Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (21): 3319-3326.doi: 10.12307/2022.638
Previous Articles Next Articles
Preparation and characterization of the composites of amino-modified artificial jaw nano-hydroxyapatite/polylactic acid
Liu Longzhu1, Long Yuanzhu2, Yang Chengxue1, Zhong Xinqi1, Wang Yifang1, Liu Jianguo1
- 1School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China; 2Institute of Life Sciences, Zunyi Medical University, Zunyi 563006, Guizhou Province, China
-
Received:2021-09-09Accepted:2021-10-28Online:2022-07-28Published:2022-01-27 -
Contact:Liu Jianguo, Professor, Doctoral supervisor, School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China -
About author:Liu Longzhu, Master candidate, Physician, School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China -
Supported by:National Key Research and Development Project, No. 2016YFC1102800 (to LJG); Guizhou Medical Biomaterials Research and Development Talent Base, No. [2018]3 (to LJG); Zunyi Medical Biomaterials Research and Development Innovative Talent Base, No. [2019]69 (to LJG)
CLC Number:
Cite this article
Liu Longzhu, Long Yuanzhu, Yang Chengxue, Zhong Xinqi, Wang Yifang, Liu Jianguo. Preparation and characterization of the composites of amino-modified artificial jaw nano-hydroxyapatite/polylactic acid[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3319-3326.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

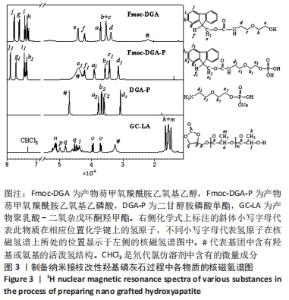
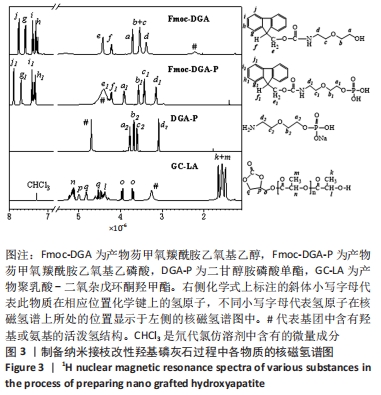
图3中,相比于Fmoc-DGA,Fmoc-DGA-P引入了磷酸基团,活泼氢#的峰面积大幅度增加。此外,和磷酸基团邻近的a1、b1、c1峰相对于a、b、c峰的位置发生了明显变化,但各个峰的面积没有发生变化,这也说明Fmoc-DGA-P对比于Fmoc-DGA只是羟基变成了磷酸基团。Fmoc-DGA-P除Fmoc基团后得到的DGA-P,在大于7×10-6处没有了典型的二苯并五环的结构峰,说明成功制备出了DGA-P。 由于实验制备的DGA-HA和LA-HA都是固体,因此不能得到相应的核磁氢谱图谱。 接枝侧链GC-LA是甘油碳酸酯和DL-丙交酯反应而成,DL-丙交酯的结构是3,6-二甲基-1,4-二氧杂环己烷-2,5-二酮的对称环状结构,只分别在1.7×10-6和5.1×10-6的位置有2个峰。对于GC-LA曲线,见图3,q、p、o三个峰来源于甘油碳酸酯结构,而2.0×10-6以下的位置的k和m两个峰,与DL-丙交酯区别明显,这属于DL-丙交酯的环状结构开环后得到的与烷基相连的-CH3峰。DL-丙交酯开环后和甘油碳酸酯反应形成了酯基,而相连的n峰就显示在5.2×10-6的位置。DL-丙交酯开环和-OH相连的烷基则在4.4 ×10-6的位置,和q峰形成了重叠峰。GC-LA的化学式显示甘油碳酸酯上基团的氢原子数是确定的,可以计算出GC-LA中源于DL-丙交酯上基团的重复单元数。以p峰为基准峰,设置峰的面积为1,则k+m为11.98,而n为2.96,通过这些数据可以看出图中的GC-LA源于DL-丙交酯的重复单元数约为3。 因此,由图3可知,不仅成功合成每一步的产物,而且计算出了GC-LA侧链的具体重复单元。 除了进行核磁氢谱测试,还对制备侧链改性HA过程中各物质进行了傅里叶红外光谱测试。图4中,Fmoc-DGA的曲线在1 694 cm-1出现C=O基团的伸缩振动峰,1 539 cm-1处出现氨基甲酸酯基团中氨基的振动峰,这2个特征峰说明芴甲氧羰酰氯与二甘醇胺的氨基反应并生成氨基甲酸酯基团。此外,Fmoc-DGA在3 324 cm-1处为-OH基团的峰,3 063 cm-1处则是苯环上的碳氢键伸缩振动形成的峰,2 882,2 940 cm-1处为亚甲基形成的峰。这些特征峰都说明成功制备出Fmoc-DGA。"
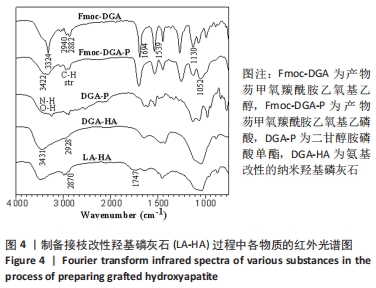
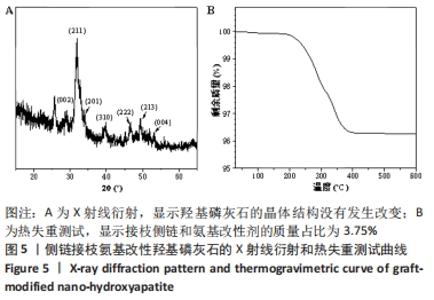
