Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (34): 5465-5472.doi: 10.12307/2021.240
Previous Articles Next Articles
Effect of modified citrus pectin on chondrocytes
Zhang Yiyun, Wang Ziqiang, Ren Ying, Du Fuchong, Du Bo, Li Xuemin
- Institute of Biomedical Engineering, Chinese Academy of Medical Sciences & Peking Union Medical College, Key Laboratory of Biomedical Materials of Tianjin, Tianjin 300192, China
-
Received:2021-01-08Revised:2021-01-11Accepted:2021-01-30Online:2021-12-08Published:2021-07-27 -
Contact:Li Xuemin, MD, Associate researcher, Institute of Biomedical Engineering, Chinese Academy of Medical Sciences & Peking Union Medical College, Key Laboratory of Biomedical Materials of Tianjin, Tianjin 300192, China -
About author:Zhang Yiyun, Master candidate, Institute of Biomedical Engineering, Chinese Academy of Medical Sciences & Peking Union Medical College, Key Laboratory of Biomedical Materials of Tianjin, Tianjin 300192, China -
Supported by:General Project of National Natural Science Foundation of China, No. 81972043 (to LXM); the Major Collaborative Innovation Project of Chinese Academy of Medical Sciences Medicine and Health Technology Innovation Project, No. 2017-I2M-1-007 (to LXM)
CLC Number:
Cite this article
Zhang Yiyun, Wang Ziqiang, Ren Ying, Du Fuchong, Du Bo, Li Xuemin. Effect of modified citrus pectin on chondrocytes[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5465-5472.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
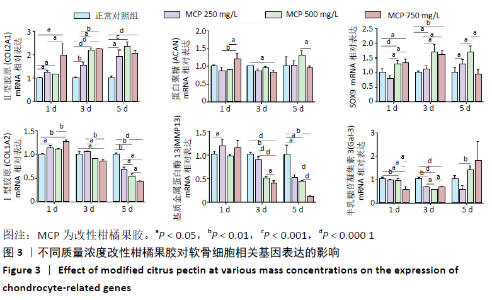
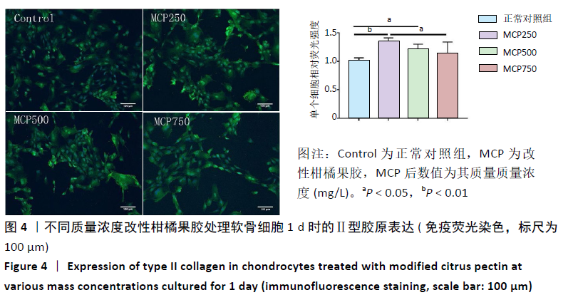
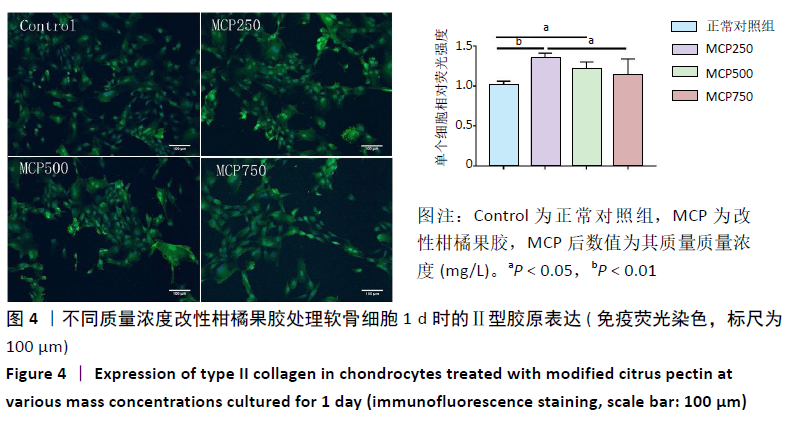
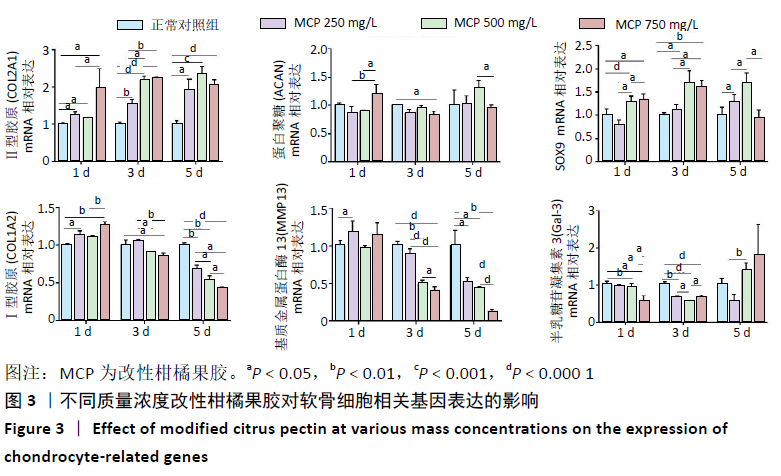
2.1.3 对软骨细胞相关基因表达的影响 图3显示了各组软骨细胞相关基因的表达情况。与正常对照组相比,改性柑橘果胶处理组的Ⅱ型胶原mRNA表达量在第1,3,5 天时均有明显升高,且在第1,3天都随着改性柑橘果胶质量浓度的升高而升高;除了750 mg/L组第3天时的蛋白聚糖mRNA表达低于正常对照组外(P=0.012),其余质量浓度组不同时间点的蛋白聚糖mRNA表达与正常对照组比较差异均无显著性意义(P > 0.05)。250 mg/L组第5天的SOX9 mRNA表达高于正常对照组(P=0.02),其余时间点与正常对照组比较差异无显著性意义(P > 0.05);500,750 mg/L组第1,3天的SOX9 mRNA表达均高于正常对照组,第5天的SOX9 mRNA表达与正常对照组比较差异无显著性意义(P > 0.05)。 