Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7237-7244.doi: 10.12307/2026.680
Function and molecular mechanism of physcion in regulating bone homeostasis
Qi Yuxin1, 2, Dang Yifan1, 2, Dai Liming2, Zhang Xiaoling1, 2
- 1Collaborative Innovation Center of Regenerative Medicine and Medical BioResource Development and Application Co-constructed by the Province and Ministry, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China; 2Department of Orthopedics, Xinhua Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 200092, China
-
Received:2025-06-26Revised:2025-10-20Online:2026-10-08Published:2026-02-07 -
Contact:Zhang Xiaoling, Professor, Doctoral supervisor, Collaborative Innovation Center of Regenerative Medicine and Medical BioResource Development and Application Co-constructed by the Province and Ministry, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China; Department of Orthopedics, Xinhua Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 200092, China -
About author:Qi Yuxin, MS candidate, Collaborative Innovation Center of Regenerative Medicine and Medical BioResource Development and Application Co-constructed by the Province and Ministry, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:National Natural Science Foundation of China, No. 32370892 (to ZXL)
CLC Number:
Cite this article
Qi Yuxin, Dang Yifan, Dai Liming, Zhang Xiaoling. Function and molecular mechanism of physcion in regulating bone homeostasis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7237-7244.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
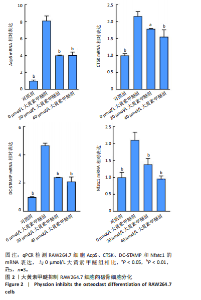
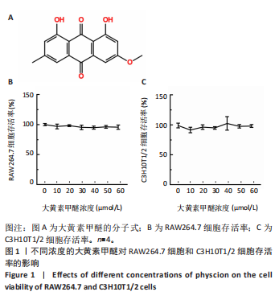
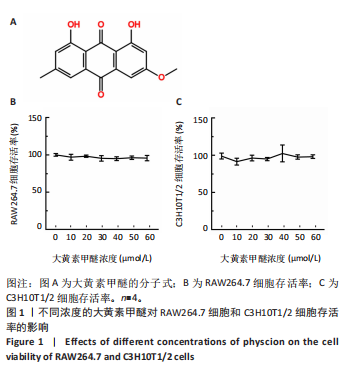
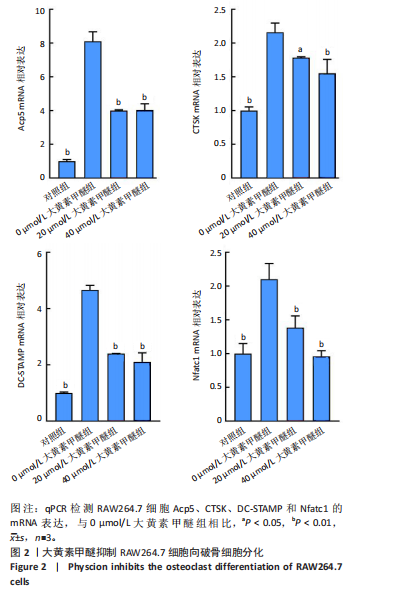
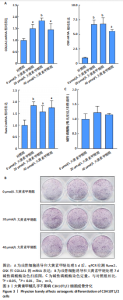
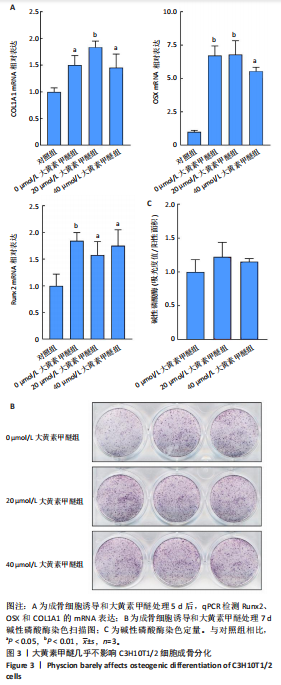
2.2 大黄素甲醚抑制破骨响应 qPCR检测结果显示,与对照组相比,添加大黄素甲醚处理的各组能显著降低破骨分化相关基因Acp5、CTSK、DC-STAMP、Nfatc1(P < 0.05)的表达,见图2。结果提示,大黄素甲醚可以抑制小鼠骨髓巨噬细胞向破骨细胞分化。 2.3 大黄素甲醚对成骨诱导无响应 qPCR检测成骨分化相关标志基因OSX、COL1A1、RunX2的表达[35]。结果显示,大黄素甲醚处理的各组之间OSX、COL1A1、RunX2基因表达差异无显著性意义(P > 0.05),见图3A。碱性磷酸酶染色检测成骨诱导7 d后矿化结节的形成,结果同样显示,大黄素甲醚处理的各组之间差异无显著性意义,见图3B,C。结果提示,大黄素甲醚对骨髓间充质干细胞向成骨细胞分化可能没有影响。 2.4 大黄素甲醚药物靶点、破骨细胞和骨质疏松症相关基因的筛选 根据上述结果,大黄素甲醚能调控骨平衡中的破骨细胞分化。因此,在Genecard数据库和OMIM数据库中以“osteoclast”和“osteoporosis”为关键词,获得相关的疾病靶点。通过"
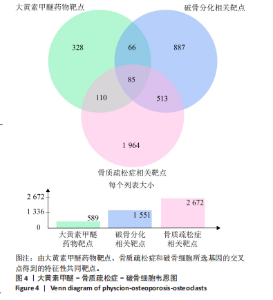
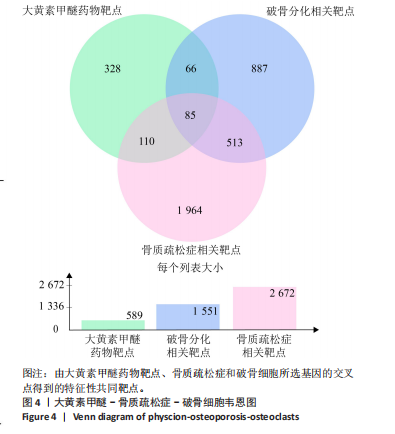
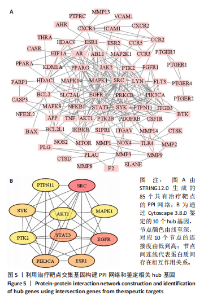
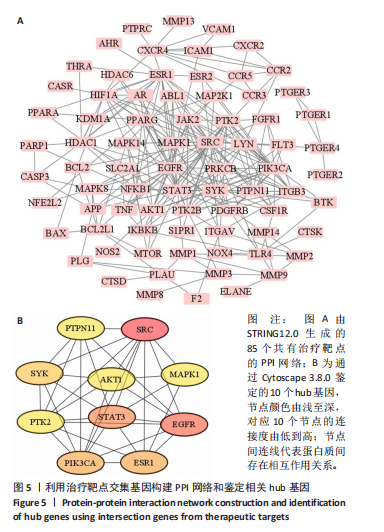
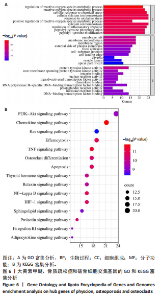
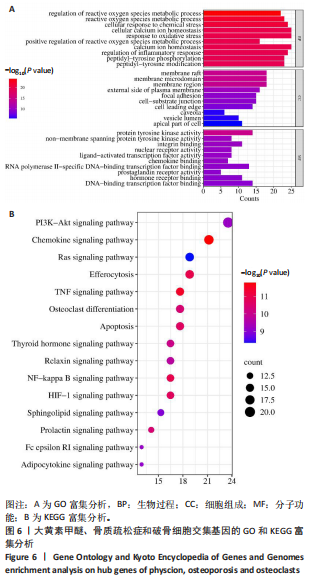
