Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (7): 1138-1143.doi: 10.3969/j.issn.2095-4344.1846
Previous Articles Next Articles
Pathogenesis of inflammatory bowel disease and mesenchymal stem cell therapy: therapeutic application and existing problems
Huang Wenwen1, 2, Li Shuo1, 2, Hou Zongliu1, 2, Wang Wenju1, 2
- the Yunnan Provincial Science and Technology Project, No. 2018IA045
-
Received:2019-04-09Revised:2019-04-18Accepted:2019-06-12Online:2020-03-08Published:2020-01-20 -
Contact:Wang Wenju, Professor, Yan’an Hospital affiliated to Kunming Medical University, Kunming 650051, Yunnan Province, China; Key Laboratory of Tumor Immunological Prevention and Treatment of Yunnan Province, Kunming 650051, Yunnan Province, China -
About author:Huang Wenwen, Master, Yan’an Hospital affiliated to Kunming Medical University, Kunming 650051, Yunnan Province, China -
Supported by:the Yunnan Provincial Science and Technology Project, No. 2018IA045
CLC Number:
Cite this article
Huang Wenwen, Li Shuo, Hou Zongliu, Wang Wenju. Pathogenesis of inflammatory bowel disease and mesenchymal stem cell therapy: therapeutic application and existing problems[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1138-1143.
share this article
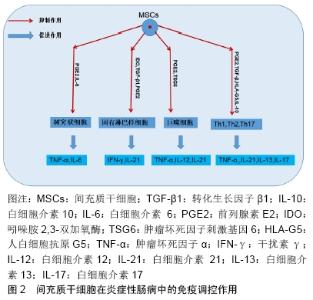
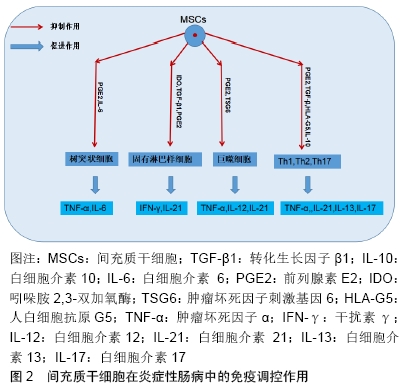
2.1 炎症性肠病的发病机制概述 炎症性肠病是一类以永久性特发性为主要特征的消化道慢性炎症性疾病[6-7],可影响结肠黏膜或者整个胃肠道。该病的临床症状主要表现为肠炎、腹泻、出血复发,甚至可能进一步造成残疾[8-9],此外炎症性肠病具有发展成为癌症的潜在风险,主要包括结直肠癌和淋巴瘤[10]。炎症性肠病在西方国家更为普遍,140万美国人和220万欧洲人深受其害[11]。近年来,在亚洲国家,其患病率与发病率正在迅速增加[12]。目前大多数的治疗策略都主要针对于抑制炎症性肠病的临床症状,深入理解炎症性肠病的发病机制能帮助研究者找到治疗炎症性肠病的最佳方法。 2.1.1 遗传易感性 炎症性肠病的病因尚不清楚,但其具有很强的遗传倾向,尤其是患者的一级亲属有更高发生炎症性肠病的危险,相较于异卵双生子,同卵双生子的患病率也较高[13-14]。FARMER等[15]在美国克利夫兰诊所进行的一项研究指出,对于克罗恩病,同卵双生子的发病率为35%,而异卵双生子的患病率仅为7%;对于溃疡性结肠炎,同卵双生子发病率为11%,异卵双生子的发病率为3%。因此,遗传病学研究得出了一致结论:遗传因素在炎症性肠病的发生发展中起着重要但非决定性作用,且与溃疡性结肠炎相比,在克罗恩病中的影响更大。 2.1.2 环境因素 毫无疑问,环境在各个疾病的发生中都起着十分重要的作用,炎症性肠病也不例外。当前,移民国家以及正在经历快速西化的国家中炎症性肠病呈高流行趋势,凸显出环境因素在该疾病发生过程中的重要性[16-17],包括吸烟、药物、饮食、精神压力等在内的大量因素都被认为是导致炎症性肠病发生的原因。吸烟仍是被研究得最为透彻的相关因素,吸烟会增加克罗恩病的风险,并且与术后疾病的复发率相关[18]。一项以小鼠为研究对象的研究指出,维生素D缺乏与结肠炎易感性增加显著相关,1,25(OH)2D3补充剂的使用改善了肠道炎症的严重程度[19]。最近的生态学和流行病学证据表明,空气污染亦可能提高克罗恩病和溃疡性结肠炎的发病风险,发展中国家的克罗恩病和溃疡性结肠炎发病率上升与工业化的发展趋势相符合[17]。同时,BITTON等[20]也提出压力水平较低的人群发生炎症性肠病的概率也越小。 2.1.3 炎症依赖性血管生成 肠黏膜免疫系统异常反应引起的炎症反应在炎症性肠病的发病机制中起重要作用[21]。有研究指出,克罗恩病和溃疡性结肠炎黏膜的局部微血管系统经历了炎症依赖性血管生成的强烈过程。DANESE等[22]对克罗恩病和溃疡性结肠炎黏膜中的黏膜血管化、内皮整合素表达、血管生成因子及其生物活性进行了评估,得出结论:与正常黏膜相比,炎症性肠病黏膜提取物在角膜和绒毛尿囊膜测定中均能有效诱导炎症依赖性血管生成。 2.1.4 肠道菌群的失调 人类的肠道微生物由约1 150种细菌组成[23],肠道中有着许多的微生物群落,如细菌、真菌以及病毒等。