与正常对照组比较,不同质量浓度改性柑橘果胶处理后第1天的Ⅰ型胶原mRNA表达均有不同程度的升高,但在第3,5天时都有降低,尤其是第5天时的Ⅰ型胶原mRNA表达明显低于正常对照组(P < 0.05),并且下降程度随改性柑橘果胶质量浓度的升高更加明显。与Ⅰ型胶原mRNA表达变化相似,250 mg/L组第1天的基质金属蛋白酶13 mRNA表达明显高于正常对照组(P < 0.05),而第3,5天的基质金属蛋白酶13 mRNA表达低于正常对照组,并且下降程度随改性柑橘果胶质量浓度升高更加明显。250 mg/L组第3天的Gal-3 mRNA表达明显低于正常对照组(P < 0.05),其余时间点比较差异均无显著性意义;500,750 mg/L组第1,3天的Gal-3 mRNA表达明显低于正常对照组(P < 0.05),第5天的Gal-3 mRNA表达与正常对照组比较差异无显著性意义(P > 0.05)。"
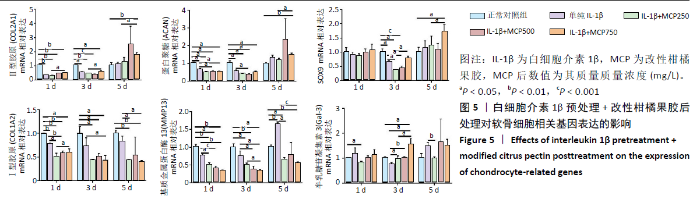
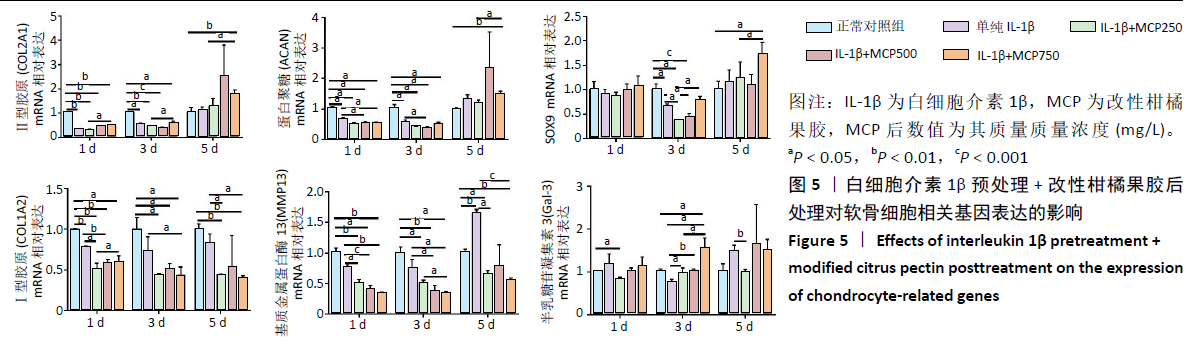
2.2.1 对软骨细胞相关基因表达的影响 白细胞介素1β预处理+改性柑橘果胶后处理对软骨细胞相关基因表达的影响,见图5。与正常对照组相比,单纯白细胞介素1β处理组第1,3天的Ⅱ型胶原和蛋白聚糖mRNA表达降低(P < 0.05),第5天时无明显变化(P > 0.05);单纯白细胞介素1β处理组第3天的SOX9 mRNA表达降低,第1,5天时无明显变化(P > 0.05)。与单纯白细胞介素1β处理组比较,改性柑橘果胶处理各组第1,3天的Ⅱ型胶原mRNA表达无明显变化(P > 0.05),蛋白聚糖和SOX9 mRNA表达会有不同程度的降低;改性柑橘果胶处理各组第5天的Ⅱ型胶原、蛋白聚糖和SOX9 mRNA表达都有一定的升高。 与正常对照组比较,单纯白细胞介素1β处理组第1天的Ⅰ型胶原mRNA表达降低(P=0.005),第3和5天时无明显变化(P > 0.05);改性柑橘果胶处理各组第1,3,5天的Ⅰ型胶原mRNA表达不同程度地低于正常对照组、单纯白细胞介素1β处理组;同时,250 mg/L改性柑橘果胶处理组第1天的Ⅰ型胶原mRNA表达低于750 mg/L改性柑橘果胶处理组(P < 0.05)。单纯白细胞介素1β处理组第1,3天的基质金属蛋白酶13 mRNA表达低于正常对照组,改性柑橘果胶处理各组第1,3天的基质金属蛋白酶13 mRNA表达低于单纯白细胞介素1β处理组,并且降低程度随改性柑橘果胶质量浓度的增加更明显;单纯白细胞介素1β处理组第5天的基质金属蛋白酶13 mRNA表达高于正常对照组(P=0.01),改性柑橘果胶处理各组第5天的基质金属蛋白酶13 mRNA表达不同程度地低于正常对照组、单纯白细胞介素1β处理组。单纯白细胞介素1β处理组不同时间点的Gal-3 mRNA表达与正常对照组比较差异无显著性意义(P > 0.05);250 mg/L改性柑橘果胶处理组第1天的Gal-3 mRNA表达低于正常对照组(P < 0.05),改性柑橘果胶处理各组第3天的Gal-3 mRNA表达高于正常对照组,改性柑橘果胶处理各组第3天的Gal-3 mRNA表达高于单纯白细胞介素1β处理组(P < 0.05),250 mg/L改性柑橘果胶处理组第5天的Gal-3 mRNA表达低于单纯白细胞介素1β处理组(P < 0.05)。"
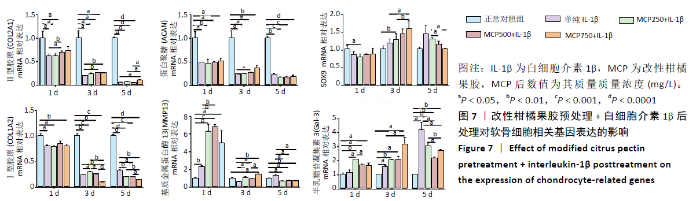
2.3.1 对软骨细胞相关基因表达的影响 改性柑橘果胶预处 理+白细胞介素1β后处理对软骨细胞相关基因表达的影响,见图7。与正常对照组相比,白细胞介素1β处理各组中软骨细胞的Ⅱ型胶原和蛋白聚糖mRNA表达在整个培养周期内均不同程度地降低;与单纯白细胞介素1β处理组相比, 500 mg/L改性柑橘果胶处理组第1,3天的Ⅱ型胶原mRNA表达升高,750 mg/L改性柑橘果胶处理组第3,5天的Ⅱ型胶原mRNA表达升高。500 mg/L改性柑橘果胶处理组第3天的蛋白聚糖mRNA表达高于单纯白细胞介素1β处理组(P=0.024)。单纯白细胞介素1β处理组整个培养周期内的SOX9 mRNA表达与正常对照组比较差异均无显著性意义(P > 0.05);250 mg/L改性柑橘果胶处理组第1天的SOX9 mRNA表达低于正常对照组(P < 0.05),第3天的SOX9 mRNA表达高于正常对照组(P < 0.05);750 mg/L改性柑橘果胶处理组第3天的SOX9 mRNA表达高于单纯白细胞介素1β处理组(P=0.028)。"
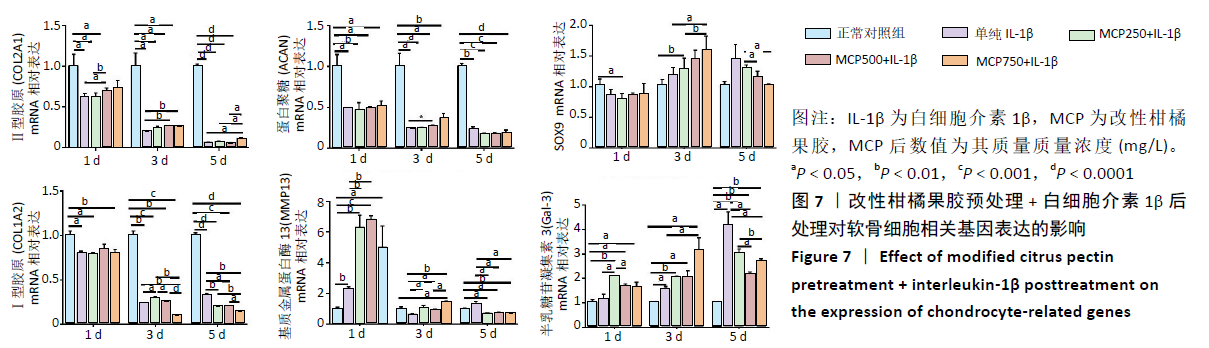
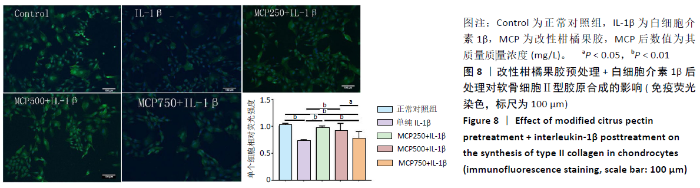
