Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6337-6344.doi: 10.12307/2026.185
Previous Articles Next Articles
Exercise-intestinal flora and aging
Wang Yinfeng, Yao Lijuan, Ma Zhennan, Chen Leqin
- Shanxi Normal University, Taiyuan 030031, Shanxi Province, China
-
Received:2025-07-11Revised:2025-08-22Online:2026-08-28Published:2026-02-04 -
Contact:Chen Leqin, Professor, Shanxi Normal University, Taiyuan 030031, Shanxi Province, China -
About author:Wang Yinfeng, MS, Shanxi Normal University, Taiyuan 030031, Shanxi Province, China
CLC Number:
Cite this article
Wang Yinfeng, Yao Lijuan, Ma Zhennan, Chen Leqin. Exercise-intestinal flora and aging[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6337-6344.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
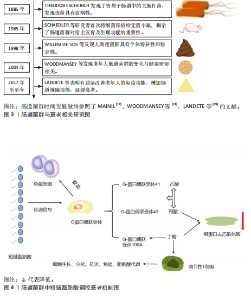
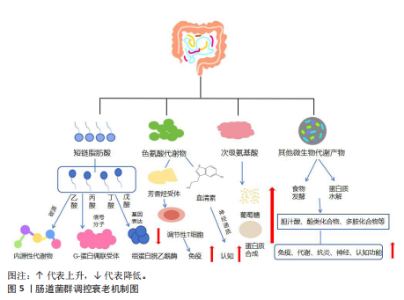
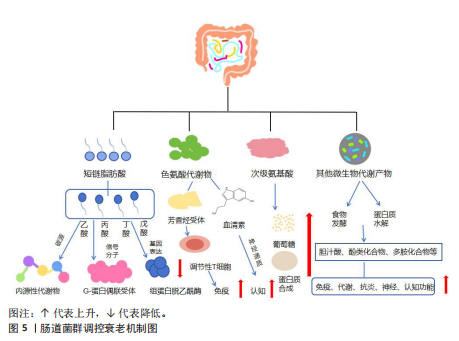
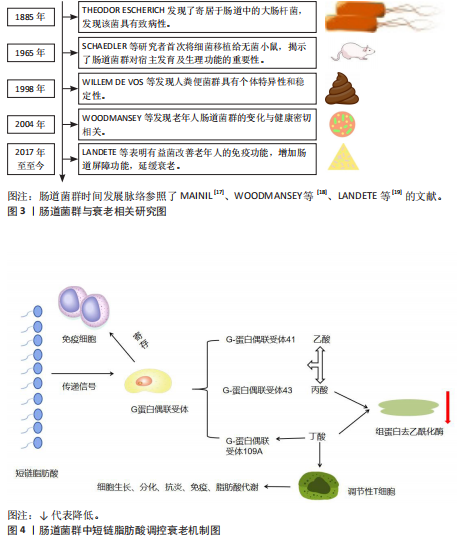
2.1 肠道菌群概述 肠道菌群是指栖息在人体消化道中的一群复杂的微生物群落,可分为有益菌、中性菌(条件致病菌)和有害菌3大类[13]。有益菌参与食物的消化和多种微生物的合成[14];有害菌的繁殖会危害人体的健康和引发多种疾病[15];而中性菌(条件致病菌)是指具有双重作用的微生物群体[16],正常情况下对人体有益,而当肠道内环境失调时,会对人体的健康造成威胁。近年来,随着多种测序技术的更新,为肠道菌群预防衰老提供了一定的思考[17-19],肠道菌群调控衰老的时间线见图3。相关研究表明衰老会引起肠道菌群中有害菌比例上升,有益菌比例下降,而运动又会引起肠道菌群有益菌比例上升,有害菌比例下降。因此,如何用运动调控肠道菌群延缓衰老的相关研究有一定的重要意义,为干预衰老提供一定的思路。 2.2 肠道菌群与衰老 相关研究表明,老年人群体的肠道菌群随着年龄增长会发生一定的改变,具体表现在:多样性下降,有益菌减少,有害菌增加,会出现多器官功能退化,能量代谢紊乱、代谢综合征、认知功能下降和肌少症等疾病[20]。一项关于长寿老年群体的调查发现老年群体肠道的克里斯滕森菌科菌群较为富集,炎症相关的菌群比例会增加[21]。 老年人肠道菌群代谢产物的变化会引起衰老相关进程的发生。肠道菌群的代谢产物主要有短链脂肪酸、色氨酸代谢物、次级氨基酸以及其他微生物代谢产物。短链脂肪酸是连接肠道菌群和调控衰老途径的桥梁,寄居于免疫细胞中的G-蛋白偶联受体接收信号,形成G-蛋白偶联受体41、G-蛋白偶联受体43和G-蛋白偶联受体109A和乙酸、丙酸、丁酸和戊酸结合调控衰老,见图4。丁酸盐与衰老的关系最为密切[22],丁酸能够刺激棕色与白色脂肪组织中赖氨酸特异性去甲基化酶1以及解偶联蛋白1的表达,进而促进机体的能量消耗,增强宿主的代谢,提高机体能力[23]。短链脂肪酸通过多种机制促进宿主-微生物群代谢:①用作产生宿主内源性代谢物的碳源;②作为激活宿主G-蛋白偶联受体的信号分子;③通过抑制组蛋白脱乙酰酶影响宿主基因的表达[24-25]。色氨酸代谢物能够激活芳香烃受体、抑制炎症反应、增强肠道屏障功能,调控免疫细胞的功能,促进调节性T细胞的分化,从而维持免疫稳态[26-27]。色氨酸代谢物——血清素可以作为神经递质,影响情绪和认知功能[24]。次级氨基酸参与调节蛋白质合成和葡萄糖代谢,影响机体的代谢功能[27],其中石胆酸和脱氧胆酸能够通过激活特定受体G蛋白偶联胆汁酸受体1调节肠道炎症反应[28]。而其他微生物代谢物:肠道菌群通过食物发酵胆汁酸,调节脂质吸收,加快糖脂代谢的速度,间接影响机体的能量代谢[29]、代谢膳食产生的多酚类化合物,这些化合物可能具有抗氧化和抗炎作用,增强机体的免疫功能[30],多胺化合物——精胺和组氨酸与肠道的屏障完整性和免疫功能相关[31]。肠道菌群通过影响哺乳动物雷帕霉素靶蛋白信号通路,调节衰老相关的代谢、炎症和自噬,影响衰老进程[32]。 综上所述,肠道菌群调控衰老的途径主要是通过肠道菌群糖发酵食物或蛋白质水解,影响衰老相关的免疫、代谢、神经功能,进而改善衰老相关疾病,见图5。"
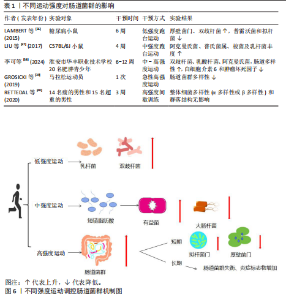
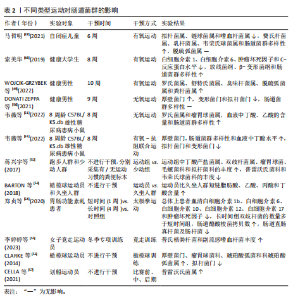
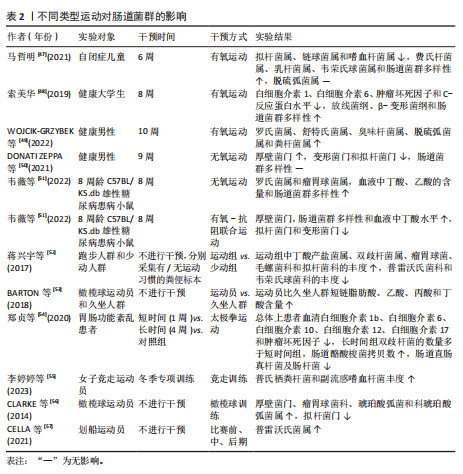
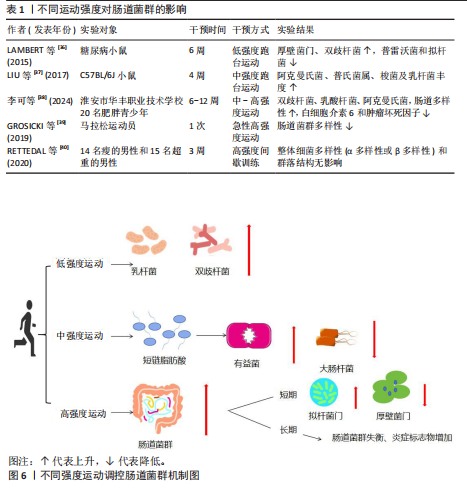
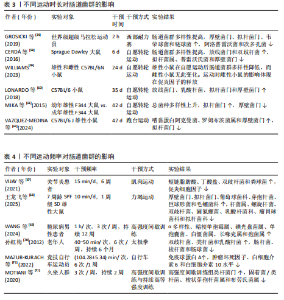
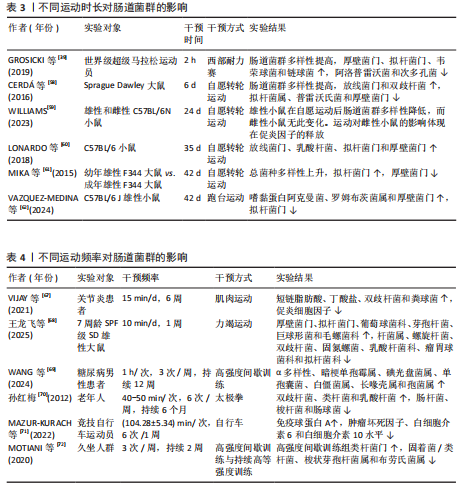
2.3 运动与肠道菌群 运动能够促进肠道菌群中的单形拟杆菌、罗氏菌属和普氏栖粪杆菌等菌群产生短链脂肪酸[33],促进血清素、色胺和犬尿氨酸合成[34],来调节牛磺酸与皮质醇代 谢[35],维护肠道屏障功能,调节肠道内部环境稳态,但是不同的运动方案对肠道菌群的表现具有差异性。 2.3.1 不同运动强度的运动调控肠道菌群 不同运动强度运动对肠道菌群的影响是不同的[36-40],见表1及图6。 研究表明低强度运动缩短肠道菌群与病原体接触的时间,减少疾病的发生,增加乳杆菌和双歧杆菌等有益菌的丰度[41],中等强度运动能够增加短链脂肪酸产生有益菌的丰度和多样性,减少大肠杆菌数量,增强肠道屏障功能[42]。在短期高强度运动(不足3个月的)期间,拟杆菌门的丰度会增加,厚壁菌门的丰度则可能减少[43]。但长时间高强度训练(大于3个月的运动)可能带来负面影响,如肠道菌群失衡和炎症标志物增加[44]。70%的运动员在剧烈运动后出现腹痛、恶心和腹泻,长时间高强度运动增加肠道通透性的压力,可能导致细菌及其有毒产物和微生物群衍生的脂多糖进入血液并激活全身炎症[45-46]。易位的脂多糖激活Toll样受体,激活单核细胞表面的Toll样受体4/分化簇14/髓样分化因子2受体复合物,触发核因子κB通路,促进炎性细胞因子的产生。 2.3.2 不同类型的运动调控肠道菌群 研究结果表明,各类运动对肠道菌群的影响呈现出不同的特点[47-57],见表2。 综上所述,不同类型的运动均能增加肠道菌群的多样性、有益菌菌群的含量,提升厚壁菌门含量和降低拟杆菌门含量。通过对比有运动习惯和久坐、少动人群的研究发现,长期规律的运动可以增加肠道菌群内代谢产物短链脂肪酸、丁酸、乙酸和丙酸的含量。而不同运动专项的运动员由于运动类别的不同,会出现不同的菌种含量增多或降低。 2.3.3 不同时长的运动调控肠道菌群 当前关于运动时长调控肠道菌群处于初步探索阶段,不同时长的运动调控肠道菌群具有一定的差异性[39,58-62],见表3。 在单次运动过程中,机体适应运动强度后其肠道菌群多样性增加,毛螺菌科、考拉杆菌属和阿克曼氏杆菌丰度上升,而瘤胃球菌科丰度下降,这些菌群的变化能促使肠道菌群在单次运动后对运动损伤启动代偿机制[63]。而短时间内的急性运动(小于60 min的运动),可以改变肠道菌群内部的结构,男性耐力运动员在完成急性运动后,6种细菌(罗氏菌属、大肠杆菌TOP498、瘤胃球菌UCG-005、经黏液真杆菌属、瘤胃球菌属9、梭状芽孢杆菌)的丰度发生变化,可以缓解炎症和维持葡萄糖的稳态[64]。慢性运动能优化肠道菌群的结构,维护肠道内壁屏障的完整,抑制肠道炎症环境,致使肠道代谢物的丰度下降,调控机体抗炎路径。长期坚持中等强度的有氧运动(如快走、慢跑或游泳)可以增加乳杆菌和阿克曼氏菌等有益菌的丰度,同时减少拟杆菌属、普雷沃氏菌和厚壁菌门等有害菌的丰度[65],降低炎症因子水平,提高抗氧化能力,增强免疫功能[66]。 2.3.4 不同频率的运动调控肠道菌群 不同频率的运动对不同人群调控菌群的变化具有一定的差异性[67-72],见表4。研究表明,不同频率的运动均能增加不同人群有益菌的数量,降低促炎因子、细胞的水平及改善炎症反应的水平。"