相对于Fmoc-DGA,Fmoc-DGA-P在3 422 cm-1处出现了磷酸根上氢的振动峰,在1 052 cm-1处出现P-O键形成的强峰,这都说明Fmoc-DGA-P制备成功。DGA-P相对于Fmoc-DGA-P在傅里叶红外光谱曲线上没有芳环的烷基峰和氨基甲酸酯基团上的C=O特征峰,表示DGA-P上已经没有了Fmoc基团。此外,DGA-P在1 000-1 132 cm-1范围内的磷酸基团仍然存在,说明DGA-P的成功制备。 DGA-HA是以DGA-P作为原料之一制备得到的。图4中代表HA的特征基团,包含3 431 cm-1处-OH的峰和1 034 cm-1处-PO3的峰相对于DGA-P得到了明显增强,而2 928 cm-1处亚甲基的峰说明DGA-P参与反应并得到DGA-HA。 图3和图4说明接枝侧链GC-LA和DGA-HA都已经制备成功。因此可以通过GC-LA的甲基二氧杂戊环酮结构和DGA-HA表面的氨基结构反应,得到GC-LA侧链改性的LA-HA。图4中LA-HA和DGA-HA红外光谱曲线十分类似,但是2 993和2 870 cm-1上甲基伸缩振动峰的强度明显增强,这是源于GC-LA侧链上的甲基基团的作用。而LA-HA在1 747 cm-1有一个很强的峰,这是源于GC-LA上的C=O基团。LA-HA含有源于GC-LA的特征峰,证实了GC-LA对DGA-HA进行接枝改性得到了LA-HA。 图5A中标记为(002)(211)(202)(310)(222)(213)(004)的7个峰都是HA在X射线衍射得出的晶体特征峰,因此LA-HA中属于HA的晶体结构没有改变。图3、4和图5A中的数据都可以说明成功制备出了LA-HA,而LA-HA中有机成分,即接枝侧链和氨基改性剂的含量可以通过图5B中的热失重测试获取。LA-HA在200 ℃之前几乎没有质量损失,而在200-400 ℃温度范围显示出有机物分解,挥发并失去质量;剩余的无机HA成分在600 ℃之内不会分解,从而可以知道失去的质量即为LA-HA中的有机成分,质量占比为LA-HA的3.75%。"
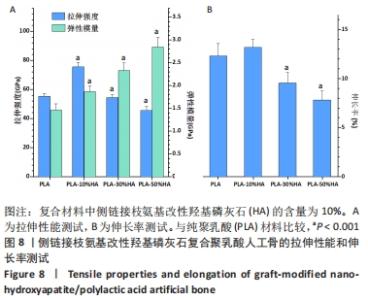
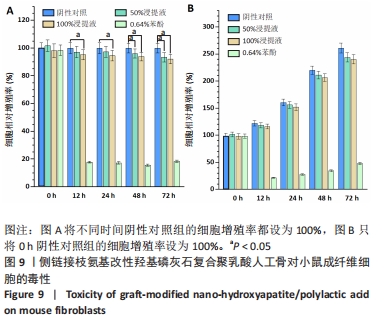
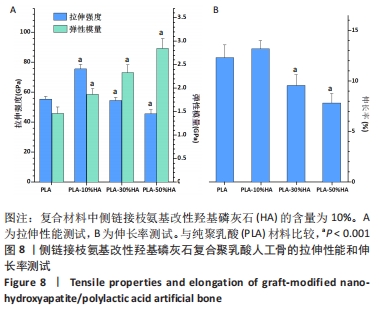
2.2.2 机械性能测试结果 对PLA-HA复合材料作为人工骨的机械性能进行表征,可以发现随着LA-HA含量的增加,材料的拉伸强度先增加再降低,见图8A。其中PLA-10%HA复合材料的拉伸强度最高,达到了75.6 MPa,比纯PLA材料的拉伸强度增加了36.3%;当LA-HA的含量达到30%时,复合材料的拉升强度相对于纯PLA材料不再增加,进一步增加LA-HA的含量,复合物的拉升强度甚至低于纯PLA材料。不同于拉升强度,随着LA-HA的增加,材料的弹性模量大幅增加。PLA-10%HA、PLA-30%HA和PLA-50%HA复合材料的弹性模量分别为1.87,2.33,2.85 GPa,分别比纯PLA材料提升了28.1%,59.3%,94.5%。图8B中PLA-HA复合材料的伸长率的变化趋势和图8A中的拉伸强度变化趋势类似,不同的是,PLA-10%HA的伸长率相对于纯PLA材料无统计学差异。 从图8可以看出,PLA-10%HA复合材料的拉伸强度和弹性模量相对于纯PLA材料有了明显提升,而且伸长率没有降低,提示PLA-10%HA复合材料作为人造颌骨的综合机械性能可能是最佳的。"
| [1] ZHAO D, ZHU T, LI J, et al. Poly(lactic-co-glycolic acid)-based composite bone-substitute materials. Bioact Mater. 2020;6(2):346-360. [2] BLUME O, BACK M, MARTIN K, et al. A customized allogenic bone block for alveolar reconstruction quantitated by a 3D matching technique: A case report. Clin Case Rep. 2021;9(9):e04771. [3] BARBU HM, LANCU SA, RAPANI A, et al. Guided bone regeneration with concentrated growth factor enriched bone graft matrix (sticky bone) vs. bone-shell technique in horizontal ridge augmentation: a retrospective study. J Clin Med. 2021;10(17):3953. [4] ORYAN A, ALIDADI S, MOSHIRI A, et al. Bone regenerative medicine: classic options, novel strategies, and future directions. J Orthop Surg Res. 2014;9(1):18. [5] ZHANG Y, LUAN J, ZHANG Y, et al. Preparation and characterization of iron-doped tricalcium silicate-based bone cement as a bone repair material. Materials (Basel). 2020;13(17):3670. [6] PAMENTIER L, RIFFAULT M, HOEY DA. Utilizing osteocyte derived factors to enhance cell viability and osteogenic matrix deposition within IPN hydrogels. Materials (Basel). 2020;13(7):1690. [7] KONG D, LIN G, SHI Y, et al. Performance of heterotopic bone elicited with bone morphogenic protein-2 microspheres as a bone repair material. Mater Design. 2020;191:108657. [8] PASCU EI, STOKES J, MCGUINNESS GB. Electrospun composites of PHBV, silk fibroin and nano-hydroxyapatite for bone tissue engineering. Mater Sci Eng C. 2013;33(8):4905-4916. [9] KHAN FSA, MUBARAK NM, KHALID M, et al. Functionalized multi-walled carbon nanotubes and hydroxyapatite nanorods reinforced with polypropylene for biomedical application. Sci Rep. 2021;11(1):843. [10] OBERDIEK F, VARGAS CI, RIDER P, et al. Ex vivo and in vivo analyses of novel 3D-Printed bone substitute scaffolds incorporating biphasic calcium phosphate granules for bone