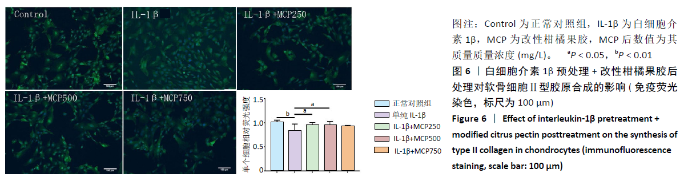
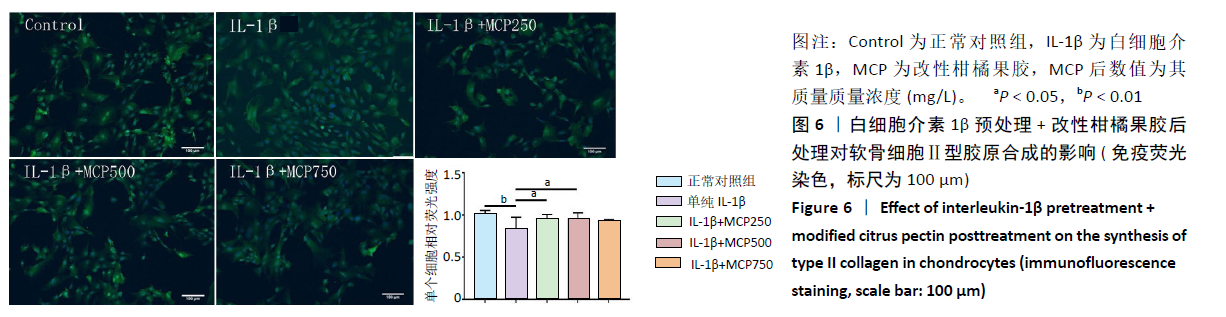
与正常对照组相比,白细胞介素1β处理各组的Ⅰ型胶原mRNA表达在整个培养周期内均明显下降;750 mg/L改性柑橘果胶处理组第3天的Ⅰ型胶原mRNA表达低于单纯白细胞介素1β处理组(P=0.002);改性柑橘果胶处理各组第5天的Ⅰ型胶原mRNA表达低于单纯白细胞介素1β处理组。与正常对照组相比,白细胞介素1β处理各组第1天的基质金属蛋白酶13 mRNA表达升高,750 mg/L改性柑橘果胶处理组第3天的基质金属蛋白酶13 mRNA表达仍升高;改性柑橘果胶处理各组第5天的基质金属蛋白酶13 mRNA表达低于正常对照组、单纯白细胞介素1β处理组。与正常对照组相比,白细胞介素1β处理各组第3,5天的Gal-3 mRNA表达升高,并且改性柑橘果胶处理各组整个周期的Gal-3 mRNA表达升高(P < 0.05);500,750 mg/L改性柑橘果胶处理组第5天的Gal-3 mRNA表达低于单纯白细胞介素1β处理组(P < 0.05)。 2.3.2 对软骨细胞Ⅱ型胶原合成的影响 改性柑橘果胶预处理+白细胞介素1β后处理对软骨细胞Ⅱ型胶原合成的影响,见图8。各组软骨细胞都呈现出Ⅱ型胶原免疫荧光染色阳性,有意思的是与正常对照组和单纯白细胞介素1β处理组相比不同,经改性柑橘果胶预处理后的软骨细胞成团生长趋势较为明显。统计分析显示,单纯白细胞介素1β处理组的Ⅱ型胶原荧光强度值低于正常对照组(P < 0.05),250,500 mg/L改性柑橘果胶处理组Ⅱ型胶原荧光强度值高于单纯白细胞介素1β处理组(P < 0.05)。"

| [1] GOLDRING MB. Chondrogenesis, chondrocyte differentiation, and articular cartilage metabolism in health and osteoarthritis. Ther Adv Musculoskelet Dis. 2012;4(4):269-285. [2] AHSAN T, SAH RL. Biomechanics of integrative cartilage repair. Osteoarthritis Cartilage. 1999;7(1):29-40. [3] WARNOCK WR, MARSH CA, HAND DR. Outcome of arthroscopic debridement of cartilage injury in the equine distal interphalangeal joint. Can Vet J. 2019;60(7):731-736. [4] GIANAKOS AL, YASUI Y, FRASER EJ, et al. The Effect of Different Bone Marrow Stimulation Techniques on Human Talar Subchondral Bone: A Micro-Computed Tomography Evaluation. Arthroscopy. 2016;32(10):2110-2117. [5] BECHER C, MALAHIAS MA, ALI MM, et al. Arthroscopic microfracture vs. arthroscopic autologous matrix-induced chondrogenesis for the treatment of articular cartilage defects of the talus. Knee Surg Sports Traumatol Arthrosc. 2019;27(9):2731-2736. [6] MERKELY G, OGURA T, BRYANT T, et al. Severe Bone Marrow Edema Among Patients Who Underwent Prior Marrow Stimulation Technique Is a Significant Predictor of Graft Failure After Autologous Chondrocyte Implantation. Am J Sports Med. 2019;47(8):1874-1884. [7] HUANG BJ, HU JC, ATHANASIOU KA. Cell-based tissue engineering strategies used in the clinical repair of articular cartilage. Biomaterials. 