Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7245-7250.doi: 10.12307/2026.810
Previous Articles Next Articles
Effects of platelet-rich fibrin on osteogenic genes and bone microstructure in rats with peri-implant bone defect
Yang Li, Wang Chao, Ma Xuliang, Yao Yao, Wang Ruicong, Zhang Yixuan, Miao Wei
- Department of Oral Implantology, Hebei Eye Hospital, Xingtai 054000, Hebei Province, China
-
Received:2025-08-01Revised:2025-12-23Online:2026-10-08Published:2026-02-09 -
Contact:Wang Chao, MS, Chief physician, Department of Oral Implantology, Hebei Eye Hospital, Xingtai 054000, Hebei Province, China -
About author:Yang Li, MS, Attending physician, Department of Oral Implantology, Hebei Eye Hospital, Xingtai 054000, Hebei Province, China
CLC Number:
Cite this article
Yang Li, Wang Chao, Ma Xuliang, Yao Yao, Wang Ruicong, Zhang Yixuan, Miao Wei. Effects of platelet-rich fibrin on osteogenic genes and bone microstructure in rats with peri-implant bone defect[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7245-7250.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
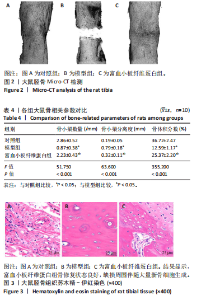
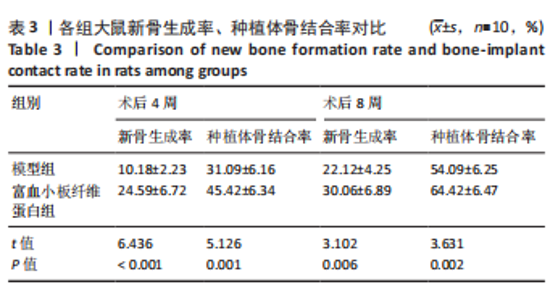
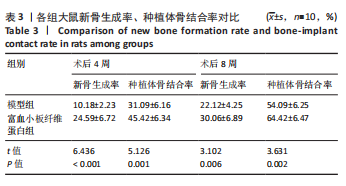
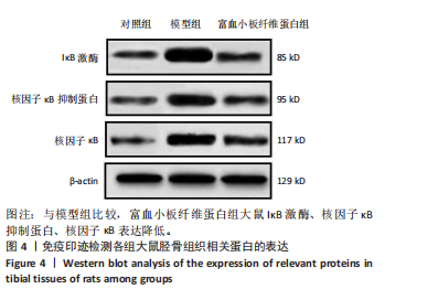
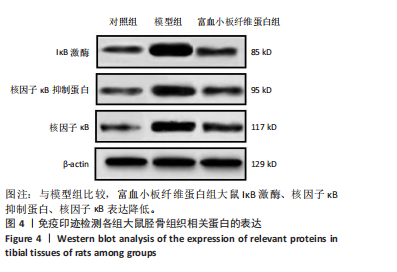
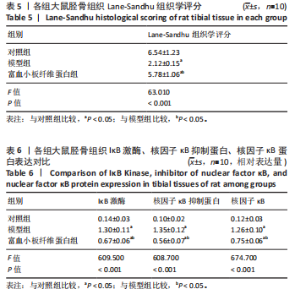
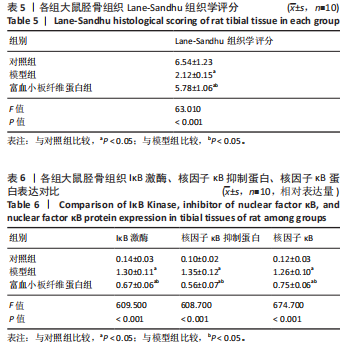
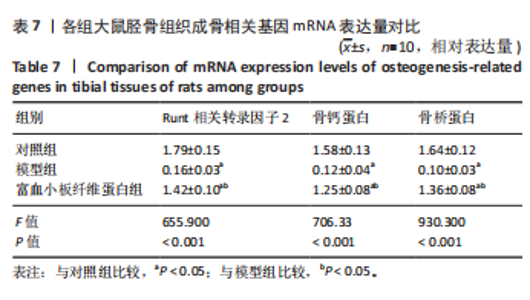
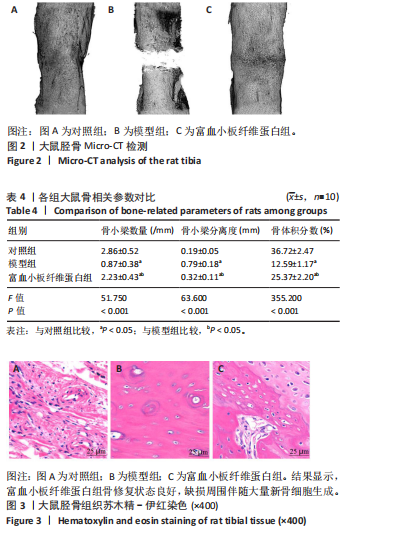
2.3 各组大鼠Micro-CT检测及相关参数对比 Micro-CT检测结果显示,模型组大鼠无显著新生骨组织形成且骨段区域仅伴随少量且分散的骨组织;富血小板纤维蛋白组大鼠伴有一定的新生骨组织生成,并可见部分骨断端相连,见图2。与对照组比较,模型组大鼠骨小梁数量、骨体积分数降低(P < 0.05),骨小梁分离度增高(P < 0.05);与模型组比较,富血小板纤维蛋白组大鼠骨小梁数量、骨体积分数升高(P < 0.05),骨小梁高度降低(P < 0.05),见表4。 2.4 苏木精-伊红染色 结果显示,富血小板纤维蛋白组骨修复状态良好,缺损周围伴随大量新骨细胞生成,说明富血小板纤维蛋白能够加速骨细胞的修复进程并具有良好的骨整合性,见图3。与对照组比较,模型组大鼠Lane-Sandhu组织学评分降低(P < "
| [1] NOMIER AS, GAWEESH ED, TAALAB MR, et al.Efficacy of low-dose cone beam computed tomography and metal artifact reduction tool for assessment of peri-implant bone defects: an in vitro study. BMC Oral Health. 2022;22(1):615. [2] TAŞDEMIR U, KIRTAY M, KELEŞ A, et al. Autogenous Tooth Bone Graft and Simvastatin Combination Effect on Bone Healing. J Craniofac Surg. 2020;31(8): 2350-2354. [3] TASDEMIR U, IYILIKÇI B, AKTÜRK MC, et al. The Effect of Autogenous Bone Graft Mixed With Recombinant Human Vascular Endothelial Growth Factor on Bone Regeneration. J Craniofac Surg. 2021;32(6):2233-2237. [4] BALDWIN P, LI DJ, AUSTON DA, et al. Autograft, Allograft, and Bone Graft Substitutes: Clinical Evidence and Indications for Use in the Setting of Orthopaedic Trauma Surgery. J Orthop Trauma. 