regeneration. Int J Mol Sci. 2021;22(7):3588. [11] BARBECK M, SCHRODE ML, ALKILDANI S, et al. Exploring the biomaterial-induced secretome: physical bone substitute characteristics influence the cytokine expression of macrophages. Int J Mol Sci. 2021;22(9):4442. [12] GIRON J, KERSTNER E, MEDDEIROS T, et al. Biomaterials for bone regeneration: an orthopedic and dentistry overview. Braz J Med Biol Res. 2021;54(9):e11055. [13] Al-AMIN M, ABDUL-RANI AM, DANISH M, et al. Investigation of coatings, corrosion and wear caracteristics of machined biomaterials through hydroxyapatite mixed-EDM process: a review. Materials (Basel). 2021; 14(13):3597. [14] GARCIA -GARETA E, COATHUP MJ, BLUNN GW. Osteoinduction of bone grafting materials for bone repair and regeneration. Bone. 2015;81:112-121. [15] JI C, BI L, LI J, et al. Salvianolic acid b-loaded chitosan/hydroxyapatite scaffolds promotes the Repair of segmental bone defect by angiogenesis and osteogenesis. Int J Nanomedicine. 2019;14:8271-8284. [16] MAZZONI E, MAZZIOTTA C, IAQUINTA MR, et al. Enhanced osteogenic differentiation of human bone marrow-derived mesenchymal stem cells by a hybrid hydroxylapatite/collagen scaffold. Front Cell Dev Biol. 2021;8: 610570. [17] RAHMATI M, STOTZEL S, KHASSAWNA TE, et al. Early osteoimmunomodulatory effects of magnesium-calcium-zinc alloys. J Tissue Eng. 2021;12:20417314211047100. [18] KARAGEORGIOU V, KAPLAN D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials. 2005;26(27):5474-5491. [19] BUTREDDY A, GADDAM RP, KOMMINENI N, et al. PLGA/PLA-based long-acting injectable depot microspheres in clinical use: production and characterization overview for protein/peptide delivery. Int J Mol Sci. 2021; 22(16):8884. [20] HAMMOUCHE S, HAMMOUCHE D, MCNICHOLAS M. Biodegradable bone regeneration synthetic scaffolds: in tissue engineering. Curr Stem Cell Res Ther. 2012;7(2):134-142. [21] ROCHA CR, CHAIVEZ-FLORES D, ZUVERZA-MENA N, et al. Surface organo-modification of hydroxyapatites to improve PLA/HA compatibility. J Appl Polym Sci. 2020:e49293. [22] 张晓明,王洪艳,李俊锋.改性MWNTs/纳米HA/PLA骨修复材料的制备[J].吉林大学学报(工学版),2008,38(4):844-847. [23] 孙帆,顾永江,孙富华,等.离子键增强水性聚氨酯/纳米羟基磷灰石复合材料的研究[J].功能材料,2015,46(11):11006-11010. [24] DONYA H, DARWESH R, AHMED MK. Morphological features and mechanical properties of nanofibers scaffolds of polylactic acid modified with hydroxyapatite/CdSe for wound healing applications. Int J Biol Macromol. 2021;186:897-908. [25] BAKIR M, MEYER JL, SUTRISNO A, et al. Aromatic thermosetting copolyester bionanocomposites as reconfigurable bone substitute materials: Interfacial interactions between reinforcement particles and polymer network. Sci Rep. 2018;8(1):14869. [26] ONAK G, KARAMAN O. Accelerated mineralization on nanofibers via non-thermal atmospheric plasma assisted glutamic acid templated peptide conjugation. Regen Biomater. 