Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (19): 2972-2979.doi: 10.3969/j.issn.2095-4344.1242
Previous Articles Next Articles
Influence of joint distraction height on cartilage repair in rabbit models of knee oteoarthritis
Shi Huanan, Zhang Yonghong, Peng Ruijian, Li Xiaohui, Guo Xiujuan
- (Department of Orthopedics, the Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China)
-
Received:2019-01-30Online:2019-07-08Published:2019-07-08 -
Contact:Zhang Yonghong, MD, Chief physician, Doctoral supervisor, Department of Orthopedics, the Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
About author:Shi Huanan, Master candidate, Department of Orthopedics, the Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
Supported by:the Research Project of Shanxi Provincial Overseas Scholarship, No. 2015-104
CLC Number:
Cite this article
Shi Huanan, Zhang Yonghong, Peng Ruijian, Li Xiaohui, Guo Xiujuan. Influence of joint distraction height on cartilage repair in rabbit models of knee oteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(19): 2972-2979.
share this article
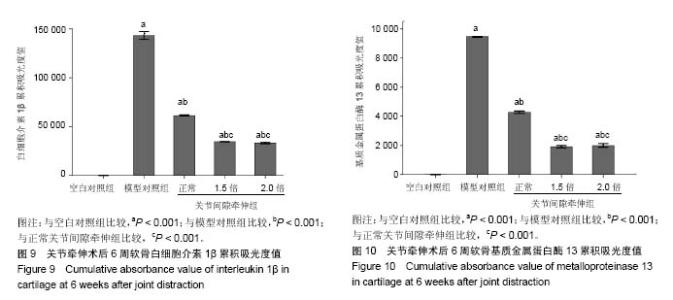
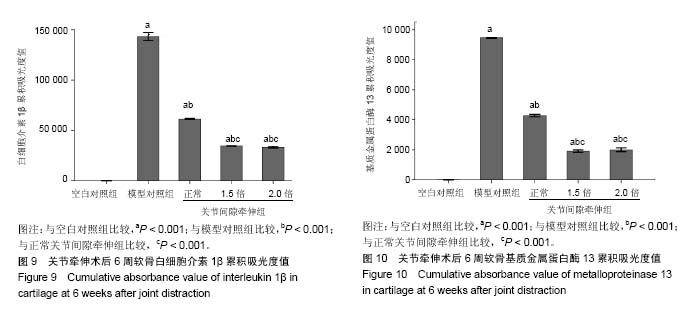
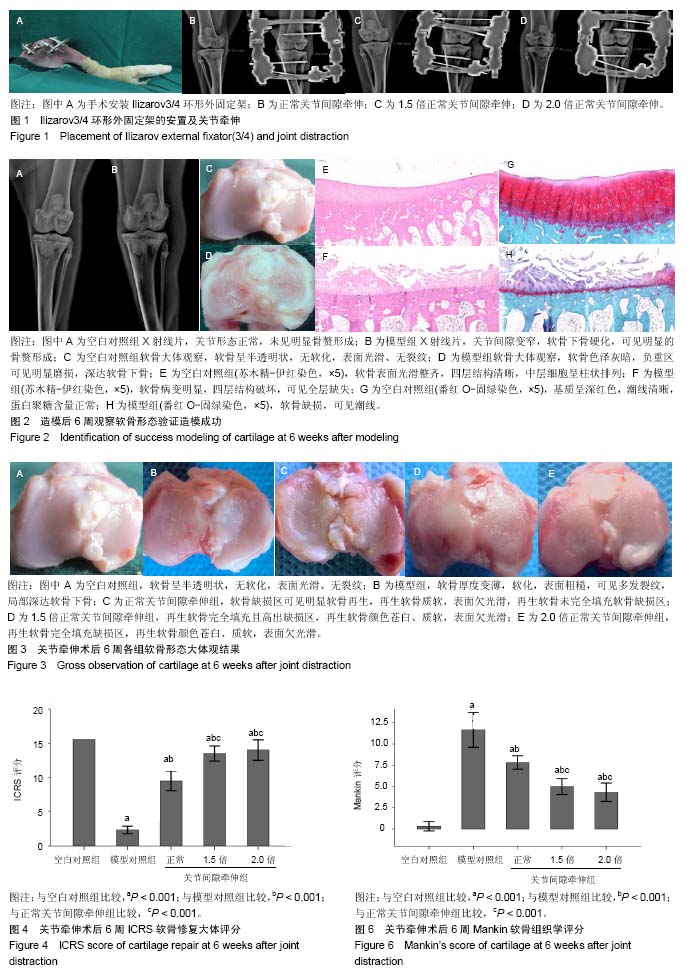
2.1 实验动物数量分析 新西兰大白兔2只用于验证造模,其余均进入结果分析,无脱落。 2.2 膝关节软骨形态验证造模情况 左膝关节X射线:可见空白对照组关节形态正常,未见明显骨赘形成(图2A);模型组关节间隙变窄,软骨下骨硬化,可见明显的骨赘形成(图2B)。大体观:可见空白对照组软骨呈半透明状,无软化,表面光滑、无裂纹,关节液清亮,滑膜未见明显增生(图2C);模型组软骨色泽灰暗,负重区可见明显磨损,深达软骨下骨,滑膜增生明显(图2D)。苏木精-伊红染色:可见空白对照组软骨表面光滑整齐,四层结构清晰,中层细胞呈柱状排列(图2E);模型组软骨病变明显,四层结构破坏,可见全层缺失(图2F)。番红O-固绿染色:可见空白对照组软骨基质呈深红色,潮线清晰,蛋白聚糖含量正常(图2G);模型组软骨缺损,潮线可见(图2H)。 2.3 关节牵伸后6周各组软骨形态大体观结果 空白对照组软骨呈半透明状,无软化,表面光滑、无裂纹,关节液清亮,滑膜未见明显增生(图3A);模型对照组软骨厚度变薄,软化,表面粗糙,可见多发裂纹,局部深达软骨下骨,滑膜增生明显(图3B);正常关节间隙牵伸组软骨缺损区可见软骨再生,再生软骨质软,表面欠光滑,再生软骨未完全填充软骨缺损区(图3C);1.5倍正常关节间隙牵伸组再生软骨完全填充且高出缺损区,再生软骨颜色苍白、质软,表面欠光滑(图3D);2.0倍正常关节间隙牵伸组再生软骨完全填充缺损区,再生软骨颜色苍白、质软,表面欠光滑(图3E)。2.0倍正常关节间隙牵伸组软骨修复ICRS评分明显高于模型对照组(P < 0.001),3个实验组评分均低于空白对照组(P < 0.01),见图4。正常关节间隙牵伸组分值显著低于1.5倍和2.0倍正常关节间隙牵伸组(P < 0.001),1.5倍和2.0倍正常关节间隙牵伸组无明显差异(P > 0.05)。 2.4 关节牵伸后6周各组软骨苏木精-伊红染色、番红O-固绿染色显微镜观察结果 空白对照组软骨表面光滑整齐,四层结构清晰,中层细胞呈柱状排列,番红O-固绿染色显示糖胺多糖均匀表达(图5A);模型对照组可见明显软骨缺损区,番红O-固绿染色显示糖胺多糖不表达(图5B);正常关节间隙牵伸组软骨缺损区再生软骨无明显四层结构,以浅层纤维组织为主,深层为较薄的透明软骨,可见成团分布的同源细胞群,番红O-固绿染色显示浅层糖胺多糖不表达,深层可见其表达(图5C);1.5倍正常关节间隙牵伸组软骨表面欠光滑,可见软骨组织的四层结构,各层软骨细胞排列不整齐,可见软骨裂隙,深层可见多个成团分布的同源细胞群,番红O-固绿染色显示糖胺多糖在软骨表层以下表达(图5D),但不均匀;2.0倍正常关节间隙牵伸组软骨表面欠光滑,可见软骨组织的四层结构,各层软骨细胞排列不整齐,深层可见多个成团分布的同源细胞群,番红O-固绿染色显示糖胺多糖在软骨全层表达,欠均匀,以深层为主,表层含量低(图5E)。 