多项研究结果显示,通过检查克罗恩病和溃疡性结肠炎患者肠管的发炎和非发炎部分,发现炎症性肠病患者的粪便微生物多样性显著低于健康对照组[24],炎症性肠病患者的肠道微生物群也远不及健康对照组稳定[25]。 2.2 间充质干细胞治疗炎症性肠病的机制 约150年前,Cohnheim发现远端血流中存在能够修复伤口的细胞,并证明了该细胞的起源,首次提出了骨髓中存在非造血干细胞[26],随后TAVASSOLI和CROSBY[27]将完整的骨髓碎片移植到啮齿动物的髓外部位重建造血进一步证明了非造血干细胞的存在。20世纪70年代,FRIEDENSTEIN等[28]将形成集落团体的成纤维细胞命名为基质细胞,标志着间充质干细胞的确切发现。ARNOLD CAPLAN于19世纪90年代引入间充质干细胞的术语,他定义间充质干细胞为“一系列能够通过独立和独特的谱系转变分化为多种末期表型”的基质细胞[29]。 间充质干细胞具有自我更新、组织迁移和多能性的能力,除此之外,它们还具有强大的免疫调节作用,包括抑制辅助性T细胞Th1和Th17的增殖,促进调节型T细胞(Tregs)分化,同时可产生抗纤维化和促进血管生成的营养因子,可以促使炎症缓解和组织功能修复[30]。目前,一些基础和临床前期实验证实,间充质干细胞能够安全有效地改善顽固性炎症性肠病的症状,甚至达到愈合黏膜和纠正患者肠道黏膜异常免疫的效果[31]。 2.2.1 免疫调节 炎症性肠病是一种自身免疫疾病,间充质干细胞则具有恢复炎症性肠病患者免疫稳态的潜力,它可以通过直接分泌抗炎因子或者与免疫细胞直接接触从而发挥免疫调节作用,使炎症性肠道内及其周围的促炎免疫反应转向为有益的抗炎免疫反应[32]。 间充质干细胞的免疫抑制能力并非一种天然特性,针对细胞旁分泌活性的研究表明,间充质干细胞发挥免疫调节作用需要提前受到促炎细胞因子干扰素、肿瘤坏死因子和白细胞介素1的激活[33]。在炎症环境中,间充质干细胞释放包括转化生长因子β1、白细胞介素10、白细胞介素6、前列腺素E2、白血病抑制因子、肿瘤坏死因子刺激基因6、人白细胞抗原G5、吲哚胺2,3-双加氧酶在内的一系列因子[34],从而以减轻炎症性肠病患者肠道炎症的程度,见图2。在没有炎症的环境下,间充质干细胞可分泌炎症趋化因子,将淋巴细胞趋化到炎症部位并增强T细胞免疫应答。 "

除了间充质干细胞释放的可溶性因子外,免疫调节也是通过细胞接触依赖性机制引起的。活化的T细胞和间充质干细胞之间的细胞-细胞接触可诱导间充质干细胞产生白细胞介素10,白细胞介素10又刺激可溶性人白细胞抗原G5的释放[30,35]。人白细胞抗原G5可以抑制T细胞增殖、T细胞的细胞毒性、自然杀伤细胞的功能和促进Tregs产生[36]。间充质干细胞还可通过FAS途径诱导T细胞凋亡,导致Tregs上调,最终出现免疫耐受。除此之外,间充质干细胞还分泌程序性死亡配体1,2(PD-L1,PD-L2),参与独立的免疫抑制机制[37]。WU等[38]经过最新的实验研究证实,间充质干细胞可通过释放外泌体及调节泛素修饰水平减轻炎症性肠病症状,发挥其功能。 2.2.2 血管生成 适当的血管生成是炎症性肠病患者受损肠道再生和修复的必需因素。在一项动物研究中,前列腺素缺陷小鼠的结肠黏膜中产生局灶性伤口,由于缺氧和平滑肌坏死,这些小鼠发展成为穿透性溃疡。研究人员发现刺激血管内皮生长因子依赖性血管生成的前列腺素I2,是结肠活检损伤后预防穿透性溃疡所必需的。此外,与静脉注射结肠间充质干细胞相比,黏膜注射的细胞可以通过最具代表性的间充质干细胞趋化因子受体CXCR4,强力迁移到肠道损伤部位[39]。因此,损伤后肠道修复和再生所需的适当血管生成取决于间充质干细胞分泌的血管内皮生长因子水平。除血管内皮生长因子外,已知成纤维细胞生长因子和单核细胞趋化蛋白也是间充质干细胞介导血管发生的主要细胞因子[40]。 2.2.3 肠上皮重生 炎症性肠病患者的肠道上皮黏膜屏障受到损伤,使肠腔内抗原移位至肠黏膜固有层,并激活肠黏膜免疫系统,引发肠黏膜免疫系统功能紊乱[41]。间充质干细胞可通过促进肠上皮细胞的增殖和肠干细胞的分化,刺激肠上皮修复。同时,间充质干细胞通过帮助这些细胞再生,对分泌肠黏蛋白的杯状细胞具有保护作用[42]。除此之外,间充质干细胞促进紧密连接蛋白、claudin-2,12,15的表达[43],从而降低炎症诱导的上皮通透性。所有上述间充质干细胞特性,共同阻止腔内抗原/病原体侵入肠黏膜和黏膜下层,从而避免炎症的发生和进一步发展。 2.3 间充质干细胞治疗炎症性肠病 由于免疫抑制特性和组织修复作用,间充质干细胞移植被认为是最有前景的治疗方法之一。低免疫原性主要是由于间充质干细胞低表达主要组织相容性复合物Ⅰ类分子,不表达主要组织相容性复合物Ⅱ类分子和共刺激分子。目前用于治疗炎症性肠病的间充质干细胞主要有3种类型:骨髓间充质干细胞、脂肪间充质干细胞以及脐带间充质干细胞。 骨髓间充质干细胞具有较强的增殖与分化潜能,较低的免疫原性,可以对慢性结肠炎发挥长期的保护作 用[44]。骨髓间充质干细胞研究广泛[45],但由于其含量较少,提取过程中患者存在痛苦,且随着年龄增长,骨髓间充质干细胞数目将越来越少,因此在临床上应用存在一定局限性[46]。此后,脂肪间充质干细胞的出现,克服了骨髓间充质干细胞的缺陷,脂肪间充质干细胞具有与骨髓间充质干细胞相似的免疫抑制功能[47]。由于无创和无痛采样功能,脐带间充质干细胞也逐渐成为治疗炎症性肠病的种子细胞[48]。间充质干细胞治疗炎症性肠病主要有2种手段:干细胞移植治疗肠腔炎症性肠病以及干细胞局部注射治疗炎症性肠病所致瘘管。 近年来不少研究对间充质干细胞移植治疗炎症性肠病的效果进行了评估。结果显示,与安慰剂相比,局部注射自体或同种异体间充质干细胞在多个病例和随机对照试验中均显示阳性结果,免疫调节剂、抗肿瘤坏死因子α药物和局部管理(包括手术)的传统疗法都疗效甚微[49-50]。 考虑到同种异体间充质干细胞治疗可能引发的免疫反应,一部分作者支持使用自体间充质干细胞进行研究[51]。一项为期6个月的第一阶段临床试验显示,用自体间充质干细胞移植治疗的12例克罗恩病患者,其完整临床愈合率和复杂瘘管有效治疗的放射学证据高达83%[52]。