Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8309-8318.doi: 10.12307/2026.207
Performance of calcium sulfate-magnesium oxide composites as anti-infective bone graft materials
Hu Liqun1, Xiao Dongqin2, Ma Chenxi3, Li Zhuohan4, Yan Jiyuan1, Li Zhong1, He Kui1, Duan Ke1
- 1Department of Orthopedics and Joint Surgery, Affiliated Hospital of Southwest Medical University, Sichuan Orthopedic Implant Device R&D and Application Technology Engineering Laboratory, Luzhou 646000, Sichuan Province, China; 2Institute of Tissue Engineering and Stem Cells, Second Clinical College of North Sichuan Medical College, Nanchong Central Hospital, Nanchong 637000, Sichuan Province, China; 3Department of Orthopedics, Luxian People’s Hospital, Luzhou 646100, Sichuan Province, China; 4Southwest Medical University, Luzhou 646000, Sichuan Province, China
-
Accepted:2025-09-08Online:2026-11-18Published:2026-04-23 -
Contact:Duan Ke, MD, Professor, Department of Orthopedics and Joint Surgery, Affiliated Hospital of Southwest Medical University, Sichuan Orthopedic Implant Device R&D and Application Technology Engineering Laboratory, Luzhou 646000, Sichuan Province, China -
About author:Hu Liqun, MS, Department of Orthopedics and Joint Surgery, Affiliated Hospital of Southwest Medical University, Sichuan Orthopedic Implant Device R&D and Application Technology Engineering Laboratory, Luzhou 646000, Sichuan Province, China -
Supported by:National Natural Science Foundation of China, No. 82002289 (to XDQ); Sichuan Provincial Science and Technology Plan, No. 2022YFS0628-C1 (to HK); Sichuan Provincial Natural Science Foundation, No. 2023NSFSC1740 (to XDQ)
CLC Number:
Cite this article
Hu Liqun, Xiao Dongqin, Ma Chenxi, Li Zhuohan, Yan Jiyuan, Li Zhong, He Kui, Duan Ke. Performance of calcium sulfate-magnesium oxide composites as anti-infective bone graft materials[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8309-8318.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
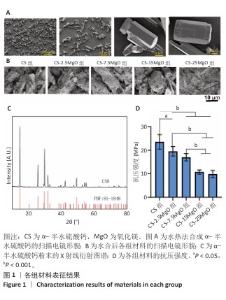
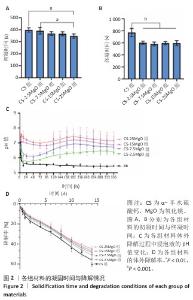
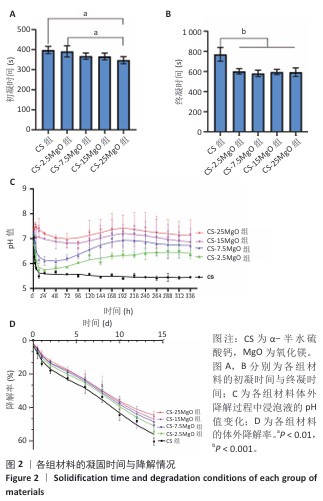
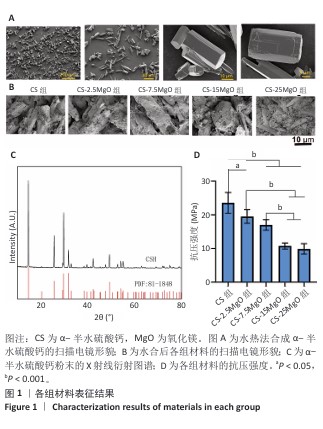