综上所述,运动是一种有效调节肠道菌群的手段,但其效果受到多种因素的综合影响而呈现出不同的结果。当前研究结果讨论了不同的运动强度、运动类型、运动时长和运动频率单独调控肠道菌群的差异性。然而,关于运动形式、运动强度、运动时长和运动频率调控肠道菌群的组合关系需要进一步的探究和验证,以便更好地根据不同的老年群体提出个性化和最优解的运动方案。 2.4 运动·肠道菌群抗衰老时的肠-器官轴 运动和肠道菌群单独对衰老的调控作用是单一的,存在一定的局限性,而规律运动调控健康的肠道菌群对衰老的整体效益是优于二者单独作用的。运动·肠道菌群的结合,能促进肠道菌群代谢产物的生成,富集有益菌的种类和数量,减少内毒素和炎症因子等有毒代谢产物进入肠道内,降低炎症水平,加强肠道屏障功能,增强能量利用,促进机体代谢和内分泌激素的调节。 肠道菌群和衰老的变化是双向协调的过程,老年群体的肠道菌群具备更强的衰老相关代谢物——苯乙酰谷氨酰胺合成能力,使得血液中苯乙酸和苯乙酰谷氨酰胺含量增加,导致细胞中线粒体功能障碍和DNA损伤,最终细胞发生衰老,导致器官组织衰老[73]。而运动能调控肠道菌群内部稳态间接影响衰老,运动刺激肠道菌群富集有益菌和增加菌群的多样性,调控衰老、延缓器官功能衰退。肠道-器官轴是指肠道微生物菌群和其他部位器官进行双向或多向交互交流的生物轴[74],运动·肠道菌群交互通过不同的肠道-器官轴调控衰老具体表现在:运动刺激机体的器官,调动相关细胞与肠道菌群进行双向交流,产生相关肠道菌群代谢物调控衰老的相关进程。 2.4.1 运动介导肠-脑轴抗衰老的通路 衰老会引起脑功能缓慢衰退,影响大脑的认知功能。而身体内环境稳态失调会导致神经退行性疾病和脑卒中等相关疾病的发生[75]。肠-脑轴是指中枢神经系统和胃肠道通过多种信号分子和通路进行双向信息交互[76]。大脑可以调控肠道的运动影响其功能,而肠道则通过影响免疫、神经和代谢通路进行大脑信号的传导。运动通过肠-脑轴影响肠道菌群和神经系统的双向交流。运动激活下丘脑-垂体-肾上腺轴释放促甲肾上腺素神经递质,调动肠道菌群产生γ-氨基丁酸、多巴胺和乙酰胆碱等神经活性物质建立反馈,加强个体的认知功能和抗炎途径,延缓衰老[77];运动通过中枢神经系统的迷走神经通路产生乙酰胆碱类的神经递质抑制肠道炎症反应,减少促炎菌(变形菌门)的增殖,促进有益菌(厚壁菌门)的定植[78];运动通过提高胆囊收缩素、胰高血糖素样肽的浓度加速肠道内皮内分泌细胞的信号传导,延缓脑衰老[79]。 2.4.2 运动介导肠-皮肤轴抗衰老的通路 衰老会引起皮肤的老化、皮下内分泌失调[80]。肠道和皮肤富含血管和神经,肠道代谢产物经过循环进入血液之中,破坏了皮肤屏障功能引起肠道屏障的受损,触发胃肠道疾病和皮肤疾病的同时发生,这种关系组成了肠-皮肤轴[81]。运动能增加肠道菌群代谢物短链脂肪酸的浓度,"
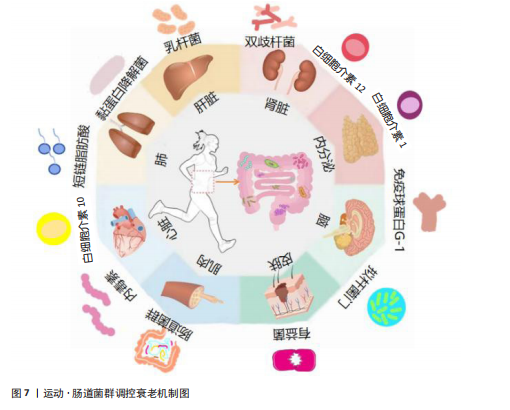
促使G-蛋白偶联受体41/G-蛋白偶联受体43受体接收信号并与短链脂肪酸结合或抑制组蛋白去乙酰化酶,下调核因子κB通路,减少促炎因子白细胞介素6、白细胞介素17和肿瘤坏死因子的释放[82],缓解炎症衰老,增强肠道屏障功能,最终补充肠膜明串珠菌,延缓皮肤衰老[83];运动能增加肠道免疫球蛋白A分泌、调节T细胞平衡Th1细胞/Th2细胞、减少辅助性T细胞和环氧化酶2细胞数量[53,84],促进肠道免疫,通过血液循环加强皮肤免疫。皮肤损伤或炎症通过释放透明质酸改变肠道菌群组成,增加大肠杆菌致病菌的定植风险,而运动通过改善肠道菌群抑制皮肤炎症发生这一过程,延缓皮肤衰老相关进程[85]。 2.4.3 运动介导肠-肌肉轴抗衰老的通路 衰老会引起肌肉质量的下降和功能的退化,加速肌肉萎缩,使身体运动功能退化[86]。肠-肌肉轴是指肠道微生物群的代谢产物短链脂肪酸影响腺苷酸活化蛋白激酶信号通路参与肌肉代谢、能量利用以及肌肉蛋白质的合成与分解,而肌肉质量和功能的改善能丰富肠道菌群的多样性,调节肠道菌群的内部稳态[87-88]。运动后乳酸被韦荣氏球菌属转化为丙酸,丙酸作为肠道菌群短链脂肪酸的代谢产物,激活腺苷酸活化蛋白激酶通路,促进肌肉脂肪酸氧化和提高胰岛素敏感性,减少内毒素进入血液循环发生炎症,加快糖脂代谢,增强肌肉力量和功能,延缓衰老[89-90]。短链脂肪酸还能抑制核因子κB等炎症通路,减少促炎因子肿瘤坏死因子、白细胞介素6的释放,缓解肌肉炎症和氧化应激,增强骨骼肌的力量[91]。 2.4.4 运动介导肠-心脏轴抗衰老的通路 衰老会引起心肌细胞的衰老和心脏功能的下降[92]。肠-心脏轴的形成是肠道代谢物——氧化三甲胺联系的,氧化三甲胺浓度的变化被认为是血管疾病的标志之一[93],高浓度的氧化三甲胺会导致肠道炎症环境,造成机体内部环境的失调,引起内部细胞的老化[94]。运动能够增加有益菌的种类和数量,提升乙酸的浓度增加短链脂肪酸产生菌的数量,改善菌群结构[95],降低血压[96];运动通过增加抗炎因子白细胞介素10的分泌,抑制NOD样受体蛋白3炎症小体通路,降低氧化三甲胺的浓度,减轻心脏的炎症损伤,延缓衰老[97]。 2.4.5 运动介导肠-肝脏轴抗衰老的通路 衰老会引起肝脏抗炎和代谢功能的下降,削弱肝脏的生殖和再生细胞的能力,引起肝损伤和肝脏疾病的易感性[98]。肠-肝脏轴是肠道和肝脏之间的双向联系。肠道菌群代谢参与肝肠循环的胆汁酸,调节肝脏的功能,而外膜囊泡和生物活性代谢物进入门静脉影响肝脏的生理[99]。运动富集丁酸菌——嗜黏蛋白阿克曼菌,减少内毒素向肝的转移,从而缓解肝脂肪堆积,促进机体的能量代谢[95];运动影响变形菌门、放线菌门和疣微菌门的富集,减少厚壁菌门和广古菌门的富集,建立胰岛素抵抗[100];运动依赖腺苷酸活化蛋白激酶促进肝脏中的脂肪吞噬从而抑制肝脏脂质积累,加快糖脂代谢[101];运动促进肠道菌群的代谢活动,合成维生素B族和维生素K进入肝脏,参与代谢途径调控衰老[102]。 2.4.6 运动介导肠-肺轴抗衰老的通路 衰老会引起肺部弹性回缩力的下降,呼吸肌力量的衰退[103]。肠-肺轴的联系是由于肠道和肺之间都来源于胚胎期的内胚层[104]。肺部微生物环境的改变会导致肠道菌群中大肠埃希菌等有害菌比例的上升,炎症因子表达提高,引起肠道炎症发生[105]。运动促进短链脂肪酸的代谢,影响拟杆菌门、厚壁菌门和变形菌门的相对丰度,增加肺部免疫因子的释放,提高肺功能[106];运动通过肠道菌群的代谢产物短链脂肪酸,分泌内毒素等代谢产物,调控呼吸道系统的免疫功能,减少呼吸系统相关疾病的发生;运动诱导肠道菌群富集分段丝状细菌和枸橼酸杆菌附着肠上皮细胞诱导产生血清淀粉样蛋白A、活性氧分泌白细胞介素22,增强肠道的抵抗能力间接影响肺的免疫功能[107]。 2.4.7 运动介导肠-肾轴抗衰老的通路 衰老会引起肾脏结构和功能的变化,造成免疫系统的失调和慢性炎症[108]。肠-肾轴是指肠道和肾脏之间的相互串扰联系,肠道菌群代谢物(短链脂肪酸、尿毒素、神经递质和炎症因子)影响肾脏的功能[109],而肾脏能排出肠道菌群中的有害菌群,调节稳态[110]。运动通过肠-肾轴优化肠道菌群组成,降低肠杆菌目、放线菌门、埃希菌-志贺菌属和双歧杆菌属的丰度,升高拟杆菌门、毛螺菌属、粪球菌属、萨特氏菌属的丰度[111],减少促炎因子和尿毒素的产生[112],通过免疫途径,抑制肾小管炎症和纤维化,延缓肾功能衰退[82]。 