2019;33(4):203-213. [5] NOELKEN R, WESTPHAL L, SCHIEGNITZ E,et al.Hard and soft tissue regeneration of severe peri-implantitis defects with the laser-assisted peri-implant defect regeneration technique: 3-year results. Int J Implant Dent. 2023;9(1):3. [6] SCOPELLITI F, CATTANI C, DIMARTINO V, et al.Platelet derivatives and the immunomodulation of wound healing. Int Mol Sci. 2022;23(15):8370. [7] SANE VD, SUNIL NAIR V, JADHAV R,et al. Comparative Evaluation of Efficacy of Platelet Rich Plasma (PRP) and Platelet Rich Fibrin (PRF) in Bone Regeneration after Surgical Removal of Impacted Bilateral Mandibular Third Molars - A Comparative Study. Indian J Otolaryngol Head Neck Surg. 2024;76(1):811-818. [8] 杨景哲,肖长栓,暴海洋,等.富血小板纤维蛋白对难愈合创面炎症反应的影响[J].河北医学,2024,30(4):580-586. [9] RAZI MA, MAHAJAN A, ZARRIN R,et al.Impact of Platelet-Rich Fibrin (PRF) Versus Freeze-Dried Bone Allograft (FDBA) on Peri-Implant Soft and Hard Tissue in Alveolar Ridge Preservation. J Pharm Bioallied Sci. 2024;16(Suppl 4):S3550-S3552. [10] KESIKA P, THANGALEELA S, SISUBALAN N, et al.The Role of the Nuclear Factor-Kappa B (NF-κB) Pathway in SARS-CoV-2 Infection. Pathogens. 2024;13(2):164. [11] HUANG F, WANG H, ZHANG Y, et al.Synergistic Effect of QNZ, an Inhibitor of NF-κB Signaling, and Bone Morphogenetic Protein 2 on Osteogenic Differentiation in Mesenchymal Stem Cells through Fibroblast-Induced Yes-Associated Protein Activation. Int J Mol Sci. 2023;24(9):7707. [12] TESSARO HM, ZAMBOM FFF, FORESTO-NETO O, et al.Chronic Nuclear Factor-Kappa B Inhibition Prevents Experimental Aging Nephropathy. Gerontology. 2024;27(1):1-13. [13] WEI GJ, LIANG X, WU F, et al.Ginkgetin attenuates bone loss in OVX mice by inhibiting the NF-κB/IκBα signaling pathway.PeerJ. 2024;10(1):12:e17722. [14] DAI Y, YI X, HUANG Y, et al.miR-345-3p Modulates M1/M2 Macrophage Polarization to Inhibit Inflammation in Bone Infection via Targeting MAP3K1 and NF-κB Pathway.J Immunol. 2024;212(5):844-854. [15] 谢亚明,周伟. 引导骨组织再生技术对牙颊侧轻度骨缺损患者种植成功率及口腔健康状况的影响[J]. 医学美学美容,2024,33(21):122-125. [16] 翁忠如,朱丽婷,周谷满.引导骨组织再生技术对牙缺失伴骨缺损患者种植修复效果及美观度的影响分析[J].慢性病学杂志,2024,25(5):784-787. [17] 季明意,季欣意,徐俊峰.褪黑素促进骨再生机制及在口腔种植中的应用[J].中国组织工程研究,2025,29(18):3868-3876. [18] 郭佳慧,焦冰玉.富血小板纤维蛋白(PRF)在急性跟腱断裂经皮微创修复手术中的应用效果评价[J].黑龙江科学,2024,15(22):131-133. [19] HILMY F, DILOGO IH, REKSODIPUTRO MH, et al.Evaluation of adipose-derived stem cells (ASCS) exosome implantation and platelet-rich fibrin (PRF) on critical long bone defects in Sprague-Dawley rats. Eur J Orthop Surg Traumatol. 