Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (9): 1439-1444.doi: 10.12307/2022.443
Previous Articles Next Articles
Human salivary components and osteoporosis/osteopenia
Zhu Chan1, 2, Han Xuke1, 2, Yao Chengjiao1, 2, Zhou Qian1, 2, Zhang Qiang3, Chen Qiu1, 2
- 1Affiliated Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, Sichuan Province, China; 2Chengdu University of Traditional Chinese Medicine, Chengdu 610075, Sichuan Province, China; 3Sichuan Provincial Orthopedic Hospital, Chengdu 610041, Sichuan Province, China
-
Received:2021-07-14Revised:2021-07-15Accepted:2021-08-14Online:2022-03-28Published:2021-12-10 -
Contact:陈秋,教授,博士生导师,成都中医药大学附属医院,四川省成都市 610072;成都中医药大学,四川省成都市 6100756 -
About author:Zhu Chan, Doctoral candidate, Attending physician, Affiliated Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, Sichuan Province, China; Chengdu University of Traditional Chinese Medicine, Chengdu 610075, Sichuan Province, China -
Supported by:National Traditional Chinese Medicine and Western Medicine Clinical Collaboration Project for Major and Intractable Diseases, No. CYW2019079 (to CQ); State Administration of Traditional Chinese Medicine Project, No. SATCM-2015-BZ(204) (to CQ); Chengdu Key Technology Application Demonstration Project, No. 2019-YF09-00094-SN (to CQ)
CLC Number:
Cite this article
Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
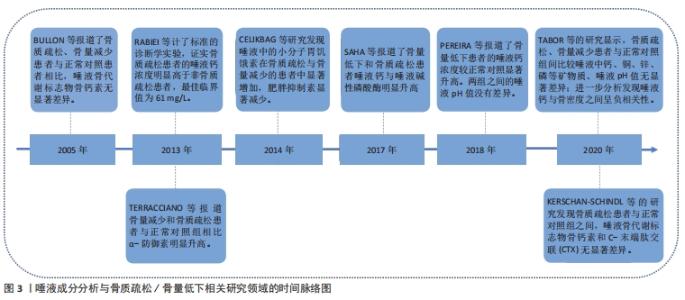
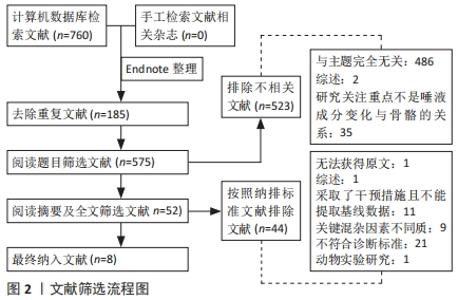
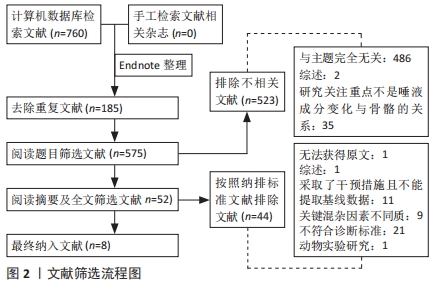
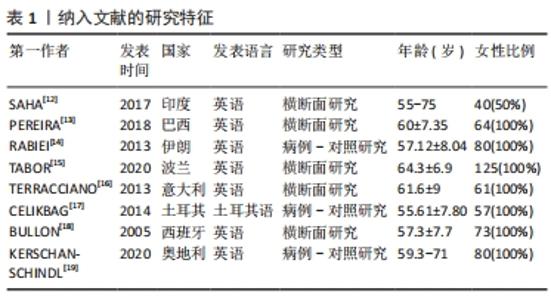
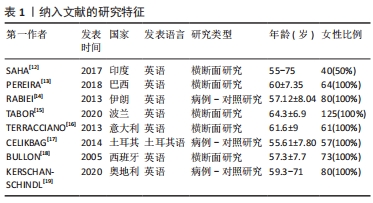
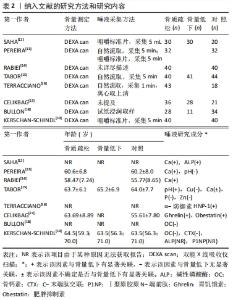
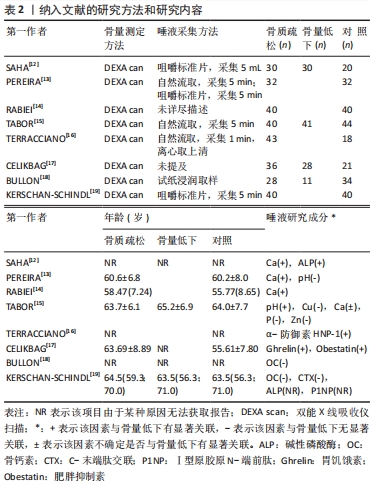
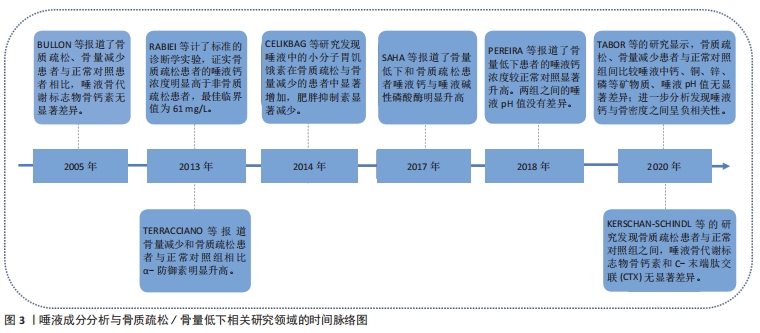
2.3 唾液成分与骨质疏松/骨量低下的关系 在唾液成分与骨量低下/骨质疏松的相关性研究中,关注了12种唾液成分指标,分别是唾液矿物质(钙Ca、锌Zn、磷P、铜Cu),唾液中的典型骨转换标志物碱性磷酸酶(alkaline phosphatase,ALP),骨钙素(osteocalcin,OC)、C-末端肽交联(C-terminal telopeptide of type 1 collagen,CTX)、Ⅰ型原胶原N-端前肽(procollagen type 1 N-peptide,P1NP),唾液中的某些生物活性肽(胃饥饿素Ghrelin、肥胖抑制素Obestatin)和唾液pH值。唾液成分分析与骨质疏松/骨量低下相关研究领域的时间脉络图,见图3。"

2.3.1 矿物质(Mineral) 骨代谢平衡是适当矿化和脱矿的结果,这些过程是动态的、终生的。离子钙和无机磷酸盐是骨矿化的底物,一直被认为是最重要的骨骼矿物质[20]。RABIEI等[14]设计了一个标准的诊断学实验,对40例绝经后骨质疏松妇女和40例非骨质疏松妇女进行病例-对照研究,采集全唾液样本测量钙浓度,结果显示病例组和对照组唾液钙值分别为68.2(61.87-76.67) mg/L vs. 56.3(62.73-50.01) mg/L,P < 0.001;根据ROC曲线确定的最佳临界值为61.0 mg/L,敏感度和特异度分别为67.5%(95%CI:52.33-82.67)和60%(95% CI:44.62-75.38),因此认为唾液钙是诊断骨质疏松的一个可能的替代方法。SAHA等[12]采集了80例受试者的唾液,根据骨密度将他们分为骨质疏松组、骨量低下组、正常组,3组的唾液钙值分别为8.42±1.33,7.67±1.06,4.25±0.45(P < 0.001),骨质疏松/骨量低下的患者唾液钙含量显著增高,因此推测唾液钙可能是一个有效的生物学标记物以用于发现潜在的骨代谢障碍。PEREIRA等[13]进行了一项控制性横断面研究,将64名绝经后妇女根据骨密度分为低骨量组和正常组,两组分别收集刺激性唾液和非刺激性唾液,低骨量组较正常组受试者有更高的唾液钙浓度(285 mg/L vs. 85 mg/L,P < 0.001),当不考虑分组效应直接将所有样本的骨密度与唾液钙浓度进行线性相关分析,却发现二者相关性很弱(rs=0.097)。