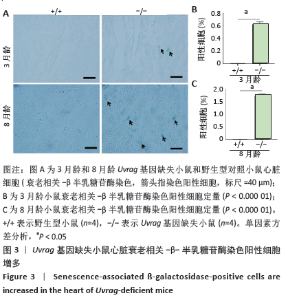
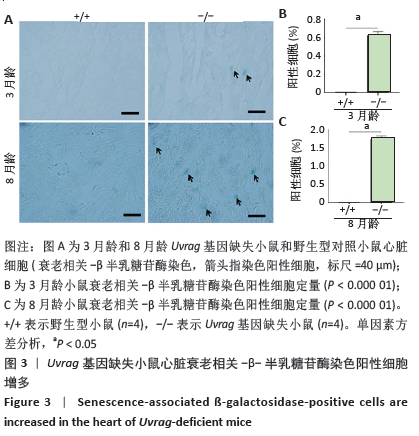
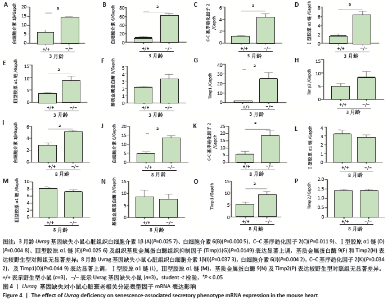
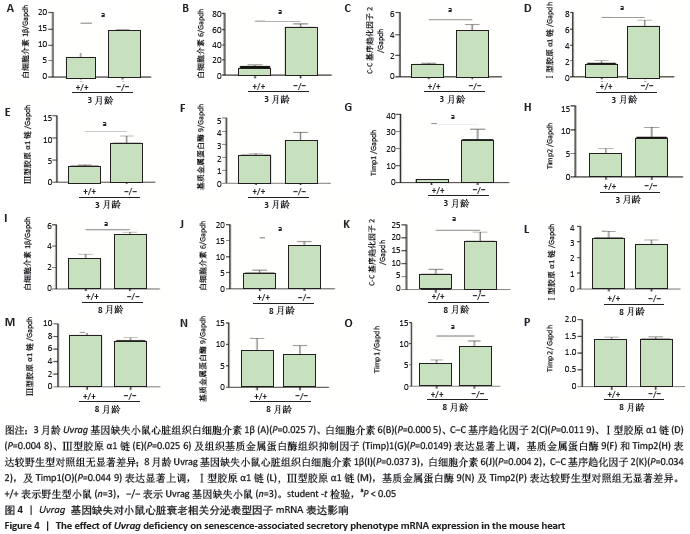
[1] DE ALMEIDA AJPO, RIBEIRO TP, DE MEDEIROS IA. Aging: molecular pathways and implications on the cardiovascular system. Oxid Med Cell Longev. 2017; 2017:7941563.
[2] CALCINOTTO A, KOHLI J, ZAGATO E, et al. Cellular senescence: aging, cancer, and injury.Physiol Rev. 2019;99:1047-1078.
[3] ABATE M, FESTA A, FALCO M, et al. Mitochondria as playmakers of apoptosis, autophagy and senescence. Semin Cell Dev Biol. 2020;98:139-153.
[4] KOROLCHUK VI, MIWA S, CARROLL B, et al. Mitochondria in Cell Senescence: Is mitophagy the weakest link? EBioMedicine.2017;21:7-13.
[5] REGULSKI MJ. Cellular Senescence: what, why, and how. Wounds. 2017;29: 168-174.
[6] MARUSYK A, WHEELER LJ, MATHEWS CK, et al. p53 mediates senescence-like arrest induced by chronic replicational stress.Mol Cell Biol. 2007;27: 5336-5351.
[7] RAYESS H, WANG MB, SRIVATSAN ES. Cellular senescence and tumor suppressor gene p16. Int J Cancer. 2012;130:1715-1725.
[8] KONG Y, CUI H, RAMKUMAR C, et al.Regulation of senescence in cancer and aging. J Aging Res. 2011;2011:963172.
[9] STORER M, MAS A, ROBERT-MORENO A, et al. Senescence is a developmental mechanism that contributes to embryonic growth and patterning. Cell. 2013;155:1119-1130.
[10] DA SILVA-ÁLVAREZ S, PICALLOS-RABINA P, ANTELO-IGLESIAS L, et al. The development of cell senescence. Exp Gerontol. 2019;128:110742.
[11] DA SILVA-ÁLVAREZ S, GUERRA-VARELA J, SOBRIDO-CAMEÁN D, et al. Cell senescence contributes to tissue regeneration in zebrafish. Aging Cell. 2020;19:e13052.
[12] YUN MH. Cellular senescence in tissue repair: every cloud has a silver lining.Int J Dev Biol. 2018;62:591-604.
[13] XU M, PIRTSKHALAVA T, FARR JN, et al. Senolytics improve physical function and increase lifespan in old age.Nat Med. 2018;24:1246-1256.
[14] CAMPISI J, KAPAHI P, LITHGOW GJ, et al. From discoveries in ageing research to therapeutics for healthy ageing. Nature. 2019;571:183-192.
[15] SHIMIZU I, MINAMINO T. Cellular senescence in cardiac diseases.J Cardiol. 2019;74:313-319.
[16] MAVROGONATOU E, PRATSINIS H, KLETSAS D. The role of senescence in cancer development. Semin Cancer Biol. 2020;62:182-191.
[17] KRITSILIS M, V RIZOU S, KOUTSOUDAKI PN, et al. Ageing, Cellular Senescence and Neurodegenerative Disease. Int J Mol Sci. 2018;19. pii: E2937.
[18] SCHAFER MJ, HAAK AJ, TSCHUMPERLIN DJ, et al. Targeting senescent cells in fibrosis: pathology, paradox, and practical considerations. Curr Rheumatol Rep. 2018;20:3.