2016;98:1-22. [8] JUNG M, KARAMPINOS D, HOLWEIN C, et al. Quantitative 3-T Magnetic Resonance Imaging After Matrix-Associated Autologous Chondrocyte Implantation With Autologous Bone Grafting of the Knee: The Importance of Subchondral Bone Parameters. Am J Sports Med. 2021:363546520980134. [9] ARBAB D, REIMANN P, BRUCKER M, et al. Alignment in total knee arthroplasty - A comparison of patient-specific implants with the conventional technique. Knee. 2018;25(5):882-887. [10] KAMENAGA T, HIRANAKA T, KIKUCHI K, et al. Influence of tibial component rotation on short-term clinical outcomes in Oxford mobile-bearing unicompartmental knee arthroplasty. Knee. 2018;25(6):1222-1230. [11] 赵晨西,刘明远.膝关节骨性关节炎的治疗进展[J].湖南中医杂志,2018, 34(11):197-200. [12] SCIACCHITANO S, LAVRA L, MORGANTE A, et al. Galectin-3: One Molecule for an Alphabet of Diseases, from A to Z. Int J Mol Sci. 2018;19(2):379. [13] SACCON F, GATTO M, GHIRARDELLO A, et al. Role of galectin-3 in autoimmune and non-autoimmune nephropathies. Autoimmun Rev. 2017;16(1):34-47. [14] SREJOVIC I, SELAKOVIC D, JOVICIC N, et al. Galectin-3: Roles in Neurodevelopment, Neuroinflammation, and Behavior. Biomolecules. 2020;10(5):798. [15] WEINMANN D, SCHLANGEN K, ANDRé S, et al. Galectin-3 Induces a Pro-degradative/inflammatory Gene Signature in Human Chondrocytes, Teaming Up with Galectin-1 in Osteoarthritis Pathogenesis. Sci Rep. 2016;6:39112. [16] HU Y, YéLéHé-OKOUMA M, EA HK, et al. Galectin-3: A key player in arthritis. Joint Bone Spine. 2017;84(1):15-20. [17] ELIAZ I, RAZ A. Pleiotropic Effects of Modified Citrus Pectin. Nutrients. 2019;11(11): 2619. [18] MERHEB R, ABDEL-MASSIH RM, KARAM MC. Immunomodulatory effect of natural and modified Citrus pectin on cytokine levels in the spleen of BALB/c mice. Int J Biol Macromol. 2019;121:1-5. [19] ABU-ELSAAD NM, ELKASHEF WF. Modified citrus pectin stops progression of liver fibrosis by inhibiting galectin-3 and inducing apoptosis of stellate cells. Can J Physiol Pharmacol. 