2.4.8 运动介导肠-内分泌轴抗衰老的通路 衰老会引起内分泌的代谢紊乱,激素调节水平发生变化,造成机体内分泌激素异常,使老年群体衰弱[113]。肠-内分泌轴是肠道和内分泌系统之间的双向调节作用,肠道菌群的代谢产物通过血液循环进入内分泌细胞和器官。内分泌细胞释放激素,传递信号至中枢神经系统,继而影响内分泌系统的调节,而内分泌系统的变化又影响内分泌细胞,间接影响肠道功能[114]。运动刺激肠道菌群生成代谢物短链脂肪酸、胆汁酸代谢物和吲哚衍生物,参与血液循环,影响下丘脑-垂体-肾上腺轴,调节压力反应和皮质醇分泌;影响下丘脑-垂体-甲状腺轴,调节甲状腺激素分泌;影响下丘脑-垂体-性腺轴,调节性激素分泌[74];通过内分泌调节加快体循环,提高代谢速度,减缓衰老。 运动作为一种干预手段,调动器官参与运动的同时通过肠-器官轴调控肠道菌群内部的变化,主要表现在菌群多样性增加、有益菌的富集、调节肠道菌群中的代谢物短链脂肪酸,提高免疫球蛋白G1水平,增强机体免疫、合成维生素K、B族维生素,加快糖脂代谢进入血液循环、增强骨骼肌肌肉质量、显著降低白细胞介素1b、白细胞介素6、白细胞介素10、白细胞介素12、白细胞介素17和肿瘤坏死因子水平,降低炎症反应、参与能量消耗、影响神经系统间接提高机体的认知和行为功能进而增强肠道屏障功能,从而实现衰老的调控。 综上所述,不同的运动方案调控肠-器官轴的生理机制尚未阐述,运动时长、运动频率、运动周期和运动强度的组合对肠道菌群的影响机制需要证实,其如何影响器官调控肠道菌群仍需进一步的研究,见图7。"
| [1] MOMENI Z, LOGAN JE, SIGAL RJ, et al. Can resistance exercise Be a tool for healthy aging in post-menopausal women with type 1 diabetes? Int J Environ Res Public Health. 2021;18(16):8716. [2] PINCKARD K, BASKIN KK, STANFORD KI. Effects of exercise to improve cardiovascular health. Front Cardiovasc Med. 2019;6:69. [3] 贾丽晔, 郭琪, 王鹏程, 等. 运动疗法对心血管疾病患者的影响和作用机理研究进展[J]. 中国康复理论与实践,2016,22(9):1041-1044. [4] MENG J, GENG Q, JIN S, et al. Exercise protects vascular function by countering senescent cells in older adults. Front Physiol. 2023;14:1138162. [5] GERRITSEN J, SMIDT H, RIJKERS GT, et al. Intestinal microbiota in human health and disease: the impact of probiotics. Genes Nutr. 2011;6(3):209-240. [6] SARASWATI S, SITARAMAN R. Aging and the human gut microbiota-from correlation to causality. Front Microbiol. 2015;5:764. [7] BRANCA JJV, GULISANO M, NICOLETTI C. Intestinal epithelial barrier functions in ageing. Ageing Res Rev. 2019;54:100938. [8] NAGPAL R, MAINALI R, AHMADI S, et al. Gut microbiome and aging: Physiological and mechanistic insights. Nutr Healthy Aging. 2018; 4(4):267-285. [9] STRASSER B, WOLTERS M, WEYH C, et al. The Effects of Lifestyle and Diet on Gut Microbiota Composition, Inflammation and Muscle Performance in Our Aging Society. Nutrients. 2021;13(6):2045. [10] ZHANG L, LIU Y, SUN Y, et al. Combined Physical Exercise and Diet: Regulation of Gut Microbiota to Prevent and Treat of Metabolic Disease: A Review. Nutrients. 2022;14(22):4774. [11] BONOMINI-GNUTZMANN R, PLAZA-DÍAZ J, JORQUERA-AGUILERA C, et al. Effect of Intensity and Duration of Exercise on Gut Microbiota in Humans: A Systematic Review. Int J Environ Res Public Health. 2022;19(15):9518. [12] KUMAR M, BABAEI P, JI B, et al. Human gut microbiota and healthy aging: Recent developments and future prospective. Nutr Healthy Aging. 2016;4(1):3-16. [13] ADAMCZYK-SOWA M, MEDREK A, MADEJ P, et al. Does the Gut Microbiota Influence Immunity and Inflammation in Multiple Sclerosis Pathophysiology? J Immunol Res. 2017;2017: 7904821. [14] ELSHAFEI AM. Potentials of microorganisms in human health. J Appl Life Sci Int. 2021; 24(4):1-9. [15] GUPTA PD. The mighty microbiota: regulator of the human body. Clin Res Clin Trials. 2021;3(5):1-8. [16] LUO YANG HG. Analysis of the influence of probiotics on intestinal micro-ecology and related diseases. J Integr Nurs. 2019;1(3):163-169. [17] MAINIL J. Escherichia coli virulence factors. Vet Immunol Immunopathol. 2013;152(1-2): 2-12. [18] WOODMANSEY EJ, MCMURDO ME, MACFARLANE GT, et al. Comparison of compositions and metabolic activities of fecal microbiotas in young adults and in antibiotic-treated and non-antibiotic-treated elderly subjects. Appl Environ Microbiol. 