Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (33): 5263-5269.doi: 10.12307/2023.485
Previous Articles Next Articles
Aspartate-glutamate carrier 1 regulates myelination in preterm rats with periventricular leukomalacia
Wang Lizhen1, Yang Caijin1, Chen Rong2, Li Ling1
- 1Department of Pediatrics, Hainan Western Central Hospital, Danzhou 571700, Hainan Province, China; 2Department of Neurology, The First Affiliated Hospital of Hainan Medical University, Haikou 570102, Hainan Province, China
-
Received:2021-10-21Accepted:2022-07-31Online:2023-11-28Published:2023-03-29 -
Contact:Li Ling, Chief physician, Department of Pediatrics, Hainan Western Central Hospital, Danzhou 571700, Hainan Province, China -
About author:Wang Lizhen, Attending physician, Department of Pediatrics, Hainan Western Central Hospital, Danzhou 571700, Hainan Province, China -
Supported by:Hainan Provincial Natural Science Foundation in 2017, No. 817325 (to WLZ)
CLC Number:
Cite this article
Wang Lizhen, Yang Caijin, Chen Rong, Li Ling. Aspartate-glutamate carrier 1 regulates myelination in preterm rats with periventricular leukomalacia[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5263-5269.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
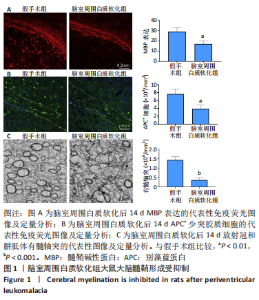
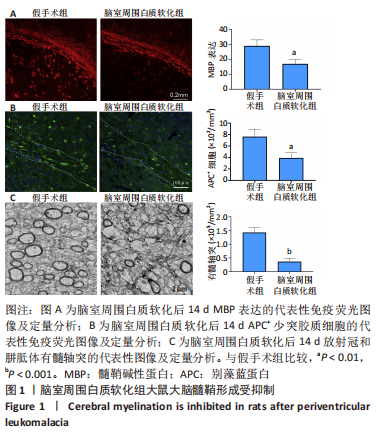
2.1 实验动物数量分析 假手术组和脑室周围白质软化组大鼠,每组30只;用于分离室周白质组织5只大鼠;pcDNA3-AGC1组和pcDNA3-NC组大鼠,每组10只。共计85只,实验过程中动物无死亡。 2.2 脑室周围白质软化抑制发育中大脑的髓鞘形成 在脑室周围白质软化后14 d,分别通过MBP检测髓鞘形成和APC免疫染色的成熟少突胶质细胞数量(图1A,B)。与假手术组相比,脑室周围白质软化组大鼠白质中MBP表达以及APC+少突胶质细胞的数量显著减少(P < 0.01)。与免疫染色结果相似,透射电子显微镜显示脑室周围白质软化组白质中有髓轴突明显少于假手术组(P < 0.001),见图1C。这些结果表明,脑室周围白质软化抑制白质中少突胶质细胞分化和髓鞘形成。"
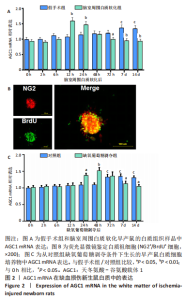
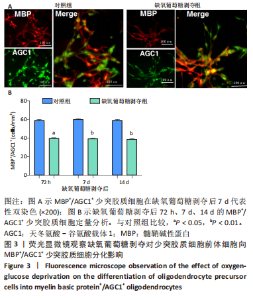
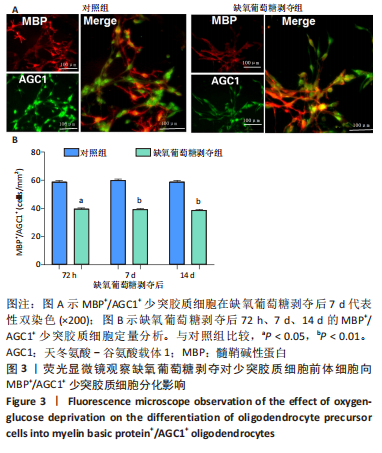
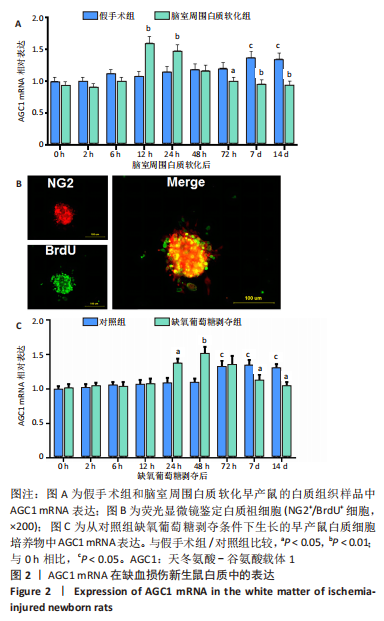
2.3 脑室周围白质软化以及缺氧葡萄糖剥夺对AGC1 mRNA表达影响 AGC1在许多神经系统疾病中失调,包括帕金森病、多发性硬化症和阿尔茨海默病[5-6]。因此,通过实时荧光定量PCR对AGC1进行定量分析。在体内假手术条件下,未成熟脑组织发育7,14 d后,AGC1 mRNA表达开始显著增加(P < 0.05);与0 h相比,体内脑室周围白质软化开始后12-24 h时AGC1 mRNA表达显著升高(P < 0.05),而在72 h-14 d时显著降低(P < 0.05),见图2A。通过荧光显微镜鉴定作为白质祖细胞标志物的NG2和作为细胞增殖标志物的BrdU,结果表明,白质祖细胞可以增殖为同时出现NG2阳性红色荧光和BrdU阳性绿色荧光的神经球,见图2B。在体外对照条件下,与0 h相比,对照组72 h-14 d时AGC1 mRNA表达显著增加(P < 0.05);与对照组相比,在缺氧葡萄糖剥夺后24 h开始显著诱导AGC1 mRNA表达(P < 0.05),并在48 h达到最大值(P < 0.05),而在缺氧葡萄糖剥夺后7-14 d时显著降低(P < 0.05),见图2C。"
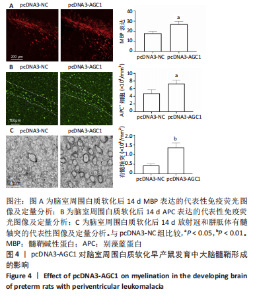
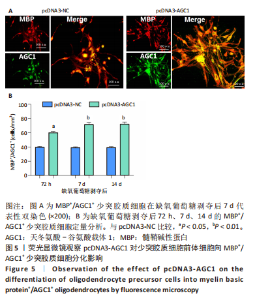
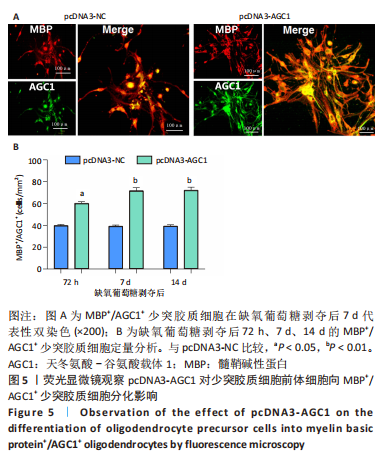
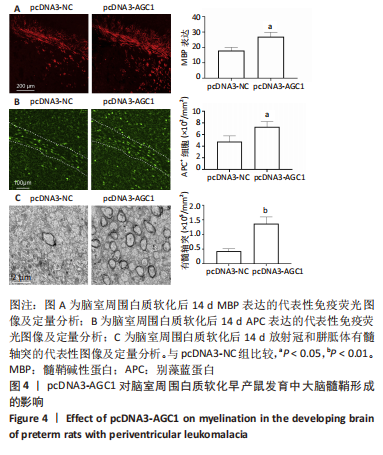
2.5 AGC1对髓鞘形成的影响 鉴于AGC1对髓鞘形成的有益影响,研究通过构建pcDNA3-AGC1质粒增强白质中AGC1表达。结果显示,pcDNA3-AGC1处理组在脑室周围白质软化后14 d处髓鞘MBP染色和APC+少突胶质细胞数量比pcDNA3-NC对照组显著增强(P < 0.05),见图4A,B。透射电子显微镜显示,在用pcDNA3-AGC1治疗后,白质中有髓轴突数量显著增加(P < 0.05),见图4C,表明AGC1促进脑室周围白质软化脑内少突胶质细胞分化和髓鞘形成。此外,在体外实验中,pcDNA3-AGC1进一步促进了缺氧葡萄糖剥夺后72?h、7 d和14 d时少突胶质细胞前体细胞分化为MBP+/AGC1+?少突胶质细胞(P < 0.05),见图5。"
