而细胞凋亡的DNA片段是有序的,细胞核是破碎成碎片的[28-29],两者之间最大的区别是焦亡期间形成质膜孔。因此,焦亡细胞可以通透7-氨基放射线霉素(7-AAD)、碘化丙啶(PI)和溴化乙锭(EtBr),因为这些染料的分子质量 低;相反,凋亡细胞保持细胞膜的完整性,因此这些染料不能对其染色[29-30]。Annexin V也能染色焦亡细胞,并与磷脂酰丝氨酸(PS)结合[31]。细胞焦亡与凋亡的区别和联系,见表1。"

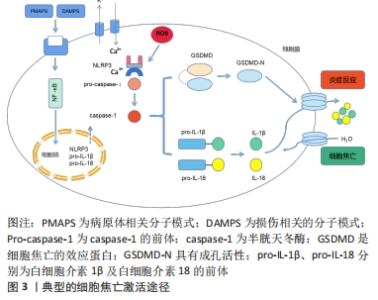
2.2.3 细胞焦亡的分子特征 Caspase家族:最初,细胞焦亡被定义为依赖caspase-1/4/5/11介导的一种程序性细胞死亡。caspase家族是一类促炎性半胱天冬酶,在先天性免疫和肿瘤的发生中发挥重要作用。最近的研究表明,包括caspase-3/4/5/6/8/9/11在内的其他caspase也可引起其他不同类型细胞的细胞焦亡。之前一直认为caspase-3是细胞凋亡的代表,但现在发现caspase-3可以通过切割GSDME诱导细胞焦亡[26]。此外,caspase-9的激活也通过激活caspase-3参与了焦亡[32],而caspase-6介导了切割GSDMC[33]。尽管目前的研究发现caspase-1和caspase-4,5,11只与焦亡有关,而caspase-2,7,10只与凋亡有关,随着研究的深入,凋亡、焦亡与caspases的关系可能会陆续报道。 Gasdermin家族:直到2015年,细胞死亡命名委员会定义焦亡为一种gasdermin介导的程序性死亡。Gasdermins主要在巨噬细胞、树突状细胞、皮肤和黏膜膜上皮中表达,是焦亡的最终效应蛋白。人类表达gasdermin A/B/C/D (GSDMA/B/C/D)、gasdermin E (GSDME)和DFNB59 (Pejvakin,PJVK),小鼠表达Gsdma1-3、Gsdmc1-4、Gsdmd、DFNA5和DFNB59[30]。其中,GSDMD和GSDME在焦亡中研究最为深入。除Pejvakin外,所有gasdermin家族成员都包含2个保守结构域,即N端孔化结构域和C端阻遏结构域[34-36]。通常情况下,Gasdermins的N端和C端相互作用保持寡聚,而当机体受到各种外源或内源性因素刺激后,Gasdermins被炎性半胱氨酸蛋白酶或颗粒酶切割,N端与C端分离,转移到细胞膜上寡聚并形成质膜孔,释放出细胞内容物和炎症因子[37]。虽然已报道许多Gasdermin家族蛋白与人类疾病有关,但其具体机制和功能仍有待研究。 炎症小体:焦亡是由病原体相关分子模式、损伤相关分子模式或稳态改变分子过程刺激触发的,包括细胞内脂多糖、细胞外三磷酸腺苷、细胞质双链DNA、细菌鞭毛和颗粒物质[38]。识别这些触发因子需要炎症小体的参与。炎症小体是炎症反应的重要组成部分,也是慢性低度炎症起始的第一步。炎症小体是细胞内的蛋白寡聚体,是感知病原体、组织损伤和体内平衡改变的信号传感器,它们由传感器蛋白、适配器蛋白和效应蛋白组成,激活后在一个大的复合物中齐聚[11]。炎症小体传感器蛋白通常是一种模式识别受体。模式识别受体有3个家族,富含亮氨酸重复序列的受体家族(包括NLRP3,NLRP1,NLRC4)、黑色素瘤缺乏因子2或炎性蛋白Pyrin[12]。富含亮氨酸重复序列的受体家族通常由1个富含亮氨酸重复序列、1个核苷酸结合寡聚域和1个caspase招募结构域或pyrin 结构域组成[27]。凋亡相关斑点样蛋白是一种含有caspase招募结构域的衔接蛋白,可以招募并激活效应蛋白caspase-1,切割GSDMD,诱导细胞焦亡。同时,激活的caspase-1将关键的促炎因子白细胞介素1β和白细胞介素18的前体加工成活性形式,因为缺乏分泌的信号肽,这些因子需要通过GSDMD形成的质膜孔来释放。白细胞介素1β激活核转录因子κB信号通路,导致其他炎症因子分泌(如白细胞介素6和肿瘤坏死因子α)和招募更多免疫细胞,从而放大炎症反应[39]。 2.2.4 细胞焦亡的分子机制 典型的细胞焦亡途径:典型的细胞焦亡主是由炎症小体介导的。NLRP3 是目前为止研究最深入的炎症小体,NLRP3炎症小体的激活过程有2个步骤:起始和激活[40]。由于NLRP3的基础表达水平不足以组装典型炎症小体,且NLRP3在正常情况下是泛素化的[41]。因此,起始需要脂多糖、肿瘤坏死因子α和白细胞介素1β等作为启动信号,通过激活核转录因子κB信号通路上调NLRP3、白细胞介素1β前体和白细胞介素18前体的转录水平[42]。再通过Lys63特异性去泛素化酶BRCC3介导NLRP3的去泛素化[41]。NLRP3炎症小体激活的机制有很多,如钾离子外排、钙离子内流、活性氧的生成、线粒体DNA的释放、溶酶体破裂以及质膜孔的形成[43]。 炎症小体被激活后,将caspase-1前体转化为活性形式。激活的caspase-1在GSDMD特定位点上进行切割,释放出N端具有成孔活性的结构域,在质膜上形成一个直径为10-20 nm的中空环状低聚体,通过破坏钠、钾离子的浓度梯度,导致渗透压发生变化。当孔隙的数量超过细胞的代偿能力时,水通过渗透梯度进入细胞,然后细胞膨胀、破裂和死亡。白细胞介素1β和白细胞介素18的前体也被caspase-1激活,并通过GSDMD孔释放,触发炎症反应,进而诱导细胞焦亡[15]。典型的细胞焦亡激活途径,见图3。"
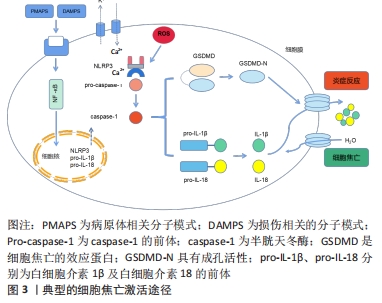