[19] PALMER AK, XU M, ZHU Y, et al. Targeting senescent cells alleviates obesity-induced metabolic dysfunction. Aging Cell. 2019;18:e12950.
[20] YOUSEFZADEH MJ, ZHU Y, MCGOWAN SJ, et al. Fisetin is a senotherapeutic that extends health and lifespan. EBioMedicine. 2018;36:18-28.
[21] GONZALEZ-VALDES I, HIDALGO I, BUJARRABAL A, et al. Bmi1 limits dilated cardiomyopathy and heart failure by inhibiting cardiac senescence.Nat Commun. 2015;6:6473.
[22] YOUNG AR, NARITA M, FERREIRA M, et al.Autophagy mediates the mitotic senescence transition. Genes Dev. 2009;23:798-803.
[23] KWON Y, KIM JW, JEOUNG JA, et al.Autophagy is pro-senescence when seen in close-up, but anti-senescence in long-shot.Mol Cells. 2017;40:607-612.
[24] GARCÍA-PRAT L, MARTÍNEZ-VICENTE M, PERDIGUERO E, et al. Autophagy maintains stemness by preventing senescence. Nature. 2016;529:37-42.
[25] Tai H, Wang Z, Gong H, et al. Autophagy impairment with lysosomal and mitochondrial dysfunction is an important characteristic of oxidative stress-induced senescence. Autophagy. 2017;13:99-113.
[26] Park JT, Lee YS, Cho KA, et al. Adjustment of the lysosomal-mitochondrial axis for control of cellular senescence.Ageing Res Rev. 2018;47:176-182.
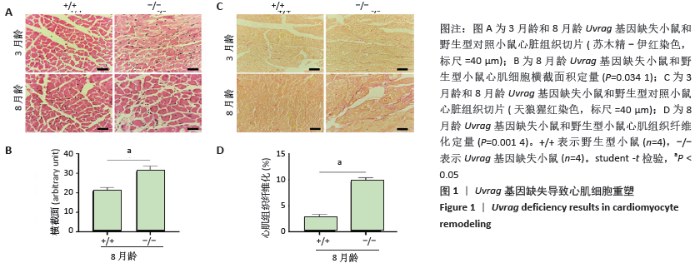
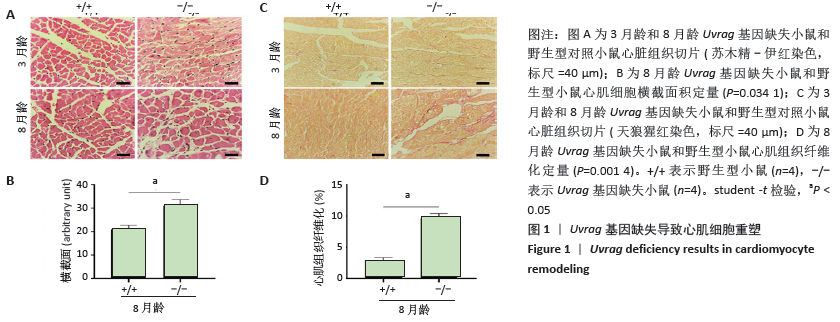
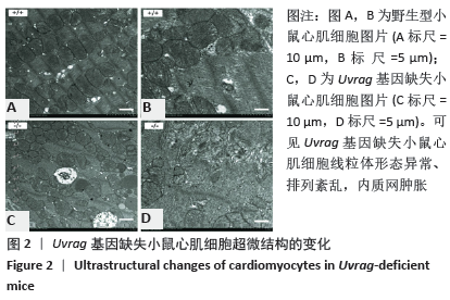
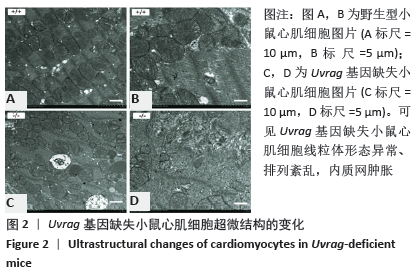
[27] ZHU H, HE L. Uvrag: A multifunctional protein with essential roles in the heart. J Cardiol Curr Res. 2014;1:00003.
[28] SONG Y, QUACH C, LIANG C. Uvrag in autophagy, inflammation, and cancer. Autophagy. 2020;16:387-388.
[29] SONG Z, AN L, YE Y, et al. Essential role for Uvrag in autophagy and maintenance of cardiac function. Cardiovasc Res. 2014;101:48-56.
[30] HUDGINS AD, TAZEARSLAN C, TARE A, et al. Age- and tissue-specific expression of senescence biomarkers in mice. Front Genet. 2018;9:59.
[31] ZI Z, SONG Z, ZHANG S, et al. Rubicon deficiency enhances cardiac autophagy and protects mice from lipopolysaccharide-induced lethality and reduction in stroke volume.J Cardiovasc Pharmacol. 2015;65:252-261.
[32] SOTO-GAMEZ A, QUAX WJ, DEMARIA M. Regulation of survival networks in senescent cells: from mechanisms to interventions.J Mol Biol. 2019;431: 2629-2643.
[33] VASILEIOU PVS, EVANGELOU K, VLASIS K, et al. Mitochondrial homeostasis and cellular senescence. Cells. 2019;8. pii: E686.
[34] NAKAMURA S, OBA M, SUZUKI M, et al. Suppression of autophagic activity by Rubicon is a signature of aging. Nat Commun.2019;10:847.
[35] HOSHINO A, MITA Y, OKAWA Y, et al. Cytosolic p53 inhibits Parkin-mediated mitophagy and promotes mitochondrial dysfunction in the mouse heart.Nat Commun. 2013;4:2308. |