TABOR等[15]测量了125名绝经后女性不同部位的骨密度,并测量唾液中的多种矿物质含量,最后发现唾液钙浓度与股骨颈骨密度呈显著负相关(r=-0.23,P < 0.05),而在其他部位(脊柱、全髋)则没发现这种显著的相关性;该研究还同时测定了唾液中铜、锌、磷的含量,发现这些指标在骨质疏松/骨量低下与正常对照之间均无显著的差异。 2.3.2 典型骨转换标志物 在不同疾病状态时,血循环或者尿液中的骨转换标志物会发生不同程度的改变。碱性磷酸酶是一种约45 kD的蛋白质,存在于多种组织当中,在矿化组织的细胞中高度表达,是骨形成中起关键作用的骨转换标志物[21]。SAHA等[12]采集了80例患者的唾液,同时测定骨密度,根据骨密度分为骨质疏松组、骨量低下组、正常组,各组碱性磷酸酶的值分别为163.44±7.79,168.94±8.11,57.84±5.90(P < 0.001),认为骨质疏松/骨量低下的患者唾液中的碱性磷酸酶含量较高。 骨钙素是一种由成骨细胞分泌的蛋白质,其在骨重塑、成骨细胞特异性表达和调控的过程中发挥重要作用[11]。血清中骨钙素是成骨细胞活性的标志物,其水平反映了骨形成的速率[22]。BULLON等[18]选择了73例绝经后女性,其中骨质疏松组28例,骨量低下组11例,正常组34例,各组唾液骨钙素水平分别为(5.2±3.9),(5.7±2.8),(5.3±2.2) μg/L(P=0.88),提示不同骨量组别之间唾液骨钙素的含量没有显著差异。 CTX是一类敏感的骨吸收标志物,KERSCHAN-SCHINDL等[19]选择了40例绝经后骨质疏松女性及与其年龄相匹配的40名绝经后非骨质疏松患者,同时采集了血液和唾液中骨代谢标志物,发现骨质疏松组与非骨质疏松组的唾液骨钙素值为:(1.63±0.35) μg/L vs. (1.58±0.25) μg/L,P=0.329;两组的CTX值分别为(0.06±0.049) μg/L vs. (0.047±0.025) μg/L,P=0.315;并且还将受试者的不同部位的骨密度与唾液中骨钙素和CTX浓度进行相关性分析,仍然提示二者与被试者任何部位的骨密度没有显著相关性。 2.3.3 其他生物活性肽 由于破骨细胞受到免疫系统在分子水平的调节,因此,骨质疏松患者的免疫细胞可能会释放特定产物[23-24]。α-防御素是一种小的抗菌肽,是先天免疫系统的一部分,并动态地参与了多种生物活动,HNP-1(human neutrophil peptide-1)是防御素的一种类型,最近被报道可作为优秀运动员免疫系统生物标志[25]。TERRACCIANO等[16]连续招募了61名绝经后妇女,同时测定骨密度和采集唾液,发现唾液中α-防御素的浓度与骨密度呈显著负相关(r=-0.26,P < 0.05)。 胃饥饿素是一种多方面的肠道激素,标志性功能是其对生长激素释放、食物摄入和脂肪沉积的刺激作用,最新的研究成果显示胃饥饿素可以通过调节成骨细胞的增殖和分化来调节骨代 谢[26]。肥胖抑制素为一种生长素释放肽相关肽,来源于前生长素释放肽原基因的翻译后加工[27]。CELIKBAG等[17]选择了85名绝经后女性行骨密度检查,其中骨质疏松组36名,骨量低下组28名,正常组21名,采集所有被试者的唾液检测胃饥饿素和肥胖抑制素,各组的胃饥饿素水平分别为(79.8±14.0),(79.74±11.76),(70.28±15.36) ng/L;各组的肥胖抑制素水平分别是(750±1 460),(160±290),(2 180±2 500) ng/L,提示绝经后的骨质疏松/骨量低下患者基线时唾液中胃饥饿素水平显著高于对照组(P < 0.05),肥胖抑制素Obestatin水平显著低于对照组(P < 0.05)。 2.3.4 酸碱度pH值 唾液pH值表示口腔的酸碱程度,是一个化学标志物。PEREIRA等[13]将64名绝经后妇女根据骨密度分为低骨量组和正常组,两组分别收集唾液,低骨量组与正常组唾液的pH值比较无显著差异(7.2±0.4 vs. 7.1±0.4,P=0.738),不考虑分组效应,直接将总样本的骨密度与唾液pH值做相关性分析,二者相关性也很弱(rs=-0.190)。TABOR等[15]测量了125名绝经后女性不同部位的骨密度,发现脊柱骨质疏松患者的唾液pH值明显低于骨密度正常者(6.65±0.67 vs. 6.96±0.58,P < 0.05),而在其他部位(股骨颈、全髋)则没有这样的显著相关。"

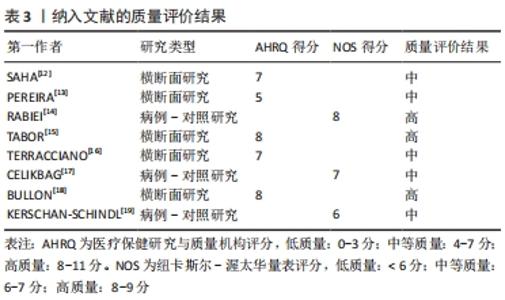
| [1] 夏维波,章振林,林华,等.原发性骨质疏松症诊疗指南(2017)[J].中国骨质疏松杂志,2019,25(3):281-309. [2] HONG SJ, YANG BE, YOO DM, et al. Analysis of the relationship between periodontitis and osteoporosis/fractures: a cross-sectional study. BMC Oral Health. 2021;21(1):125. [3] TAYEB Y, GOULTSCHIN J, FOGEL M, et al. The relationship between osteoporosis, osteopenia and periodontitis. Refuat Hapeh Vehashinayim (1993). 2003;20(1):8-22,78. [4] CHOU HH, LU SL, WANG ST. The Association between Bone Mineral Density and Periodontal Disease in Middle-Aged Adults. Int J Environ Res Public Health. 2021;18(6):3321. [5] INAGAKI K, KUROSU Y, SAKANO M, et al. Oral osteoporosis: a review and its dental implications. Clin Calcium. 