Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (13): 2018-2024.doi: 10.3969/j.issn.2095-4344.2992
Previous Articles Next Articles
A downtrend of mesenchymal cells derived from the second heart field and cardiac neural crest in the outflow tract of Cx43 knockout embryonic mouse heart
Li Hang, Jing Ya, Li Yunhua, Li Hairong, Yang Yanping
- Department of Histology and Embryology, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China
-
Received:2019-12-23Revised:2019-12-28Accepted:2020-02-26Online:2021-05-08Published:2020-12-28 -
Contact:Yang Yanping, MD, Professor, Department of Histology and Embryology, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
About author:Li Hang, Master candidate, Department of Histology and Embryology, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 31200899 (to YYP)
CLC Number:
Cite this article
Li Hang, Jing Ya, Li Yunhua, Li Hairong, Yang Yanping. A downtrend of mesenchymal cells derived from the second heart field and cardiac neural crest in the outflow tract of Cx43 knockout embryonic mouse heart[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2018-2024.
share this article
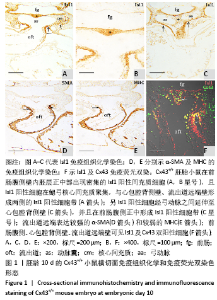
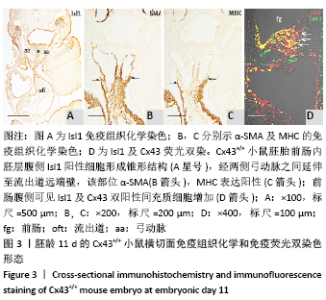
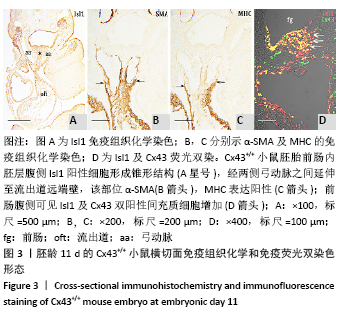
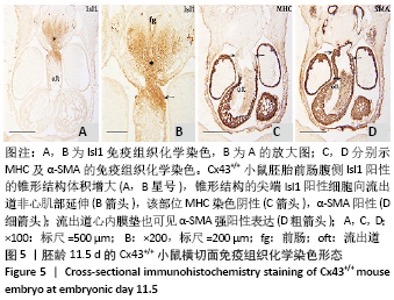
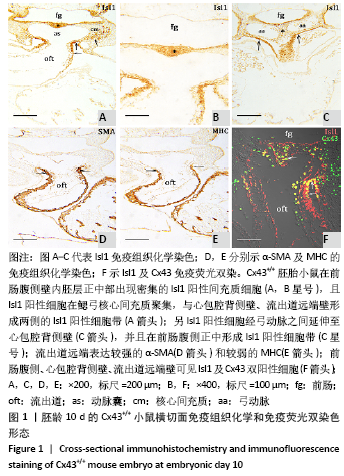
2.1 实验动物数量分析 实验选取胚龄10-13 d的 Cx43+/+和Cx43-/-小鼠胚胎共40只,Cx43+/+小鼠作为对照组共25只,舍去5只未达标准胚胎,其他胚龄10,11,11.5,13 d小鼠各5只全部纳入实验结果进行分析;Cx43-/-小鼠作为实验组共15只,胚龄10,11,13 d各5只。在整个实验过程中小鼠无脱失,全部纳入实验结果进行分析。 2.2 Cx43基因敲除小鼠Isl1阳性细胞的迁移和分布变化 小鼠胚龄10 d,心管向右弯曲成襻,可分辨心室、心房和流出道。流出道壁由心肌、内皮及二者之间的心胶质构成。流出道远端与动脉囊相连,动脉囊背侧与前肠腹侧内胚层相邻,两侧与弓动脉相通。Cx43+/+小鼠胚胎前肠腹侧壁内胚层的正中部与两侧相比,表达较强的Isl1,且内胚层正中部的腹侧出现密集的间充质细胞,同样为Isl1强阳性,见图1A,B,连续切片观察这些Isl1阳性间充质细胞经弓动脉之间延伸至心包腔背侧壁,在前肠腹侧正中形成Isl1阳性细胞带,见图1C。鳃弓核心间充质Isl1阳性细胞聚集,并与心包腔背侧壁、流出道远端壁形成两侧的Isl1阳性细胞带,见图1A。相邻切片染色显示流出道远端壁的Isl1阳性区表达较强的α-SMA和较弱的MHC,见图1D,E,提示Isl1阳性细胞正在向心肌细胞分化。前肠腹侧、心包腔背侧壁及流出道远端壁部分Isl1阳性细胞同时表达Cx43,见图1F。"
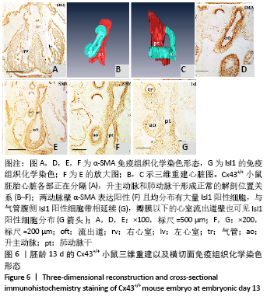
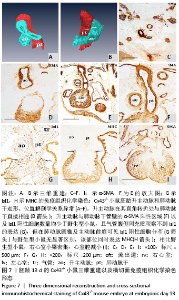
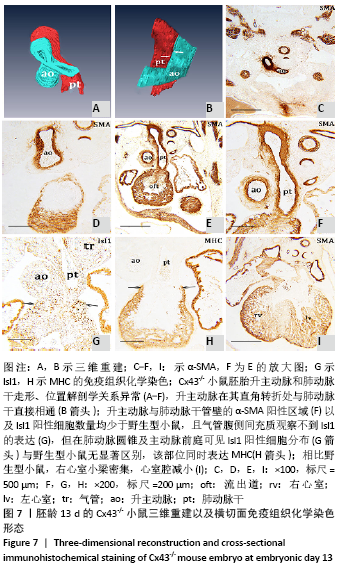
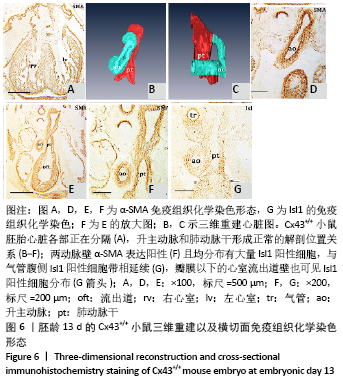
胚龄13 d,心脏各部正在分隔,见图6A。流出道缩短,在Cx43+/+和Cx43-/-小鼠胚胎均可观察到流出道的非心肌部已分隔为升主动脉和肺动脉干。瓣膜以下的流出道心内膜垫正在融合形成流出道隔,将流出道分隔为左右两部分,即主动脉前庭和肺动脉圆锥。野生型小鼠胚胎升主动脉由肺动脉干右侧经腹侧斜上走行到达其左侧,形成成体心脏升主动脉、肺动脉干正常的解剖位置关系,见图6B-F。两动脉壁均分布有大量Isl1阳性细胞,与气管腹侧Isl1阳性细胞带相延续,见图6G,相邻切片显示α-SMA的强阳性表达,见图6F。瓣膜以下的心室流出道壁也可见Isl1阳性细胞分布,见图6G。而在Cx43-/-小鼠胚胎,连续切片与三维重建显示升主动脉由肺动脉干右侧首先直行离开心包腔,再横向左行形成主动脉弓,见图7A-F,而升主动脉在其直角转折处与肺动脉干直接相通,见图7B,升主动脉与肺动脉干管壁内Isl1阳性细胞数量明显少于野生型小鼠,气管腹侧间充质观察不到Isl1的表达,见图6G,7G,两动脉管壁近内皮侧为α-SMA强阳性,但α-SMA阳性区域少于野生型小鼠,见图6F,7F。在肺动脉圆锥、主动脉前庭Isl1阳性细胞分布与野生型小鼠无显著区别,见图6G,7G,该部位同时表达MHC,见图7H。与野生型小鼠胚胎相比,Cx43基因敲除小鼠胚胎右心室小梁密集,心室腔减小,见图6A,7I。 "