对8例患有1个或多个难治性、复杂的克罗恩病瘘管患者进行了一项不同阶段的研究,采用一次性自体骨髓间充质干细胞进行瘘管内注射治疗。8周时,8例瘘管中有6例愈合,2例残余瘘出现部分闭合,引流减少。随访12-22个月无不良反应[53]。该研究者又进一步研究了Ⅱ期试验中瘘管对骨髓间充质干细胞治疗的反应,49例患有复杂肛周瘘的患者被随机分配到2组,病灶内分别注射纤维蛋白胶或纤维蛋白胶+脂肪间充质干细胞,若8周未达到瘘管愈合则第2次治疗,改为2倍剂量的脂肪间充质干细胞。接受脂肪间充质干细胞治疗的24例患者中有17例瘘管完全消退,其中11例患者在第1次注射后愈合,6例患者需要第2次注射,而对照组25例患者中仅有4例缓解,与对照组相比,治疗组的生活质量有显著改善[54]。同时,另一项局部注射自体骨髓间充质干细胞治疗克罗恩病瘘管的研究表明,骨髓间充质干细胞在克罗恩病瘘管的治疗中表现出良好效果[55]。10例对现有治疗方法不敏感或不适合的克罗恩病瘘管患者进行了4次局部注射治疗,自体骨髓间充质干细胞总数为20×106,间隔4周,观察到所有患者直肠黏膜都有良好愈合,克罗恩病活动指数和肛周病活动指数均得到改善,并且无任何不良反应,70%的患者瘘管完全闭合。 对于常规治疗、生物治疗或两者皆疗效不好的复杂肛周瘘而言,同种异体间充质干细胞移植是一种有效且安全的治疗方式。一项Ⅲ期、随机双盲、平行、安慰剂组对照、多中心临床试验对212例顽固性、复杂、活动性肛周瘘患者进行手术,闭合瘘管内部开口,然后将单独的间充质干细胞或安慰剂注射到邻近所有瘘管和内部开口的组织中,随访24周,间充质干细胞组与安慰剂组相比具有更高的瘘闭合率,所有外部开口闭合的时间明显缩短[56]。不仅如此,SOONTARARAK等[57]最近进行的一项研究显示,诱导多能干细胞衍生的间充质干细胞也在炎症性肠病小鼠模型中展现出有效性,刺激肠道上皮细胞增殖和肠道干细胞数量增加,并增加了肠道血管的生成,可以显著改善小鼠的临床症状和肠道内的炎症反应。 2.4 间充质干细胞治疗炎症性肠病的安全性 LIANG等[58]团队报道了7例炎症性肠病患者(4例克罗恩病,3例溃疡性结肠炎)静脉输注同种异体间充质干细胞移植治疗的经验。间充质干细胞剂量1×106个/kg。移植治疗后,所有患者继续使用类固醇和/或免疫抑制剂进行常规治疗。结果显示,7例患者的临床状况逐渐改善,输注后无不良反应。同时也有报道指出,大鼠间充质干细胞在体外培养的过程可能自发产生基因突变并在移植该细胞后诱发纤维肉瘤的形成,因此间充质干细胞移植会否诱发新生肿瘤值得探讨。但到目前为止,无论是在治疗炎症性肠病患者的临床试验中,还是在其他疾病中都没有报道过主要的相关不良事件[59-61],只是一小部分研究表明在注射期间或注射后会发生轻度和短暂的发热,但并无组织异位生长的发生[62]。因此,炎症性肠病治疗中的安全性仍需更多长期随访。 "

| [1] STONE CD. The economic burden of inflammatory bowel disease: clear problem, unclear solution. Dig Dis Sci. 2012;57(12):3042-3044. [2] BARNES EL, KAPPELMAN MD. Editorial: Increasing Incidence of Pediatric Inflammatory Bowel Disease in France: Implications for Etiology, Diagnosis, Prognosis, and Treatment. Am J Gastroenterol. 2018;113(2):273-275. [3] UNGARO R, MEHANDRU S, ALLEN PB, et al. Ulcerative colitis. Lancet. 2017;389(10080):1756-1770. [4] YANG Q, JIA L, LI X, et al. Long Noncoding RNAs: New Players in the Osteogenic Differentiation of Bone Marrow- and Adipose-Derived Mesenchymal Stem Cells. Stem Cell Rev. 2018;14(3):297-308. [5] KIM N, CHO SG. Clinical applications of mesenchymal stem cells. Korean J Intern Med. 2013;28(4):387-402. [6] FARRELL D, MCCARTHY G, SAVAGE E. Self-reported Symptom Burden in Individuals with Inflammatory Bowel Disease. J Crohns Colitis. 2016;10(3):315-322. [7] KASER A, ZEISSIG S, BLUMBERG RS. Inflammatory bowel disease. Annu Rev Immunol. 2010;28:573-621. [8] NOWAKOWSKI J, CHROBAK AA, DUDEK D. Psychiatric illnesses in inflammatory bowel diseases - psychiatric comorbidity and biological underpinnings. Psychiatr Pol. 2016;50(6):1157-1166. [9] ABRAHAM BP. Symptom management in inflammatory bowel disease. Expert Rev Gastroenterol Hepatol. 2015;9(7):953-967. [10] ABRAHAM C, CHO JH. Inflammatory bowel disease. N Engl J Med. 2009;361(21):2066-2078. [11] LOFTUS EV JR. Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology. 2004;126(6):1504-1517. [12] YE Y, PANG Z, CHEN W, et al. The epidemiology and risk factors of inflammatory bowel disease. Int J Clin Exp Med. 2015;8(12): 22529-22542. [13] SILVERBERG MS, MIREA L, BULL SB, et al. A population- and family-based study of Canadian families reveals association of HLA DRB1*0103 with colonic involvement in inflammatory bowel disease. Inflamm Bowel Dis. 2003;9(1):1-9. [14] SAMADDER NJ, VALENTINE JF, GUTHERY S, et al. Family History Associates With Increased Risk of Colorectal Cancer in Patients With Inflammatory Bowel Diseases. Clin Gastroenterol Hepatol. 2018 Sep 26. doi: 10.1016/j.cgh.2018.09.038. [Epub ahead of print] [15] FARMER RG, MICHENER WM, MORTIMER EA. Studies of family history among patients with inflammatory bowel disease. Clin Gastroenterol. 1980;9(2):271-277. [16] TSIRONI E, FEAKINS RM, PROBERT CS, et al. Incidence of inflammatory bowel disease is rising and abdominal tuberculosis is falling in Bangladeshis in East London, United Kingdom. Am J Gastroenterol. 2004;99(9):1749-1755. [17] THIA KT, LOFTUS EV JR, SANDBORN WJ, et al. An update on the epidemiology of inflammatory bowel disease in Asia. Am J Gastroenterol. 2008;103(12):3167-3182. [18] BIRRENBACH T, BÖCKER U. Inflammatory bowel disease and smoking: a review of epidemiology, pathophysiology, and therapeutic implications. Inflamm Bowel Dis. 2004;10(6):848-859. [19] CANTORNA MT, MUNSICK C, BEMISS C, et al. 1,25-Dihydroxycholecalciferol prevents and ameliorates symptoms of experimental murine inflammatory bowel disease. J Nutr. 2000; 130(11):2648-2652. [20] BITTON A, DOBKIN PL, EDWARDES MD, et al. Predicting relapse in Crohn's disease: a biopsychosocial model. Gut. 2008;57(10): 1386-1392. [21] GONÇALVES P, ARAÚJO JR, DI SANTO JP. A Cross-Talk Between Microbiota-Derived Short-Chain Fatty Acids and the Host Mucosal Immune System Regulates Intestinal Homeostasis and Inflammatory Bowel Disease. Inflamm Bowel Dis. 2018;24(3):558-572. [22] DANESE S, SANS M, DE LA MOTTE C, et al. Angiogenesis as a novel component of inflammatory bowel disease pathogenesis. Gastroenterology. 2006;130(7):2060-2073. [23] QIN J, LI R, RAES J, et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature.2010;464(7285): 59-65. [24] JOOSSENS M, HUYS G, CNOCKAERT M, et al. Dysbiosis of the faecal microbiota in patients with Crohn's disease and their unaffected relatives. Gut. 2011;60(5):631-637. [25] ANDOH A, IMAEDA H, AOMATSU T, et al. Comparison of the fecal microbiota profiles between ulcerative colitis and Crohn's disease using terminal restriction fragment length polymorphism analysis. J Gastroenterol. 