Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 4843-4852.doi: 10.12307/2026.763
Previous Articles Next Articles
Quercetin promotes osteogenic differentiation of senescent jaw bone marrow mesenchymal stem cells
Wang Hengxin1, 2, Li Hongkun1, 2, Xu Nuo1, 2, Li Anping1, 2, Wang Xinjing1, 2, Zhang Tong2
- 1Chinese PLA Medical School, Beijing 100853, China; 2Department of Stomatology, First Medical Center, Chinese PLA General Hospital, Beijing 100853, China
-
Received:2025-08-04Accepted:2025-10-17Online:2026-07-08Published:2026-02-13 -
Contact:Zhang Tong, MD, Chief physician, Department of Stomatology, First Medical Center, Chinese PLA General Hospital, Beijing 100853, China -
About author:Wang Hengxin, Master candidate, Chinese PLA Medical School, Beijing 100853, China; Department of Stomatology, First Medical Center, Chinese PLA General Hospital, Beijing 100853, China -
Supported by:Beijing Natural Science Foundation, No. 7232154 (to ZT)
CLC Number:
Cite this article
Wang Hengxin, Li Hongkun, Xu Nuo, Li Anping, Wang Xinjing, Zhang Tong. Quercetin promotes osteogenic differentiation of senescent jaw bone marrow mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4843-4852.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
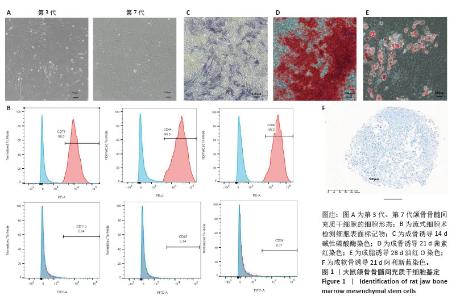
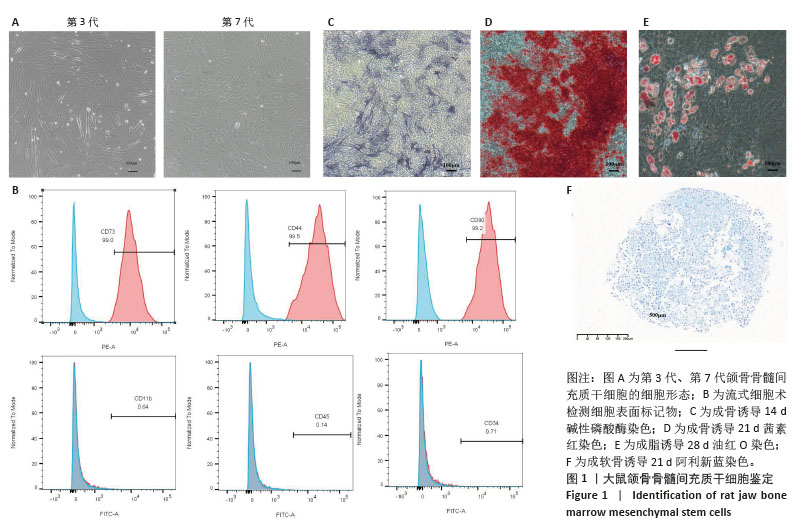
2.1 颌骨骨髓间充质干细胞培养形态变化 细胞分离培养3 d后,镜下可见少量贴壁生长的长梭形或三角形细胞,以及大量圆形、较亮的血细胞;经过换液,血细胞数量逐渐减少,颌骨骨髓间充质干细胞不断分裂增殖,数量增加,汇合度达80%以上时,按1∶2的比例进行传代,第3代细胞呈现典型的未分化间充质干细胞形态特征,细胞表面光滑,呈细长纺锤形,并呈现有序的旋涡状生长;相比之下,第7代细胞表现出明显的衰老相关形态学改变,表现为体积增大,呈不规则扁平状或“煎蛋状”形态,边界模糊不清,排列方向紊乱(图1A)。 2.2 颌骨骨髓间充质干细胞表面抗原、三系分化能力 采用流式细胞术对细胞表面抗原的表达进行检测,间充质干细胞表面抗原CD90、CD44、CD73阳性表达,而造血干细胞相关抗原 CD34、CD45、CD11b阴性表达(图1B),表明提取的细胞为颌骨来源间充质干细胞即颌骨骨髓间充质干细胞。成骨诱导14 d碱性磷酸酶染色、成骨诱导21 d茜素红染色,显微镜下可见明显蓝染和红染的钙结节(图1C,D),结果显示细胞有成骨诱导能力;成脂诱导28 d油红O染色,显微镜下可见大量红染脂滴(图1E),证明细胞有成脂分化能力;成软骨诱导21 d阿利新蓝染色,显微镜下可见蓝染软骨球(图1F),表明细胞有成软骨分化能力。"
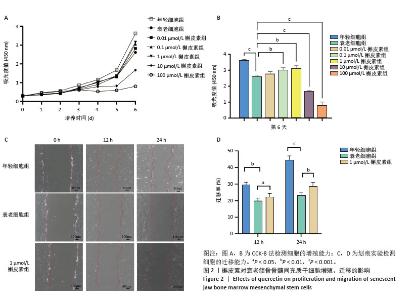
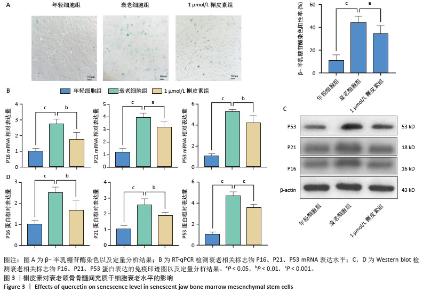
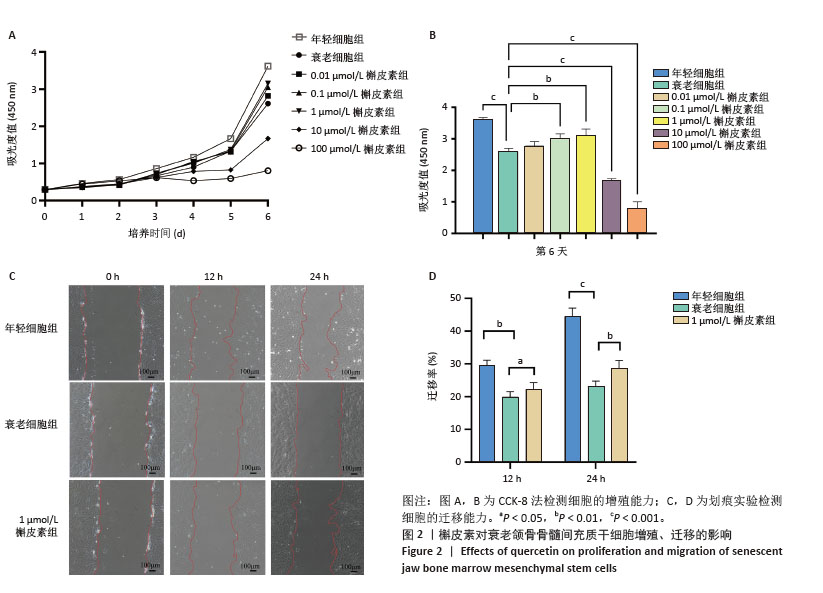
2.3 不同浓度槲皮素对衰老颌骨骨髓间充质干细胞增殖的影响 0.01,0.1,1,10,100 μmol/L含槲皮素完全培养基处理衰老颌骨骨髓间充质干细胞,第1-6天用CCK-8试剂盒测定细胞增殖活力。如图2A,B所示,第7代衰老细胞的增殖能力较第3代年轻细胞显著降低(P < 0.001),成功验证了衰老细胞模型。实验结果显示槲皮素的作用呈现明显的浓度依赖性,高浓度组(10,100 μmol/L)从处理第2天开始即显著抑制颌骨骨髓间充质干细胞增殖(P < 0.05),而低浓度组(0.01,0.1,1 μmol/L)在整个观察周期内未显示明显抑制作用;值得注意的是,1 μmol/L槲皮素组表现出最佳的促增殖作用(P < 0.01)。 2.4 槲皮素对衰老颌骨骨髓间充质干细胞迁移的影响 为了排除高浓度药物细胞毒性的影响,选用1 μmol/L槲皮素对衰老颌骨骨髓间充质干细胞进行干预,细胞划痕结果显示第7代衰老细胞的迁移能力较第3代年轻细胞显著下降(P < 0.01),成功验证了衰老细胞模型。1 μmol/L槲皮素组较衰老细胞组迁移率显著提高(P < 0.05),见图2C,D。"