2007;17(2):157-63. [6] ZHANG C Z, CHENG XQ, LI JY, et al. Saliva in the diagnosis of diseases. Int J Oral Sci. 2016;8(3):133-137. [7] WOŹNIAK M, PALUSZKIEWICZ C, KWIATEK WM. Saliva as a non-invasive material for early diagnosis. Acta Biochim Pol. 2019;66(4):383-388. [8] WATTS NB, CAMACHO PM, LEWIECKI EM, et al. AACE/ACE Postmenopausal Osteoporosis Guidelines Task Force. American Association of Clinical Endocrinologists/American College of Endocrinology Clinical Practice Guidelines for the Diagnosis and Treatment of Postmenopausal Osteoporosis-2020 Update. Endocr Pract. 2021;27(4):379-380. [9] ZENG X, ZHANG Y, KWONG JS, et al. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J Evid Based Med. 2015;8(1):2-10. [10] LO CK, MERTZ D, LOEB M. Newcastle-Ottawa Scale: comparing reviewers’ to authors’ assessments. BMC Med Res Methodol. 2014;14:45. [11] DENT O. Methodological index for non-randomized studies. ANZ J Surg. 2003;73(9):675-676. [12] SAHA MK, AGRAWAL P, SAHA SG, et al. Evaluation of Correlation between Salivary Calcium, Alkaline Phosphatase and Osteoporosis- A Prospective, Comparative and Observational Study. J Clin Diagn Res. 2017;11(3):ZC63-ZC66. [13] PEREIRA IF, BRASILEIRO CB, KLEPERON NP, et al. Comparative study of oral and salivary parameters in patients with and without loss of bone mass. Braz Oral Res. 2018;32:e54. [14] RABIEI M, MASOOLEH IS, LEYLI EK, et al. Salivary calcium concentration as a screening tool for postmenopausal osteoporosis. Int J Rheum Dis. 2013; 16(2):198-202. [15] TABOR E, HŰPSCH H, ROKICKA J, et al. Salivary Content Might be Associated With Skeletal Status in Postmenopausal Women: SilesiaOsteoActive Study Results. J Clin Densitom. 2021;24(1):14-21. [16] TERRACCIANO R, MIGLIACCIO V, SAVINO R, et al. Association between low bone mineral density and increased α-defensin in salivary fluid among postmenopausal women. Menopause. 2013;20(12):1275-1279. [17] CELIKBAG B, AKGOL G, AYDIN S, et al. Ghrelin and Obestatin Levels in Serum and Saliva of Patients with Osteoporosis and the Effect of Strontium Ranelate. J Phys Med Rehabil. 2014;60(1):7-11. [18] BULLON P, GOBERNA B, GUERRERO JM, et al. Serum, saliva, and gingival crevicular fluid osteocalcin: their relation to periodontal status and bone mineral density in postmenopausal women. J Periodontol. 2005;76(4):513-519. [19] KERSCHAN-SCHINDL K, BOSCHITSCH E, MARCULESCU R, et al. Bone turnover markers in serum but not in saliva correlate with bone mineral density. Sci Rep. 2020;10(1):11550. [20] MURSHED M. Mechanism of Bone Mineralization. Cold Spring Harb Perspect Med. 2018;8(12):a031229. [21] VIMALRAJ S. Alkaline phosphatase: Structure, expression and its function in bone mineralization. Gene. 2020;754:144855. [22] KARSENTY G. Update on the biology of osteocalcin. Endocr Pract. 