2004;70(10):6113-6122. [19] LANDETE JM, GAYA P, RODRÍGUEZ E, et al. Probiotic Bacteria for Healthier Aging: Immunomodulation and Metabolism of Phytoestrogens. Biomed Res Int. 2017; 2017:5939818. [20] CHOI J, HUR TY, HONG Y. Influence of Altered Gut Microbiota Composition on Aging and Aging-Related Diseases. J Lifestyle Med. 2018;8(1):1-7. [21] DONATI ZEPPA S, AGOSTINI D, FERRINI F, et al. Interventions on Gut Microbiota for Healthy Aging. Cells. 2022;12(1):34. [22] 王倩楠, 李玉兰, 莫刚, 等. 肠道菌群与病理性衰老的研究进展[J].华夏医学,2024, 37(1):18-23. [23] WANG D, LIU CD, LI HF, et al. LSD1 mediates microbial metabolite butyrate-induced thermogenesis in brown and white adipose tissue. Metabolism. 2020;102:154011. [24] NICOLAS GR, CHANG PV. Deciphering the Chemical Lexicon of Host-Gut Microbiota Interactions. Trends Pharmacol Sci. 2019;40(6):430-445. [25] XIAO H, KANG S. The Role of the Gut Microbiome in Energy Balance With a Focus on the Gut-Adipose Tissue Axis. Front Genet. 2020;11:297. [26] WANG J, ZHU N, SU X, et al. Gut-Microbiota-Derived Metabolites Maintain Gut and Systemic Immune Homeostasis. Cells. 2023;12(5):793. [27] AGUS A, CLÉMENT K, SOKOL H. Gut microbiota-derived metabolites as central regulators in metabolic disorders. Gut. 2021;70(6):1174-1182. [28] WANG G, HUANG S, WANG Y, et al. Bridging intestinal immunity and gut microbiota by metabolites. Cell Mol Life Sci. 2019;76(20):3917-3937. [29] SCHOELER M, CAESAR R. Dietary lipids, gut microbiota and lipid metabolism. Rev Endocr Metab Disord. 2019;20(4):61-472. [30] GHOSH S, PRAMANIK S. Structural diversity, functional aspects and future therapeutic applications of human gut microbiome. Arch Microbiol. 2021;203(9):5281-5308. [31] FENG W, AO H, PENG C. Gut Microbiota, Short-Chain Fatty Acids, and Herbal Medicines. Front Pharmacol. 2018;9:1354. [32] GHOSH TS, SHANAHAN F, O’TOOLE PW. The gut microbiome as a modulator of healthy ageing. Nat Rev Gastroenterol Hepatol. 2022;19(9):565-584. [33] 孟先峰, 李晟, 冯贵山, 等. 男子橄榄球运动员肠道菌群特征和短链脂肪酸水平及其抗炎作用分析[J]. 中国微生态学杂志,2024,36(9):1016-1023. [34] CHEN YH, YUAN W, MENG LK, et al .The Role and Mechanism of Gut Microbiota in Pulmonary Arterial Hypertension. Nutrients. 2022;14(20):4278. [35] 刘阳, 赵纯纯, 李致潇. 运动训练通过改善肠道菌群防治代谢疾病的研究进展[J]. 中国细胞生物学学报,2024,46(7):1468-1476. [36] LAMBERT JE, MYSLICKI JP, BOMHOF MR, et al. Exercise training modifies gut microbiota in normal and diabetic mice. Appl Physiol Nutr Metab. 2015; 40(7):749-752. [37] LIU Z, LIU HY, ZHOU H, et al. Moderate-Intensity Exercise Affects Gut Microbiome Composition and Influences Cardiac Function in Myocardial Infarction Mice. Front Microbiol. 2017;8:1687. [38] 李可, 李宁川, 丁一凡, 等. 12周中高强度健身操改善肥胖青少年代谢紊乱效果分析[J]. 中国学校卫生,2024,45(6):788-793. [39] GROSICKI GJ, DURK RP, BAGLEY JR. Rapid gut microbiome changes in a world-class ultramarathon runner. Physiol Rep. 2019;7(24): e14313. [40] RETTEDAL EA, CREE JME, ADAMS SE, et al. Short-term high-intensity interval training exercise does not affect gut bacterial community diversity or composition of lean and overweight men. Exp Physiol. 2020; 105(8):1268-1279. [41] 梁家琪, 刘恒旭, 阳金鑫, 等. 运动与肠道菌健康效益的关系[J]. 中国组织工程研究,2023, 27(8):1292-1299. [42] RESENDE AS, LEITE GSF, LANCHA JUNIOR AH. Changes in the Gut Bacteria Composition of Healthy Men with the Same Nutritional Profile Undergoing 10-Week Aerobic Exercise Training: A Randomized Controlled Trial. Nutrients. 2021; 13(8):2839. [43] 徐山茸, 龚莉, 储文文, 等. 12周高强度间歇性训练对人体肠道菌群的影响[J]. 微生物学通报,2021,48(4):1215-1226. [44] 杨展, 孙学伟, 秦明珂, 等. Akkermansia muciniphila对大鼠高温运动后器官损伤的影响[J]. 