非典型的细胞焦亡途径:此途径是在2011年提出的,由caspase-4/5(人)和caspase-11(小鼠)介导[44]。非典型炎性小体中没有模式识别受体和适配器蛋白,非典型炎性小体的关键成分是caspase-4/5/11,它们通过caspase招募结构域特异性的识别胞质中的脂多糖,并切割下游效应蛋白GSDMD,从而导致细胞焦亡。有趣的是,caspase-4/5/11不能直接切割白细胞介素1β和白细胞介素18的前体,而是通过NLRP3/caspase-1途径介导白细胞介素1β和白细胞介素18的成熟和分泌[13]。有人提出了2种可能的机制:①GSDMD‐N端形成的质膜孔直接诱导K+外排,激活了NLRP3炎症小体,导致炎症因子释放;②激活的caspase-11可特异地切割和修饰Pannexin-1, 引起细胞三磷酸腺苷的释放,进一步激活细胞膜上的P2X7受体并诱导钾离子外排,最终激活NLRP3炎症小体[45]。 特殊的细胞焦亡途径:最近的研究发现了一些其他的焦亡途径。凋亡相关的caspase(如caspase-3,8)也能通过切割gasdermin蛋白(GSDMC,GSDMD和GSDME)诱导细胞焦亡,其触发因子包括化疗药物、肿瘤坏死因子α和细菌等[26,33]。 化疗药物可以诱导caspase-3介导的GSDME高表达并形成GSDME‐N端,引起肿瘤细胞焦亡。在肿瘤坏死因子α的刺激下,caspase-8特异性切割GSDMC产生GSDMC-N端,在细胞膜上形成孔隙,诱导细胞焦亡。在某些细胞中,焦亡甚至可以不依赖caspases。一项研究发现,在衰老的中性粒细胞中,GSDMD被中性粒细胞特异性丝氨酸蛋白酶弹性蛋白酶切割,形成质膜孔,诱发焦亡[46]。Ninjurin-1最初被发现是一种具有炎症和肿瘤抑制作用的黏附分子。最近的研究将其确定为另一种重要的焦亡执行蛋白[47]。 2.3 细胞焦亡与椎间盘退变的关系 "

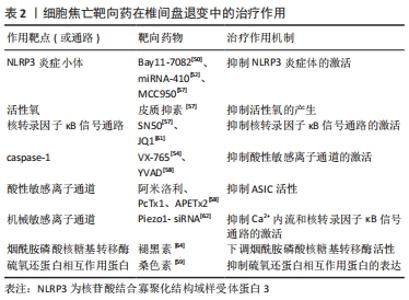
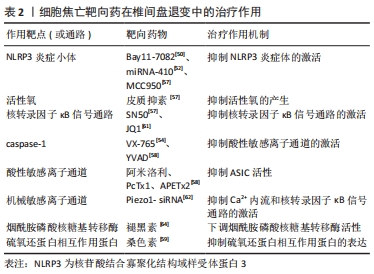
2.3.1 细胞焦亡相关分子在退变椎间盘中表达量升高 CHEN等[48]对45例退变的椎间盘和7例正常的椎间盘样本比较发现,退变组NLRP3炎症小体及其下游靶点caspase-1和白细胞介素1β的表达水平显著上调,NLRP3、caspase-1和白细胞介素1β一般局限于髓核细胞的胞浆内。此外,BAI等[49]发现与Ⅳ级椎间盘退变患者的髓核相比,Ⅴ级椎间盘退变患者的caspase-1、白细胞介素1β和白细胞介素18的表达量更高,且退变程度越高的髓核组织中活性氧的水平也越高。TANG等[50]为了研究焦亡在软骨终板中的作用,分别从MRI影像上观察到Modic改变的腰痛患者与MRI影像上没有退行性改变的椎体爆裂性骨折的年轻患者中收集软骨终板。与对照组相比,Modic改变组软骨形成标志物Ⅱ型胶原纤维和SOX9基因下调,NLRP3、caspase-1和白细胞介素1β的转录水平上调。此外,在Modic改变组中可见到人腰椎软骨终板中大量NLRP3、caspase-1和白细胞介素1β的免疫组化阳性细胞,而对照组中仅见到少量免疫阳性细胞。BRAND等[51]认为,髓核细胞的炎症起源于纤维环、浸润的炎症细胞或者背根神经节。ZHANG等[52]将髓核植入到大鼠背根神经节中,发现NLRP3、caspase-1、白细胞介素1β、白细胞介素18、p-IκBα和p-P65的活性在术后第1天开始升高,第7天达到高峰,表明核转录因子κB通路和NLRP3炎症小体参与了髓核诱导的神经根性疼痛。ZHANG等[53]用纤维环穿刺法建立小鼠椎间盘退变模型,与假手术组相比,模型组中NLRP3、活化的caspase-1和GSDMD的表达水平显著升高,这表明细胞焦亡在椎间盘退变过程中的激活是由NLRP3介导的。FU等[54]用腰椎不稳定手术建立小鼠椎间盘退变模型,结果显示,模型组小鼠纤维环和软骨终版的异位骨形成区中NLRP3的表达显著升高且模型组小鼠的髓核和纤维环中caspase-1和白细胞介素1β表达的显著升高。进一步研究表明,腰椎不稳定手术可能通过激活Wnt/β?catenin信号通路来促进椎间盘细胞的焦亡和神经生长。 2.3.2 细胞焦亡的激活信号 SONG等[55]发现椎间盘退行性疾病患者的髓核组织中NLRP3炎症小体被激活。体外研究表明,晚期糖基化终末产物作为内源性DAMPs,通过NLRP3炎症小体促进髓核细胞线粒体损伤并诱导炎症反应,敲低NLRP3或者抑制caspase-1和白细胞介素1β可以减弱晚期糖基化终末产物诱导的合成和分解代谢活性。这些研究结果表明,髓核组织中晚期糖基化终末产物的积累可能通过激活NLRP3炎症小体从而引起椎间盘炎症反应。 ZHANG等[53]发现脂多糖可以诱导髓核细胞焦亡。体外实验中,脂多糖处理可显著提高髓核细胞中NLRP3、caspase-1和GSDMD的表达水平,在与间充质干细胞共培养后,这一现象显著逆转,提示间充质干细胞处理可抑制脂多糖诱导的髓核细胞焦亡。进一步的研究显示,间充质干细胞对焦亡的影响可能主要是由其衍生的外泌体引起的,间充质干细胞来源的外泌体miRNA-410是焦亡的关键调控因子。 BAI等[49]研究了氧化应激条件下髓核细胞焦亡的机制。体外实验证实,活性氧可以诱导髓核细胞焦亡,退变髓核中NLRP3和PYCARD的表达显著升高;此外还发现,在活性氧诱导的髓核细胞焦亡过程中转录因子Nrf2和自噬水平显著升高,并对焦亡起负调控作用。此研究初步探索了髓核细胞中焦亡与自噬的关系,为椎间盘退变的机制提供了一种新的研究思路。 XU等[56]对正常人和椎间盘退变患者的髓核进行了微阵列分析,确定了miRNA-141在椎间盘退变过程中起关键作用。此外,在动物实验中,退变髓核中的焦亡相关蛋白活化的caspase-1、GSDMD、白细胞介素1β和白细胞介素18的表达水平明显高于正常髓核;miRNA-141的表达在退变髓核中显著上调,miRNA-141模拟物抑制了髓核细胞基质的合成功能。miRNA-141通过增加活性氧和硫氧还蛋白相互作用蛋白的生成诱导髓核细胞的焦亡和细胞外基质分解代谢。 TANG等[57]的研究表明,痤疮丙酸杆菌可以通过活性氧/NLRP3信号通路诱导髓核细胞焦亡,从而引起椎间盘退变。将人髓核细胞与痤疮丙酸杆菌共培养后,白细胞介素1β和白细胞介素18显著过表达,并且NLRP3、白细胞介素1β前体、caspase-1前体和GSDMD也成时间和剂量依赖性增加。此外,将正常的髓核细胞与焦亡的髓核细胞共培养时,正常髓核细胞中炎症因子和分解代谢酶的基因表达显著增加,而当细胞焦亡被抑制时,结果则相反。并且,髓核细胞与痤疮丙酸杆菌共培养可以诱导活性氧生成和NLRP3 的过表达,抑制这两种因素可减少髓核细胞焦亡。因此,抑制痤疮丙酸杆菌引起的髓核细胞焦亡可能成为椎间盘退变的新治疗策略。 