2011;46(4):479-486. [26] TAVASSOLI M, CROSBY WH. Transplantation of marrow to extramedullary sites. Science. 1968;161(3836):54-56. [27] TAVASSOLI M, CROSBY WH. Bone marrow histogenesis: a comparison of fatty and red marrow. Science.1970;169(3942): 291-293. [28] FRIEDENSTEIN AJ, DERIGLASOVA UF, KULAGINA NN, et al. Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp Hematol. 1974; 2(2):83-92. [29] CAPLAN AI. Mesenchymal stem cells. J Orthop Res.1991;9(5): 641-650. [30] RAHIMZADEH A, MIRAKABAD FS, MOVASSAGHPOUR A, et al. Biotechnological and biomedical applications of mesenchymal stem cells as a therapeutic system. Artif Cells Nanomed Biotechnol. 2016; 44(2):559-570. [31] RICART E. Current status of mesenchymal stem cell therapy and bone marrow transplantation in IBD. Dig Dis. 2012;30(4):387-391. [32] ENGLISH K. Mechanisms of mesenchymal stromal cell immunomodulation. Immunol Cell Biol. 2013;91(1):19-26. [33] CASTRO-MANRREZA ME, MONTESINOS JJ. Immunoregulation by mesenchymal stem cells: biological aspects and clinical applications. J Immunol Res. 2015;2015:394917. [34] KYURKCHIEV D, BOCHEV I, IVANOVA-TODOROVA E, et al. Secretion of immunoregulatory cytokines by mesenchymal stem cells. World J Stem Cells. 2014;6(5):552-570. [35] PREVOSTO C, ZANCOLLI M, CANEVALI P, et al. Generation of CD4+ or CD8+ regulatory T cells upon mesenchymal stem cell-lymphocyte interaction. Haematologica. 2007;92(7):881-888. [36] SELMANI Z, NAJI A, ZIDI I, et al. Human leukocyte antigen-G5 secretion by human mesenchymal stem cells is required to suppress T lymphocyte and natural killer function and to induce CD4+CD25highFOXP3+ regulatory T cells. Stem Cells. 2008;26(1): 212-222. [37] DAVIES LC, HELDRING N, KADRI N, et al. Mesenchymal Stromal Cell Secretion of Programmed Death-1 Ligands Regulates T Cell Mediated Immunosuppression. Stem Cells. 2017;35(3):766-776. [38] WU Y, QIU W, XU X, et al. Exosomes derived from human umbilical cord mesenchymal stem cells alleviate inflammatory bowel disease in mice through ubiquitination. Am J Transl Res. 2018;10(7):2026-2036. [39] MANIERI NA, MACK MR, HIMMELRICH MD, et al. Mucosally transplanted mesenchymal stem cells stimulate intestinal healing by promoting angiogenesis. J Clin Invest. 2015;125(9):3606-3618. [40] HUNG SC, POCHAMPALLY RR, CHEN SC, et al. Angiogenic effects of human multipotent stromal cell conditioned medium activate the PI3K-Akt pathway in hypoxic endothelial cells to inhibit apoptosis, increase survival, and stimulate angiogenesis. Stem Cells. 