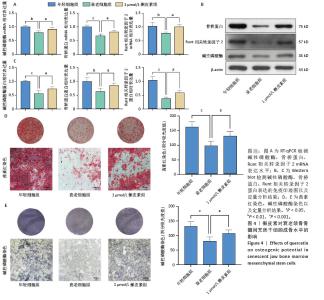
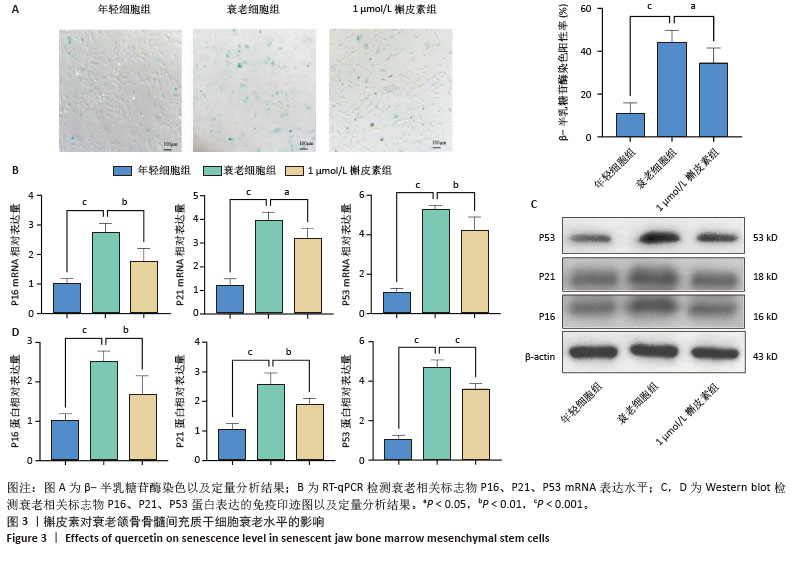
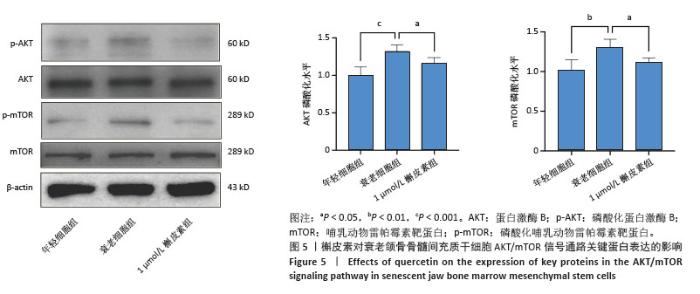
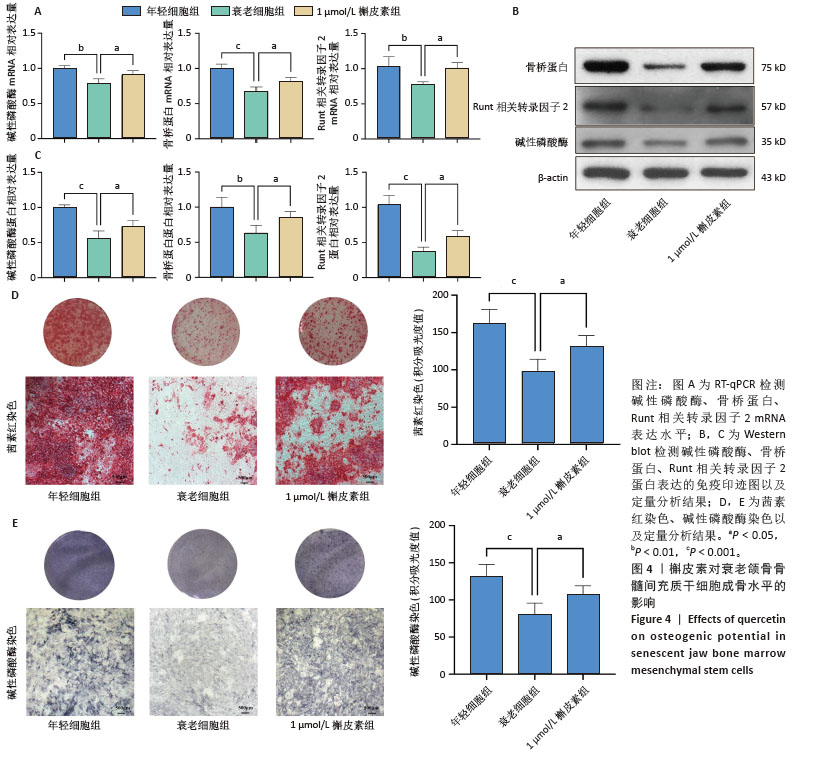
2.6 槲皮素对衰老颌骨骨髓间充质干细胞成骨的影响 成骨诱导7 d,第7代衰老细胞组成骨相关分子碱性磷酸酶、骨桥蛋白、Runt相关转录因子2的mRNA和蛋白表达水平较第3代年轻细胞组显著下降,成功验证了衰老细胞模型;1 μmol/L槲皮素组成骨相关分子碱性磷酸酶、骨桥蛋白、Runt相关转录因子2的mRNA和蛋白表达水平高于衰老细胞组;成骨诱导14 d,碱性磷酸酶染色显示1 μmol/L槲皮素组蓝紫色染色颗粒分布较密集,蓝染面积及密度高于衰老细胞组;成骨诱导21 d,茜素红染色结果显示1 μmol/L槲皮素组钙结节颗粒数量及红染面积明显多于衰老细胞组。这些结果表明1 μmol/L槲皮素提高了衰老颌骨骨髓间充质干细胞的成骨能力,见图4。"
| [1] 程钊,宇传华.全球视角的中国老年人口与疾病负担现状及趋势[J].公共卫生与预防医学,2025,36(3):1-6. [2] DU J, QIN W, WEN F, et al. Curculigoside is a Promising Osteoprotective Agent for Osteoporosis: Review. Drug Des Devel Ther. 2025;19:3323-3336. [3] ZUPAN J, STRAZAR K, KOCIJAN R, et al. Age-related alterations and senescence of mesenchymal stromal cells: Implications for regenerative treatments of bones and joints. Mech Ageing Dev. 2021;198:111539. [4] NERI S, BORZÌ RM. Molecular Mechanisms Contributing to Mesenchymal Stromal Cell Aging. Biomolecules. 2020;10(2):340. [5] WENG Z, WANG Y, OUCHI T, et al. Mesenchymal Stem/Stromal Cell Senescence: Hallmarks, Mechanisms, and Combating Strategies. Stem Cells Transl Med. 2022;11(4):356-371. [6] WANG B, HAN J, ELISSEEFF JH, et al. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat Rev Mol Cell Biol. 2024;25(12):958-978. [7] PAN Y, GU Z, LYU Y, et al. Link Between Senescence and Cell Fate: Senescence-Associated Secretory Phenotype and Its Effects on Stem Cell Fate Transition. Rejuvenation Res. 2022;25(4):160-172. [8] LOPES-PACIENCIA S, SAINT-GERMAIN E, ROWELL MC, et al. The senescence-associated secretory phenotype and its regulation. Cytokine. 2019;117:15-22. [9] ZHANG D, YU K, YANG J, et al. Senolytic controls bone marrow mesenchymal stem cells fate improving bone formation. Am J Transl Res. 2020;12(6):3078-3088. [10] CARRILLO-MARTINEZ EJ, FLORES-HERNÁNDEZ FY, SALAZAR-MONTES AM, et al. Quercetin, a Flavonoid with Great Pharmacological Capacity. Molecules. 2024;29(5):1000. [11] YANG SY, HU Y, ZHAO R, et al. Quercetin-loaded mesoporous nano-delivery system remodels osteoimmune microenvironment to regenerate alveolar bone in periodontitis via the miR-21a-5p/PDCD4/NF-κB pathway. J Nanobiotechnology. 