中国微生态学杂志,2024,36(2):125-134,146. [45] CRANDALL CG, WILSON TE. Human cardiovascular responses to passive heat stress. Compr Physiol. 2015;5(1):17-43. [46] DOKLADNY K, ZUHL MN, MOSELEY PL. Intestinal epithelial barrier function and tight junction proteins with heat and exercise. J Appl Physiol (1985). 2016;120(6):692-701. [47] 马哲明. 有氧运动对自闭症儿童肠道菌群的影响[D]. 广州: 广州体育学院,2021. [48] 索美华. 8周有氧运动对青年肥胖者部分血清炎性因子的调节作用及对其肠道菌群的影响[D]. 济南: 山东体育学院,2019. [49] WOJCIK-GRZYBEK D, HUBALEWSKA-MAZGAJ M, SURMIAK M, et al. The Combination of Intestinal Alkaline Phosphatase Treatment with Moderate Physical Activity Alleviates the Severity of Experimental Colitis in Obese Mice via Modulation of Gut Microbiota, Attenuation of Proinflammatory Cytokines, Oxidative Stress Biomarkers and DNA Oxidative Damage in Colonic Mucosa. Int J Mol Sci. 2022;23(6):2964. [50] DONATI ZEPPA S, AMATORI S, SISTI D, et al. Nine weeks of high-intensity indoor cycling training induced changes in the microbiota composition in non-athlete healthy male college students. J Int Soc Sports Nutr. 2021;18(1):74. [51] 韦薇, 张秋, 黄燕凤, 等. 不同运动方式对2型糖尿病小鼠肠道菌群及短链脂肪酸的影响[J]. 广西医科大学学报,2022,39(4):643-648. [52] 蒋兴宇, 赵霞, 邹凌云, 等. 适度运动对人体肠道菌群结构的影响[J]. 第三军医大学学报, 2017,39(18):1824-1831. [53] BARTON W, PENNEY NC, CRONIN O, et al. The microbiome of professional athletes differs from that of more sedentary subjects in composition and particularly at the functional metabolic level. Gut. 2018;67(4):625-633. [54] 郑贞, 罗杨, 鄢显明, 等.不同训练时间太极拳运动康复方案对肠道菌群功能的影响[J].中国病原生物学杂志,2020,15(9):1071-1074. [55] 李婷婷, 赵如珍, 徐玉婷. 山东省女子竞走运动员冬训前、后肠道菌群特征与运动表现分析[J]. 山东体育科技,2023,45(6):46-52. [56] CLARKE SF, MURPHY EF, O’SULLIVAN O, et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut. 2014; 63(12):1913-1920. [57] CELLA V, BIMONTE VM, SABATO C, et al. Nutrition and Physical Activity-Induced Changes in Gut Microbiota: Possible Implications for Human Health and Athletic Performance. Foods. 2021; 10(12):3075. [58] CERDÁ B, PÉREZ M, PÉREZ-SANTIAGO JD, et al. Gut Microbiota Modification: Another Piece in the Puzzle of the Benefits of Physical Exercise in Health? Front Physiol. 2016;7:51. [59] WILLIAMS ZAP, SZYSZKOWICZ JK, OSBORNE N, et al. Sex-specific effects of voluntary wheel running on behavior and the gut microbiota-immune-brain axis in mice. Brain Behav Immun Health. 2023;30:100628. [60] LONARDO A, TARGHER G. Non-alcoholic fatty liver disease research 2016. Switzerland, ST: MDPI - Multidisciplinary Digital Publishing Institute,2018. [61] MIKA A, VAN TREUREN W, GONZÁLEZ A, et al. Exercise is More Effective at Altering Gut Microbial Composition and Producing Stable Changes in Lean Mass in Juvenile versus Adult Male F344 Rats. PLoS One. 2015;10(5):e0125889. [62] VAZQUEZ-MEDINA A, RODRIGUEZ-TRUJILLO N, AYUSO-RODRIGUEZ K, et al. Exploring the interplay between running exercises, microbial diversity, and tryptophan metabolism along the microbiota-gut-brain axis. Front Microbiol. 2024;15:1326584. [63] 李小英, 唐齐, 陈颖. 肠道菌群与运动后的代谢获益[J]. 上海医学,2021,44(10):717-721. [64] TABONE M, BRESSA C, GARCÍA-MERINO JA, et al. The effect of acute moderate-intensity exercise on the serum and fecal metabolomes and the gut microbiota of cross-country endurance athletes. Sci Rep. 2021;11(1):3558. [65] PARMAR S, HANKUS K, BARZAK B, et al. The effect of physical activity on gut microbiota. A review. Med J Cell Biol. 2023;10(4):138-143. [66] YUAN X, XU S, HUANG H, et al. Influence of excessive exercise on immunity, metabolism, and gut microbial diversity in an overtraining mice model. Scand J Med Sci Sports. 