2007; 25(9):2363-2370. [41] ANDO Y, INABA M, SAKAGUCHI Y, et al. Subcutaneous adipose tissue-derived stem cells facilitate colonic mucosal recovery from 2,4,6-trinitrobenzene sulfonic acid (TNBS)-induced colitis in rats. Inflamm Bowel Dis. 2008;14(6):826-838. [42] FAWZY SA, EL-DIN ABO-ELNOU RK, ABD-EL-MAKSOUD EL-DEEB DF, et al. The possible role of mesenchymal stem cells therapy in the repair of experimentally induced colitis in male albino rats. Int J Stem Cells. 2013;6(2):92-103. [43] YABANA T, ARIMURA Y, TANAKA H, et al. Enhancing epithelial engraftment of rat mesenchymal stem cells restores epithelial barrier integrity. J Pathol. 2009;218(3):350-359. [44] LEE HJ, OH SH, JANG HW, et al. Long-Term Effects of Bone Marrow-Derived Mesenchymal Stem Cells in Dextran Sulfate Sodium-Induced Murine Chronic Colitis. Gut Liver. 2016;10(3): 412-419. [45] NEOFYTOU E, DEUSE T, BEYGUI RE, et al. Mesenchymal stromal cell therapy: different sources exhibit different immunobiological properties. Transplantation. 2015;99(6):1113-1118. [46] CICCOCIOPPO R, CANGEMI GC, KRUZLIAK P, et al. Concise Review: Cellular Therapies: The Potential to Regenerate and Restore Tolerance in Immune-Mediated Intestinal Diseases. Stem Cells. 2016; 34(6):1474-1486. [47] BASSI G, PACELLI L, CARUSONE R, et al. Adipose-derived stromal cells (ASCs). Transfus Apher Sci. 2012;47(2):193-198. [48] WANG M, CAI J, HUANG A, et al. Pre-treatment of human umbilical cord-derived mesenchymal stem cells with interleukin-6 abolishes their growth-promoting effect on gastric cancer cells. Int J Mol Med. 2015; 35(2):367-375. [49] HAWKEY CJ, ALLEZ M, CLARK MM, et al. Autologous Hematopoetic Stem Cell Transplantation for Refractory Crohn Disease: A Randomized Clinical Trial. JAMA. 2015;314(23):2524-2534. [50] LINDSAY JO, ALLEZ M, CLARK M, et al. Autologous stem-cell transplantation in treatment-refractory Crohn's disease: an analysis of pooled data from the ASTIC trial. Lancet Gastroenterol Hepatol. 2017; 2(6):399-406. [51] SERENA C, KEIRAN N, MADEIRA A, et al. Crohn's Disease Disturbs the Immune Properties of Human Adipose-Derived Stem Cells Related to Inflammasome Activation. Stem Cell Reports. 2017;9(4):1109-1123. [52] DIETZ AB, DOZOIS EJ, FLETCHER JG, et al. Autologous Mesenchymal Stem Cells, Applied in a Bioabsorbable Matrix, for Treatment of Perianal Fistulas in Patients With Crohn's Disease. Gastroenterology. 