2024; 34(5):2805-2810. [20] 李谊,胡浩磊,张秀静,等.3D生物打印自体骨结合骨髓间充质干细胞修复兔鼻骨骨缺损的基础研究[J].生物医学工程与临床,2025,29(3):291-299. [21] WANG T , HUOJIA M , LI J , et al Experimental study of transforming growth factor-β3 combined with dental pulp stem cells in promoting the implant’s osseointegration. Zhonghua Kou Qiang Yi Xue Za Zhi. 2017;52(6):367-373. [22] 牛婉琼,汪振华,周志伟.n-HA/壳聚糖/米诺环素复合支架在种植体周围炎骨缺损模型中的应用效果评价[J].上海口腔医学,2024,33(5):449-454. [23] 张昊,刘传宏,于苗苗,等.钛网结合Masquelet技术治疗创伤后干骺端大段骨缺损临床效果分析[J].临床军医杂志,2023,51(1):8-11. [24] 张松,陈步国,唐兆麟,等. Masquelet技术结合游离皮瓣移植技术在手部软组织伴掌骨缺损中的可行性及临床疗效[J]. 中华手外科杂志,2024,40(5):426-430. [25] LUCA C, MARCO F, BENEDETTA S, et al.Is core decompression and bone marrow concentrate with demineralized bone matrix and platelet-rich fibrin suitable for treating femoral head osteonecrosis?. J Hip Preserv Surg. 2024;11(4):263-270. [26] 宁晔,唐丽宇,龚飞飞,等.富血小板纤维蛋白联合Bio-Oss骨粉修复颌骨囊肿术后骨缺损的疗效[J].蚌埠医学院学报,2024,49(2):187-191. [27] 郭佳慧,焦冰玉.富血小板纤维蛋白(PRF)在急性跟腱断裂经皮微创修复手术中的应用效果评价[J].黑龙江科学,2024,15(22):131-133. [28] 柴纪华,汤明语,苗德田,等.冻干水平富血小板纤维蛋白(Ly-H-PRF)通过调控巨噬细胞M2型极化促进伤口愈合[J].临床口腔医学杂志,2024,40(11):648-652. [29] GAUTAM K, KAPOOR A, MATHUR S, et al.Comparative Evaluation of Autogenous Bone Graft and Autologous Platelet-Rich Fibrin with and Without 1.2 mg in situ Rosuvastatin Gel in the Surgical Treatment of Intrabony Defect in Chronic Periodontitis Patients.Contemp Clin Dent. 2022;13(1):69-77. [30] 黄艳玲,董樾,侯团结,等.脂肪间充质干细胞联合可注射富血小板纤维蛋白修复耳软骨缺损的实验研究[J].中国美容整形外科杂志,2024,35(11):665-669+696. [31] GAN S, ZHENG Z, ZHANG M. Lyophilized Platelet-Rich Fibrin Exudate-Loaded Carboxymethyl Chitosan/GelMA Hydrogel for Efficient Bone Defect Repair. ACS Appl Mater Interfaces. 2023;15(22):26349-26362. [32] BIN L , CEWEN H , XINYUE H ,et al.3D printing nacre powder/sodium alginate scaffold loaded with PRF promotes bone tissue repair and regeneration. Biomater Sci. 2024;12(9):2418-2433. [33] 梁玉祥,郎志刚,郭强,等.富血小板纤维蛋白联合泡沫敷料治疗慢性感染伤口的疗效及抑菌作用分析[J].实用临床医药杂志,2023,27(21):69-72. [34] 王婷,何俐.富白细胞-血小板纤维蛋白在大鼠牙髓修复中的应用[J].实用医学杂志,2024,40(14):1941-1946. [35] 宁晔,唐丽宇,龚飞飞,等.富血小板纤维蛋白联合Bio-Oss骨粉修复颌骨囊肿术后骨缺损的疗效[J].蚌埠医学院学报,2024,49(2):187-191. [36] 李文怡,张琬琳,肖西峰.富血小板纤维蛋白应用于子宫内膜损伤修复研究进展[J].中国妇产科临床杂志,2025,26(2):156-158. [37] 王世振,周军,李荣荣,等.磷酸钙修饰钛植入物结合富血小板血浆促进骨质疏松大鼠种植体骨结合的实验研究[J].临床和实验医学杂志,2023, 22(3):237-242. [38] 诸建华.富血小板纤维蛋白治疗慢性创面的应用价值探讨[J].中国实用乡村医生杂志,2024,31(8):72-74+78. [39] 高也,芦帅,孙勇.β-磷酸三钙复合改良型富血小板纤维蛋白对兔成骨细胞增殖与分化的影响[J].