Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (25): 3975-3980.doi: 10.3969/j.issn.2095-4344.0929
Previous Articles Next Articles
HGF/c-Met axis regulates human adipose tissue-derived stem cells in burn wound healing
Li Xue-yang1, 2, Zheng Wan-ling2, Yang Chao1, Xia Si-zhan2, Xing Xin1
- 1Department of Plastic Surgery, Changhai Hospital, Second Military Medical University, Shanghai 200433, China; 2Department of Plastic Surgery, Affiliated Hospital of Xuzhou Medical University, Xuzhou 221002, Jiangsu Province, China
-
Revised:2018-03-03Online:2018-09-08Published:2018-09-08 -
Contact:Xing Xin, M.D., Professor, Department of Plastic Surgery, Changhai Hospital, Second Military Medical University, Shanghai 200433, China -
About author:Li Xue-yang, Doctoral candidate, Associate chief physician, Department of Plastic Surgery, Changhai Hospital, Second Military Medical University, Shanghai 200433, China; Department of Plastic Surgery, Affiliated Hospital of Xuzhou Medical University, Xuzhou 221002, Jiangsu Province, China -
Supported by:the Project of Xuzhou Municipal Office of Science and Technology, No. KC15SH087
CLC Number:
Cite this article
Li Xue-yang, Zheng Wan-ling, Yang Chao, Xia Si-zhan, Xing Xin. HGF/c-Met axis regulates human adipose tissue-derived stem cells in burn wound healing[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(25): 3975-3980.
share this article
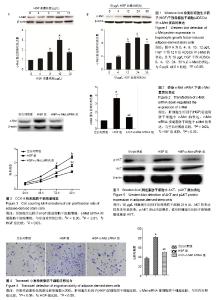
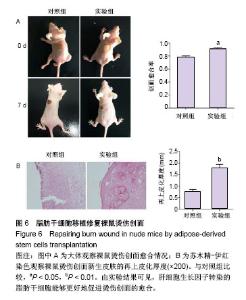
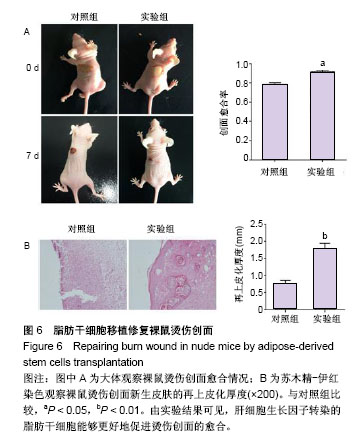
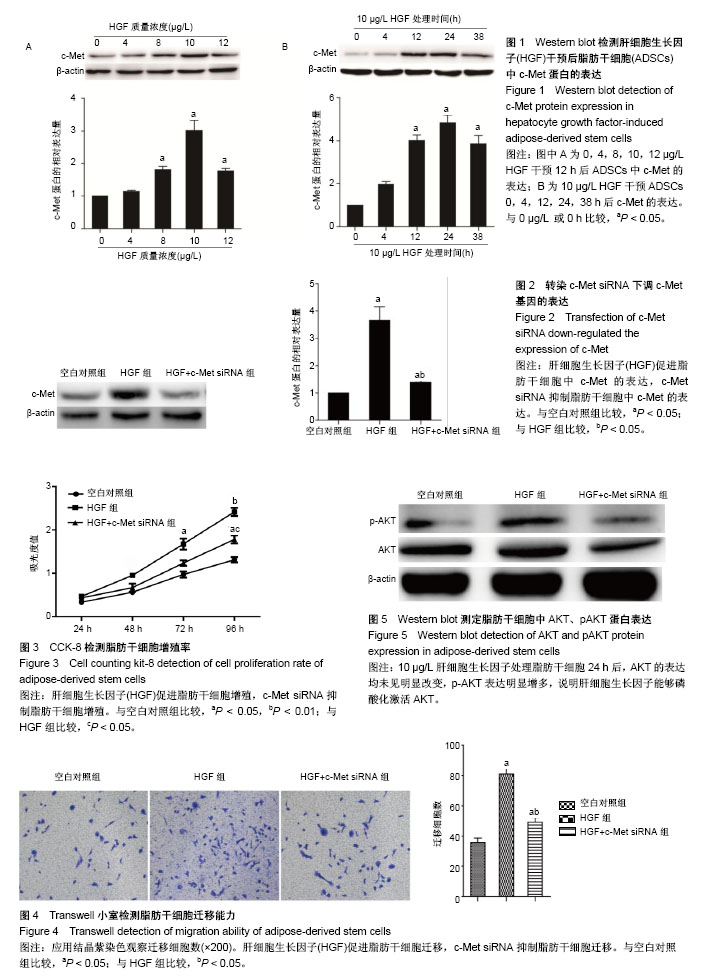
2.1 HGF诱导c-Met蛋白的表达 采用0,4,8,10,12 μg/L HGF处理ADSCs 12 h后通过Western blot检测c-Met蛋白的表达,差异有显著性意义(P < 0.05),与0 μg/L组相比较,4 μg/L组c-Met表达无明显变化(P=0.235),8,10 μg/L组c-Met的表达逐渐上调,12 μg/L组c-Met表达较10 μg/L组下降(P < 0.05,图1A)。因此,选择10 μg/L HGF处理ADSCs 0,4,12,24,38 h后通过Western blot检测c-Met蛋白的表达,差异有显著性意义(P < 0.05),与0 h组相比较,4 h组c-Met表达无明显变化(P=0.523),12,24 h组c-Met的表达明显逐渐上调,38 h组较24 h组c-Met表达下调(P < 0.05,图1B),综上实验结果作者认为以10 μg/L HGF处理ADSCs 24 h为最适浓度及时间。 2.2 c-Met siRNA抑制c-Met蛋白的表达 3组ADSCs中c-Met蛋白的表达差异有显著性意义(P < 0.05),HGF(10 μg/L)处理ADSCs 24 h后,HGF组与空白对照组比较,c-Met蛋白表达明显上调(P < 0.05);与HGF组比较,HGF+c-Met siRNA组c-Met蛋白表达明显下调(P < 0.05,图2)。 2.3 HGF促进ADSCs增殖,c-Met siRNA抑制ADSCs增殖 通过重复测量数据的方差分析,不同因素处理ADSCs后,比较不同时间点的细胞增殖情况,差异有显著性意义(P < 0.05);与空白对照组比较, HGF组细胞增殖能力升高,差异有显著性意义(P < 0.05);与HGF组比较,HGF+c-Met siRNA组细胞增殖能力降低,差异有显著性意义(P < 0.05),见图3。 2.4 HGF促进ADSCs迁移,c-Met siRNA抑制ADSCs迁移 采用重复数据方差分析,不同因素处理ADSCs后,细胞迁移能力差异有显著性意义(P < 0.05)。与空白对照组比较,HGF组细胞迁移能力增强,差异有显著性意义(P < 0.05);与HGF组比较,HGF+c-Met siRNA组细胞迁移能力降低,差异有显著性意义(P < 0.05),见图4。 2.5 HGF/c-Met激活PI3K/AKT信号通路 HGF(10 μg/L)处理c-Met siRNA ADSCs及ADSCs 24 h,AKT的表达均未见明显改变;与空白对照组比较,HGF组p-AKT表达明显增多;与HGF组比较,HGF+c-Met siRNA组p-AKT表达明显减少(图5)。 2.6 HGF预处理ADSCs移植对裸鼠烫伤创伤愈合的影响 在移植后第7天进行裸鼠模型的创面比较(图6)。实验组在创伤后第7天的创面愈合率和上皮化厚度显著高于对照组,差异有显著性意义(P < 0.05)。"