2016;94(5):554-562. [20] LU Y, ZHANG M, ZHAO P, et al. Modified citrus pectin inhibits galectin-3 function to reduce atherosclerotic lesions in apoE-deficient mice. Mol Med Rep. 2017; 16(1):647-653. [21] LI HY, YANG S, LI J C, et al. Galectin 3 inhibition attenuates renal injury progression in cisplatin-induced nephrotoxicity. Biosci Rep. 2018;38(6):BSR20181803. [22] TIAN Y, LV W, LU C, et al. Galectin-3 inhibition attenuates doxorubicin-induced cardiac dysfunction by upregulating the expression of peroxiredoxin-4. Can J Physiol Pharmacol. 2020;98(10):700-707. [23] 魏子淏,杨伟,刘夫国,等.改性柑橘果胶研究进展[J].中国食品添加剂, 2014(3):194-200. [24] SYNYTSYA A, POUČKOVá P, ZADINOVá M, et al. Hydrogels based on low-methoxyl amidated citrus pectin and flaxseed gum formulated with tripeptide glycyl-l-histidyl-l-lysine improve the healing of experimental cutting wounds in rats. Int J Biol Macromol. 2020;165:3156-3168. [25] YIN Q, CHEN J, MA S, et al. Pharmacological Inhibition of Galectin-3 Ameliorates Diabetes-Associated Cognitive Impairment, Oxidative Stress and Neuroinflammation in vivo and in vitro. J Inflamm Res. 2020;13:533-542. [26] CAO J, YANG J, WANG Z, et al. Modified citrus pectins by UV/HO oxidation at acidic and basic conditions: Structures and in vitro anti-inflammatory, anti-proliferative activities. Carbohydr Polym. 2020;247:116742. [27] RAHMANI DEL BAKHSHAYESH A, BABAIE S, TAYEFI NASRABADI H, et al. An overview of various treatment strategies, especially tissue engineering for damaged articular cartilage. Artif Cells Nanomed Biotechnol. 2020;48(1):1089-1104. [28] KWON H, PASCHOS NK, HU JC, et al. Articular cartilage tissue engineering: the role of signaling molecules . Cell Mol Life Sci. 2016;73(6):1173-1194. [29] SZWEDOWSKI D, SZCZEPANEK J, PACZESNY Ł, et al. Genetics in Cartilage Lesions: Basic Science and Therapy Approaches. Int J Mol Sci. 2020;21(15):5430. [30] YU Y, WANG S, ZHOU Z. Cartilage Homeostasis Affects Femoral Head Necrosis Induced by Methylprednisolone in Broilers. Int J Mol Sci. 2020;21(14):4841. [31] GUILAK F, NIMS RJ, DICKS A, et al. Osteoarthritis as a disease of the cartilage pericellular matrix. Matrix Biol. 2018;71-72:40-50. [32] LIU C, WANG B, XIAO L, et al. Protective effects of the pericellular matrix of chondrocyte on articular cartilage against the development of osteoarthritis. Histol Histopathol. 