| [1] 张玉,王向鹏,刘佳,等. 脑室周围白质软化大鼠脑组织Toll样受体4表达变化与细胞凋亡的关系[J]. 中华实用儿科临床杂志, 2019,34(16):1254-1258. [2] HAO L, SHEN L, YIN Q. Clinical Diagnosis of White Matter Softening in Premature Infants Based on Electroencephalogram (EEG). J Healthc Eng. 2021;2021(98):1-10. [3] HUANG XM. WEN SY, NG E, et al. Periventricular white matter hyperintensity burden and cognitive impairment in early Parkinson’s disease. Eur J Neurol. 2020;27(6):876-886. [4] 叶孝严,王大雨,徐艳,等. 普仑司特对脑室周围白质软化新生大鼠的作用[J]. 中国当代儿科杂志,2020,22(6):129-134. [5] REVIN VV, PINYAEV SI, PARCHAYKINA MV, et al. The Effect of Resveratrol on the Composition and State of Lipids and the Activity of Phospholipase A2 During the Excitation and Regeneration of Somatic Nerves. Front Physiol. 2019;10:384. [6] BORHANI-HAGHIGHI M, MOHAMADI Y, KASHANI IR. In utero transplantation of neural stem cells ameliorates maternal inflammation-induced prenatal white matter injury. J Cell Biochem. 2019;120(8): 12785-12795. [7] FU XM, WANG Y, FU WL, et al. The Combination of Adipose-derived Schwann-like Cells and Acellular Nerve Allografts Promotes Sciatic Nerve Regeneration and Repair through the JAK2/STAT3 Signaling Pathway in Rats. Neuroscience. 2019;422(6):134-145. [8] YU J, YE K, LI J, et al. Schwann-like cell conditioned medium promotes angiogenesis and nerve regeneration. Cell Tissue Bank. 2021;23(1): 1-18. [9] GROTH SL, DONAHUE SP, REDDY A, et al. Periventricular Leukomalacia in Patients With Pseudo-glaucomatous Cupping. Am J Ophthalmol. 2020;211:31-41. [10] FORBES TA, GOLDSTEIN EZ, DUPREE JL, et al. Environmental enrichment ameliorates perinatal brain injury and promotes functional white matter recovery. Nature Commun. 2020;11(1):67-74. [11] PETRALLA S, PEÑA-ALTAMIRA LE, POETA E, et al. Deficiency of mitochondrial aspartate-glutamate carrier 1 leads to oligodendrocyte precursor cell proliferation defects both in vitro and in vivo. Int J Mol Sci. 2019;20(18):4486. [12] 何亚芳, 张建华, 陈惠金, 等. 美金胺对缺血诱导未成熟脑白质谷氨酸异常信号传输的体内外阻断研究[J]. 中华实用儿科临床杂志, 2020,35(14):1093-1097. [13] PROFILO E, PEÑA-ALTAMIRA LE, CORRICELLI M, et al. Down-regulation of the mitochondrial aspartate-glutamate carrier isoform 1 AGC1 inhibits proliferation and N-acetylaspartate synthesis in Neuro2A cells. Biochim Biophys Acta Mol Basis Dis. 2017;1863(6):1422-1435. [14] SHELTON GD, MINOR KM, LI K, et al. A Mutation in the Mitochondrial Aspartate/Glutamate Carrier Leads to a More Oxidizing Intramitochondrial Environment and an Inflammatory Myopathy in Dutch Shepherd Dogs. J Neur Dis. 2019;6(4):485-501. [15] CONTRERAS L, RAMIREZ L, DU J, et al. Deficient glucose and glutamine metabolism