2.3.3 细胞焦亡靶向药物 BRAND等[51]发现核转录因子κB通路抑制剂Bay11-7082可抑制NLRP3、凋亡相关斑点样蛋白、caspase-1、白细胞介素1β、白细胞介素18、磷酸化核转录因子κB抑制蛋白及p-p65蛋白等的表达和活化,减轻髓核诱导的神经根性疼痛。JIN等[7]研究发现纳米颗粒富勒醇通过调节NLRP3炎症小体和抑制神经多张性肽的释放来抑制椎间盘突出引起的神经元炎症。ZHAO等[58]研究发现外源性皮质抑素可逆转肿瘤坏死因子α诱导的髓核细胞变性。核转录因子κB抑制剂SN50和NLRP3抑制剂MCC950都能有效减轻髓核细胞退变程度。 ZHAO等[59]研究了酸性敏感离子通道对髓核细胞焦亡的调控机制,发现细胞外乳酸可以促进NLRP3炎症小体的活化和髓核细胞焦亡,这个过程可以被焦亡抑制剂甘氨酸和YVAD阻断。进一步研究发现,乳酸诱导的NLRP3炎症小体激活可以被酸性敏感离子通道抑制剂(阿米洛利、PcTx1及APETx2)和NLRP3 siRNA阻断;细胞外乳酸通过酸性敏感离子通道调节活性氧水平;活性氧激活核转录因子κB信号通路,进一步促进NLRP3的激活和白细胞介素1β的释放,导致椎间盘退变。 ZHOU等[60]研究发现硫氧还蛋白相互作用蛋白通过典型的细胞焦亡激活途径促进髓核细胞焦亡,并通过体内外实验证明桑色素可以抑制这种蛋白,从而抑制髓核细胞焦亡。TANG等[61]也发现和厚朴酚可降低硫氧还蛋白相互作用蛋白的表达,抑制NLRP3炎症小体激活,从而抑制下游炎症因子白细胞介素1β的激活,延缓椎间盘的退变。 HONG等[62]的研究发现NLRP3炎症小体介导的焦亡可由肿瘤坏死因子α诱导,溴结构域蛋白4抑制剂JQ1通过抑制核转录因子κB信号通路的激活,从而减轻NLRP3炎症小体介导的焦亡,而核转录因子κB通路的抑制剂Bay11-7082则限制了NLRP3炎症小体介导的焦亡的激活。此外,通过检测溴结构域蛋白4的mRNA在人髓核组织中的表达情况,发现溴结构域蛋白4的表达水平随着髓核退变程度分级的增加而显著增加。 SUN等[63]发现了一种新型机械敏感离子通道,它是调节NLRP3炎症小体活性的重要上游因子。机械牵张增加了机械敏感离子通道蛋白的表达和细胞内钙负荷,并通过激活核转录因子κB信号通路上调NLRP3的转录水平。此外,它还直接作为第二刺激,促进NLRP3的组装、caspase-1活性增加和白细胞介素1β分泌增加。Piezo1-siRNA被用来下调机械敏感离子通道的表达,抑制NLRP3炎症小体中Ca2+内流和核转录因子κB通路的激活,最终减缓椎间盘退变。 褪黑激素作为一种抗炎性神经内分泌激素,在骨关节炎中起抗炎作用。CHEN等[64]将褪黑素注射到大鼠退变椎间盘中,发现治疗组蛋白聚糖和Ⅱ型胶原蛋白的水平升高。体外实验证实,白细胞介素1β诱导NLRP3炎症小体启动和激活,而褪黑素通过抑制核转录因子κB信号通路的激活和减少线粒体活性氧的产生来抑制炎症反应。HUANG等[65]认为褪黑素通过下调烟酰胺磷酸核糖基转移酶来降低NLRP3炎症小体活性,从而逆转肿瘤坏死因子α诱导的细胞外基质降解,烟酰胺磷酸核糖基转移酶通过MAPK和核转录因子κB信号通路抑制NLRP3炎症小体活性。 吴子健等[66]研究发现通督活血汤含药血清可抑制椎间盘纤维环细胞的焦亡,他们使用脂多糖/三磷酸腺苷制造细胞焦亡模型,分别给予5%,10%,20%通督活血汤含药血清干预。结果显示,与模型组相比,各治疗组白细胞介素1β和白细胞介素18的表达量降低,caspase-1和GSDMD的基因及蛋白表达量显著降低;结果显示,通督活血汤含药血清干预椎间盘纤维环细胞焦亡的最佳干预浓度为10%,最佳干预时间为24 h;但通督活血汤为成方,组方中的有效成分复杂,在细胞焦亡中具体起作用的成分有待于深入研究。 细胞焦亡靶向药物在椎间盘退变中的治疗作用,见表2。"
| [1] GBD 2017 DISEASE AND INJURY INCIDENCE AND PREVALENCE COLLABORATORS. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1789-1858. [2] HOY D, MARCH L, BROOKS P, et al. The global burden of low back pain: estimates from the Global Burden of Disease 2010 study. Ann Rheum Dis. 2014;73(6):968-974. [3] KATZ JN. Lumbar disc disorders and low-back pain: socioeconomic factors and consequences. J Bone Joint Surg Am. 2006;88 Suppl 2:21-24. [4] OICHI T, TANIGUCHI Y, OSHIMA Y, et al. Pathomechanism of intervertebral disc degeneration. JOR spine. 2020;3(1):e1076. [5] CHENG X, ZHANG G, ZHANG L, et al. Mesenchymal stem cells deliver exogenous miR-21 via exosomes to inhibit nucleus pulposus cell apoptosis and reduce intervertebral disc degeneration. J Cell Mol Med. 2018;22(1):261-276. [6] GALLUZZI L, VITALE I, AARONSON SA, et al. Molecular mechanisms of cell death:recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018;25(3):486-541. [7] JIN L, DING M, OKLOPCIC A, et al. Nanoparticle fullerol alleviates radiculopathy via NLRP3 inflammasome and neuropeptides. Nanomedicine. 