Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (11): 1788-1793.doi: 10.3969/j.issn.2095-4344.0587
Previous Articles Next Articles
Interaction between satellite glial cells and neurons in sensory neuropathic pain
Wang Xianbin, Li Liyan
- (Institute of Neuroscience, Kunming Medical University, Kunming 650500, Yunnan Province, China)
-
Received:2018-08-07Online:2019-04-18Published:2021-04-28 -
Contact:Li Liyan, MD, Professor, Institute of Neuroscience, Kunming Medical University, Kunming 650500, Yunnan Province, China -
About author:Wang Xianbin, Institute of Neuroscience, Kunming Medical University, Kunming 650500, Yunnan Province, China -
Supported by:the National Natural Science Foundation of China, No. 31560295; the Health Science and Technology Program of Yunnan Province, No. 2014NS202; the Combined Project of Kunming Medical University, No. 2015FB098
CLC Number:
Cite this article
Wang Xianbin, Li Liyan. Interaction between satellite glial cells and neurons in sensory neuropathic pain[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(11): 1788-1793.
share this article
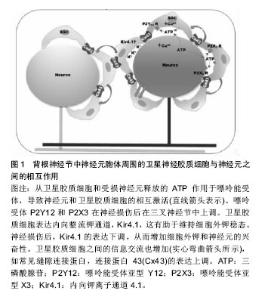
2.1 背根节神经节的解剖结构 背根神经节位于脊柱两椎骨之间,向中枢连接脊髓前角和后角神经元,其向外周沿神经纤维接受躯体感觉信号和输出中枢运动控制信号。在背根节中主要由感觉神经元、卫星胶质细胞以及许旺细胞组成。卫星胶质细胞包绕在感觉神经元胞体周围,与神经元共同组成了一个功能相对独立的结构单位[16]。 2.2 卫星胶质细胞在维持神经元外环境的静息状态中的作用 2.2.1 缝隙连接 是几乎存在于所有动物的细胞膜上,是细胞间进行直接交流的唯一信息通道。缝隙连接的通道可让部分小分子通过从而进入另一个细胞实现代谢偶联或电偶联,如细胞内的无机盐离子、氨基酸、三磷酸腺苷等小分子物质[17]。缝隙连接广泛存在,两种同类型的细胞间或者不同类型的细胞间均存在缝隙连接。例如,在中枢神经系统中,缝隙连接就存在于神经元之间、胶质细胞之间以及神经元与胶质细胞之 间[18]。在外周神经系统同样有类似的缝隙连接存在,在神经冲动信息传递及各种反应过程中,通过缝隙连接调节细胞外离子浓度,神经元与神经胶质细胞间的电活动等作用,维持着细胞内外环境的稳定和信息交流的功 能[19-20]。缝隙连接的基础是缝隙连接蛋白[21],在神经元上的连接蛋白主要是Cx26、Cx32和Cx36,而星形胶质细胞和卫星胶质细胞表面主要是Cx43[18]。神经病理性疼痛大部分是由于外周神经损伤导致,外周神经损伤后将导致痛觉过敏,是由于增强了感觉神经元的兴奋性,降低了神经元的兴奋阈值。神经损伤后,卫星胶质细胞也可能在脊髓伤害信号的传递中发挥重要作用[14]。研究表明,胶质细胞间的缝隙连接可能在神经病理性疼痛中发挥重要作用[15]。当神经损伤后,卫星胶质细胞间的缝隙连接数目明显增多[22]。卫星胶质细胞间缝隙连接数量在神经受损后将会明显增多,但过一段时间后又逐渐恢复到正常水平,然而神经受损导致的神经病理性疼痛并未痊愈,因此神经病理性疼痛的发生发展机制不仅仅只有缝隙连接发挥作用。 2.2.2 离子通道和神经递质 神经损伤后,损伤部位大量的炎性细胞聚集释放炎性递质,使化学信号诱发产生电信号传递到背根节或三叉神经节,电信号在背根节或三叉神经节转换。研究证明,ATP是参与神经元与卫星胶质细胞通讯的主要信号传送者之一[23],当神经冲动到达背根节时,卫星胶质细胞和神经元会大量释放ATP,从而激活嘌呤能(P2)受体(例如P2X和P2Y),增加细胞间钙离子浓度[24-27]。嘌呤能受体P2Y12 R的激活,可增加卫星胶质细胞的钙离子内流,反过来增加细胞的兴奋 性[28-30]。背根节中,细胞间存在大量的离子以维持细胞电位的稳定,神经节中的卫星胶质细胞在钾离子缓冲中起着重要作用[31]。细胞间钾离子的稳态维持着神经元的兴奋性,当细胞外钾离子浓度增加时,神经元兴奋性增加[32]。卫星胶质细胞表达内向整流钾通道Kir4.1,其缓冲神经节中的钾浓度[31]。研究表明,当神经损伤后,Kir4.1的表达下调,并且siRNA沉默Kir4.1在自由移动的大鼠中诱导自发和诱发的面部疼痛样行为,见图1[33]。"
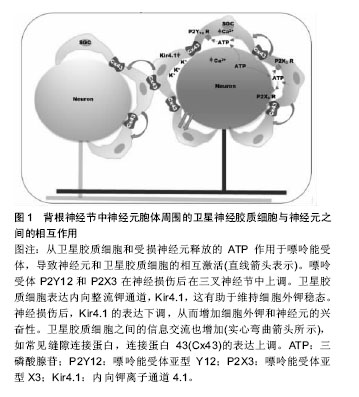
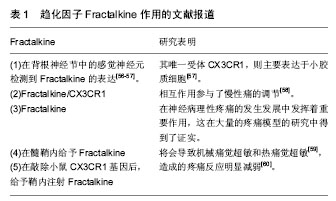
