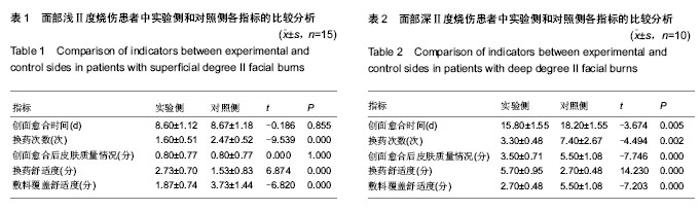
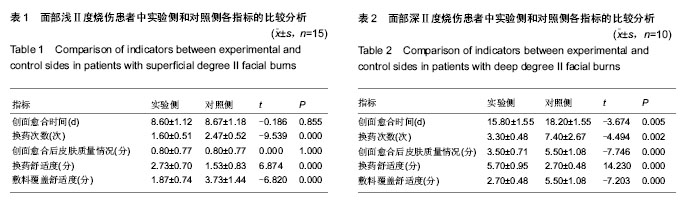
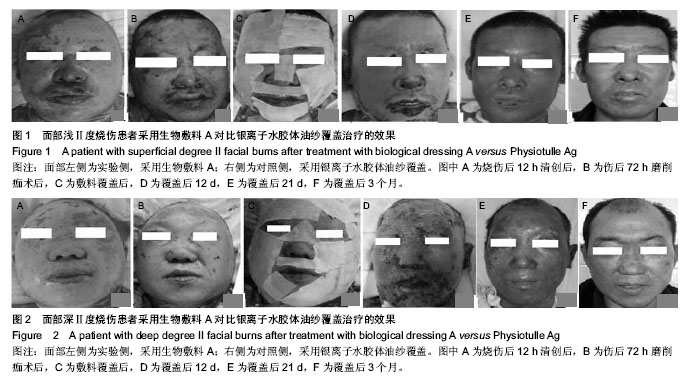
| [1] Wu J, Chen J. Pay close attention to the quality of life of patients and look to the future of burn rehabilitation. Zhonghua Shaoshang Zazhi.2013;29(2):119-121.
[2] Mabrouk A,Boughdadi NS,Helal HA,et al.Moist occlusive dressing (Aquacel® Ag) versus moist open dressing (MEBO®) in the management of partial-thickness facial burns: A comparative study in Ain Shams University.Burns. 2012;38(3):396-403.
[3] Hoogewerf CJ,Van BAr ME,Hop MJ,et al.Topical treatment for facial burns. Cochrane Database Syst Rev. 2013;1:CD008058.
[4] Hoeksema H,Vandekerckhove D,Verbelen J,et al.A comparative study of 1% silver sulphadiazine (Flammazine®) versus an enzyme alginogel (Flaminal®) in the treatment of partial thickness burns.Burns.2013;39(6):1234-1241.
[5] Dougherty WR,Spence RJ.Reconstruction of the burned face/cheek: acute and delayed. Achauer and Sood's burn surgery: reconstruction and rehabilitation,2006:234-253.
[6] Vloemans A,Soesman AM,Suijker M,et al.A randomised clinical trial comparing a hydrocolloid-derived dressing and glycerol preserved allograft skin in the management of partial thickness burns.Burns.2003;29(7):702-710.
[7] Djaprie SM,Wardhana A.Dressing for Partial Thickness Burn Using Microbial Cellulose and Transparent Film Dressing: A Comparative Study. Jurnal Plastik Rekonstruksi.2013.
[8] Mirastschijski U,Sander JT,Weyand B,et al.Rehabilitation of burn patients: An underestimated socio-economic burden. Burns.2013;39(2):262-268.
[9] Bull JP,Squire JR,Topley E.Experiments with occlusive dressings of a new plastic.Lancet.1948;252(6519):213-215.
[10] Schilling RS,Roberts M,Goodman N.Clinical trial of occlusive plastic dressings.Lancet.1950;1(6599):293-296.
[11] Winter GD.Formation of the scab and the rate of epithelization of superficial wounds in the skin of the young domestic pig. Nature.1962;193:293-294.
[12] Wasiak J,Cleland H,Campbell F,et al.Dressings for superficial and partial thickness burns.Cochrane Database Syst Rev. 2013; 3:CD002106.
[13] Caruso DM,Foster KN,Blome-Eberwein SA,et al.Randomized clinical study of Hydrofiber dressing with silver or silver sulfadiazine in the management of partial-thickness burns.J Burn Care Res.2006;27(3):298-309.