2018;28(5):1541-1551. [67] VIJAY A, KOURAKI A, GOHIR S, et al. The anti-inflammatory effect of bacterial short chain fatty acids is partially mediated by endocannabinoids. Gut Microbes. 2021; 13(1):1997559. [68] 王龙飞, 江大雷. 6周力竭运动对大鼠肠道菌群结构的影响[J]. 四川体育科学,2025,44(1): 39-44,60. [69] WANG Y, CHEN J, NI Y, et al. Exercise-changed gut mycobiome as a potential contributor to metabolic benefits in diabetes prevention: an integrative multi-omics study. Gut Microbes. 2024;16(1):2416928. [70] 孙红梅. 健身气功·八段锦练习对老年人肠道菌群的影响[J]. 中国运动医学杂志,2012, 31(11):973-977. [71] MAZUR-KURACH P, FRĄCZEK B, KLIMEK AT. Does Multi-Strain Probiotic Supplementation Impact the Effort Capacity of Competitive Road Cyclists? Int J Environ Res Public Health. 2022;19(19):12205. [72] MOTIANI KK, COLLADO MC, ESKELINEN JJ, et al. Exercise Training Modulates Gut Microbiota Profile and Improves Endotoxemia. Med Sci Sports Exerc. 2020;52(1):94-104. [73] YANG H, WANG T, QIAN C, et al. Gut microbial-derived phenylacetylglutamine accelerates host cellular senescence. Nat Aging. 2025;5(3):401-418. [74] LIN X, YU Z, LIU Y, et al. Gut-X axis. Imeta. 2025; 4(1):e270. [75] LOSSI L, CASTAGNA C, MERIGHI A. An Overview of the Epigenetic Modifications in the Brain under Normal and Pathological Conditions. Int J Mol Sci. 2024;25(7):3881. [76] LIN B, WANG Y, ZHANG P, et al. Gut microbiota regulates neuropathic pain: potential mechanisms and therapeutic strategy. J Headache Pain. 2020; 21(1):103. [77] CLARK A, MACH N. Exercise-induced stress behavior, gut-microbiota-brain axis and diet: a systematic review for athletes. J Int Soc Sports Nutr. 2016;13(1):43. [78] MARTIN CR, OSADCHIY V, KALANI A, et al. The Brain-Gut-Microbiome Axis. Cell Mol Gastroenterol Hepatol. 2018;6(2):133-148. [79] 王拴, 杨子懿, 宋诗语, 等. 微生物-肠-脑轴在运动减轻神经炎症中的作用[J]. 生命科学, 2025,37(2):186-195. [80] AGRAWAL R, HU A, BOLLAG WB. The Skin and Inflamm-Aging. Biology (Basel). 2023;12(11):1396. [81] SALEM I, RAMSER A, ISHAM N, et al. The Gut Microbiome as a Major Regulator of the Gut-Skin Axis. Front Microbiol. 2018;9:1459. [82] 刘伊依, 邱俊强. 运动通过调节SCFAs延缓衰老及其作用机制的研究进展[J]. 生命科学, 2023,35(6):779-789. [83] GAO T, LI Y, WANG X, et al. Alginate oligosaccharide-mediated butyrate-HIF-1α axis improves skin aging in mice. J Pharm Anal. 2024;14(5):100911. [84] CODELLA R, LUZI L, TERRUZZI I. Exercise has the guts: How physical activity may positively modulate gut microbiota in chronic and immune-based diseases. Dig Liver Dis. 2018;50(4):331-341. [85] DOKOSHI T, CHEN Y, CAVAGNERO KJ, et al. Dermal injury drives a skin to gut axis that disrupts the intestinal microbiome and intestinal immune homeostasis in mice. Nat Commun. 2024;15(1):3009. [86] COEN PM, MUSCI RV, HINKLEY JM, et al. Mitochondria as a Target for Mitigating Sarcopenia. Front Physiol. 2019;9:1883. [87] LIU C, CHEUNG WH, LI J, et al. Understanding the gut microbiota and sarcopenia: a systematic review. J Cachexia Sarcopenia Muscle. 2021; 12(6):1393-1407. [88] ZHOU Y, CHU Z, LUO Y, et al. Dietary Polysaccharides Exert Anti-Fatigue Functions via the Gut-Muscle Axis: Advances and Prospectives. Foods. 2023;12(16):3083. [89] DONATI ZEPPA S, AGOSTINI D, GERVASI M, et al. Mutual Interactions among Exercise, Sport Supplements and Microbiota. Nutrients. 2019;12(1):17. [90] GIZARD F, FERNANDEZ A, DE VADDER F. Interactions between gut microbiota and skeletal muscle. Nutr Metab Insights. 