神经损伤后,电信号传递到神经节中,导致神经元大量释放神经递质以及神经和免疫因子,如谷氨酸,ATP,P物质,CGRP,脑源性神经营养因子,白细胞介素6和CCL2等[34-36]。这些介质增加突触后神经元的敏感性,并激活神经元周围的卫星胶质细胞。外周释放的免疫因子,如促炎性细胞因子(如白细胞介素6)也可能激活中枢神经胶质细胞[37-38]。外周白细胞介素6可通过循环转运到中枢神经系统,增加大脑血管内皮细胞中的COX-2活性和PGE2的释放,从而导致中枢免疫应答[37-38]。 2.2.3 卫星胶质细胞谷氨酸-谷氨酰胺循环调节神经元活动 谷氨酸是中枢神经和外周神经系统中重要的兴奋性神经递质,过多的谷氨酸会导致神经元的兴奋性增加,从而导致疼痛。卫星胶质细胞摄取细胞外谷氨酸,维持神经元外谷氨酸的稳态平衡。胶质细胞和神经元之间的谷氨酸-谷氨酰胺循环在增加神经损伤后的神经元兴奋性方面也起着关键作用[39-41]。胶质细胞摄取已经释放到突触中的谷氨酸,然后谷氨酰胺合成酶将其合成谷氨酰胺。神经元使用谷氨酰胺生产谷氨酸,补充谷氨酸供应[39-41]。谷氨酸转运蛋白GLT-1参与谷氨酸转运入胶质细胞。神经损伤后,GLT-1下调,导致谷氨酸积累,从而增加突触后神经元的兴奋性[42]。研究表明,病理性疼痛大鼠模型中,感觉神经元胞体中的谷氨酸含量增 加[43]。在外周神经系统中并不含有降解谷氨酸的酶,所以谷氨酸的清除主要依靠卫星胶质细胞上高亲和力的谷氨酸转运体来进行。卫星胶质细胞上也表达谷氨酸的运载体谷氨酸-天冬氨酸受体(glutamate-aspartate transporter,GLAST) 和谷氨酰胺合成酶(glutamine synthetase,GS),因此,卫星胶质细胞可以摄取细胞外的谷氨酸,并且在细胞内将谷氨酸合成谷氨酰胺。卫星胶质细胞摄取谷氨酸的作用可以维持细胞外谷氨酸的正常水平,从而维持神经元的兴奋性[44]。 2.2.4 卫星胶质细胞促进交感芽生以调节神经元活动 1993年,McLachlan和Chung分别报道在神经病理性疼痛模型建立后,背根神经节内的交感神经轴突(酪氨酸羟化酶阳性,THIR)末梢出芽增生并在一些大中直径胞体感觉神经元周围形成所谓“篮状结构(basket structure)”[45-46]。交感发芽和神经病理疼痛的机制主要有直接耦联和间接耦联两种假说[47],但这两种假说都忽视了背根节中的胶质细胞,现研究发现胶质细胞在交感芽生从而导致神经病理性疼痛的过程中可能起着至关重要的作用[48]。有研究发现,在电镜下交感轴突在卫星细胞层形成的曲张体结构,这些交感轴突末梢与卫星胶质细胞间可以形成类似突触的结构,猜测卫星胶质细胞与感觉神经元之间可能存在交感-感觉偶联,从而起着中介的作用[49]。此外,有研究发现,卫星胶质释放的神经营养因子能够促进交感出芽结构的形成,如神经生长因子,NT-3,脑源性神经营养因子等[50-51]。神经损伤后,背根节中的卫星胶质细胞神经生长因子 mRNA表达明显升高,且背根神经节中神经元并不能合成神经生长因子,用神经生长因子抗体干预后发现交感芽生明显减少[52]。此外,在神经病理性疼痛模型鞘内给予神经生长因子,背根节中有大量交感出芽和篮状结构形成[53],外周神经损伤后,背根节中卫星胶质细胞释放神经生长因子或NT-3,启动了交感轴突的再生过程,使得交感轴突向卫星细胞发芽,与卫星胶质细胞形成交感-感觉偶联,从而调节神经元的活动[34]。 2.2.5 趋化因子介导背根神经节神经元与卫星胶质细胞的相互作用 趋化因子是指一类低分子量的蛋白质,因其趋化细胞的迁移而得名[54]。在神经系统中,趋化因子发挥着重要的作用,参与各种生物学过程,包括控制免疫细胞趋化,刺激新血管形成、刺激释放炎症细胞因子、调节细胞间信息传递等[55]。Fractalkine是一种与神经元细胞外表面相连的独特趋化因子,见表1。"
| [1] Li D, Yan Y, Yu L, et al. Procaine Attenuates Pain Behaviors of Neuropathic Pain Model Rats Possibly via Inhibiting JAK2/STAT3. Biomolecules AND Therapeutics. 2016;24(5): 489-494. [2] Pozeg P, Palluel E, Ronchi R, et al. Virtual reality improves embodiment and neuropathic pain caused by spinal cord injury. Neurology. 2017;89(18):1894-1903.[3] López Chiriboga AS, Cheshire WPJr. Trigeminal Neuralgia Following Lightning Injury. J Oral Facial Pain Headache. 2017;31(4):e7.[4] Galánarriero I, Serranomuñoz D, Gómezsoriano J, et al. The role of Omega-3 and Omega-9 fatty acids for the treatment of neuropathic pain after neurotrauma. Biochim Biophys Acta. 2017;1859(9 Pt B). [5] Taha O, Opitz T, Mueller M, et al. Neuropathic pain in experimental autoimmune neuritis is associated with altered electrophysiological properties of nociceptive DRG neurons. Exp Neurol. 2017;():.[6] Ran R, Gu J, Fu J, et al. The role of the GABA-A receptor of the adjacent intact dorsal root ganglion neurons in rats with neuropathic pain. Acta Neurobiol Exp (Wars). 2014;74(4): 405-414.[7] Tan AM, Samad OA, Dibhajj SD, et al. Virus-Mediated Knockdown of Nav1. 3 in Dorsal Root Ganglia of STZ-Induced Diabetic Rats Alleviates Tactile Allodynia. Mol Med. 2014;21(1):544-552.[8] Jia T, Rao J, Zou L, et al. Nanoparticle-Encapsulated Curcumin Inhibits Diabetic Neuropathic Pain Involving the P2Y12 Receptor in the Dorsal Root Ganglia. Front Neurosci. 