2024;22(1):94. [12] FENG R, WANG Q, YU T, et al. Quercetin ameliorates bone loss in OVX rats by modulating the intestinal flora-SCFAs-inflammatory signaling axis. Int Immunopharmacol. 2024;136:112341. [13] ZHANG F, ZHANG Y, ZHOU J, et al. Metabolic effects of quercetin on inflammatory and autoimmune responses in rheumatoid arthritis are mediated through the inhibition of JAK1/STAT3/HIF-1α signaling. Mol Med. 2024;30(1):170. [14] LORENZO EC, TORRANCE BL, HAYNES L. Impact of senolytic treatment on immunity, aging, and disease. Front Aging. 2023;4:1161799. [15] WANG X, ZHOU Y, LUO C, et al. Senolytics ameliorate the failure of bone regeneration through the cell senescence-related inflammatory signalling pathway. Biomed Pharmacother. 2024;175:116606. [16] ZHOU Y, XIN X, WANG L, et al. Senolytics improve bone forming potential of bone marrow mesenchymal stem cells from aged mice. NPJ Regen Med. 2021;6(1):34. [17] GEORGIOU N, KAKAVA MG, ROUTSI EA, et al. Quercetin: A Potential Polydynamic Drug. Molecules. 2023;28(24):8141. [18] BI Y, QIAO X, CAI Z, et al. Exosomal miR-302b rejuvenates aging mice by reversing the proliferative arrest of senescent cells. Cell Metab. 2025;37(2): 527-541.e6. [19] ZHANG Y, ZHANG J, LESANI P, et al. Osteopontin Rejuvenates Senescent Adipose-Derived Stem Cells and Restores their Bone Tissue Regenerative Function. Stem Cell Rev Rep. 2024;20(4):1106-1120. [20] LI T, MENG X, LI S, et al. Technique for Isolation and Culture of Rat Jaw Bone Marrow Mesenchymal Stem Cells. J Vis Exp. 2024;(207):e66765. [21] ZHANG Y, YU X, ZHOU C, et al. Targeting cellular senescence in senile osteoporosis: therapeutic potential of traditional Chinese medicine. Front Med (Lausanne). 2023;10:1288993. [22] TIAN RC, ZHANG RY, MA CF. Rejuvenation of Bone Marrow Mesenchymal Stem Cells: Mechanisms and Their Application in Senile Osteoporosis Treatment. Biomolecules. 2025;15(2):276. [23] LI TQ, MENG XB, SHI Q, et al. Research progress in biological characteristics and influencing factors of jaw bone marrow mesenchymal stem cell. Zhonghua Kou Qiang Yi Xue Za Zhi. 2022;57(1): 107-112. [24] 范永晶,王姝,金武龙.颌骨来源骨髓间充质干细胞成骨分化的特点、优势与应用[J].中国组织工程研究,2024,28(1):100-106. [25] YAMAZA T, REN G, AKIYAMA K, et al. Mouse mandible contains distinctive mesenchymal stem cells. J Dent Res. 2011;90(3):317-324. [26] LEE DJ, KWON J, CURRENT L, et al. Osteogenic potential of mesenchymal stem cells from rat mandible to regenerate critical sized calvarial defect. J Tissue Eng. 2019;10:2041731419830427. [27] WATTS G. Leonard Hayflick and the limits of ageing. Lancet. 