2018;33(8):757-764. [33] DANALACHE M, ERLER AL, WOLFGART JM, et al. Biochemical changes of the pericellular matrix and spatial chondrocyte organization-Two highly interconnected hallmarks of osteoarthritis. J Orthop Res. 2020;38(10):2170-2180. [34] GENCOGLU H, ORHAN C, SAHIN E, et al. Undenatured Type II Collagen (UC-II) in Joint Health and Disease: A Review on the Current Knowledge of Companion Animals. Animals (Basel). 2020;10(4):697. [35] FISCHENICH KM, WAHLQUIST JA, WILMOTH RL, et al. Human articular cartilage is orthotropic where microstructure, micromechanics, and chemistry vary with depth and split-line orientation. Osteoarthritis Cartilage. 2020;28(10):1362-1372. [36] LUO L, CHU J, ESWARAMOORTHY R, et al. Engineering Tissues That Mimic the Zonal Nature of Articular Cartilage Using Decellularized Cartilage Explants Seeded with Adult Stem Cells.ACS Biomater Sci Eng. 2017;3(9):1933-1943. [37] LEFEBVRE V, ANGELOZZI M, HASEEB A. SOX9 in cartilage development and disease. Curr Opin Cell Biol. 2019;61:39-47. [38] DAI J, ZHANG Y, CHEN D, et al. Glabridin inhibits osteoarthritis development by protecting chondrocytes against oxidative stress, apoptosis and promoting mTOR mediated autophagy. Life Sci. 2021:118992. [39] 张煜珅,张普华,刘鑫成,等.丹参酮ⅡA对软骨细胞去分化影响的研究[J].中国骨与关节损伤杂志,2017,32(9):942-945. [40] MEHANA EE, KHAFAGA AF, EL-BLEHI SS. The role of matrix metalloproteinases in osteoarthritis pathogenesis: An updated review. Life Sci. 2019;234:116786. [41] LI H, WANG D, YUAN Y, et al. New insights on the MMP-13 regulatory network in the pathogenesis of early osteoarthritis. Arthritis Res Ther. 2017;19(1):248. [42] XU G, ZHANG C, YANG H, et al. Modified citrus pectin ameliorates myocardial fibrosis and inflammation via suppressing galectin-3 and TLR4/MyD88/NF-κB signaling pathway. Biomed Pharmacother. 