2.1 材料表征结果 扫描电镜观察显示,α-半水硫酸钙粉末大部分呈短棒状晶体,有少量长条状晶体(长度50-80 μm,直径20-40 μm),表面光滑(图1A);经过水合硬化后的样品排列紧密,并且除CS组外,CS-2.5MgO、CS-7.5MgO、CS-15MgO和CS-25MgO组均可见氧化镁颗粒团聚体分布在晶体表面及晶体之间,并且颗粒密度随氧化镁比例的增加而上升(图1B)。 X射线衍射图像显示,α-半水硫酸钙粉末的主要衍射峰与半水硫酸钙的典型衍射角相匹配(图1C)。随着材料中氧化镁比例的增加,材料的抗压强度呈降低趋势(图1D);与CS组相比,CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组抗压强度分别降低17%,28%,55%和58%。 5组材料的初凝和终凝时间范围分别为5-7 min和10-13 min,相比CS组,CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组初凝时间分别缩短2%,8%,8%和13%,终凝时间分别缩短22%,25%,23%,23%(图2A,B)。 各组浸泡液pH值变化见图2C。相同时间点下,随着材料中氧化镁比例的增加,浸泡液pH值增加。CS组浸泡液pH值从7.3迅速下降,在12 h达到5.49,随后保持相对稳定至实验结束(336 h);其他组均呈现pH值先下降再上升趋势,最后逐渐稳定。CS-2.5MgO组浸泡液pH值在24 h降至5.69,随后逐渐上升,于240 h稳定在6.39;CS-7.5MgO组、CS-15MgO组、CS-25MgO组浸泡液pH值分别在24 h或48 h降至最低值,随后逐渐上升,于240 h分别稳定在6.82,7.13和7.45。各组材料在PBS中基本以匀速降解,相同时间点下,材料降解率与氧化镁比例呈负相关,至336 h,CS组、CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组材料降解率分别为60%,54%,51%,49%和45%(图2D)。 "
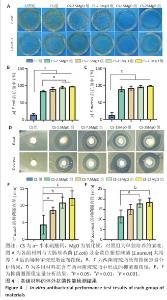
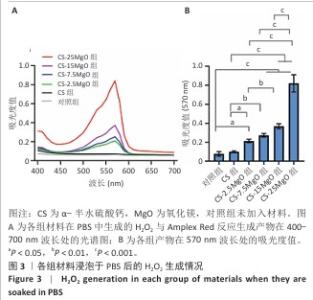
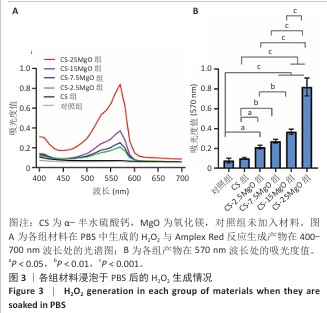
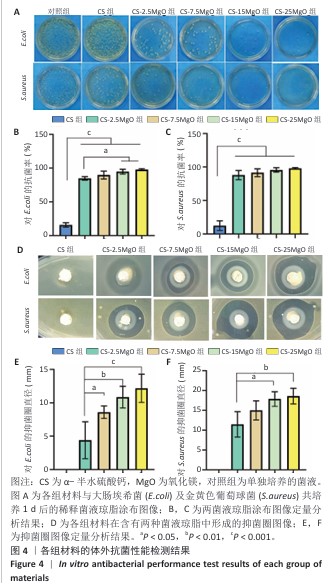
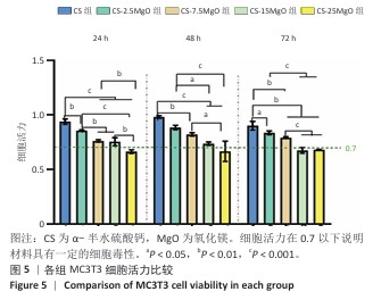
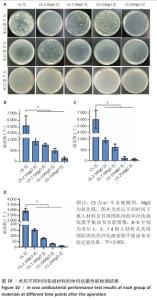
2.3 材料的体外抗菌性能检测结果 各组大肠埃希菌(或金黄色葡萄球菌)菌液琼脂平板涂布实验结果,见图4A-C。随着氧化镁比例的增加,材料对两种细菌的抗菌率均呈上升趋势。CS组对大肠埃希菌、金黄色葡萄球菌的抑菌率分别为16%和12%,各硫酸钙-氧化镁复合材料组对大肠埃希菌的抗菌率在85%-98%之间,对金黄色葡萄球菌的抗菌率在88%-98%之间。 各组材料抑菌圈实验结果见图4D-F。CS组均未观察到抑菌圈形成,其他4组均形成抑菌圈,并且抑菌圈直径随氧化镁比例的增加而增大。但是,观察到硫酸钙-氧化镁复合材料周围有白色不透明圈,具体机制不明,可能是因为硫酸钙-氧化镁复合材料的样品吸水性更强,使周围琼脂水分减少从而不透明化,具体原因有待进一步研究。 "
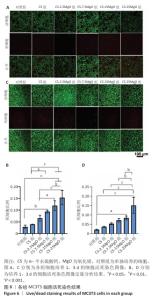
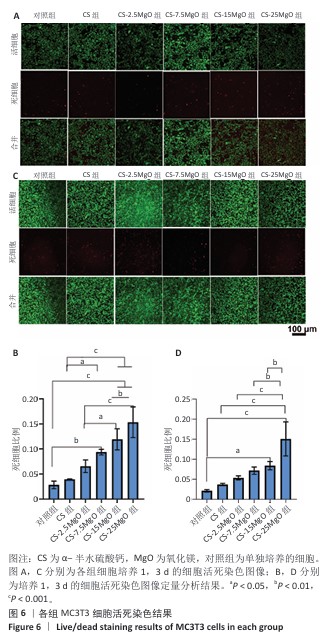
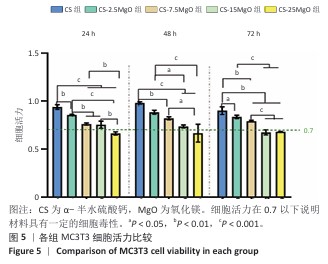
2.4 材料的体外细胞相容性评估结果 各组细胞CCK-8检测结果见图5。培养24,48,72 h,CS组、CS-2.5MgO组和CS-7.5MgO组细胞活力均> 77%,按照ISO10993-5标准判定该3组材料无细胞毒性。CS-15MgO组培养24,48 h的细胞活力> 72%,培养72 h的细胞活力下降至69%,表明该材料具有轻微细胞毒性。CS-25MgO组细胞活力活力一直保持在65%-69%之间,表明该组材料具有轻度细胞毒性。 各组细胞活死染色结果见图6。相同培养时间下,随着材料中氧化镁比例的增加,死细胞比例增加。培养24 h后,对照组、CS组、CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组死细胞比例分别为3%,4%,7%,9%,12%,15%;培养3 d后,对照组、CS组、CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组死细胞比例分别为2%,4%,5%,7%,8%,15%。"
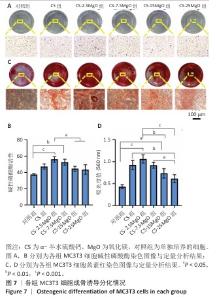
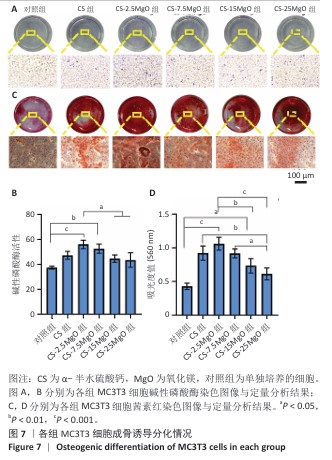
2.5 材料的成骨诱导分化检测结果 各组细胞碱性磷酸酶染色结果见图7A,B。各组孔底均染为蓝色,染色最深的为CS-2.5MgO组,其次为CS-7.5MgO组、CS组。与对照组比,CS组、CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组碱性磷酸酶活性分别增加26%,50%,41%,19%和16%。 各组细胞茜素红染色结果见图7C,D。各组钙结节均被茜素红染为红色,CS-2.5MgO组染色最深,其次为CS-7.5MgO组、CS组。与对照组相比,CS组、CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组染色液吸光度值分别增加了115%,147%,113%,71%,43%。"