2011; 377(9783):2075. [28] BRITO KNL, TRENTIN AG. Role of mesenchymal stromal cell secretome on recovery from cellular senescence: an overview. Cytotherapy. 2025; 27(4):422-437. [29] POSPELOVA TV, LEONTIEVA OV, BYKOVA TV, et al. Suppression of replicative senescence by rapamycin in rodent embryonic cells. Cell Cycle. 2012;11(12):2402-2407. [30] LI Y, YU P, GAO Y, et al. Effects of the combination of Epimedii Folium and Ligustri Lucidi Fructus on apoptosis and autophagy in SOP rats and osteoblasts via PI3K/AKT/mTOR pathway. Biomed Pharmacother. 2024; 173:116346. [31] XIA TS, XU SY, LAI LY, et al. Bitter acids from Humulus lupulus L. alleviate D-galactose induced osteoblastic senescence and bone loss via regulating AKT/mTOR-mediated autophagy. J Food Drug Anal. 2024;32(4):506-519. [32] YANCHEN F, YALI L, XUE D, et al. Exploring the multicomponent synergy mechanism of Zuogui Wan on postmenopausal osteoporosis by a systems pharmacology strategy. J Tradit Chin Med. 2024;44(3):489-495. [33] QIU Y, ZHAO Y, JIA L, et al. Combination of dasatinib and quercetin promotes osteogenic differentiation and stemness maintenance of hPDLSCs via YAP/TAZ. Anim Cells Syst (Seoul). 2025;29(1):19-29. [34] WANG Y, CHE L, CHEN X, et al. Repurpose dasatinib and quercetin: Targeting senescent cells ameliorates postmenopausal osteoporosis and rejuvenates bone regeneration. Bioact Mater. 2023;25:13-28. [35] JIA L, XIAO H, HAO Z, et al. Senolytic elimination of senescent cells improved periodontal ligament stem cell-based bone regeneration partially through inhibiting YAP. Biochim Biophys Acta Mol Cell Res. 2025;1872(3):119921. [36] PANG XG, CONG Y, BAO NR, et al. Quercetin Stimulates Bone Marrow Mesenchymal Stem Cell Differentiation through an Estrogen Receptor-Mediated Pathway. Biomed Res Int. 2018;2018:4178021. [37] LIN H, SOHN J, SHEN H, et al. Bone marrow mesenchymal stem cells: Aging and tissue engineering applications to enhance bone healing. Biomaterials. 