2017; 23(10):1270-1274. [23] WADA S, FUKAWA T, KAMIYA S. Osteocalcin and bone. Clin Calcium. 2007; 17(11):1673-1677. [24] BOYCE BF. Advances in the regulation of osteoclasts and osteoclast functions. J Dent Res. 2013;92(10):860-867. [25] PERO R, BRANCACCIO M, MENNITTI C, et al. HNP-1 and HBD-1 as Biomarkers for the Immune Systems of Elite Basketball Athletes. Antibiotics (Basel). 2020;9(6):306. [26] PRADHAN G, SAMSON SL, SUN Y. Ghrelin: much more than a hunger hormone. Curr Opin Clin Nutr Metab Care. 2013;16(6):619-624. [27] LI J B, ASAKAWA A, CHENG K, et al. Biological effects of obestatin. Endocrine. 2011;39(3):205-211. [28] SEW N L, LAINE M, KARJALAINEN S, et al. Salivary calcium reflects skeletal bone density of heavy smokers. Arch Oral Biol. 2004;49(5): 355-358. [29] WASTI A, WASTI J, SINGH R. Estimation of salivary calcium level as a screening tool for the osteoporosis in the post-menopausal women: A prospective study. Indian J Dent Res. 2020;31(2):252-256. [30] SEWóNL, LAINE M, KARJALAINEN S, et al. The effect of hormone replacement therapy on salivary calcium concentrations in menopausal women. Arch Oral Biol. 2000;45(3):201-206. [31] BUDUNELI N, SAYGAN BH, KARADUMAN U, et al. Calcium, vitamin D supplements with or without alendronate and supragingival calculus formation in osteoporotic women: a preliminary study. Expert Opin Pharmacother. 2008;9(12):2015-2020. [32] SEV N L, LAINE MA, KARJALAINEN S, et al. Effect of age on flow-rate, protein and electrolyte composition of stimulated whole saliva in healthy, non-smoking women. Open Dent J. 2008;2:89-92. [33] RAI B, KAUR J, CATALINA M. Bone mineral density, bone mineral content, gingival crevicular fluid (matrix metalloproteinases, cathepsin K, osteocalcin), and salivary and serum osteocalcin levels in human mandible and alveolar bone under conditions of simulated microgravity. J Oral Sci. 2010;52(3):385-390. [34] KAWADA T. Serum alkaline phosphatase level and metabolic syndrome. Clin Chim Acta. 2020;506:187. [35] No authors listed. Serum alkaline phosphatase (AP) is perhaps the most widely used biomarker since it was discovered. Introduction. Subcell Biochem. 2015;76:v-vii. [36] OSELLA G, VENTURA M, ARDITO A, et al. Cortisol secretion, bone health, and bone loss: a cross-sectional and prospective study in normal non-osteoporotic women in the early postmenopausal period. Eur J Endocrinol. 2012;166(5):855-860. [37] RAFF H, RAFF JL, DUTHIE EH, et al. Elevated salivary cortisol in the evening in healthy elderly men and women: correlation with bone mineral density. J Gerontol A Biol Sci Med Sci. 1999;54(9):M479-483. [38] HARDY R, COOPER M S. Adrenal gland and bone . Arch Biochem Biophys. 