2020;126:110071. [43] GOLDRING MB, MARCU KB. Cartilage homeostasis in health and rheumatic diseases . Arthritis Res Ther. 2009;11(3):224. [44] ZHANG L, MA S, SU H, et al. Isoliquiritigenin Inhibits IL-1β-Induced Production of Matrix Metalloproteinase in Articular Chondrocytes. Mol Ther Methods Clin Dev. 2018;9:153-159. [45] HE YK, CEN XT, LIU SS, et al. Protective effects of ten oligostilbenes from Paeonia suffruticosa seeds on interleukin-1β-induced rabbit osteoarthritis chondrocytes. BMC Chem. 2019;13(1):72. [46] YI H, ZHANG W, CUI ZM, et al. Resveratrol alleviates the interleukin-1β-induced chondrocytes injury through the NF-κB signaling pathway. J Orthop Surg Res. 2020;15(1):424. [47] ZHENG W, TAO Z, CAI L, et al. Chrysin Attenuates IL-1β-Induced Expression of Inflammatory Mediators by Suppressing NF-κB in Human Osteoarthritis Chondrocytes. Inflammation. 2017;40(4):1143-1154. [48] SHI S, MAN Z, LI W, et al. Silencing of Wnt5a prevents interleukin-1β-induced collagen type II degradation in rat chondrocytes. Exp Ther Med. 2016;12(5): 3161-3166. [49] ZENG YF, WANG R, BIAN Y, et al. Catalpol Attenuates IL-1β Induced Matrix Catabolism, Apoptosis and Inflammation in Rat Chondrocytes and Inhibits Cartilage Degeneration. Med Sci Monit. 2019;25:6649-6659. [50] GAO GC, CHENG XG, WEI QQ, et al. Long noncoding RNA MALAT-1 inhibits apoptosis and matrix metabolism disorder in interleukin-1β-induced inflammation in articular chondrocytes via the JNK signaling pathway. J Cell Biochem. 2019;120(10):17167-17179. [51] MA C, ZHOU X, XU K, et al. Specnuezhenide Decreases Interleukin-1β-Induced Inflammation in Rat Chondrocytes and Reduces Joint Destruction in Osteoarthritic Rats. Front Pharmacol. 2018;9:700. |
| [1] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [2] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [3] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [4] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [5] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [6] | Xie Chongxin, Zhang Lei. Comparison of knee degeneration after anterior cruciate ligament reconstruction with or without remnant preservation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 735-740. |