2019;203:96-110. [38] MARIE PJ. Bone cell senescence: mechanisms and perspectives. J Bone Miner Res. 2014;29(6):1311-1321. [39] FARR JN, KHOSLA S. Cellular senescence in bone. Bone. 2019;121:121-133. [40] KIM M, KIM C, CHOI YS, et al. Age-related alterations in mesenchymal stem cells related to shift in differentiation from osteogenic to adipogenic potential: implication to age-associated bone diseases and defects. Mech Ageing Dev. 2012;133(5):215-225. [41] DING J, ZHANG R, LI H, et al. ASIC1 and ASIC3 mediate cellular senescence of human nucleus pulposus mesenchymal stem cells during intervertebral disc degeneration. Aging (Albany NY). 2021;13(7):10703-10723. [42] HU X, LEI X, LIN W, et al. Quercetin promotes osteogenic differentiation of bone marrow mesenchymal stem cells by modulating the miR-214-3p/Wnt3a/β-catenin signaling pathway. Exp Cell Res. 2025;444(2):114386. [43] ZHANG W, JIA L, ZHAO B, et al. Quercetin reverses TNF α induced osteogenic damage to human periodontal ligament stem cells by suppressing the NF κB/NLRP3 inflammasome pathway. Int J Mol Med. 2021;47(4):39. [44] YU J, JING Z, SHEN D, et al. Quercetin promotes autophagy to alleviate cigarette smoke-related periodontitis. J Periodontal Res. 2023;58(5): 1082-1095. [45] SAXTON RA, SABATINI DM. mTOR Signaling in Growth, Metabolism, and Disease. Cell. 2017;168(6):960-976. [46] MANNING BD, CANTLEY LC. AKT/PKB signaling: navigating downstream. Cell. 2007;129(7):1261-1274. [47] LOPICCOLO J, BLUMENTHAL GM, BERNSTEIN WB, et al. Targeting the PI3K/Akt/mTOR pathway: effective combinations and clinical considerations. Drug Resist Updat. 2008;11(1-2):32-50. [48] CHU Q, LIU F, HE Y, et al. mTORC2/RICTOR exerts differential levels of metabolic control in human embryonic, mesenchymal and neural stem cells. Protein Cell. 2022;13(9):676-682. [49] FU W, WU G. Targeting mTOR for Anti-Aging and Anti-Cancer Therapy. Molecules. 2023;28(7):3157. [50] MARAFIE SK, AL-MULLA F, ABUBAKER J. mTOR: Its Critical Role in Metabolic Diseases, Cancer, and the Aging Process. Int J Mol Sci. 2024;25(11):6141. [51] WANG X, PROUD CG. The mTOR pathway in the control of protein synthesis. Physiology (Bethesda). 2006;21:362-369. [52] TAN P, WANG YJ, LI S, et al. The PI3K/Akt/mTOR pathway regulates the replicative senescence of human VSMCs. Mol Cell Biochem. 2016;422(1-2): 1-10. [53] LI Y, LIU Z, YAN H, et al. Polygonatum sibiricum polysaccharide ameliorates skeletal muscle aging and mitochondrial dysfunction via PI3K/Akt/mTOR signaling pathway. Phytomedicine. 