2017;13(6):2049-259. [8] 胡宝阳,杨学军. MicroRNA影响椎间盘退变过程的研究进展及可发展空间[J].中国组织工程研究,2020,24(21):3372-3378. [9] GRUBER HE, HOELSCHER GL, INGRAM JA, et al. Autophagy in the degenerating human intervertebral disc: in vivo molecular and morphological evidence, and induction of autophagy in cultured annulus cells exposed to proinflammatory cytokines-implications for disc degeneration. Spine. 2015;40(11):773-782. [10] MCKENZIE BA, DIXIT VM, POWER C. Fiery cell death: pyroptosis in the central nervous system. Trends Neurosci. 2020;43(1):55-73. [11] DE MIGUEL C, PELEGR NP. Emerging role of the inflammasome and pyroptosis in hypertension. Int J Mol Sci. 2021;22(3):1064. [12] RUAN J, WANG S, WANG J. Mechanism and regulation of pyroptosis-mediated in cancer cell death. Chem Biol Interact. 2020. doi: 10.1016/j.cbi.2020109052. [13] MAN SM, KARKI R, KANNEGANTI TD. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol Rev. 2017;277(1):61-75. [14] JOHNSON ZI, SCHOEPFLIN ZR, CHOI H, et al. Disc in flames: roles of TNF-α and IL-1β in intervertebral disc degeneration. Eur Cell Mater. 2015;30:104-116; discussion 16-17. [15] CHAO-YANG G, PENG C, HAI-HONG Z. Roles of NLRP3 inflammasome in intervertebral disc degeneration. Osteoarthritis Cartilage. 2021;29(6):793-801. [16] RISBUD MV, SCHOEPFLIN ZR, MWALE F, et al. Defining the phenotype of young healthy nucleus pulposus cells: recommendations of the Spine Research Interest Group at the 2014 annual ORS meeting. J Orthop Res. 2015;33(3):283-293. [17] WU Y, CISEWSKI S, SACHS B L, et al. Effect of cartilage endplate on cell based disc regeneration: a finite element analysis. Mol Cell Biomech. 2013;10(2):159-182. [18] TONG W, LU Z, QIN L, et al. Cell therapy for the degenerating intervertebral disc. Transl Res. 2017;181:49-58. [19] LI P, HOU G, ZHANG R, et al. High-magnitude compression accelerates the premature senescence of nucleus pulposus cells via the p38 MAPK-ROS pathway. Arthritis Res Ther. 2017;19(1):209. [20] WANG SZ, RUI YF, TAN Q, et al. Enhancing intervertebral disc repair and regeneration through biology: platelet-rich plasma as an alternative strategy. Arthritis Res Ther. 2013;15(5):220. [21] FENG Y, EGAN B, WANG J. Genetic factors in intervertebral disc degeneration. Genes Dis. 2016;3(3):178-185. [22] ZYCHLINSKY A, PREVOST MC, SANSONETTI PJ. Shigella flexneri induces apoptosis in infected macrophages. Nature. 1992;358(6382):167-169. [23] COOKSON BT, BRENNAN MA. Pro-inflammatory programmed cell death. Trends Microbiol. 