2010;503(1):137-145. [39] JEFFERIES WM. Cortisol and immunity. Med Hypotheses. 1991;34(3):198-208. [40] IR R, 徐隆绍. 类固醇性骨质疏松的发病学和治疗 [J]. 国外医学内分泌学分册, 1990(1):22-25. [41] CHIODINI I, MASCIA M L, MUSCARELLA S, et al. Subclinical hypercortisolism among outpatients referred for osteoporosis. Ann Intern Med. 2007;147(8): 541-548. [42] TURPEINEN U, HÄMÄLÄINEN E. Determination of cortisol in serum, saliva and urine. Best Pract Res Clin Endocrinol Metab. 2013;27(6):795-801. [43] HART K A, KITCHINGS KM, KIMURA S, et al. Measurement of cortisol concentration in the tears of horses and ponies with pituitary pars intermedia dysfunction. Am J Vet Res. 2016;77(11):1236-1244. [44] WESTER VL, VAN ROSSUM EF. Clinical applications of cortisol measurements in hair. Eur J Endocrinol. 2015;173(4):M1-10. [45] GOZANSKY WS, LYNN JS, LAUDENSLAGER ML, et al. Salivary cortisol determined by enzyme immunoassay is preferable to serum total cortisol for assessment of dynamic hypothalamic--pituitary--adrenal axis activity. Clin Endocrinol (Oxf). 2005;63(3):336-341. [46] REIMONDO G, PIA A, BOVIO S, et al. Laboratory differentiation of Cushing’s syndrome. Clin Chim Acta. 2008;388(1-2):5-14. [47] ROMERO MF, ROSSANO AJ. Acid-Base Basics. Semin Nephrol. 2019;39(4): 316-327. [48] GALGUT PN. The relevance of pH to gingivitis and periodontitis. J Int Acad Periodontol. 2001;3(3):61-67. [49] WANG O, HU Y, GONG S, et al. A survey of outcomes and management of patients post fragility fractures in China. Osteoporos Int. 2015;26(11): 2631-2640. [50] CHANDRAN M, BHADADA SK, EBELING PR, et al. IQ driving QI: the Asia Pacific Consortium on Osteoporosis (APCO): an innovative and collaborative initiative to improve osteoporosis care in the Asia Pacific. Osteoporos Int. 2020;31(11):2077-2081. [51] KACZOR-URBANOWICZ KE, MARTIN CARRERAS-PRESAS C, ARO K, et al. Saliva diagnostics - Current views and directions. Exp Biol Med (Maywood). 2017;242(5):459-472. [52] GREABU M, BATTINO M, MOHORA M, et al. Saliva--a diagnostic window to the body, both in health and in disease. Med Life. 2009;2(2):124-132. |
| [1] | Jiang Huanchang, Zhang Zhaofei, Liang De, Jiang Xiaobing, Yang Xiaodong, Liu Zhixiang. Comparison of advantages between unilateral multidirectional curved and straight vertebroplasty in the treatment of thoracolumbar osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1407-1411. |
| [2] | Li Wei, Zhu Hanmin, Wang Xin, Gao Xue, Cui Jing, Liu Yuxin, Huang Shuming. Effect of Zuogui Wan on bone morphogenetic protein 2 signaling pathway in ovariectomized osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1173-1179. |