各牵伸组Mankin骨关节病组织评分明显低于模型组(P < 0.001),显著高于空白对照组(P < 0.001),其中正常关节间隙牵伸组分值显著高于1.5倍和2.0倍正常关节间隙牵伸组(P < 0.001),1.5倍和2.0倍正常关节间隙牵伸组间无明显差异(P > 0.05),见图6。 2.5 关节牵伸后6周各组白细胞介素1β、基质金属蛋白酶13在软骨组织的表达 光镜下可见各组白细胞介素1β、基质金属蛋白酶13的表达情况:空白对照组仅见软骨表层极弱表达;模型对照组软骨全层基质、软骨细胞胞浆高表达,呈深棕色,阳性结果较空白对照组显著增强;正常关节间隙牵伸组再生软骨全层表达,以表、中层显著;1.5倍正常关节间隙牵伸组再生软骨以表层表达为主,中层可见散在表达;2.0倍正常关节间隙牵伸组软骨表层仍可见表达,阳性结果高于空白对照组。见图7,8。 经Image Pro Plus 6.0图像分析软件分析累积吸光度值,白细胞介素1β、基质金属蛋白酶13累积吸光度值越大,则阳性结果越高。各牵伸组累积吸光度值明显低于模型对照组(P < 0.001),正常关节间隙牵伸组吸光度值显著高于1.5倍和2.0倍正常关节间隙牵伸组(P < 0.001),1.5倍和2.0倍正常关节间隙牵伸组间无明显差异(P > 0.05)。各组白细胞介素1β累积吸光度值见图9,基质金属蛋白酶13累积吸光度值见图10。"
| [1] 夏玲,王欢,窦晓莉,等.膝骨性关节炎的非手术治疗现状[J].中国老年学杂志,2011,4(31):1491-1494.[2] Hootman JM, Helmick CG. Projections of US prevalence of arthritis and associated activity limitations. Arthritis Rheum. 2006;54:226-229. [3] Kurtz SM, Lau E, Ong K, et al. Future young patient demand for primary and revision joint replacement:national projections from 2010 to 2030. Clin Orthop Relat Res.2009;467: 2606-2612. [4] Ethgen O, Bruyère O, Richy F, et al. Health-relatedquality of life in totalhip and total knee arthroplasty.A qualitative and systematic review of the literature. J Bone Joint Surg Am. 2004;86-A(5):963-974. [5] Deirmengian CA, Lonner JH. What's new in adult reconstructive knee surgery. J Bone Joint Surg Am.2012; 94(2):182-188. [6] Mastbergen SC, Saris DB, Lafeber FPJG. Functional articular cartilage repair: here,near,or is the best approach not yet clear? Nat Rev Rheumatol.2013;9(5):277-290. [7] van Valburg AA, van Roy H, Lafeber FP, et al. Beneficial effects of intermittent fluid pressure of low physiological magnitude on cartilage and inflammation in osteoarthritis. An in vitro study. J Rheumatol.1998;25(3):515-520. [8] Judet R, Judet T. The use of a hinge distraction apparatus after arthrolysis and arthroplasty. Rev Chir Orthop Reparatrice Appar Mot.1978,64(5):353-365.[9] Intema F, van Roermund PM, Marijnissen AC, et al. Tissue structure modification in knee osteoarthritis by use of joint distraction:an open 1-year pilot study. Ann Rheum Dis. 2011;70:1441-1446.[10] Wiegant K, van Roermund PM, Intema F, et al. Sustained clinical and structural benefit after joint distraction in the treatment of severe knee osteos rthritis.Osteoarthr Cartil. 2013;21:1660-1667.