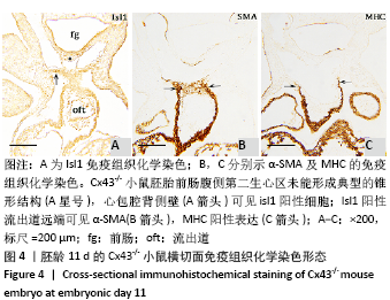
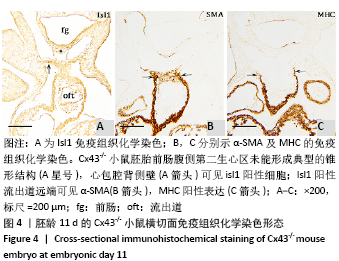
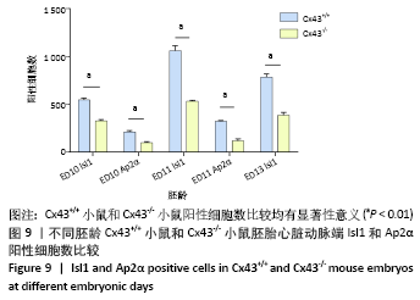
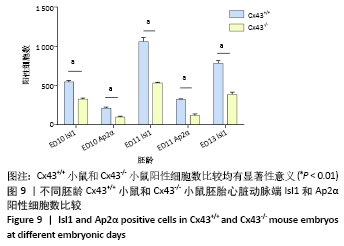
2.3 Cx43基因敲除小鼠神经嵴阳性细胞的迁移和分布 小鼠胚龄10 d,野生型小鼠胚胎Ap2α阳性神经嵴细胞呈鞘状包裹弓动脉壁,一侧延伸至动脉囊腹侧及其与之相连的流出道远端的心胶质内,提示流出道心内膜垫开始形成。另一侧延伸至动脉囊背侧壁开始形成间充质隔,具体可见图8A,B。Cx43-/-小鼠动脉囊腹侧与背侧壁Ap2α阳性细胞明显少于野生型小鼠胚胎,流出道远端心胶质内未观察到Ap2α阳性细胞,见图8C。 胚龄11 d,Ap2α阳性神经嵴细胞继续沿弓动脉壁向流出道的心内膜垫迁移,弓动脉壁周围可见Cx43与Ap2α双阳性细胞,见图8D。Cx43-/-小鼠胚胎神经管两侧Ap2α阳性的间充质细胞迁移至弓动脉壁周围,呈散在分布。在动脉囊背侧、腹侧以及与之相连的流出道心内膜垫远端仅可观察到少量Ap2α阳性细胞,见图8E。至胚龄11.5 d,大量Ap2α阳性分布于弓动脉周围、前肠腹侧Isl1阳性的锥形结构的两侧,见图5B,8F。此期动脉囊成为流出道的非心肌部,该部分并无心内膜垫,因此弓动脉壁周围的Ap2α不再向此处迁移,见图8F。流出道心内膜垫内间充质细胞数量达高峰,这些细胞不表达Ap2α,而是α-SMA强阳性,见图5D,8F。 "

| [1] KOTINI M, BARRIGA EH, LESLIE J, et al. Gap junction protein connexin-43 is a direct transcriptional regulator of N-cadherin in vivo. Nat Commun. 2018;9(1):3846. [2] WAXSE BJ, SENGUPTA P, HESKETH GG, et al. Myosin VI facilitates connexin 43 gap junction accretion. J Cell Sci. 2017;130(5):827-840. [3] GHAMESI TAHRIR F, GUPTA M, MYERS V, et al. Role of bcl2-associated athanogene 3 in turnover of gap junction protein,connexin 43, in neonatal cardiomyocytes. Sci Rep. 2019;9(1):7658. [4] PERVOLARAKI E, DACHTLER J, ANDERSON RA, et al. Ventricular myocardium development and the role of connexins in the human fetal heart. Sci Rep. 2017;7(1):12272. [5] KABA RA, COPPEN SR, DUPONT E, et al. Comparison of connexin 43, 40 and 45 expression patterns in the developing human and mouse hearts. Cell Commun Adhes. 2001;8(4-6):339-343. [6] REAUME AG, SOUSA PAD, KULKARNI S, et al. Cardiac malformation in neonatal mice lacking connexin 43. Science. 1995;5205(267): 1831-1834. [7] MATSUYAMA D, KAWAHARA K. Proliferation of neonatal cardiomyocytes by connexin 43 knockdown via synergistic inactivation of p38 MAPK and increased expression of FGF1. Basic Res Cardiol. 2009;104(6): 631-642. [8] KRETZ M, ECKARDT D, KRUGER O, et al. Normal embryonic development and cardiac morphogenesis in mice with Wnt1-Cre-mediated deletion of connexin43. Genesis. 2006;44(6):269-276. [9] GREENBERG JM, LUMBRERAS V, PELAEZ D, et al. Neural crest stem cells can differentiate to a cardiomyogenic lineage with an ability to contract in response to pulsed infrared stimulation. Tissue Eng PartC Methods. 2016;22(10):982-990. [10] AGHAJANIAN H, CHO YK, RIZER NW, et al. Pdgfralpha functions in endothelial-derived cells to regulate neural crest cells and the development of the great arteries. Dis Models Mech. 2017;10(9): 1101-1108. [11] HUANG GY, WESSELS A, SMITH BR, et al. Alteration in connexin 43 gap junction gene dosage impairs conotruncal heart development. Dev Biol. 1998;32-44. [12] ANDERSEN P, TAMPAKAKIS E, JIMENEZ DV, et al. Precardiac organoids form two heart fields via Bmp/Wnt signaling. Nat Commun. 