2018;11:755.[9] Lagares A, Li HY, Zhou XF, et al. Primary sensory neuron addition in the adult rat trigeminal ganglion: evidence for neural crest glio-neuronal precursor maturation. J Neurosci. 2007;27(30):7939-7953. [10] 杨悦橙,马柯.卫星胶质细胞参与神经病理性痛的研究进展[J].中国疼痛医学杂志,2013,19(6):2766-2770.[11] Hanani M. Satellite glial cells in sympathetic and parasympathetic ganglia: In search of function. Brain Res Rev. 2010;64(2):304-327.[12] Liu FY, Sun YN, Wang FT, et al. Activation of satellite glial cells in lumbar dorsal root ganglia contributes to neuropathic pain after spinal nerve ligation. Brain Res. 2012;1427(1):65-77.[13] Nascimento DS, Castro-Lopes JM, Moreira Neto FL. Satellite glial cells surrounding primary afferent neurons are activated and proliferate during monoarthritis in rats: is there a role for ATF3? PLoS One. 2014;9(9):e108152.[14] Ji RR, Berta T, Nedergaard M. Glia and pain: Is chronic pain a gliopathy? Pain. 2013;():S10-S28.[15] Yoon SY, Robinson CR, Zhang H, et al. Spinal astrocyte gap junctions contribute to oxaliplatin-induced mechanical hypersensitivity. J Pain. 2013;14(2):205-214.[16] 焦晓翠,张会然,张璇,等.卫星胶质细胞对外周神经元作用的研究进展[J].中国药理学通报,2014;30(5):612-614.[17] Eugenin EA, Basilio D, Sáez JC, et al. The Role of Gap Junction Channels During Physiologic and Pathologic Conditions of the Human Central Nervous System. J Neuroimmune Pharmacol. 2012;7(3):499-518. [18] Cui Y, Masaki K, Yamasaki R, et al. Extensive dysregulations of oligodendrocytic and astrocytic connexins are associated with disease progression in an amyotrophic lateral sclerosis mouse model. J Neuroinflammation. 2014;11:42..[19] Hanani M. Satellite glial cells in sensory ganglia: from form to function. Brain Res Rev. 2005;48(3):457-476.[20] Hanani M, Huang TY, Cherkas PS, et al. Glial cell plasticity in sensory ganglia induced by nerve damage. Neuroscience. 2002;114(2):279-283.[21] Vit JP, Jasmin L, Bhargava A, et al. Satellite glial cells in the trigeminal ganglion as a determinant of orofacial neuropathic pain. Neuron Glia Biol. 2006;2(4):247-257.[22] Ohara PT, Vit JP, Bhargava A, et al. Evidence for a role of connexin 43 in trigeminal pain using RNA interference in vivo. J Neurophysiol. 2008;100(6):3064-3073.[23] Hanani M. Intercellular communication in sensory ganglia by purinergic receptors and gap junctions: implications for chronic pain. Brain Res. 2012;1487(17):183-191.[24] Gu Y, Chen Y, Zhang X, et al. Neuronal soma-satellite glial cell interactions in sensory ganglia and the participation of purinergic receptors. Neuron Glia Biol. 2010;6(1):53-62.[25] Suadicani SO, Cherkas PS, Zuckerman J, et al. Bidirectional calcium signaling between satellite glial cells and neurons in cultured mouse trigeminal ganglia. Neuron Glia Biol. 2010; 6(1):43-51.