super-Pred、CTD、STP和TargetNet等数据库,共确定了589个药物靶点基因。利用韦恩图将药物作用靶点与疾病相关靶点比对交集,确定了85个共有治疗靶点基因,见图 4。 2.5 PPI网络的构建与核心靶点筛选 将药物与疾病交集的85个基因导入String数据库中,将置信度设置为中等置信度0.4,在移除网络中的游离节点后,构建了一个包含85个节点和257条边的网络,见图5A。随后,将STRING数据库中获得的PPI网络数据导入Cytoscape 3.8.0软件,并应用插件cytohubba进行筛选与可视化处理。通过degree算法,识别出Top10的hub靶点:SRC、PIK3CA、EGFR、STAT3、PTK2、PTPN11、AKT1、ESR1、SYK、MAPK1,见图5B。 2.6 GO与KEGG通路富集分析 针对85个共有靶点进行了GO功能富集分析。从生物过程、细胞组成和分子功能3个维度中,各挑选了前10位的显著条目。GO功能图表显示靶点与活性氧代谢调节过程、氧化应激反应等生物过程相关,见图6A。通过KEGG富集分析,一共获得了145条相关通路,选取与骨质疏松症关系最相关的15条通路绘制条形图,其中包含磷脂酰肌醇-3"
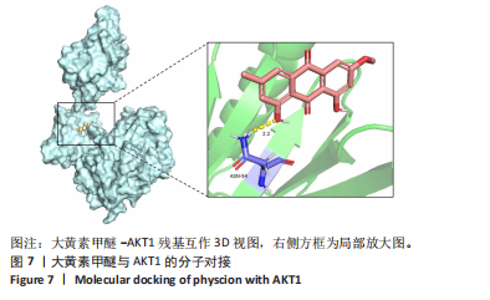
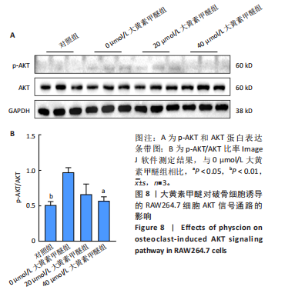
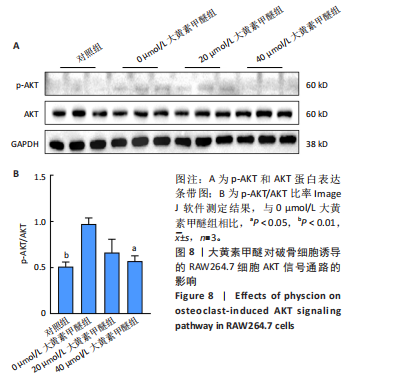
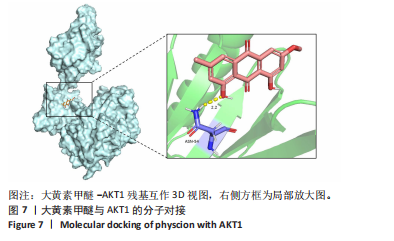
2.7 大黄素甲醚与AKT1的分子对接结果 利用PDB数据库筛选得到AKT1的3D结构 (PDB ID:7apj),与大黄素甲醚进行分子对接,两者的结合口袋形成了合适的空间互补。在结合口袋内,大黄素甲醚与 AKT1的氨基酸残基ASN54形成氢键,结合能为 -10.72 kJ/mol,见图7。一般情况下,当结合能 < 0 J/mol ,认为受配体对之间能自发结合[37]。因此认为大黄素甲醚与AKT1具有一定的结合稳定性。 2.8 大黄素甲醚通过AKT途径抑制破骨细胞分化 根据hub靶点和KEGG通路富集分析,猜测大黄素甲醚的作用可能与AKT1及PI3K-AKT通路相关,并且分子对接结果预测两者存在相互结合影响。 由于AKT是破骨细胞分化的关键,因此验证AKT的磷酸化水平[38]。 Western Blot检测结果显示,与对照组相比,0 μmol/L大黄素甲醚组RAW264.7细胞的p-AKT/AKT显著上升(P < 0.05);而与0 μmol/L大黄素甲醚组比,20 μmol/L大黄素甲"
| [1] BERENDSEN AD, OLSEN BR. Bone development. Bone. 2015;80:14-18. [2] SONG S, GUO Y, YANG Y, et al. Advances in pathogenesis and therapeutic strategies for osteoporosis. Pharmacol Ther. 2022;237:108168. [3] TETI A, WHYTE MP. Osteopetrosis: Gene-based nosology and significance. Bone. 2023;175:116854. [4] WALKER MD, SOLOMON CG, SHANE E. Postmenopausal Osteoporosis. N Engl J Med. 2023;389(21):1979-1991. [5] CHENG CH, CHEN LR, CHEN KH. Osteoporosis Due to Hormone Imbalance: An Overview of the Effects of Estrogen Deficiency and Glucocorticoid Overuse on Bone Turnover. Int J Mol Sci. 2022;23(3):1376. [6] GAO S, ZHAO Y. Quality of life in postmenopausal women with osteoporosis: a systematic review and meta-analysis. Qual Life Res. 2023;32(6):1551-1565. [7] SALARI N, DARVISHI N, BARTINA Y, et al. Global prevalence of osteoporosis among the world older adults: a comprehensive systematic review and meta-analysis. J Orthop Surg Res. 2021;16(1):669. [8] REID IR, BILLINGTON EO.Drug therapy for osteoporosis in older adults. Lancet. 2022;399(10329):1080-1092. [9] WANG LT, CHEN LR, CHEN KH. Hormone-Related and Drug-Induced Osteoporosis: A Cellular and Molecular Overview. Int J Mol Sci. 2023;24(6):5814. [10] GEHRKE B, ALVES COELHO MC, BRASIL D’ALVA C, et al. Long-term consequences of osteoporosis therapy with bisphosphonates. Arch Endocrinol Metab. 