[11] Kuchuk NO, Wiegant K, van der Woude JAD, et al. Clinical benefit and cartilaginous tissue repair after knee joint distraction:5 years follow-up. Ann Rheum Dis.2015;74:359 [12] Chen Y, Sun Y, Pan X, et al. Joint distraction attenuates osteoarthritis by reducing secondary inflammation, cartilage degeneration and subchondral bone aberrant change. Osteoarthritis Cartilage.2015;23(10):1728-1735.[13] Nishino T, Ishii T, Chang F, et al. Effect of gradual weight-bearing on regenerated articular cartilage after joint distraction and motion in a rabbit model. J Orthop Res. 2010;28(5):600-606.[14] Benito MJ, Veale DJ, FitzGerald O, et al. Synovial tissue inflammation in early and late osteoarthritis. Ann Rheum Dis.2005;64(9):1263-1267. [15] Wayne JS, Mcdowell CL, Shields KJ, et al. In vivo response of polylactic acid-alginate scaffolds and bone marrow-derived cells for cartilage tissue engineering. Tissue Engineering. 2005;11(5-6):953-963. [16] Mankin HJ, Johnson ME, Lippiello L. Biochemical and metabolic abnormalities in articular cartilage from osteoarthritic humanhips. J bone Joint Surg(Am).1971; 53(3):523.[17] Ding L, Heying E, Nicholson N, et al. Mechanical impact induces cartilage degradation via mitogen activated protein kinases. Osteoarthritis Cartilage.2010;18:1509-1517.[18] Wei L, Fleming BC, Sun X, et al. Comparison of differential biomarkers of osteoa rthritis with and without posttraumatic injury in the Hartley guinea pig model. J Orthop Res.2010; 28:900-906.[19] Tochigi Y, Buckwalter JA, Martin JA, et al. Distribution and progression of chondrocyte damage in a whole-organ model of human ankle intraarticular fracture. J Bone Joint Surg Am. 2011;93:533-539.[20] van Roermund PM, Marijnissen AC, Lafeber FP. Joint distraction as an alternative for the treatment of osteoarthritis. Foot Ankle Clin.2002;7:515-527.[21] van Valburg AA, van Roermund PM, Lammens J, et al. Can Ilizarov joint distraction delay the need for an arthrodesis of the ankle? A preliminary report. J Bone Joint Surg Br.1995; 77(5):720-725.[22] Lafeber F, Veldhuijzen JP, Vanroy JL, et al. Huber-Bruning O,Bijlsma JW.Intermittent hydrostatic compressive force stimulates exclusively the proteoglycan synthesis of osteoarthritic human cartilage. Br J Rheumatol.1992; 31:437-442.[23] van Valburg AA, van Roermund PM, Marijnissen AC, et al. Joint distraction in treatment of osteoarthritis (II): effects on cartilage in a canine model. Osteoarthritis Cartilage.2000; 8(1):1-8.[24] Havdrup T, Telhag H. Papain-induced changes in the knee joints of adult rabbits. Acta Orthop Scand.1977;48(2): 143-149.