| [7] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [8] | Li Yonghua, Feng Qiang, Tan Renting, Huang Shifu, Qiu Jinlong, Yin Heng. Molecular mechanism of Eucommia ulmoides active ingredients treating synovitis of knee osteoarthritis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 765-771. |
| [9] | Song Shan, Hu Fangyuan, Qiao Jun, Wang Jia, Zhang Shengxiao, Li Xiaofeng. An insight into biomarkers of osteoarthritis synovium based on bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 785-790. |
| [10] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [11] | Fang Xin, Guo Jinming, Liu Pinduan. Different concentrations of platelet rich plasma in the repair of cartilage defects in rabbits with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5588-5593. |
| [12] | Fan Shuai, Lin Jiebin, Wu Chunfei, Xu Zhaohui. Effect of serum containing Bushen Tiaogan prescription on interleukin-1beta-induced chondrocyte apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5594-5598. |
| [13] | Sun Mingshuai, Fan Chongshan, Li Kaijie, Gao Ruiyong, Wang Yudong, Shen Ruixue, Wang Lihe. Role of chondrocyte autophagy in osteoarthritis and its targeted therapy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5688-5693. |
| [14] | Wang Weikang, Liu Xiaodong, Zhou Changlin, Tian Jun. Regulatory role of microRNAs in the occurrence and development of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5709-5715. |
| [15] | Qiao Renqiu, Yin Li, Zhang Yi, Wang Haitao, Xia Peige, Kong Zhiheng. Medium- and long-term assessment of lateral compartment and patellofemoral compartment osteoarthritis after medial unicompartmental knee arthroplasty with Oxford mobile-bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(33): 5318-5323. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||