重庆医学,2023,52(24):3686-3692. [40] TSENG KF, SHIU ST, HUNG CY, et al.Osseointegration Potential Assessment of Bone Graft Materials Loaded with Mesenchymal Stem Cells in Peri-Implant Bone Defects. Int J Mol Sci. 2024;25(2):862. [41] MITO K, LACHNISH J, YAO CJ. Scaffold-Free Bone Marrow-Derived Mesenchymal Stem Cell Sheets Enhance Bone Formation in a Weight-Bearing Rat Critical Bone Defect Model. Tissue Eng Part A. 2024;30(3-4):107-114. [42] 徐云容,王志华,段浩,等.锶掺杂生物玻璃/丝素蛋白复合支架在骨质疏松骨缺损修复中的应用机制及其可能临床价值[J].骨科,2025,16(1):77-81. [43] 姚文超,陈垍航,蒋臻,等.丹参提取物对感染性骨缺损大鼠炎症水平的影响[J].中华医院感染学杂志,2024,34(3):400-404. [44] 卡米尔.富白细胞-血小板纤维蛋白对软组织愈合及炎症调节的研究[D]. 长春:吉林大学,2019. [45] DESAI K, THAKKAR R, PATEL J, et al.Comparative evaluation of DFDBA versus PRF with DFDBA in treatment of grade-II furcation defects–A clinical trial. J Pharm Bioallied Sci. 2024;16(1):S794-S796. [46] 张磊.KGN联合富血小板血浆通过抑制炎症反应促进前交叉韧带重建术后腱骨愈合和应用加强线技术提高其临床疗效的研究[D].石家庄:河北医科大学,2023. [47] 李名禄,马兵.富血小板血浆通过NF-κB p65/BMP-7信号通路促进骨髓基质干细胞成骨分化的研究[J].临床口腔医学杂志,2022,38(2):78-81. [48] 张悦,董红宾,张玮,等.Bio-Oss骨复合富血小板纤维蛋白修复兔牙周骨缺损[J].中华口腔医学研究杂志(电子版),2020,14(5):280-287. [49] 柴纪华,汤明语,苗德田,等.冻干水平富血小板纤维蛋白(Ly-H-PRF)通过调控巨噬细胞M2型极化促进伤口愈合[J].临床口腔医学杂志,2024,40(11):648-652. [50] 范文娟,杨明,曹直,等.富血小板纤维蛋白联合柚皮苷对牙髓干细胞成骨分化及BMPs-ERK5通路的影响[J].中国老年学杂志,2024,44(8):1907-1911. |
| [1] | Li Zhenyu, Zhang Siming, Bai Jiaxiang, Zhu Chen. Osthole improves osteogenic differentiation function of bone marrow mesenchymal stem cells under high-glucose conditions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1641-1648. |
| [2] | Yan Chengbo, Luo Qiuchi, Fan Jiabing, Gu Yeting, Deng Qian, Zhang Junmei. Effect of type 2 diabetes mellitus on orthodontic tooth movement and bone microstructure parameters on the tension side in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 824-831. |
| [3] | Zhan Lei, Wu Lina, Li Huan, Liu Min, Chen Tao, Pu Xiaobing, Zhou Changchun. Silk fibroin hydrogel loaded with icariin to promote tendon-bone healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5178-5787. |
| [4] | Du Chuanjia, Jiao Jiao, Zhou Jihe, Wang Shuai, Liu Guohui, Wang Yang, Shang Huayu, Wang Huiming, Zhang Xiaopei. Influence of different trabecular bone volume fractions on crack propagation in femoral neck fractures: a finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(15): 3753-3759. |
| [5] | Wei Dongdong, Shi Meikun, Wang Lei. Gelatin methacryloyl hydrogel loaded with injectable platelet-rich fibrin promotes skin wound repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(14): 3643-3651. |