[25] Karadam B, Karatosun V, Murat N, et al. No beneficial effects of jointdistraction on early microscopical changes in osteoarthritic knees. A study in rabbits. Acta Orthop.2005; 76(1):95-98.[26] Blom AB, van der Kraan PM, van den Berg WB. Cytokine targeting in osteoarthritis. Curr Drug Targets.2007;8:283-292. [27] Dahlberg L, Billinghurst RC, Manner P, et al. Selective enhancement of collagenase-mediated cleavage of resident type II collagen in cultured osteoarthritic cartilage and arrest with a synthetic inhibitor that spares collagenase 1 (matrix metalloproteinase.Arthritis Rheum.2000;43(3):673-682.[28] Shlopov BV, Gumanovskaya ML, Hasty KA. Autocrine regulation of collagenase 3(matrix metalloproteinase 13) during osteoarthritis. Arthritis Rheum.2000; 43(1):195-205.[29] Kobayashi M, Squires GR, Mousa A, et al. Role of interleukin-1 and tumor necrosis factor alpha in matrix degradation of human osteoarthritic cartilage. Arthritis Rheum.2005;52(1):128-135.[30] Elsaid KA, Jay GD, Chichester CO. Reduced expression and proteolytic susceptibility of lubricin/superficial zone protein may explain early elevation in the coefficient of friction in the joints of rats with antigen-induced arthritis. Arthritis Rheum. 2007;56:108-116. |
| [1] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [2] | Li Yonghua, Feng Qiang, Tan Renting, Huang Shifu, Qiu Jinlong, Yin Heng. Molecular mechanism of Eucommia ulmoides active ingredients treating synovitis of knee osteoarthritis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 765-771. |
| [3] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [4] | Yang Wei, Chen Zehua, Yi Zhiyong, Huang Xudong, Han Qingmin, Zhang Ronghua. Effectiveness of intra-articular injection of hyaluronic acid versus placebo in the treatment of early and mid-stage knee osteoarthritis: a Meta-analysis based on randomized, double-blind, controlled, clinical trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3760-3766. |
| [5] | Liu Jinwei, Chen Yunzhen, Wan Chunyou. Changes of osteogenic growth factors in the broken end of bone nonunion under stress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3619-3624. |
| [6] | Zuo Xiuqin, Yin Sasa, Xie Huimin, Jia Zishan, Zhang Lining. Applicability and specifications of platelet-rich plasma in musculoskeletal repair [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3239-3245. |