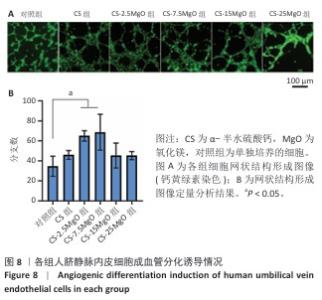
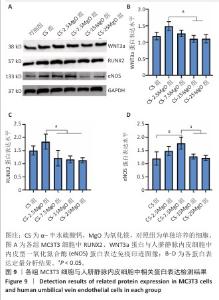
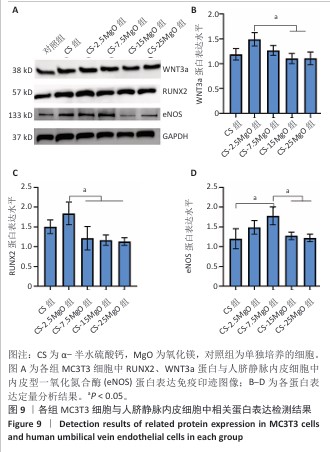
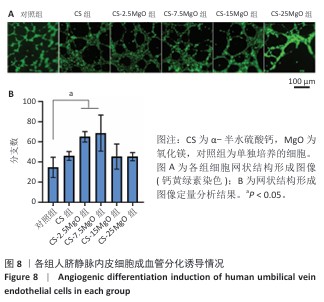
2.6 材料的促成血管形成能力检测结果 各组基质胶上的人脐静脉内皮细胞钙黄绿素染色后在荧光显微镜下呈绿色,细胞交织形成网(图8A)。定量分析结果显示,与对照组相比,CS组、CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组成管分支数量分别增加33%,88%,98%,31%和31%(图8B)。 2.7 Western Blot检测成骨相关蛋白与成血管相关蛋白结果 各组细胞中RUNX2、WNT3a、内皮型一氧化氮合酶蛋白表达检测结果,见图9。与对照组相比,CS组、CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组WNT3a蛋白表达分别增加18%,50%,27%,10%和11%,RUNX2蛋白表达分别增加了51%,84%,22%,17%和13%,内皮型一氧化氮合酶蛋白表达增加20%,49%,78%,28%和22%。"
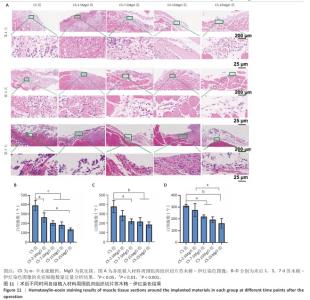
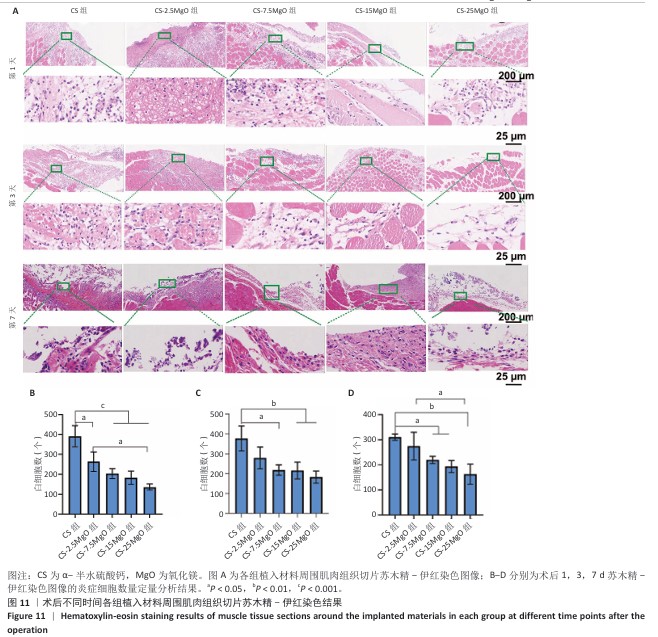
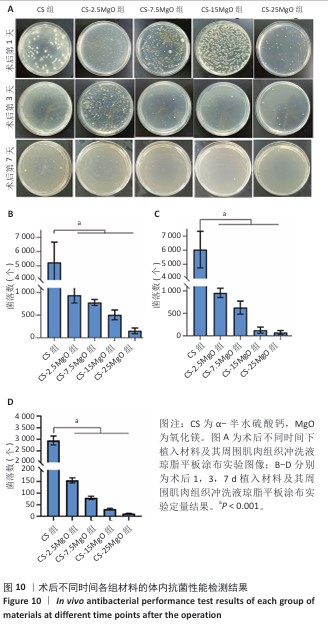
2.8 材料体内抗菌活性检测结果 2.8.1 实验动物数量分析 45只SD大鼠全部进入结果分析。 2.8.2 冲洗液琼脂平板涂布实验结果 各组冲洗液琼脂平板涂布实验结果,见图10。术后相同时间下,与CS组相比,各硫酸钙-氧化镁复合材料组冲洗液中的菌落形成减少,并且随着材料中氧化镁比例的增加逐渐减少。 2.8.3 材料周围肌肉组织苏木精-伊红染色结果 各组材料周围肌肉组织苏木精-伊红染色结果,见图11。 5组材料附近的肌肉细胞未出现坏死或崩解,与其他4组相比,CS组肌肉组织有更严重的炎症细胞聚集和炎性渗出。定量分析结果显示,术后1 d,与CS组相比,CS-2.5MgO组、CS-7.5MgO组、CS-15MgO组和CS-25MgO组术后1 d的白细胞计数分别降低33%,48%,53%和65%,术后3 d的白细胞计数分别降低26%,42%,43%和52%,术后7 d的白细胞计数比CS组分别降低12%,29%,38%和48%。 "
| [1] LI C, LV H, DU Y, et al. Biologically modified implantation as therapeutic bioabsorbable materials for bone defect repair. Regen Ther. 2021;19:9-23. [2] KURIEN T, PEARSON RG, SCAMMELL BE. Bone graft substitutes currentlyavailable in orthopaedic practice: the evidence for their use. Bone Joint J. 2013;95(5): 583-597. [3] 程玮璐,张译丹,刘英慧.骨填充材料的临床应用进展[J].中国医疗器械信息,2023,29(21):43-47. [4] PUTRA NE, MIRZAALI MJ, APACHITEI I, et al. Multi-material additive manufacturing technologies for Ti-,Mg-,and Fe-based biomaterials for bone substitution. Acta Biomater. 2020;16(109):1-20. [5] 谢广渊,潘伟城,谭志斌,等.硫酸钙人工骨在良性骨肿瘤刮除术后骨缺损填充中的愈合情况及其重建作用研究[J].生物骨科材料与临床研究,2017, 14(2):45-48. [6] 杨国敬,林勉,张雷,等.骨损伤修复用硫酸钙及其无机复合材料的研究进展[J].无机材料学报,2013,28(8):795-803. [7] CHAMANSARA A, BEHNAMGHADER A, ZAMANIAN A. Preparation and characterization of injectable gelatin/alginate/chondroitin sulfate/alphacalcium sulfate hemihydrate composite paste for bone repair application. J Biomater Appl. 2022;36(10):1758-1774. [8] ZIRAN BH, SMITH WR, MORGAN SJ. Use of calcium-based demineralized bone matrix/allograft for nonunions and posttraumatic reconstruction of the appendicular skeleton: preliminary results and complications. J Trauma. 