2023;68: e220334. [11] LI H, XIAO Z, QUARLES LD, et al. Osteoporosis: Mechanism, Molecular Target and Current Status on Drug Development. Curr Med Chem. 2021;28(8):1489-1507. [12] MUKWAYA E, XU F, WONG MS, et al. Chinese herbal medicine for bone health. Pharm Biol. 2014;52(9):1223-1228. [13] ZHOU G, ZHANG X, GU Z, et al. Research progress on the treatment of knee osteoarthritis combined with osteoporosis by single-herb Chinese medicine and compound.Front Med (Lausanne). 2023;10:1254086. [14] ZHAO J, ZHANG Q, ZOU G, et al. Arenobufagin, isolated from toad venom, inhibited epithelial-to-mesenchymal transition and suppressed migration and invasion of lung cancer cells via targeting IKKβ/NFκB signal cascade. J Ethnopharmacol. 2020;250:112492. [15] SUN C, CHEN X, YANG S, et al. LBP1C-2 from Lycium barbarum alleviated age-related bone loss by targeting BMPRIA/BMPRII/Noggin. Carbohydr Polym. 2023;310:120725. [16] HUANG H, CHEN J, LIN X, et al. Morinda Officinalis Polysaccharides Inhibit Osteoclast Differentiation by Regulating miR-214-3p/NEDD4L in Postmenopausal Osteoporosis Mice. Calcif Tissue Int. 2024;115(5): 673-685. [17] SOOMRO NA, WU Q, AMUR SA, et al. Natural drug physcion encapsulated zeolitic imidazolate framework, and their application as antimicrobial agent. Colloids Surf B Biointerfaces. 2019;182:110364. [18] TRYBUS W, KR L T, TRYBUS E, et al. Physcion Induces Potential Anticancer Effects in Cervical Cancer Cells. Cells. 2021;10(8):2029. [19] DONG X, WANG L, SONG G, et al.Physcion Protects Rats Against Cerebral Ischemia-Reperfusion Injury via Inhibition of TLR4/NF-kB Signaling Pathway. Drug Des Devel Ther. 2021;15:277-287. [20] LEE SJ, CHO SJ, KWON EY, et al. Physcion reduces lipid accumulation and prevents the obesity in mice. Nutr Metab (Lond). 2019;16:31. [21] 杨璐, 赵国宏, 白莉敏. 大黄素甲醚通过调节RAGE/MAPK信号通路减轻糖尿病性骨质疏松损伤 [J]. 山西医科大学学报,2023,54(5): 607-615. [22] 李利华, 李欢. 大黄活性成分大黄素甲醚联合振动训练对骨质疏松大鼠骨生物力学和骨代谢的影响[J].分子植物育种,2024,22(19): 6485-6492. [23] YIN P, HUANG J, YANG K, et al. Myricitrin versus EGCG in the Treatment of Obesity: Target Mining and Molecular Mechanism Exploring based on Network Pharmacology.Curr Pharm Des. 2023;29(24):1939-1957. [24] XIAOYING M, ZHIMING H, TAO Y, et al. Elucidating the molecular mechanisms underlying anti-inflammatory effects of Morchella esculenta in the arachidonic acid metabolic pathway by network pharmacology and molecular docking. Sci Rep. 2023;13(1):15881. [25] SADAQAT M, QASIM M, TAHIR UL QAMAR M, et al. Advanced network pharmacology study reveals multi-pathway and multi-gene regulatory molecular mechanism of Bacopa monnieri in liver cancer based on data mining, molecular modeling, and microarray data analysis. Comput Biol Med. 2023;161:107059. [26] JI L, SONG T, GE C, et al. Identification of bioactive compounds and potential mechanisms of scutellariae radix-coptidis rhizoma in the treatment of atherosclerosis by integrating network pharmacology and experimental validation. Biomed Pharmacother. 