2001;9(3):113-114. [24] SHI J, ZHAO Y, WANG K, et al. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature. 2015;526(7575):660-665. [25] KAYAGAKI N, STOWE IB, LEE BL, et al. Caspase-11 cleaves gasdermin D for non-canonical inflammasome signalling. Nature. 2015;526(7575):666-671. [26] WANG Y, GAO W, SHI X, et al. Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a gasdermin. Nature. 2017;547(7661):99-103. [27] LI MY, ZHU XL, ZHAO BX, et al. Adrenomedullin alleviates the pyroptosis of Leydig cells by promoting autophagy via the ROS-AMPK-mTOR axis. Cell Death Dis. 2019; 10(7):489. [28] JORGENSEN I, MIAO EA. Pyroptotic cell death defends against intracellular pathogens. Immunol Rev. 2015;265(1):130-142. [29] ZHANG Y, CHEN X, GUEYDAN C, et al. Plasma membrane changes during programmed cell deaths. Cell Res. 2018;28(1):9-21. [30] YU P, ZHANG X, LIU N, et al. Pyroptosis:mechanisms and diseases. Signal Transduct Target Ther. 2021;6(1):128. [31] VANDEN BERGHE T, HASSANNIA B, VANDENABEELE P. An outline of necrosome triggers. Cell Mol Life Sci. 2016;73(11-12):2137-2152. [32] ZHOU B, ZHANG JY, LIU XS, et al. Tom20 senses iron-activated ROS signaling to promote melanoma cell pyroptosis. Cell Res. 2018;28(12):1171-1185. [33] HOU J, ZHAO R, XIA W, et al. PD-L1-mediated gasdermin C expression switches apoptosis to pyroptosis in cancer cells and facilitates tumour necrosis. Nat Cell Biol. 2020;22(10):1264-1275. [34] ROGERS C, ERKES DA, NARDONE A, et al. Gasdermin pores permeabilize mitochondria to augment caspase-3 activation during apoptosis and inflammasome activation. Nat Commun. 2019;10(1):1689. [35] KUANG S, ZHENG J, YANG H, et al. Structure insight of GSDMD reveals the basis of GSDMD autoinhibition in cell pyroptosis. Proc Natl Acad Sci U S A. 2017;114(40): 10642-10647. [36] LIU Z, WANG C, YANG J, et al. Crystal Structures of the Full-Length Murine and Human Gasdermin D Reveal Mechanisms of Autoinhibition, Lipid Binding, and Oligomerization. Immunity. 2019;51(1):43-49.e4. [37] DING J, WANG K, LIU W, et al. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature. 2016;535(7610):111-116. [38] XIA S, HOLLINGSWORTH LRT, WU H. Mechanism and Regulation of Gasdermin-Mediated Cell Death. Cold Spring Harb Perspect Biol. 2020;12(3):31451512. [39] ZHANG Z, ZHANG Y, LIEBERMAN J. Lighting a fire: can we harness pyroptosis to ignite antitumor immunity? Cancer Immunol Res. 2021;9(1):2-7. [40] 胡新力,周凯亮,吴晨宇,等.脊髓损伤中细胞焦亡的研究进展[J].中国脊柱脊髓杂志,2020,30(7):657-662. [41] PY BF, KIM MS, VAKIFAHMETOGLU-NORBERG H, et al. Deubiquitination of NLRP3 by BRCC3 critically regulates inflammasome activity. Mol Cell. 2013;49(2):331-338. [42] HE B, NIE Q, WANG F, et al. Role of pyroptosis in atherosclerosis and its therapeutic implications. J Cell Physiol. 