| [7] | Yang Tengyun, Li Yanlin, Liu Dejian, Wang Guoliang. Application prospects and problems of peripheral blood derived mesenchymal stem cells in cartilage repair of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3071-3076. |
| [8] | Yin Yuhui, Zhan Jiawen, Wang Shangquan, Shao Chenchen, Zhou Liang. Meta-analysis of acupuncture combined with massage in the treatment of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2719-2726. |
| [9] | Zhu Shiqiang, Xu Jianfeng, Hei Xiaoyan, Chen Yundong, Tian Xinbao, Zhang Jinchen, Lin Ruizhu. Effect of internal heat-type acupuncture needle therapy on the expression of type I collagen, matrix metalloproteinase-3 and osteopontin in the subchondral bone of rabbit knee osteoarthritis model [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2636-2642. |
| [10] | Meng Maohua, Li Ying, Chen Xin, Cheng Lu, Dong Qiang. Effects and mechanisms of enamel matrix derivatives on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2108-2113. |
| [11] |
Xie Fei, Li Yanle, Lin Xinxiao, Hu Haiwei, Sang Zhicheng, Sun Yongsheng, Jiang Kewei, Cheng Ying, Wen Guannan, Wen Jianmin, Sun Weidong.
Mechanism underlying mechanical stress regulating fibroblasts-derived
exosomes at the osteotomized end following hallux valgus correction |
| [12] | Jing Xiaowei, Mu Yingxiu. A study on anterior cruciate ligament injury and knee stability with a silicon mesh brace [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 505-510. |
| [13] | Huo Shaochuan, Wang Haibin, Tang Hongyu, Wang Yueqi, Chen Qunqun, Feng Ziyu, Li Yikai. Treatment of knee osteoarthritis by tonifying kidney and spleen and activating blood circulation herbs via chondrocyte IL-1beta/ERR-alpha/SOX9/Col2alpha1 signaling pathway in a rat model [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(35): 5577-5581. |
| [14] | Xiao Lei, Hou Ligang. Effect of glucosamine capsule on cartilage metabolism-related genes in peripheral blood mononuclear cells of patients with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5007-5012. |
| [15] | Deng Kaifeng, Chen Rilan, Zhu Shengwang, Wei Xingcheng, Zhang Lijuan, Zhu Ying. Electroacupuncture combined with thunder-fire moxibustion in the treatment of cold-dampness knee osteoarthritis: a randomized controlled clinical trial [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4638-4642. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||