[14] Skórkowska-Telichowska K,Czemplik M,Kulma A,et al.The local treatment and available dressings designed for chronic wounds.J Am Acad Dermatol.2013;68(4):e117-e126.
[15] Lavery LA,La Fontaine J,Thakral G,et al.Randomized Clinical Trial to Compare Negative-Pressure Wound Therapy Approaches with Low and High Pressure, Silicone-Coated Dressing, and Polyurethane Foam Dressing. Plast Reconstr Surg. 2014;133(3):722-726.
[16] Meaume S,Dissemond J,Addala A,et al.Evaluation of two fibrous wound dressings for the management of leg ulcers: Results of a European randomised controlled trial (EARTH RCT).J Wound Care.2014;23(3):105-116.
[17] Nyame TT,Chiang HA,Orgill DP.Clinical Applications of Skin Substitutes. Surg Clin North Am.2014;94(4):839-850.
[18] Chiu T,Burd A.“Xenograft” dressing in the treatment of burns.Clin Dermatol.2005;23(4):419-423.
[19] Hartstein B,Gausche-Hill M,Cancio LC.Burn Injuries in Children and the Use of Biological Dressings.Pediatr Emerg Care.2013;29(8):939-948.
[20] Hosseini SN,Mousavinasab SN,Fallahnezhat M.Xenoderm dressing in the treatment of second degree burns. Burns. 2007; 33(6):776-781.
[21] Eriksson A,Burcharth J,Rosenberg J.Animal derived products may conflict with religious patients' beliefs. BMC Med Ethics. 2013;14(1):48.
[22] Verbelen J, Hoeksema H, Heyneman A, et al. Aquacel (®) Ag dressing versus Acticoat™ dressing in partial thickness burns: A prospective, randomized, controlled study in 100 patients. Part 1: Burn wound healing.Burns: journal of the International Society for Burn Injuries, 2013.
[23] Sullivan T,Smith J,Kermode J,et al.Rating the burn scar.J Burn CareRes.1990;11(3):256-260.
[24] Revill SI,Robinson JO,Rosen M,et al.The reliability of a linear analogue for evaluating pain. Anaesthesia. 1976;31(9): 1191-1198.
[25] Gordon M. Burn care protocols: infection control. J Burn Care Rehabil.1997;8:67-71.
[26] Tompkins D. Burn nursing. In: Herndon D, editor. Total burn care. Philadelphia, PA: WB Saunders,1996:46.
[27] Hinman CD,Maibach H.Effect of air exposure and occlusion on experimental human skin wounds.Nature.1963;200:377-378.
[28] Gibran NS,Committee on Organization and Delivery of Burn Care, American Burn Association. Practice guidelines for burn care, 2006. J Burn Care Res.2006;27(4):437-438.
[29] Wright JB,Lam K,Burrell RE.Wound management in an era ofincreasing bacterial antibiotic resistance: a role for topical silver treatment. Am J Infect Control.1998;26(6):572-577.
[30] 陈炯,韩春茂,林小玮,等.纳米银敷料在修复Ⅱ度烧伤创面的应用研究[J].中华外科杂志,2006,44(1):50-52.
[31] 王强,贾成钢.美宝湿润烧伤膏与磺胺嘧啶银霜治疗热镁渣烧伤的比较[J].中华烧伤疮疡杂志,2009,21(3):182-184.
[32] Sasse KC,Ackerman EM,Brandt JR.MatriStem xenograft material: Case series and cost analysis.Surgery.2013;1(1):3.
[33] Junkins-Hopkins JM.Biologic dressings.J Am Acad Dermatol. 2011;64(1):e5-e7.
[34] Hermans MH.Porcine xenografts vs.(cryopreserved) allografts in the management of partial thickness burns: Is there a clinical difference?Burns.2014;40(3): 408-415.
[35] Barone AA,Mastroianni M,Farkash EA,et al.Genetically modified porcine split-thickness skin grafts as an alternative to allograft for provision of temporary wound coverage: preliminary characterization.Burns.2014.pⅡ: S0305-4179(14)00290-3. doi:10.1016/j.burns.2014.09.003. [Epub ahead of print]
[36] Gierek M,Kawecki M,Miku? K,et al.Biological Dressings as a Substitutes of the Skin in the Treatment of Burn Wounds.Pol Przegl Chir.2013;85(6):354-359.
[37] Raimer DW,Petitt MS,Nosrati N,et al.Porcine xenograft biosynthetic wound dressings for the management of postoperative Mohs wounds. Dermatol Online J. 2011;17(9):1. |