[26] Weick M, Cherkas PS, Härtig W, et al. P2 receptors in satellite glial cells in trigeminal ganglia of mice. Neuroscience. 2003; 120(4):969-977. [27] Villa G, Fumagalli M, Verderio C, et al. Expression and contribution of satellite glial cells purinoceptors to pain transmission in sensory ganglia: an update. Neuron Glia Biol. 2010;6(1):31-42.[28] Ceruti S, Fumagalli M, Villa G, et al. Purinoceptor-mediated calcium signaling in primary neuron-glia trigeminal cultures. Cell Calcium. 2008;43(6):576-590.[29] Takeda M, Takahashi M, Matsumoto S. Contribution of the activation of satellite glia in sensory ganglia to pathological pain. Neurosci Biobehav Rev. 2009;33(6):784-792.[30] Katagiri A, Shinoda M, Honda K, et al. Satellite glial cell P2Y12 receptor in the trigeminal ganglion is involved in lingual neuropathic pain mechanisms in rats. Molecular Pain. 2012;8(1):23.[31] Tang X, Schmidt TM, Perezleighton CE,et al. Kir4. 1 is Responsible for the Native Inward Potassium Conductance of Satellite Glial Cells in Sensory Ganglia. Neuroscience. 2010; 166(2):397-407. [32] Bellot-Saez A, Kã©Kesi O, Morley JW, et al. Astrocytic modulation of neuronal excitability through K+ spatial buffering. Neurosci Biobehav R. 2017;(77):87-97.[33] Vit JP, Ohara PT, Bhargava A, et al. Silencing the Kir4.1 potassium channel subunit in satellite glial cells of the rat trigeminal ganglion results in pain-like behavior in the absence of nerve injury.J Neurosci. 2008;28(16):4161-4171.[34] Scholz J, Woolf CJ. The neuropathic pain triad: neurons, immune cells and glia. Nat Neurosci. 2007;10(11):1361.[35] Ren K, Dubner R. Neuron-glia crosstalk gets serious: role in pain hypersensitivity. Current Opinion in Anaesthesiology. 2008;21(5):570.[36] Milligan E, Watkins L. Pathological and protective roles of glia in chronic pain. Nature Reviews Neuroscience. 2008;10(1): 23.[37] Schöbitz B, Voorhuis DA, De Kloet ER.Localization of interleukin 6 mRNA and interleukin 6 receptor mRNA in rat brain. Eur J Neurosci.1992;136(2):189-192. [38] Vallières L, Rivest S. Regulation of the genes encoding interleukin-6, its receptor, and gp130 in the rat brain in response to the immune activator lipopolysaccharide and the proinflammatory cytokine interleukin-1beta. J Neurochem. 1997;69(4):1668-1683.[39] Chiang CY, Wang J, Xie YF, et al. Astroglial glutamate- glutamine shuttle is involved in central sensitization of nociceptive neurons in rat medullary dorsal horn.J Neurosci. 2007;27(34):9068-9076.[40] Fonseca LL, Monteiro MA, Alves PM, et al. Cultures of rat astrocytes challenged with a steady supply of glutamate: new model to study flux distribution in the glutamate-glutamine cycle. Glia. 2005;51(4):286-296.