2007; 63(6):1324-1328. [9] MORLEY R, ROTHWELL M, STEPHENSON J, et al. Complex foot infection treated with surgical debridement and antibiotic loaded calcium sulfate—a retrospective cohort study of 137 cases. J Foot Ankle Surg. 2022;61(2):239-247. [10] LIU H, LI P, TANG Z, et al. Study on injectable silver-incorporated calcium phosphate composite with enhanced antibacterial and biomechanical properties for fighting bone cement-associated infections. Colloids Surf B Biointerfaces. 2023;227:113382. [11] 倪方方,王博林,宋腾蛟,等.纳米银颗粒的毒性效应及作用机制研究进展[J].中国药理学通报,2016,32(5):593-597. [12] WEI S, MA JX, XU L, et al. Biodegradable materials for bone defect repair. Mil Med Res. 2020;7:54. [13] SAWAI J, IGARASHI H, HASHIMOTO A, et al. Evaluation of growth inhibitory effect of ceramics powder slurry on bacteria by conductance method. J Chem Eng Japan. 1995;28(3):288-293. [14] SAWAI J, YOSHIKAWA T. Quantitative evaluation of antifungal activity of metallic oxide powders (MgO, CaO and ZnO) by an indirect conductimetric assay. J Appl Microbiol. 2004;96(4):803-809. [15] WU X, JIAO Y, JIA C, et al. Joule heating-induced active Mg0 into nano-Mg composites for boosted oxidation and antiviral performance. ACS EST Eng. 2024;4(6):1302-1311. [16] HAYAT S, MUZAMMIL S, RASOOL MH, et al. In vitro antibiofilm and anti‐adhesion effects of magnesium oxide nanoparticles against antibiotic resistant bacteria. Microbiol Immunol. 2018;62(4):211-220. [17] SAWAI J, IGARASHI H, HASHIMOTO A, et al. Evaluation of growth inhibitory effect of ceramics powder slurry on bacteria by conductance method. J Chem Eng Japan. 1995;28(3):288-293. [18] JIN T, HE Y. Antibacterial activities of magnesium oxide (MgO) nanoparticles against foodborne pathogens. J Nanopart Res. 2011;13:6877-6885. [19] LI X, LIU H, HE Y, et al. Enhanced antibacterial effect with MgO nanoplates: Role of oxygen vacancy and alkalinity. Ceram Int. 2024;50(21):42877-42885. [20] LEUNG YH, NG AMC, XU X, et al. Mechanisms of antibacterial activity of MgO: non‐ROS mediated toxicity of MgO nanoparticles towards Escherichia coli. Small. 2014;10(6):1171-1183. [21] YIN Z, LI S, LI X, et al. Effect of pH on the microstructure and antibacterial properties of MgO nanoparticles by microwave-assisted solution combustion. J Alloys Compd. 