2018;9(1): 3140. [13] CORTES C, FRANCOU A, DE BONO C, et al. Epithelial properties of the second heart field. Circ Res. 2018;122(1):142-154. [14] ANDERSON RH, CHAUDHRY B, MOHUN TJ, et al. Normal and abnormal development of the intrapericardial arterial trunks in humans and mice. Cardiovasc Res. 2012;95(1):108-115. [15] KEYTE A, HUTSON MR. The neural crest in cardiac congenital anomalies. Differentiation. 2012;84(1):25-40. [16] GIBBS BC, DAMERLA RR, VLADAR EK, et al. Prickle1 mutation causes planar cell polarity and directional cell migration defects associated with cardiac outflow tract anomalies and other structural birth defects. Biol Open. 2016;5(3):323-335. [17] OKAMOTO N, AKIMOTO N, HIDAKA N, et al. Formal genesis of the outflow tracts of the heart revisited: previous works in the light of recent observations. Congenit Anom. 2010;50(3):141-158. [18] AGHAJANIAN H, CHO YK, RIZER NW, et al. Pdgfralpha functions in endothelial-derived cells to regulate neural crest cells and the development of the great arteries. Dis Models Mech. 2017;10(9): 1101-1108. [19] ZHANG J, JULIA YF, CHANGYQ, et al. The FGF-BMP signaling axis regulates outflow tract valve primordium formation by promoting cushion neural crest cell differentiation. Circ Res. 2010;107(10): 1209-1219. [20] 师亮,李慧超,景雅,等.鼠胚呼吸内胚层相关第二生心区发育中的上皮-间充质转化[J].解剖学报,2018,49(4):480-485. [21] RAMIREZ A, ASTROF S. Vualization and analysis of pharyngeal arch arteries using whole-mount immunohistochemistry and 3D reconstruction. J Vis Exp. 2020. Doi:10.3791/60797. [22] SONG YC, DOHN TE, RYDEEN AB, et al. HDAC1-mediated repression of the retinoic acid-responsive gene ripply3 promotes second heart field development. PLoS Genet. 2019;15(5):e1008165. [23] WITZEL HR, JUNGBLUT B, CHOE CP, et al. The LIM protein ajuba restricts the second heart field progenitor pool by regulating Isl1 activity. Dev Cell. 2012;23(1):58-70. [24] PIVEN OO, WINATA CL. The canonical way to make a heart: β-catenin and plakoglobin in heart development and remodeling. Exp Biol and Med. 2017;242(18):1735-1745. [25] ANTHONY J, HORTON AJ, BROOKER J, et al. Nkx2.5 second heart field target gene Ccdc117 regulates DNA metabolism and proliferation. Sci Rep. 2019;9(1):1738. [26] Colombo S, de Sena-Tomas C, George V, et al. Nkx2.5 genes establish second heart field cardiomyocyte progenitors at the arterial pole and pattern the venous pole through Isl1 repression. Development. 