2020;13: 1178638820980490. [91] 史益凡, 王新颖. 肠道菌群对骨骼肌的影响及SCFAs介导“肠-肌轴”的机制研究进展[J]. 解放军医学杂志,2021,46(9): 849-854. [92] TANG X, LI PH, CHEN HZ. Cardiomyocyte Senescence and Cellular Communications Within Myocardial Microenvironments. Front Endocrinol (Lausanne). 2020;11:280. [93] NAM HS. Gut Microbiota and Ischemic Stroke: The Role of Trimethylamine N-Oxide. J Stroke. 2019;21(2):151-159. [94] XU H, WANG X, FENG W, et al. The gut microbiota and its interactions with cardiovascular disease. Microb Biotechnol. 2020;13(3):637-656. [95] GAO C, WEI J, LU C, et al. A new perspective in intestinal microecology: lifting the veil of exercise regulation of cardiometabolic diseases. Gut Microbes. 2024;16(1):2404141. [96] YAN Q, ZHAI W, YANG C, et al. The Relationship among Physical Activity, Intestinal Flora, and Cardiovascular Disease. Cardiovasc Ther. 2021; 2021:3364418. [97] 田振军, 王涛. 运动与心脏保护的整合生理学研究进展及问题思考[J]. 上海体育大学学报, 2025,49(2):11-28. [98] SCHMUCKER DL, SANCHEZ H. Liver regeneration and aging: a current perspective. Curr Gerontol Geriatr Res. 2011;2011:526379. [99] ZHANG YL, LI ZJ, GOU HZ, et al. The gut microbiota-bile acid axis: a potential therapeutic target for liver fibrosis. Front Cel Infect Microbiol. 2022;12:945368. [100] HOYLES L, FERNÁNDEZ-REAL JM, FEDERICI M, et al. Molecular phenomics and metagenomics of hepatic steatosis in non-diabetic obese women. Nat Med. 2018;24(7):1070-1080. [101] GAO Y, ZHANG W, ZENG LQ, et al. Exercise and dietary intervention ameliorate high-fat diet-induced NAFLD and liver aging by inducing lipophagy. Redox Biol. 2020;36:101635. [102] BIDELL MR, HOBBS ALV, LODISE TP. Gut microbiome health and dysbiosis: A clinical primer. Pharmacotherapy. 2022;42(11): 849-857. [103] KUKRETY SP, PAREKH JD, BAILEY KL. Chronic obstructive pulmonary disease and the hallmarks of aging. Lung India. 2018;35(4):321-327. [104] WRIGHT PF, ACKERMAN ME, BRICKLEY EB. Mucosal Immunity: The Forgotten Arm of the Immune System. J Pediatric Infect Dis Soc. 2019; 8(1):53-54. [105] MINDT BC, DIGIANDOMENICO A. Microbiome Modulation as a Novel Strategy to Treat and Prevent Respiratory Infections. Antibiotics (Basel). 2022;11(4): 474. [106] CLEMENTE F, BRAVINI E, CORNA S, et al. Relazione tra esercizio fisico e microbiota intestinale nell’essere umano: revisione sistematica [The relationship between physical exercise and gut microbiota in the human being: a systematic review]. Epidemiol Prev. 2021;45(4):245-253. [107] PRICE AE, SHAMARDANI K, LUGO KA, et al. A Map of Toll-like Receptor Expression in the Intestinal Epithelium Reveals Distinct Spatial, Cell Type-Specific, and Temporal Patterns. Immunity. 2018; 49(3):560-575.e6. [108] 王海萍, 孙晶, 王荣. 肾脏衰老与老年肾脏疾病的研究进展[J]. 老年医学研究, 2021,2(5):51-55. [109] CAO C, ZHU H, YAO Y, et al. Gut Dysbiosis and Kidney Diseases. Front Med (Lausanne). 2022;9: 829349. [110] GUO Y, CHEN X, GONG P, et al. The Gut-Organ-Axis Concept: Advances the Application of Gut-on-Chip Technology. Int J Mol Sci. 2023; 24(4):4089. [111] 包文晗, 唐雯. 初诊IgA肾病患者的肠道菌群及其与疾病进展因素的相关分析[J]. 北京大学学报(医学版),2023,55(1):124-132. [112] SUN L, LI Z, HU C, et al. Age-dependent changes in the gut microbiota and serum metabolome correlate with renal function and human aging. Aging Cell. 2023;22(12):e14028. [113] 任卓, 姚晓东. 内分泌激素与老年衰弱相关性的研究进展[J]. 国际老年医学杂志,2023, 44(6):743-746. [114] WORTHINGTON JJ, REIMANN F, GRIBBLE FM. Enteroendocrine cells-sensory sentinels of the intestinal environment and orchestrators of mucosal immunity. Mucosal Immunol. 2018; 11(1):3-20. |