2019;6(4):231-240. [27] LUO JB, QIU SX, WANG YL, et al. Preparation and physicochemical properties of hydroxyapatite/polyurethane nanocomposites. Chin. J Polym Sci. 2014;32:467-475. [28] SUPOVA M. Problem of hydroxyapatite dispersion in polymer matrices: a review. J Mater Sci Mater Med. 2009;20:1201-1213. [29] KO HS, LEE S, JHO JY. Synthesis and modification of hydroxyapatite nanofiber for poly(lactic acid) composites with enhanced mechanical strength and bioactivity. Nanomaterials (Basel). 2021;11(1):213. [30] KO HS, LEE S, LEE D, et al. Mechanical properties and bioactivity of poly(lactic acid) composites containing poly(glycolic acid) fiber and hydroxyapatite particles. Nanomaterials (Basel). 2021;11(1):249. [31] ZHAO L, CHENG SM, LIU SH, et al. Improvement of the addition amount and dispersion of hydroxyapatite in the poly(lactic acid) matrix by the compatibilizer-epoxidized soybean oil. J Mater Res. 2020;35(12):1523-1530. [32] SUN J, SHEN J, Chen S, et al. Nanofiller reinforced biodegradable PLA/PHA composites: current status and future trends. Polymers (Basel). 2018;10(5): 505. [33] 李刚,刘晓南,张道海,等.亲水改性剂表面修饰纳米羟基磷灰石对其分散性的影响[J].高分子材料科学与工程,2018,34(10):66-72. [34] ZHENG YT, FU G, WANG B, et al. Physico-chemical, thermal, and mechanical properties of PLA-nHA nanocomposites: effect of glass fiber reinforcement. J Appl Polym Sci. 2020:e49286. https://doi.org/10.1002/app.49286 [35] JIANG YR, SUN FH, ZHOU XY, et al. Water dispersible hydroxyapatite nanoparticles functionalized by a family of aminoalkyl phosphates. Chin Chem Lett. 2015;26(9):1121-1128. [36] KONG WR, ZHOU XY, YANG Y, et al. A facile Synthesis of ω-aminoalkyl ammonium hydrogen phosphates. Chin Chem Lett. 2012;23:923-926. [37] GONG XY, LIU YY, WANG YS, et al. Amino graphene oxide/dopamine modified aramid fibers: preparation, epoxy nanocomposites and property analysis. Polymer. 2019;168:131-137. [38] CHEN SK, HORI N, KAJIYAMA M, et al. Compatibilities and properties of poly lactide/poly (methyl acrylate) grafted chicken feather composite: effects of graft chain length. J Appl Polym Sci. 2020;137:e48981. [39] LIAO CZ, LI K, WONG HM, et al. Novel polypropylene biocomposites reinforced with carbon nanotubes and hydroxyapatite nanorods for bone replacements. Mater Sci Eng C. 2013;33(3):1380-1388. [40] FATHI MH V, MORTAZAVI, et al. Bioactivity evaluation of synthetic nano-crystalline hydroxyapatite. Dent Res J. 2008;5:81-87. [41] LIN LW, CHOW KL, LENG Y, et al. Study of hydroxyapatite osteoinductivity with anosteogenic differentiation of mesenchymal stem cells. J Biomed Mater Res. 2009;A89A:326-335. [42] ZHI W, ZHANG C, DUAN K, et al. A novel porous bioceramics scaffold by accumulating hydroxyapatite spherulites for large bone tissue engineering in vivo. II. Construct large volume of bone grafts. J Biomed Mater Res. 2014; A102:2491-2501. [43] WATAI JS,CALVAO PS, RIGOLIN TR, et al. Retardation effect of nanohydroxyapatite on the hydrolytic degradation of poly (lactic acid). Polym Eng Sci. 2020:1-11. doi: org/10.1002/pen.25459 |