in Aralar/AGC1/Slc25a12 knockout mice contributes to altered visual function. Mol Vis. 2016;22(3):1198. [16] PROFILO E, PEÑA-ALTAMIRA LE, CORRICELLI M, et al. Down-regulation of the mitochondrial aspartate-glutamate carrier isoform 1 AGC1 inhibits proliferation and N-acetylaspartate synthesis in Neuro2A cells. BBA Mol Basis Dis. 2017;1863(6):1422-1435. [17] PFEIFFER B, SEN K, KAUR S, et al. Expanding Phenotypic Spectrum of Cerebral Aspartate-Glutamate Carrier Isoform 1 (AGC1) Deficiency. Neuropediatrics. 2019;51(2):160-163. [18] QIAO L, FU J, XUE X, et al. Neuronalinjury and roles of apoptosis and autophagy in a neonatal rat model of hypoxia-ischemia-induced periventricular leukomalacia. Mol Med Rep. 2018;17(4):5940-5949. [19] MLODY B, LORENZ C, INAK G, et al. Energy metabolism in neuronal/glial induction and in iPSC models of brain disorders. Semin Cell Dev Biol. 2016;52(9):102-109. [20] CAICEDO A, ZAMBRANO K, SANON S, et al. Extracellular mitochondria in the cerebrospinal fluid (CSF): Potential types and key roles in central nervous system (CNS) physiology and pathogenesis. Mitochondrion. 2021;58(12):1123-1132. [21] VISCOMI C, BOTTANI E, ZEVIANI M. Emerging concepts in the therapy of mitochondrial disease. Biochim Biophys Acta. 2015;1847(6-7): 544-557. [22] RANGARAJU V, LEWIS TL, HIRABAYASHI Y, et al. Pleiotropic Mitochondria: The Influence of Mitochondria on Neuronal Development and Disease. J Neurosci. 2019;39(42):8200-8208. [23] FALK MJ, LI D, GAI X, et al. AGC1 deficiency causes infantile epilepsy, abnormal myelination, and reduced N-acetylaspartate. JIMD Rep. 2014;14(5):77-85. [24] CASIMIR M, RUBI B, FRIGERIO F, et al. Silencing of the mitochondrial NADH shuttle component aspartate-glutamate carrier AGC1/Aralar1 in INS-1E cells and rat islets. Biochem J. 2009;424(3):459-466 [25] PAN S, CHAN JR. Regulation and dysregulation of axon infrastructure by myelinating glia. J Cell Biol. 2017;216(12):3903-3916. [26] ETXEBERRIA A, HOKANSON KC, DAO DQ, et al. Dynamic modulation of myelination in response to visual stimuli alters optic nerve conduction velocity. J Neurosci. 2016;36(26):6937-6948. [27] MUSAH AS, BROWN TL, JEFFRIES MA, et al. Mechanistic Target of Rapamycin Regulates the Oligodendrocyte Cytoskeleton during Myelination. J Neurosci. 2020;40(15):JN-RM-1434-18. [28] TOMASSY GS, BERGER DR, CHEN HH, et al. Distinct profiles of myelin distribution along single axons of pyramidal neurons in the neocortex. Science. 2014;344(6181):319-324. [29] ZUCCOLI GS, REIS-DE-OLIVEIRA G, GARBES B, et al. Linking proteomic alterations in schizophrenia hippocampus to NMDAr hypofunction in human neurons and oligodendrocytes. Eur Arch Psychiatry Clin Neurosci. 