2024;1009:176858. [22] NIE X, ZHANG X, LEI B, et al. Regulation of magnesium matrix composites materials on bone immune microenvironment and osteogenic mechanism. Front Bioeng Biotechnol. 2022;10:842706. [23] MONFOULET LE, BECQUART P, MARCHAT D, et al. The pH in the microenvironment of human mesenchymal stem cells is a critical factor for optimal osteogenesis in tissue-engineered constructs. Tissue Eng Part A. 2014;20(13-14):1827-1840. [24] ZHANG Y, XU J, RUAN Y, et al. Implant-derived magnesium induces local neuronal production of CGRP to improve bone-fracture healing in rats. Nat Med. 2016;22:1160-1169. [25] CHEN L, ZHU J, GE N, et al. A biodegradable magnesium alloy promotes subperiosteal osteogenesis via interleukin-10-dependent macrophage immunomodulation. Biomaterials. 2025;318:122992. [26] SUN L, LI X, XU M, et al. In vitro immunomodulation of magnesium on monocytic cell toward anti-inflammatory macrophages. Regen Biomater. 2020;7(4):391-401. [27] 中国居民膳食常量元素参考摄入量[C]//中国营养学会微量元素营养分会.中国营养学会微量元素营养第十二次学术会议暨第六届微量元素营养分会会员大会论文集,2014:164. [28] 赵颖,曾利兰,梁涛.可降解镁基金属的生物相容性研究进展[J].金属学报, 2017,53(10):1181-1196. [29] 张丹.Mg-ZnO-HA可降解镁合金生物材料的生物相容性研究[D].长沙:中南大学,2011. [30] GANDOLFI MG, IACONO F, AGEE K, et al. Setting time and expansion in different soaking media of experimental accelerated calcium-silicate cements and ProRoot MTA. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(6):e39-e45. [31] ZHANG X, WANG Y, GUO J, et al. Comparing two functions for optical density and cell numbers in bacterial exponential growth phase. J Pure Appl Microbiol. 2015;9(1):299-305. [32] ISO 10993-12:2021. Biological evaluation of medical devices - Part 12: Sample preparation and reference materials. International Organization for Standardization, Geneva, Switzerland, 2021. [33] YUAN K, CHAN YJ, KUNG KC, et al. Comparison of osseointegration on various implant surfaces after bacterial contamination and cleaning: a rabbit study. Int J Oral Maxillofac Implants. 2014;29(1):32-40. [34] STRAUB J, SEWING A, WALTER N, et al. Calcium Sulfate Bone Substitutes in Clinical Use: History, Material Properties, Application, and Outlook for the Future. J Biomed Mater Res B Appl Biomater. 