2019. Doi: 10.1242/dev.161497. [27] ZHOU L, LIU JL, XIANG ML, et al. Gata4 potentiates second heart field proliferation and Hedgehog signaling for cardiac septation. Proc Natl Acad Sci U S A. 2017;114(8):E1422-E1431. [28] SAWADA H, RATERI DL, MOORLEGHEN JJ, et al. Smooth muscle cells derived from second heart field and cardiac neural crest reside in spatially distinct domains in the media of the ascending aorta-brief report. Arter Thromb Vasc Biol. 2017;37:1722-1726. [29] MOLIN DG, POELMANN RE, DERUITER MC, et al. Transforming growth factor beta-SMAD2 signaling regulates aortic arch innervation and development. Circ Res. 2004;95(11):1109-1117. [30] KAREN LW, CECILIA WL, MARGARET L, et al. Connexin 43 expression reflflects neural crest patterns during cardiovascular development. Dev Biol. 1999;208:307-323. [31] XU X, FRANCIS R, WEI CJ, et al. Connexin 43-mediated modulation of polarized cell movement and the directional migration of cardiac neural crest cells. Development. 2006;133(18):321-324. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Min Youjiang, Yao Haihua, Sun Jie, Zhou Xuan, Yu Hang, Sun Qianpu, Hong Ensi. Effect of “three-tong acupuncture” on brain function of patients with spinal cord injury based on magnetic resonance technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-8. |
| [3] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [4] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [5] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [6] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [7] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [8] | Li Jing, Xie Jianshan, Cui Huilin, Cao Ximei, Yang Yanping, Li Hairong. Expression and localization of diacylglycerol kinase zeta and protein kinase C beta II in mouse back skin with different coat colors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1196-1200. |
| [9] | Li Jiacheng, Liang Xuezhen, Liu Jinbao, Xu Bo, Li Gang. Differential mRNA expression profile and competitive endogenous RNA regulatory network in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1212-1217. |
| [10] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [11] | Tan Jingyu, Liu Haiwen. Genome-wide identification, classification and phylogenetic analysis of Fasciclin gene family for osteoblast specific factor 2 [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1243-1248. |
| [12] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [13] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [14] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [15] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||