| [1] Sitton E. Early and late radiation-induced skin alterations. Part II: Nursing care of irradiated skin. Oncol Nurs Forum. 1992;19(6):907-912.[2] Noble-Adams R. Radiation-induced reactions. 1: An examination of the phenomenon. Br J Nurs. 1999;8(17): 1134-1140.[3] Porock D. Factors influencing the severity of radiation skin and oral mucosal reactions: development of a conceptual framework. Eur J Cancer Care (Engl). 2002;11(1):33-43.[4] Maduro JH, Pras E, Willemse PH, et al. Acute and long-term toxicity following radiotherapy alone or in combination with chemotherapy for locally advanced cervical cancer. Cancer Treat Rev. 2003;29(6):471-488.[5] Vaz AF, Pinto-Neto AM, Conde DM, et al. Quality of life of women with gynecologic cancer: associated factors. Arch Gynecol Obstet. 2007;276(6):583-589.[6] Naylor W, Mallett J. Management of acute radiotherapy induced skin reactions: a literature review. Eur J Oncol Nurs. 2001;5(4):221-233.[7] Morgan K. Radiotherapy-induced skin reactions: prevention and cure. Br J Nurs. 2014;23(16):S24, S26-32.[8] De Ruyck K, Van Eijkeren M, Claes K, et al. Radiation-induced damage to normal tissues after radiotherapy in patients treated for gynecologic tumors: association with single nucleotide polymorphisms in XRCC1, XRCC3, and OGG1 genes and in vitro chromosomal radiosensitivity in lymphocytes. Int J Radiat Oncol Biol Phys. 2005;62(4):1140-1149.[9] Hymes SR, Strom EA, Fife C. Radiation dermatitis: clinical presentation, pathophysiology, and treatment 2006. J Am Acad Dermatol. 2006;54(1):28-46.[10] Yarnold J, Brotons MC. Pathogenetic mechanisms in radiation fibrosis. Radiother Oncol. 2010;97(1):149-161.[11] Ahmed EA, Agay D, Schrock G, et al. Persistent DNA damage after high dose in vivo gamma exposure of minipig skin. PLoS One. 2012;7(6):e39521.[12] Valente S, Ciavarella C, Pasanisi E, et al. Hepatocyte Growth Factor Effects on Mesenchymal Stem Cells Derived from Human Arteries: A Novel Strategy to Accelerate Vascular Ulcer Wound Healing. Stem Cells Int. 2016;2016:3232859.[13] Broekman W, Amatngalim GD, de Mooij-Eijk Y, et al. TNF-α and IL-1β-activated human mesenchymal stromal cells increase airway epithelial wound healing in vitro via activation of the epidermal growth factor receptor. Respir Res. 2016; 17:3.[14] Sun B, Meng XH, Liu R, et al. Mechanism study for hypoxia induced differentiation of insulin-producing cells from umbilical cord blood-derived mesenchymal stem cells. Biochem Biophys Res Commun. 2015;466(3):444-449.[15] Fuse MA, Plati SK, Burns SS, et al. Combination Therapy with c-Met and Src Inhibitors Induces Caspase-Dependent Apoptosis of Merlin-Deficient Schwann Cells and Suppresses Growth of Schwannoma Cells. Mol Cancer Ther. 2017;16(11): 2387-2398.[16] Yen BL, Yen ML, Hsu PJ, et al. Multipotent human mesenchymal stromal cells mediate expansion of myeloid-derived suppressor cells via hepatocyte growth factor/c-met and STAT3. Stem Cell Reports. 