2025;136:156316. [54] SHU Z, LI X, ZHANG W, et al. MG-132 activates sodium palmitate-induced autophagy in human vascular smooth muscle cells and inhibits senescence via the PI3K/AKT/mTOR axis. Lipids Health Dis. 2024;23(1):282. [55] HAN W, GUAN M, LIAO B, et al. Low-intensity pulsed ultrasound inhibits chondrocyte senescence by inhibiting PI3K/AKT/mTOR signaling. Braz J Med Biol Res. 2025;58:e14408. [56] YANG M, TENG S, MA C, et al. Ascorbic acid inhibits senescence in mesenchymal stem cells through ROS and AKT/mTOR signaling. Cytotechnology. 2018;70(5):1301-1313. [57] MENG D, FRANK AR, JEWELL JL. mTOR signaling in stem and progenitor cells. Development. 2018;145(1):dev152595. [58] LIU G, LI X, YANG F, et al. C-Phycocyanin Ameliorates the Senescence of Mesenchymal Stem Cells through ZDHHC5-Mediated Autophagy via PI3K/AKT/mTOR Pathway. Aging Dis. 2023;14(4):1425-1440. [59] GUO Z, ZHANG J, LI M, et al. Mechanism of action of quercetin in regulating cellular autophagy in multiple organs of Goto-Kakizaki rats through the PI3K/Akt/mTOR pathway. Front Med (Lausanne). 2024;11:1442071. [60] FODOR P, KIRÁLY J, SZABÓ Z, et al. Low-Dose Quercetin Dephosphorylates AKT and Suppresses Proteins Related to Migration in Human Metastatic Uveal Melanoma Cells. Life (Basel). 2025;15(6):979. |
| [1] | Sun Lei, Zhang Qi, Zhang Yu. Pro-osteoblastic effect of chlorogenic acid protein microsphere/polycaprolactone electrospinning membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1877-1884. |
| [2] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [3] | Jiang Xinghai, Song Yulin, Li Dejin, Shao Jianmin, Xu Junzhi, Liu Huakai, Wu Yingguo, Shen Yuehui, Feng Sicheng. Vascular endothelial growth factor 165 genes transfected into bone marrow mesenchymal stem cells to construct a vascularized amphiphilic peptide gel module [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1903-1911. |
| [4] | Wang Qisa, Lu Yuzheng, Han Xiufeng, Zhao Wenling, Shi Haitao, Xu Zhe. Cytocompatibility of 3D printed methyl acrylated hyaluronic acid/decellularized skin hydrogel scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1912-1920. |
| [5] | Hu Xiongke, Liu Shaohua, Tan Qian, Liu Kun, Zhu Guanghui. Shikonin intervention with bone marrow mesenchymal stem cells improves microstructure of femur in aged mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1609-1615. |