2021;24(3):409-416. [30] REDLICH MJ, PRALL B, CANTO-SAID E, et al. High-pulse-energy multiphoton imaging of neurons and oligodendrocytes in deep murine brain with a fiber laser. Sci Rep. 2021;11(1):306-315. [31] RAMOS M, PARDO B, LLORENTE-FOLCH I, et al. Deficiency of the mitochondrial transporter of aspartate/glutamate aralar/AGC1 causes hypomyelination and neuronal defects unrelated to myelin deficits in mouse brain. J Neurosci Res. 2011;89(12):2008-2017. [32] SAKURAI T, RAMOZ N, BARRETO M, et al. Slc25a12 disruption alters myelination and neurofilaments: a model for a hypomyelination syndrome and childhood neurodevelopmental disorders. Biol Psychiatry. 2010;67(9):887-894. [33] LLORENTE-FOLCH I, RUEDA CB, PÉREZ-LIÉBANA I, et al. L-Lactate-Mediated Neuroprotection against Glutamate-Induced Excitotoxicity Requires ARALAR/AGC1. J Neurosci. 2016;36(16):4443-4456. [34] PÉREZ-LIÉBANA I, CASAREJOS MJ, ALCAIDE A, et al. βOHB Protective Pathways in Aralar-KO Neurons and Brain: An Alternative to Ketogenic Diet. J Neurosci. 2020;40(48):9293-9305. [35] NAPOLIONI V, PERSICO AM, PORCELLI V, et al. The mitochondrial aspartate/glutamate carrier AGC1 and calcium homeostasis: physiological links and abnormalities in autism. Mol Neurobiol. 2011; 44(1):83-92. [36] PUERTAS-FRÍAS G, DEL ARCO A, PARDO B, et al. Mitochondrial movement in Aralar/Slc25a12/AGC1 deficient cortical neurons. Neurochem Int. 2019;131:104541. [37] FALK MJ, LI D, GAI X, et al. AGC1 Deficiency Causes Infantile Epilepsy, Abnormal Myelination, and Reduced N-Acetylaspartate. JIMD Rep. 2014;14:77-85. [38] PARDO B, CONTRERAS L, SATRÚSTEGUI J. De novo Synthesis of Glial Glutamate and Glutamine in Young Mice Requires Aspartate Provided by the Neuronal Mitochondrial Aspartate-Glutamate Carrier Aralar/AGC1. Front Endocrinol (Lausanne). 2013;4:149. [39] PROFILO E, PEÑA-ALTAMIRA LE, CORRICELLI M, et al. Down-regulation of the mitochondrial aspartate-glutamate carrier isoform 1 AGC1 inhibits proliferation and N-acetylaspartate synthesis in Neuro2A cells. Biochim Biophys Acta Mol Basis Dis. 2017;1863(6):1422-1435. [40] JUARISTI I, GARCÍA-MARTÍN ML, RODRIGUES TB, et al. ARALAR/AGC1 deficiency, a neurodevelopmental disorder with severe impairment of neuronal mitochondrial respiration, does not produce a primary increase in brain lactate. J Neurochem. 2017;142(1):132-139. |
| [1] | Gao Yue, Lin Jianwen, Li Di, Lan Xiaoyan, Li Shen, Chu Chengyan. Oligodendrocyte progenitor cells prolong the survival of glioblastoma-bearing rats after high-dose radiotherapy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2669-2674. |
| [2] | Song Shengjiao, Li Juan, Wu Wencheng, Xiao Yun, Liao Baoying, Li Xing. Function of oligodendrocytes and demyelinating disease [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3108-3116. |