| [1] | Zhang Qingtong, Chen Leqin, Liu Chang, Chen Yuting, Guo Ruiwu. Neuromechanism of the endocannabinoid system in regulating exercise motivation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(在线): 1-11. |
| [2] | Li Sa, Sun Ning, Sun Zhaozhong, Feng Zhimeng, Li Xuedong. Evaluation parameters and specific region of C6 nerve oppression by uncinate process degeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2294-2302. |
| [3] | Liu Jinlong, Abuduwupuer·Haibier, Bai Zhen, Su Danyang, Miao Xin, Li Fei, Yang Xiaopeng. Efficacy of different nonsurgical treatments for adolescent idiopathic scoliosis: a systematic review and network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2370-2379. |
| [4] | Yang Lixia, Diao Liqin, Li Hua, Feng Yachan, Liu Xin, Yu Yuexin, Dou Xixi, Gu Huifeng, Xu Lanju. Regulatory mechanism of recombinant type III humanized collagen protein improving photoaging skin in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1988-2000. |
| [5] | Ye Qianqian, Pan Hang, Tian Chuan, Zhu Xiangqing, Ye Li, Zhao Xiaojuan, Shu Liping, Pan Xinghua. Effects of highly active umbilical cord mesenchymal stem cells on structure and function of thymus in elderly tree shrews [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1720-1729. |
| [6] | Pan Dong, Yang Jialing, Tian Wei, Wang Dongji, Zhu Zheng, Ma Wenchao, Liu Na, Fu Changxi. Resistance exercise activates skeletal muscle satellite cells in aged rats: role of adiponectin receptor 1 pathway [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1736-1746. |
| [7] | Cao Yong, Teng Hongliang, Tai Pengfei, Li Junda, Zhu Tengqi, Li Zhaojin. Interactions between cytokines and satellite cells in muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1808-1817. |
| [8] | Sun Yaotian, Xu Kai, Wang Peiyun. Potential mechanisms by which exercise regulates iron metabolism in immune inflammatory diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1486-1498. |
| [9] | Liu Huan, Zeng Shaopeng, Chen Jun, He Linqian, Yang Ying, Zhang Jing. Aging-related dysregulation of glucose metabolism: crossroads of cancer and neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1527-1538. |
| [10] | Hou Chaowen, Li Zhaojin, Kong Jianda, Zhang Shuli. Main physiological changes in skeletal muscle aging and the multimechanism regulatory role of exercise [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1464-1475. |
| [11] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [12] | Wen Fan, Xiang Yang, Zhu Huan, Tuo Yanfang, Li Feng. Exercise improves microvascular function in patients with type 2 diabetes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1225-1235. |
| [13] | Chen Qiang, Wu Wenjuan, Jiang Shuhua, Huang Da. Physical exercise improves physical function in burn patients: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1269-1281. |
| [14] | Peng Tuanhui, Song Hongming, Yang Ling, Ding Xiaoge, Meng Pengjun. Effects of long-term endurance exercise on kl/FGF23 axis and calcium-phosphorus metabolism in naturally aging mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1089-1095. |
| [15] | Jiang Yang, Peng Hao, Song Yanping, Yao Na, Song Yueyu, Yin Xingxiao, Li Yanqi, Chen Qigang. Isometric exercise reduces resting blood pressure: a meta-analysis of moderating factors and dose effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 975-986. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||