| [3] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [4] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [5] | Peng Kun. Improvement of the treatment effect of osteoporotic fractures: research status and strategy analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 980-984. |
| [6] | Shen Song, Xu Bin. Diffuse distribution of bone cement in percutaneous vertebroplasty reduces the incidence of refracture of adjacent vertebral bodies [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 499-503. |
| [7] | Ou Liang, Kong Dezhong, Xu Daoqing, Ni Jing, Fu Xingqian, Huang Weichen. Comparative clinical efficacy of polymethyl methacrylate and self-solidifying calcium phosphate cement in vertebroplasty: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 649-656. |
| [8] | Li Haoliang, Li Dongfang. Emodin promotes fracture healing in osteoporotic fracture rats by inhibiting miR-338-3p expression [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5155-5161. |
| [9] | Zhang Haiyong, Huang Jingwen, Xie Bingying, Chen Sainan, Xie Lihua, Chen Xuan, Li Shengqiang, Ge Jirong. Anti-osteoporosis effect of Gushukang in ovariectomized rats: a lumbar metabonomic analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5185-5190. |
| [10] | Gu Chao, Chen Weikai, Liu Tao, Yang Huilin, He Fan. Mitochondrial dysfunction affects osteogenic differentiation potential of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 4921-4927. |
| [11] | Xiang Qianru, Deng Xuejian, Chen Huafeng, Liang Jiamin, An Min, Yang Li. Gene-modified stem cells therapy for osteoporosis: a meta-analysis of preclinical studies [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5053-5061. |
| [12] | Huang Gao, Xu Jun, Chen Wenge. Implantation of bone marrow mesenchymal stem cells-loaded platelet-rich plasma combined with extracorporeal shock wave in the repair of bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4812-4818. |
| [13] | Huang Wei, Dong Panfeng, Huang Yourong, Xia Tian. Epimedium in regulating bone marrow mesenchymal stem cell differentiation and preventing osteoporosis related signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4889-4895. |
| [14] | Cai Feng, Yu Bo, Zeng Duo, Chen Qincan, Liao Qi. Cortical bone trajectory in elderly patients with osteoporosis of lumbar disease [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 403-407. |
| [15] | Zhong Yuanming, He Bingkun, Wu Zhuotan, Wu Sixian, Wan Tong, Zhong Xifeng. Meta-analysis of efficacy and safety of Jack kyphoplasty for osteoporotic vertebral compression fractures [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 487-492. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||