| [1] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1051-1056. |
| [2] | Tan Guozhong, Tu Xinran, Guo Liyang, Zhong Jialin, Zhang Yang, Jiang Qianzhou. Biosafety evaluation of three-dimensional printed gelatin/sodium alginate/58S bioactive glass scaffolds for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 521-527. |
| [3] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [4] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [5] | Chen Jinmin, Chen Suisheng, Ding Jing, Xia Baoquan, Luo Xiaojia, Lu Chenghai. Stability of balloon dilation with injectable calcium sulfate cement for tibial plateau fractures [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 414-418. |
| [6] | Ji Hangyu, Gu Jun, Xie Linghan, Bao Junping, Peng Xin, Wu Xiaotao. Chitosan/poly(lactic-co-glycolic acid)/polylactic acid scaffold with sustained release of nerve growth factor promotes the differentiation of bone marrow mesenchymal stem cells into neurons [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 3974-3979. |
| [7] | Zhang Haonan, Wang Xingran, Li Meimei, Ma Jinjin, Ma Yanxia, Saijilafu. Effect of N-arachidonylethanolamine on axon regeneration of the dorsal root ganglion [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 3999-4003. |
| [8] | Liu Ming, Wang Kai. Theaflavin-3-gallate modified nano-hydroxyapatite/polycaprolactone composite porous scaffold in bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3480-3486. |
| [9] | Meng Lulu, Liu Hao, Liu Han, Zhang Jun, Li Ruixin, Gao Lilan. Mechanical properties of silk fibroin/type I collagen/hydroxyapatite scaffolds based on low-temperature 3D printing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3550-3555. |
| [10] | Meng Zengdong, Zhu Bin, Zhang Yanan, Luo Lilin, Zhang Yuqin. Mechanical properties and biocompatibility of porous ZnO/hydroxyapatite composites with different porosities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3498-3504. |
| [11] | Guo Xiaopeng, Liu Yingsong, Shang Hui. Silk fibroin/nano hydroxyapatite composite combined with icariin can promote the proliferation and differentiation of bone marrow mesenchymal stem cells into nucleus pulposus like cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3528-3534. |
| [12] | Yang Jiaojiao, Hu Ming, Li Yan, Xia Delin. Effect of hydroxyapatite three-dimensional scaffolds with different apertures on the biological properties of MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3338-3344. |
| [13] | Li Yu, Pan Yuancheng, Lin Ran, Chen Shunyou. Evaluation on the preparation and performance of lysine modified polylactic acid nanofiber stent [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2557-2561. |
| [14] | Tang Yanan, Gao Teng, Ren Guiyun. Preparation and biological properties of zinc-doped hydroxyapatite [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2602-2607. |
| [15] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1530-1536. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||