2023;165:115210. [27] WANG Y, YUAN Y, WANG W, et al. Mechanisms underlying the therapeutic effects of Qingfeiyin in treating acute lung injury based on GEO datasets, network pharmacology and molecular docking. Comput Biol Med. 2022;145:105454. [28] LU S, SUN X, ZHOU Z, et al. Mechanism of Bazhen decoction in the treatment of colorectal cancer based on network pharmacology, molecular docking, and experimental validation.Front Immunol. 2023;14:1235575. [29] ZOU J, XU W, LI Z, et al. Network pharmacology-based approach to research the effect and mechanism of Si-Miao-Yong-An decoction against thromboangiitis obliterans. Ann Med. 2023;55(1):2218105. [30] KIM JH, SEONG S, KIM K, et al. Rac1-dependent regulation of osteoclast and osteoblast differentiation by developmentally regulated GTP-binding 2. Cell Death Discov. 2025;11(1):48. [31] LIU RX, GU RH, LI ZP, et al. Trim21 depletion alleviates bone loss in osteoporosis via activation of YAP1/β-catenin signaling. Bone Res. 2023;11(1):56. [32] LIU F, YUAN Y, BAI L, et al. LRRc17 controls BMSC senescence via mitophagy and inhibits the therapeutic effect of BMSCs on ovariectomy-induced bone loss. Redox Biol. 2021;43:101963. [33] WU Z, ZHU J, WEN Y, et al. Hmga1‐overexpressing lentivirus protects against osteoporosis by activating the Wnt/β‐catenin pathway in the osteogenic differentiation of BMSCs.FASEB J. 2023;37(9):e22987. [34] WANG L, LUO R, ZHAO YX, et al.[Ergosterol peroxide inducing apoptosis of human hepatocellular carcinoma by regulating mitochondrial apoptosis pathway]. Zhongguo Zhong Yao Za Zhi. 2024;49(12):3365-3372. [35] QI JL, ZHANG ZD, DONG Z, et al. mir-150-5p inhibits the osteogenic differentiation of bone marrow-derived mesenchymal stem cells by targeting irisin to regulate the p38/MAPK signaling pathway. J Orthop Surg Res. 2024;19(1):190. [36] LI J, WEI JJ, WU CH, et al. Epimedin A inhibits the PI3K/AKT/NF-κB signalling axis and osteoclast differentiation by negatively regulating TRAF6 expression.Mol Med. 2024;30(1):125. [37] SUN Z, WANG Y, PANG X, et al.Mechanisms of polydatin against spinal cord ischemia–reperfusion injury based on network pharmacology, molecular docking and molecular dynamics simulation. Bioorg Chem. 2023;140:106840. [38] CHEN X, CHEN X, CHAO R, et al. Dlk2 interacts with Syap1 to activate Akt signaling pathway during osteoclast formation. Cell Death Dis. 2023;14(9):589. [39] JIN Y, ZHOU BH, ZHAO J, et al. Fluoride-induced osteoporosis via interfering with the RANKL/RANK/OPG pathway in ovariectomized rats: Oophorectomy shifted skeletal fluorosis from osteosclerosis to osteoporosis.Environ Pollut. 