2025;113(4):e35555. [35] PETTAUER M, BALDERMANN A, EDER S, et al. Hydration of MgO: reaction kinetics and pH control on brucite crystal morphology. Crystal Growth Design. 2024;24(7):3085-3092. [36] GALOW AM, REBL A, KOCZAN D, et al. Increased osteoblast viability at alkaline pH in vitro provides a new perspective on bone regeneration. Biochem Biophys Rep. 2017;10:17-25. [37] HARADA M, UDAGAWA N, FUKASAWA K, et al. Inorganic pyrophosphatase activity of purified bovine pulp alkaline phosphatase at physiological pH. J Dent Res. 1986;65(2):125-127. [38] 李政垚,刘洁颖,吴狄,等.高镁离子浓度对人骨髓间充质干细胞增殖与成骨分化的影响[J].中华骨与关节外科杂志,2020,13(5): 419-426. [39] QIAO W, WONG KHM, SHEN J, et al. TRPM7 kinase-mediated immunomodulation in macrophage plays a central role in magnesium ion-induced bone regeneration. Nat Commun. 2021;12(1):2885. [40] NYGREN H, CHAUDHRY M, GUSTAFSSON S, et al. Increase of compact bone thickness in rat tibia after implanting MgO into the bone marrow cavity. J Funct Biomater. 2014;5(3):158-166. [41] MATHEW AA, PANONNUMMAL R. ‘Magnesium’-the master cation-as a drug—possibilities and evidences. Biometals. 2021;34(5):955-986. [42] WANG J, MA XY, FENG YF, et al. Magnesium ions promote the biological behaviour of rat calvarial osteoblasts by activating the PI3K/Akt signalling pathway. Biol Trace Elem Res. 2017;179:284-293. [43] RESNICK LM. Cellular calcium and magnesium metabolism in the pathophysiology and treatment of hypertension and related metabolic disorders. Am J Med. 1992;93(2):S11-S20. [44] BURBRIDGE MF, WEST DC, ATASSI G, et al. The effect of extracellular pH on angiogenesis in vitro. Angiogenesis. 1999;3:281-288. [45] LIU W, GUO S, TANG Z, et al. Magnesium promotes bone formation and angiogenesis by enhancing MC3T3-E1 secretion of PDGF-BB. Biochem Biophys Res Commun. 2020;528(4):664-670. [46] SARIS NEL, MERVAALA E, KARPPANEN H, et al. Magnesium: an update on physiological, clinical and analytical aspects. Clin Chim Acta. 2000;294(1-2):1-26. [47] HANG R, TIAN X, QU G, et al. Exosomes derived from magnesium ion—stimulated macrophages inhibit angiogenesis. Biomed Mater. 2022;17(4):045008. |
| [1] | Chen Shichao, Deng Yunyi, Zhao Renshengjie, Yu Ke, Li Guangwen. Antibacterial properties of photocrosslinkable hydrogel loaded with quercetin-silver nanoparticles for infected wounds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8436-8442. |