[41] Chiang CY, Li Z, Dostrovsky JO, et al. Glutamine uptake contributes to central sensitization in the medullary dorsal horn. Neuroreport. 2008; 19(11):1151-1154. [42] Sung B, Lim G, Mao J. Altered expression and uptake activity of spinal glutamate transporters after nerve injury contribute to the pathogenesis of neuropathic pain in rats.J Neurosci. 2003;23(7):2899-910.[43] Kung LH, Gong K, Adedoyin M, et al. Evidence for Glutamate as a Neuroglial Transmitter within Sensory Ganglia. PLoS One.2013; 8(7):e68312. [44] Ohara PT, Vit JP, Bhargava A,et al.Gliopathic pain: when satellite glial cells go bad.Neuroscientist. 2009;15(5):450-463.[45] Mclachlan EM, Jänig W, Devor M, et al. Peripheral nerve injury triggers noradrenergic sprouting within dorsal root ganglia. Nature.1993;363(6429):543-546.[46] Chung K, Kim HJ, Na HS, et al. Abnormalities of sympathetic innervation in the area of an injured peripheral nerve in a rat model of neuropathic pain. Neurosci Lett.1993;162(1-2): 85-88.[47] Rubin G, Kaspi T, Rappaport ZH, et al. Adrenosensitivity of injured afferent neurons does not require the presence of postganglionic sympathetic terminals. Pain.1997; 72(1-2): 183. [48] 闫栋,蒋劲,张德仁.背根节交感芽生与神经病理性疼痛关系的总结和思考[J].中国疼痛医学杂志,2010;16(3):169-171.[49] Shinder V, Govrin-Lippmann R, Cohen S, et al. Structural basis of sympathetic-sensory coupling in rat and human dorsal root ganglia following peripheral nerve injury. J Neurocytol.1999;28(9):743-761.[50] Deng YS, Zhong JH, Zhou XF. Effects of endogenous neurotrophins on sympathetic sprouting in the dorsal root ganglia and allodynia following spinal nerve injury. Exp Neurol. 2000;164(2):344-350.[51] Ramer MS, Thompson SW, Mcmahon SB. Causes and consequences of sympathetic basket formation in dorsal root ganglia. Pain. Pain. 1999;Suppl 6:S111-120. [52] Zhou XF, Deng YS, Chie E, et al. Satellite-cell-derived nerve growth factor and neurotrophin-3 are involved in noradrenergic sprouting in the dorsal root ganglia following peripheral nerve injury in the rat. Eur J Neurosci.1999;11(5): 1711-1722.[53] Jones MG, Munson JB, Thompson SW. A role for nerve growth factor in sympathetic sprouting in rat dorsal root ganglia. Pain.1999;79(1):21.[54] Asensio VC, Campbell IL. Campbell IL. Chemokines in the CNS: plurifunctional mediators in diverse states. Trends Neurosci.1999;22(11):504.[55] Deiva K, Geeraerts T, Salim H, et al. Fractalkine reduces N-methyl-d-aspartate-induced calcium flux and apoptosis in human neurons through extracellular signal-regulated kinase activation. Eur J Neurosci. 2004;20(12):3222-3232.[56] Yang JL, Bo X, Li SS,et al.Gabapentin reduces CX3CL1 signaling and blocks spinal microglial activation in monoarthritic rats. Molecular Brain, 5, 1(2012-05-30). 