2013;1(2): 139-151.[17] Branski LK, Gauglitz GG, Herndon DN, et al. A review of gene and stem cell therapy in cutaneous wound healing. Burns. 2009;35(2):171-180.[18] Peyvandi AA, Abbaszadeh HA, Roozbahany NA, et al. Deferoxamine promotes mesenchymal stem cell homing in noise-induced injured cochlea through PI3K/AKT pathway. Cell Prolif. 2018 Jan 17. doi: 10.1111/cpr.12434. [Epub ahead of print][19] Reddy LVK, Sen D. DADLE enhances viability and anti-inflammatory effect of human MSCs subjected to 'serum free' apoptotic condition in part via the DOR/PI3K/AKT pathway. Life Sci. 2017;191:195-204.[20] Lv B, Hua T, Li F, et al. Hypoxia-inducible factor 1 α protects mesenchymal stem cells against oxygen-glucose deprivation-induced injury via autophagy induction and PI3K/AKT/mTOR signaling pathway. Am J Transl Res. 2017; 9(5):2492-2499.[21] Rahmani M, Aust MM, Attkisson E, et al. Dual inhibition of Bcl-2 and Bcl-xL strikingly enhances PI3K inhibition-induced apoptosis in human myeloid leukemia cells through a GSK3- and Bim-dependent mechanism. Cancer Res. 2013;73(4): 1340-1351.[22] Li J, Huang S, Zhang J, et al. Mesenchymal stem cells ameliorate inflammatory cytokine-induced impairment of AT-II cells through a keratinocyte growth factor-dependent PI3K/Akt/mTOR signaling pathway. Mol Med Rep. 2016;13(5): 3755-3762.[23] Zhang R, Yang G, Wang Q, et al. Acylated ghrelin protects hippocampal neurons in pilocarpine-induced seizures of immature rats by inhibiting cell apoptosis. Mol Biol Rep. 2013; 40(1):51-58.[24] Kim JW, Lee MN, Jeong BC, et al. Chemical inhibitors of c-Met receptor tyrosine kinase stimulate osteoblast differentiation and bone regeneration. Eur J Pharmacol. 2017;806:10-17.[25] Deying W, Feng G, Shumei L, et al. CAF-derived HGF promotes cell proliferation and drug resistance by up-regulating the c-Met/PI3K/Akt and GRP78 signalling in ovarian cancer cells. Biosci Rep. 2017;37(2): BSR20160470. [26] Ahn SY, Kim J, Kim MA, et al. Increased HGF Expression Induces Resistance to c-MET Tyrosine Kinase Inhibitors in Gastric Cancer. Anticancer Res. 2017;37(3):1127-1138.[27] An Y, Liu WJ, Xue P, et al. Autophagy promotes MSC-mediated vascularization in cutaneous wound healing via regulation of VEGF secretion. Cell Death Dis. 2018;9(2): 58.[28] Du HC, Jiang L, Geng WX, et al. Growth Factor-Reinforced ECM Fabricated from Chemically Hypoxic MSC Sheet with Improved In Vivo Wound Repair Activity. Biomed Res Int. 2017;2017:2578017.[29] Nuschke A. Activity of mesenchymal stem cells in therapies for chronic skin wound healing. Organogenesis. 2014;10(1): 29-37.[30] Herberg S, Shi X, Johnson MH, et al. Stromal cell-derived factor-1β mediates cell survival through enhancing autophagy in bone marrow-derived mesenchymal stem cells. PLoS One. 2013;8(3):e58207. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||