2017;153(1):59-62.e2. [53] GARCÍA-OLMO D, GARCÍA-ARRANZ M, HERREROS D, et al. A phase I clinical trial of the treatment of Crohn's fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum. 2005;48(7): 1416-1423. [54] GARCIA-OLMO D, HERREROS D, PASCUAL I, et al. Expanded adipose-derived stem cells for the treatment of complex perianal fistula: a phase II clinical trial. Dis Colon Rectum. 2009;52(1):79-86. [55] CICCOCIOPPO R, BERNARDO ME, SGARELLA A, et al. Autologous bone marrow-derived mesenchymal stromal cells in the treatment of fistulising Crohn's disease. Gut.2011;60(6):788-798. [56] PANÉS J, GARCÍA-OLMO D, VAN ASSCHE G, et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn's disease: a phase 3 randomised, double-blind controlled trial. Lancet. 2016;388(10051):1281-1290. [57] SOONTARARAK S, CHOW L, JOHNSON V, et al. Mesenchymal Stem Cells (MSC) Derived from Induced Pluripotent Stem Cells (iPSC) Equivalent to Adipose-Derived MSC in Promoting Intestinal Healing and Microbiome Normalization in Mouse Inflammatory Bowel Disease Model. Stem Cells Transl Med. 2018;7(6):456-467. [58] LIANG J, ZHANG H, WANG D, et al. Allogeneic mesenchymal stem cell transplantation in seven patients with refractory inflammatory bowel disease. Gut. 2012;61(3):468-469. [59] WAKITANI S, OKABE T, HORIBE S, et al. Safety of autologous bone marrow-derived mesenchymal stem cell transplantation for cartilage repair in 41 patients with 45 joints followed for up to 11 years and 5 months. J Tissue Eng Regen Med. 2011;5(2):146-150. [60] TAN J, WU W, XU X, et al. Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. JAMA. 2012;307(11):1169-1177. [61] LE BLANC K, FRASSONI F, BALL L, et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet. 2008;371(9624):1579-1586. [62] LALU MM, MCINTYRE L, PUGLIESE C, et al. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic review and meta-analysis of clinical trials. PLoS One. 2012;7(10):e47559. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [4] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [5] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [6] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [7] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [8] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [9] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [10] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [11] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [12] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [13] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [14] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [15] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||