| [2] |
Zhang Jing, He Liping, Wen Yu, Fu Hang.
Hydrogel loaded with fibroblast exosomes promotes endothelial cell function recovery and diabetic wound healing#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6798-6806.
|
| [3] | He Zhenzhen, Huang Hanji, Wang Jiawei, Xie Qingtiao, Jiang Xianfang. Role of bioscaffolds in the repair of inflammation-driven bone and cartilage destruction and structural damage in temporomandibular joint [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5312-5320. |
| [4] | Wang Jieyan, Yao Jiayi, Xin Yingtong, Zhang Xinwen, Li Riwang, Liu Dahai. Chitosan hydrogel drug delivery system is a safer and more effective solution for treating oral ulcers [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5331-5340. |
| [5] | Chen Ying, Sun Xuheng, Liu Qing, Xiao Cong, Jiang Hongjing, Lin Zhanyi. A serum-free culture medium for the early-stage formation of tissue-engineered vascular grafts [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5093-5102. |
| [6] | Jia Wei, Zhang Mandong, Chen Weiyi, Wang Chenyan, Guo Yuan. Effects of femoral prosthetic materials on artificial knee arthroplasty performance [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1477-1481. |
| [7] | Wang Qian, Li Lu, Shu Jingyuan, Dong Zhiheng, Jin Youshi, Wang Qingshan. Micro-morphology and phase of zirconia-based nano-hydroxyapatite functional gradient biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1517-1521. |
| [8] | Zhou Pengfei, Lin Jing, Chen Yuying, Lin Minkui. Canine dental pulp stem cells-polyglycolic acid scaffold complex for canine periodontal tissue defect [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5526-5531. |
| [9] | Liu Xin, Du Bin, Sun Guangquan, Cao Jinxing, Jiang Xiaohong. Porous beta-tricalcium phosphate-polypyrrole-biotin-icariin composite scaffold promotes recruitment of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5532-5537. |
| [10] | Liao Jian, Huang Xiaolin, Zhou Qian, Huo Hua, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Yunmao, Liang Xing. Calcined bone/chitosan composite promotes osteogenic differentiation of bone marrow mesenchymal stem cells in Sprague-Dawley rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4941-4947. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||