2021. doi: 10.1002/jcp.30366. [43] JIN Y, FU J. Novel Insights Into the NLRP 3 Inflammasome in Atherosclerosis. J Am Heart Assoc. 2019;8(12):e012219. [44] KAYAGAKI N, WARMING S, LAMKANFI M, et al. Non-canonical inflammasome activation targets caspase-11. Nature. 2011;479(7371):117-121. [45] RÜHL S, BROZ P. Caspase-11 activates a canonical NLRP3 inflammasome by promoting K(+) efflux. Eur J Immunol. 2015;45(10):2927-2936. [46] KAMBARA H, LIU F, ZHANG X, et al. Gasdermin d exerts anti-inflammatory effects by promoting neutrophil death. Cell Rep. 2018;22(11):2924-2936. [47] KAYAGAKI N, KORNFELD OS. NINJ1 mediates plasma membrane rupture during lytic cell death. Nature. 2021;591(7848):131-136. [48] CHEN ZH, JIN SH, WANG MY, et al. Enhanced NLRP3, caspase-1, and IL- 1β levels in degenerate human intervertebral disc and their association with the grades of disc degeneration. Anat Rec (Hoboken). 2015;298(4):720-726. [49] BAI Z, LIU W, HE D, et al. Protective effects of autophagy and NFE2L2 on reactive oxygen species-induced pyroptosis of human nucleus pulposus cells. Aging (Albany NY). 2020;12(8):7534-7548. [50] TANG P, ZHU R, JI WP, et al. The NLRP3/Caspase-1/interleukin-1β axis is active in human lumbar cartilaginous endplate degeneration. Clin Orthop Relat Res. 2016; 474(8):1818-1826. [51] BRAND FJ 3rd, FOROUZANDEH M, KAUR H, et al. Acidification changes affect the inflammasome in human nucleus pulposus cells. J Inflamm (Lond). 2016;13(1):29. [52] ZHANG A, WANG K, DING L, et al. Bay11-7082 attenuates neuropathic pain via inhibition of nuclear factor-kappa B and nucleotide-binding domain-like receptor protein 3 inflammasome activation in dorsal root ganglions in a rat model of lumbar disc herniation. J Pain Res. 2017;10:375-382. [53] ZHANG J, ZHANG J, ZHANG Y, et al. Mesenchymal stem cells-derived exosomes ameliorate intervertebral disc degeneration through inhibiting pyroptosis. J Cell Mol Med. 2020;24(20):11742-11754. [54] FU F, BAO R, YAO S, et al. Aberrant spinal mechanical loading stress triggers intervertebral disc degeneration by inducing pyroptosis and nerve ingrowth. Sci Rep. 2021;11(1):772. [55] SONG Y, WANG Y, ZHANG Y, et al. Advanced glycation end products regulate anabolic and catabolic activities via NLRP3-inflammasome activation in human nucleus pulposus cells. J Cell Mol Med. 2017;21(7):1373-1387. [56] XU Q, XING H, WU J, et al. miRNA-141 induced pyroptosis in intervertebral disk degeneration by targeting ROS generation and activating TXNIP/NLRP3 signaling in nucleus pulpous cells. Front Cell Dev Biol. 2020;8:871. [57] TANG G, HAN X, LIN Z, et al. Propionibacterium acnes Accelerates Intervertebral Disc Degeneration by Inducing Pyroptosis of Nucleus Pulposus Cells via the ROS-NLRP3 Pathway. Oxid Med Cell Longev. 