2012, 5(1):1-12. [57] Verge GM, Milligan ED, Maier SF, et al. Fractalkine (CX3CL1) and fractalkine receptor (CX3CR1) distribution in spinal cord and dorsal root ganglia under basal and neuropathic pain conditions. Eur J Neurosci. 2004;20(5):1150-1160.[58] Clark AK, Staniland AA, Malcangio M. Fractalkine/CX3CR1 signalling in chronic pain and inflammation. Curr Pharm Biotechnol. 2011;12(10):1707-1714.[59] Milligan E, Zapata V, Schoeniger D, et al. An initial investigation of spinal mechanisms underlying pain enhancement induced by fractalkine, a neuronally released chemokine. Eur J Neurosci. 2005;22(11):2775-2782.[60] Staniland AA, Clark AK, Wodarski R, et al. Reduced inflammatory and neuropathic pain and decreased spinal microglial response in fractalkine receptor (CX3CR1) knockout mice. J Neurochem. 2010;114(4):1143-1157.[61] Souza GR, Talbot J, Lotufo CM, et al. Fractalkine mediates inflammatory pain through activation of satellite glial cells. Proc Natl Acad Sci U S A. 2013;110(27):11193-11198. |
| [1] | Min Youjiang, Yao Haihua, Sun Jie, Zhou Xuan, Yu Hang, Sun Qianpu, Hong Ensi. Effect of “three-tong acupuncture” on brain function of patients with spinal cord injury based on magnetic resonance technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-8. |
| [2] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [3] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [4] | Chai Le, Lü Jianlan, Hu Jintao, Hu Huahui, Xu Qingjun, Yu Jinwei, Quan Renfu. Signal pathway variation after induction of inflammatory response in rats with acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1218-1223. |
| [5] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| [6] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [7] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [8] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [9] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [10] | Xie Yang, Zhang Shujiang, Liu Menglan, Luo Ying, Yang Yang, Li Zuoxiao. Mechanism by which rapamycin protects spinal cord neurons in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 695-700. |
| [11] | Ma Binxiang, He Wanqing, Zhou Guangchao, Guan Yonglin. Triptolide improves motor dysfunction in rats following spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 701-706. |
| [12] | Luo Xuanxiang, Jing Li, Pan Bin, Feng Hu. Effect of mecobalamine combined with mouse nerve growth factor on nerve function recovery after cervical spondylotic myelopathy surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 719-722. |
| [13] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [14] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [15] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||