| [6] | Yuan Xiaoshuang, Yang Xu, Yang Bo, Chen Xiaoxu, Tian Ting, Wang Feiqing, Li Yanju, Liu Yang, Yang Wenxiu. Effect of conditioned medium of diffuse large B-cell lymphoma cells on proliferation and apoptosis of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1632-1640. |
| [7] | Li Zhenyu, Zhang Siming, Bai Jiaxiang, Zhu Chen. Osthole improves osteogenic differentiation function of bone marrow mesenchymal stem cells under high-glucose conditions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1641-1648. |
| [8] | Han Nianrong, Huang Yifei, Akram · Osman, Liu Yanlu, Hu Wei . Programmed cell death receptor-1 suppresses osteogenic differentiation of rat bone marrow mesenchymal stem cells in a high-glucose microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1649-1657. |
| [9] | Jin Dongsheng, Zhao Zhanghong, Zhu Ziyin, Zhang Sen, Sun Zuyan, Deng Jiang. Effects of icariin-loaded microsphere-three-dimensional scaffold on osteogenic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1658-1668. |
| [10] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [11] | Zhang Haiwen, Zhang Xian, Xu Taichuan, Li Chao. Bibliometric and visual analysis of the research status and trends of senescence in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1580-1591. |
| [12] | Lai Jiaming, , Song Yuling, Chen Zixi, Wei Jinghuan, Cai Hao, , Li Guoquan, . Screening of diagnostic markers for endothelial cell Senescence in mice with radiation-induced heart disease and analysis of immune infiltration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1450-1463. |
| [13] | Huang Liuyan, Zhang Wenxi, Chen Shuwen, Yu Shimei, Dai Zhong, Zuo Changqing. Forskolin promotes C2C12 myoblast differentiation via regulating the ERK and Akt signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1114-1121. |
| [14] | Zou Rongji, Yu Fangfang, Wang Maolin, Jia Zhuopeng. Triptolide inhibits ferroptosis and improves cerebral ischemia-reperfusion injury in a rat model of cerebral artery occlusion/reperfusion [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 873-881. |
| [15] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanism by which vascular endothelial growth factor A targets regulation of angiogenesis in the treatment of steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 671-679. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||