2023;336:122407. [40] MARINI F, GIUSTI F. Role of Wnt signaling and sclerostin in bone and as therapeutic targets in skeletal disorders. Osteoporos Int. 2023;34(2):213-238. [41] CHAI S, YANG Y, WEI L, et al. Luteolin rescues postmenopausal osteoporosis elicited by OVX through alleviating osteoblast pyroptosis via activating PI3K-AKT signaling.Phytomedicine. 2024;128:155516. [42] KIM JM, LIN C, STAVRE Z, et al. Osteoblast-Osteoclast Communication and Bone Homeostasis. Cells. 2020;9(9):2073. [43] RACHNER TD, KHOSLA S, HOFBAUER LC. Osteoporosis: now and the future. Lancet. 2011;377(9773):1276-1287. [44] LINDSAY R, KREGE JH, MARIN F, et al. Teriparatide for osteoporosis: importance of the full course]. Osteoporos Int. 2016;27(8):2395-2410. [45] CUI Z, ZHAO X, AMEVOR FK, et al. Therapeutic application of quercetin in aging-related diseases: SIRT1 as a potential mechanism. Front Immunol. 2022; 13:943321. [46] HU X, WANG Z, WANG W, et al. Irisin as an agent for protecting against osteoporosis: A review of the current mechanisms and pathways. J Adv Res. 2024;62:175-186. [47] JI W, LIU S, ZHAO X, et al. Triptolide inhibits proliferation, differentiation and induces apoptosis of osteoblastic MC3T3-E1 cells. Mol Med Rep. 2017; 16(5):7391-7397. [48] 陈俊泽, 俞辉, 肖进. 雷公藤甲素通过抑制破骨细胞生成预防骨丢失的研究[J].中国临床解剖学杂志,2023,41(1):77-80. [49] INCHINGOLO AD, INCHINGOLO AM, MALCANGI G. Effects of Resveratrol, Curcumin and Quercetin Supplementation on Bone Metabolism-A Systematic Review. Nutrients. 2022;14(17): 3519. [50] CHENG S, SHAO H, YIN D, et al. Molecular Mechanism Underlying the Action of a Celastrol-Loaded Layered Double Hydroxide-Coated Magnesium Alloy in Osteosarcoma Inhibition and Bone Regeneration. ACS Biomater Sci Eng. 2023;9(8):4940-4952. [51] 刘磊,林家茂,王荣.大黄有效成分抗肿瘤作用的研究进展[J]. 中成药, 2024,46(12):4070-4074. [52] 吴凌凤,谢华松.不同产地巴戟天药材中大黄素甲醚的含量测定[J]. 山东化工,2017,46(14):76-77,79. |
| [1] | Wen Guangwei, Zhen Yinghao, Zheng Taikeng, Zhou Shuyi, Mo Guoye, Zhou Tengpeng, Li Haishan, Lai Yiyi. Effects and mechanisms of isoginkgetin on osteoclastogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1348-1358. |
| [2] | Xu Jiamu, Yang Cheng, Li Weimin, Wang Chunqing. Role and pathogenesis of pyroptosis and inflammatory factors in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 691-700. |
| [3] | Zhang Hongrui, Wu Ruiqi, Wang Wenchi, Peng Qinglin, Cui Wei. Molecular mechanisms of active compounds from Tripterygium wilfordii in prevention and treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7332-7339. |