2021;2021:4657014. [58] ZHAO Y, QIU C, WANG W, et al. Cortistatin protects against intervertebral disc degeneration through targeting mitochondrial ROS-dependent NLRP3 inflammasome activation. Theranostics. 2020;10(15):7015-7033. [59] ZHAO K, AN R, XIANG Q, et al. Acid-sensing ion channels regulate nucleus pulposus cell inflammation and pyroptosis via the NLRP3 inflammasome in intervertebral disc degeneration. Cell Prolif. 2021;54(1):e12941. [60] ZHOU Y, CHEN Z, YANG X, et al. Morin attenuates pyroptosis of nucleus pulposus cells and ameliorates intervertebral disc degeneration via inhibition of the TXNIP/NLRP3/Caspase-1/IL-1β signaling pathway. Biochem Biophys Res Commun. 2021;559:106-112. [61] TANG P, GU JM, XIE ZA, et al. Honokiol alleviates the degeneration of intervertebral disc via suppressing the activation of TXNIP-NLRP3 inflammasome signal pathway. Free Radic Biol Med. 2018;120:368-379. [62] HONG J, LI S, MARKOVA DZ, et al. Bromodomain-containing protein 4 inhibition alleviates matrix degradation by enhancing autophagy and suppressing NLRP3 inflammasome activity in NP cells. J Cell Physiol. 2020;235(7-8):5736-5749. [63] SUN Y, LENG P, SONG M, et al. Piezo1 activates the NLRP3 inflammasome in nucleus pulposus cell-mediated by Ca(2+)/NF-κB pathway. Int Immunopharmacol. 2020;85:106681. [64] CHEN F, JIANG G, LIU H, et al. Melatonin alleviates intervertebral disc degeneration by disrupting the IL-1β/NF-κB-NLRP3 inflammasome positive feedback loop. Bone Res. 2020;8:10. [65] HUANG Y, PENG Y, SUN J, et al. Nicotinamide phosphoribosyl transferase controls NLRP3 Inflammasome Activity Through MAPK and NF-κB signaling in nucleus pulposus cells, as suppressed by melatonin. Inflammation. 2020;43(3):796-809. [66] 吴子健,胡昭端,周晓红,等.通督活血汤含药血清可抑制椎间盘纤维环细胞的焦亡[J].中国组织工程研究,2021,25(14):2148-2154. |
| [1] | Lu Pan, Zhang Chunlin, Wang Yongkui, Yan Xu, Dong Chao, Yue Yisen, Li Long, Zhu Andi. Volume changes of cervical herniated discs after open-door laminoplasty and conservative treatment as assessed by three-dimensional volume method [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1395-1401. |
| [2] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1180-1186. |