| [6] | Zhao Hongxia, Sun Zhengwei, Han Yang, Wu Xuechao , Han Jing. Osteogenic properties of platelet-rich fibrin combined with gelatin methacryloyl hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 809-817. |
| [7] | Wang Jingshuai, Zhang Xiaotong, Zhang Yange, Wan Zedong, Kong Lingwei, Cao Haiying, Jin Yu. Comparison of Mg-Li-Gd alloy and stainless steel intramedullary nail for fixation of femoral annular hemi-defects in rats [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(34): 7261-7268. |
| [8] | Wang Kaigang, Hao Dongsheng, Ma Pei, Zhou Shuo, Li Ruimin. Comparison of efficacy of different biological scaffolds for pulp regeneration therapy in immature permanent teeth: a Bayesian network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(34): 7447-7460. |
| [9] | Zhang Zhibo, Wang Zhaolin, Wang Zhigang, Li Peng, Jiang Jianhao, Zhang Kai, Yang Shuye, Du Gangqiang. Ilizarov bone transport combined with antibiotic bone cement promotes junction healing of large tibial bone defect [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(10): 2038-2043. |
| [10] | Liu Zilue, Wang Zhi, Song Wenshang, Li Suna, Cai Shixin. Autogenous bone and platelet-rich fibrin in repair of severe alveolar bone defects [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(10): 2044-2051. |
| [11] | Hou Zengtao, Dong Zhiwei, Zhang Jinfeng, Yang Xiaohui, Fan Xiao. Platelet-rich fibrin regulates apoptosis to promote cartilage repair in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5167-5171. |
| [12] | Wang Xinmin, Yan Wenkai, Song Yahui, Liu Fei. Leukocyte- and platelet-rich fibrin with autologous hamstring tendon for traumatic patella dislocation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 404-410. |
| [13] | Xi Hongzhong, Liu Xin, Sun Guangquan, Du Bin, Yuan Xinwei, Huang Yixuan, Guo Mingbin, Mai Jianbin. Application of bioelectric effect materials in design of bone tissue engineering scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3569-3575. |
| [14] | Wang Xu, Wu Yajie, Zhang Xinfu, Shi Zhi, Yang Tengyun, Xiong Bohan, Lu Xiaojun, Zhao Daohong. Expression and action mechanism of stromal cell-derived factor 1 in tendon-bone healing of rabbit rotator cuff [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 3049-3054. |
| [15] | Wang Xiaolong, Huang Haoran, Zhang Zhongxin, Wang Limin, Hu Yongcheng. Biomaterials and pore characteristics of tendon tissue engineering scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2398-2403. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||