| [3] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [4] | Xie Yang, Lü Zhiyu, Zhang Shujiang, Long Ting, Li Zuoxiao. Effects of recombinant adeno-associated virus mediated nerve growth factor gene transfection on oligodendrocyte apoptosis and myelination in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3678-3683. |
| [5] | Ye Dou, , Ma Xuexia , Guan Qian, , Luan Zuo , Yang Yinxiang , Wang Zhaoyan , Wang Qian , He Ying , Yao Ruiqin. Proportion and morphological characteristics of human oligodendrocyte precursor cells in different cell culture vessels [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 44-49. |
| [6] | Wang Guoyu, Cheng Zhijian, Yang Baohui, Li Haopeng, He Xijing. Olfactory ensheathing cell transplantation promotes the ultrastructure repair at the lesion site of rat models of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(5): 699-703. |
| [7] | Zhao Hong, Wang Suping, Lian Jianwen. Insight into white matter changes in Alzheimer’s disease: a focus on myelin and oligodendrocyte [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(13): 2120-2125. |
| [8] | Wang Zhaoyan, Yang Yinxiang, Wang Qian, Guan Qian, Luan Zuo. Effects of different preservation media on the biological properties of human oligodendrocyte precursor cells [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(29): 4651-4655. |
| [9] | Wu Cheng-jun, Su Xue-wen, Wang Zhao-yan, Yang Yin-xiang, Luan Zuo. Oligodendrocyte progenitor cell transplantation in combination with miconazole for myelin repair in mice with leukodystrophy [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(25): 4041-4046. |
| [10] | Chang Biao, Quan Qi, Sun Xun, Liu Ruo-xi, Wang Yu, Lu Shi-bi, Peng Jiang. Wallerian degeneration after peripheral nerve injury: research advance in nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(10): 1596-1603. |
| [11] | Xiong Le1, Zhang Bei1, Shen Ruo-wu2, Ji Ai-yu3, Sun Guang-qiang1, Bian Hong-lin3, Zhang Feng-yu1, Wang Yi1, Huang Heng4, Li Hua-qiao1, Zhou Shan-yu5, Shen Zhao-kang6, Wang Zhong1 . Toll-like receptor 4 antagonist protects against Wallerian degeneration after peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(42): 6308-6316. |
| [12] | Suo Lei, Yang Yin-xiang, Tang Wen-yan, Luan Zuo . Dynamic expression of Lingo-1 in a model of white matter injury [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(27): 4043-4048. |
| [13] | Li Min, Liu Jia, Zhong Yu-hua, Peng Fu-hua. Changes of gliacytes after spinal cord transection in Ambystoma mexicanum [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(11): 1993-2000. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||