| [3] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [4] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [5] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [6] | Yang Kuangyang, Wang Changbing. MRI evaluation of graft maturity and knee function after anterior cruciate ligament reconstruction with autogenous bone-patellar tendon-bone and quadriceps tendon [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 963-968. |
| [7] | Liu Jin, Li Zhen, Hao Huiqin, Wang Ze, Zhao Caihong, Lu Wenjing. Ermiao san aqueous extract regulates proliferation, migration, and inflammatory factor expression of fibroblast-like synovial cells in collagen-induced arthritis rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 688-693. |
| [8] | Wang Shihui, Cheng Yang, Zhu Yunjie, Cheng Shaodan, Mao Jianying. Effect of arc edge needle-scalpel therapy on inflammatory factors and histomorphology of the frozen shoulder in rabbit models [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 706-711. |
| [9] | Li Jiajun, Xia Tian, Liu Jiamin, Chen Feng, Chen Haote, Zhuo Yinghong, Wu Weifeng. Molecular mechanism by which icariin regulates osteogenic signaling pathways in the treatment of steroid-induced avascular necrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 780-785. |
| [10] | Dong Miaomiao, Lai Han, Li Manling, Xu Xiuhong, Luo Meng, Wang Wenhao, Zhou Guoping. Effect of electroacupuncture on expression of nucleotide binding oligomerization domain-like receptor protein 3/cysteinyl aspartate specific proteinase 1 in rats with cerebral ischemia/reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 749-755. |
| [11] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [12] | Lin Zhiyu, Han Jie, Ren Guowu, Chai Yuan, Wen Shuaibo, Wu Yukun, Xie Xiaozhong, Jin Wanqing. Active components of flemingia in regulating the signaling pathways related to knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5889-5896. |
| [13] | Zhao Yunan, Zhang Haobo, Sun Tao, Yang Xuejun. Hydrogel-based growth factors and drugs in the treatment of intervertebral disc degeneration: problems and prospects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5525-5533. |
| [14] | Li Yi, Yang Yanjun, Peng Songyun, Cheng Zhigang, Zhong Kai, Yin Tianping, Tang Lianghua. Effect of Miao medicine Jiuxian Luohan Jiegu Decoction on osteogenic differentiation and fracture healing in tibial fracture rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5350-5356. |
| [15] | Qian Xiaofen, Zeng Ping, Liu Jinfu, Wang Hao, Zhou Shulong, Pan Haida. Screening key genes in synovium of osteoarthritis by a combination of differentially expressed genes and weighted co-expression network analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5342-5349. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