| [4] | Gao Jiabin, Li Tianqi, Xu Kun, Zhu Hanmin, Zhou Xi, Li Wei. Mitophagy regulates osteoclasts: a new perspective for osteoporosis treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5982-5991. |
| [5] | Wu Lingjie, Zheng Kaiyuan, Wang Guangrong, Yin Chong . Strategies for the application of miRNA-targeted therapy in the treatment of osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5792-5803. |
| [6] | Han Jie, Hu Tianfa, Wu Yachao, Nong Bin, Yu Kailong. Forkhead box transcription factor O3 affects bone metabolism and participates in the pathological processes of various bone-related diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5770-5781. |
| [7] | Zhang Ye, An Zheqing, Xi Xin, Liu Xiaoyan, Hong Wei, Liao Jian. Zoledronic acid-loaded dissolvable microneedle patch inhibits lipopolysaccharide-induced osteoclast differentiation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5115-5124. |
| [8] | Li Jiayin, Sui Lei, Li Yanjing. microRNA-146a regulates bone metabolism and its application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4702-4712. |
| [9] | Han Yapeng, Gao Jun, Niu Yunwei, Deng Enjia. Mechanism of programmed cell death mediated by total flavonoids of Rhizoma Drynariae [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(12): 3091-3099. |
| [10] | Sun Long, Wu Haiyang, Tong Linjian, Liu Rui, Yang Weiguang, Xiao Jian, Liu Lice, Sun Zhiming. Regulatory mechanism of leptin in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(12): 3100-3108. |
| [11] | Yu Manya, Cui Xing. Contribution and interaction of various cells in bone marrow microenvironment to exosomal circular RNA associated with multiple myeloma bone disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 101-110. |
| [12] | Han Haihui, Ran Lei, Meng Xiaohui, Xin Pengfei, Xiang Zheng, Bian Yanqin, Shi Qi, Xiao Lianbo. Targeting fibroblast growth factor receptor 1 signaling to improve bone destruction in rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(9): 1905-1912. |
| [13] | Zhao Jiyu, Wang Shaowei. Forkhead box transcription factor O1 signaling pathway in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(9): 1923-1930. |
| [14] | Zhu Hanmin, Wang Song, Xiao Wenlin, Zhang Wenjing, Zhou Xi, He Ye, Li Wei, . Mitophagy regulates bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1676-1683. |
| [15] | Wang Wentao, Hou Zhenyang, Wang Yijun, Xu Yaozeng. Apelin-13 alleviates systemic inflammatory bone loss by inhibiting macrophage M1 polarization [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1548-1555. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||