Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (41): 6708-6713.doi: 10.3969/j.issn.2095-4344.2014.41.026
Previous Articles Next Articles
Mitochondrial transfer mechanism of bone marrow mesenchymal stem cells for rescue of tissue injury
Peng Yi, Shu Chang, Fu Zhou
- Department of Respiratory Medicine, Children’s Hospital of Chongqing Medical University, Chongqing 400014, China
-
Revised:2014-09-07Online:2014-10-01Published:2014-10-01 -
Contact:Fu Zhou, M.D., Chief physician, Professor, Department of Respiratory Medicine, Children’s Hospital of Chongqing Medical University, Chongqing 400014, China -
About author:Peng Yi, Studying for master’s degree, Department of Respiratory Medicine, Children’s Hospital of Chongqing Medical University, Chongqing 400014, China -
Supported by:the National Natural Science Foundation of China, No. 30700915
CLC Number:
Cite this article
Peng Yi, Shu Chang, Fu Zhou. Mitochondrial transfer mechanism of bone marrow mesenchymal stem cells for rescue of tissue injury [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(41): 6708-6713.
share this article
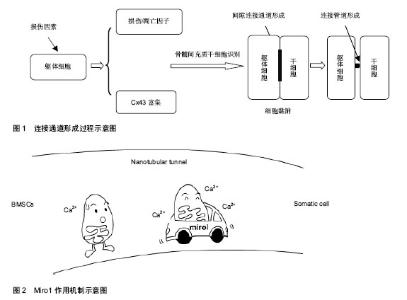
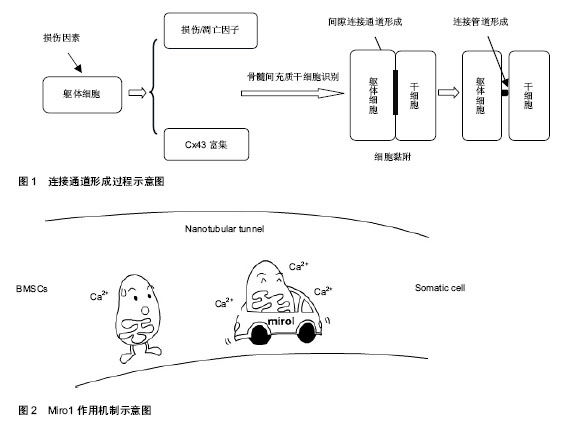
2.1 线粒体转移的可能机制 线粒体转移机制有以下几种假说:①受体细胞被动吞噬间充质干细胞出芽产生的含线粒体的微泡。②细胞完整融合后选择性丢失供体细胞核。③通过转移线粒体DNA而非完整功能性线粒体。④在受体细胞及供体细胞间形成连接管道(tunnelling nanotubes,TNT)主动转运完整线粒体。 研究者向含有损伤细胞的培养基中添加人血小板(含功能性线粒体但不含细胞核)或者分离出的骨髓间充质干细胞源性线粒体[2,5],两个实验都没有观察到线粒体进入损伤细胞中。此外,有研究发现,细胞外游离线粒体可以激活模式识别受体[14],加重组织损伤,均提示被动吞噬微泡转移线粒体可能性不大。成人和胚胎干细胞被报道可以同完全分化的躯体细胞,如心肌细胞、干细胞、神经元细胞相融合[15-18],但有意义的完整细胞融合继而选择性丢失供体细胞核的发生率很低,不足以解释骨髓间充质干细胞灌注之后所达到的治疗效果。永久细胞融合可能使细胞含有特殊核型(双核异核体或者单核超倍体)[19]。研究者对受体细胞进行基因分析没有发现任何供体细胞核基因标记[2,5,7],基本除外线粒体通过细胞融合方式转移。此外,线粒体DNA介导的损伤修复也不太可能[7]。多篇文章报道连接管道存在于不同细胞的培养模型中[20-24],通过染色标记与显微技术,研究者在连接管道结构中直接观察到线粒体转移过程[6,25],因此,目前多数学者认为,细胞间通过形成连接管道进行线粒体转移。 连接管道的形成可能通过以下两种方式[5,26-27]:①细胞间形成原始紧密连接之后再分离。②细胞间富含肌动蛋白微粒的扩展。Liu等[5]在实验中发现,微粒先于连接管道形成且参与连接管道的早期形成,因此他们提出,损伤细胞与间充质干细胞产生和互相交换微粒,是连接管道结构形成的重要机制。 以上研究表明,细胞间通过形成连接管道进行线粒体转移。供体细胞与受体细胞间产生和互相交换肌动蛋白微粒,是连接管道形成的重要机制(图1)。 2.2 线粒体转移的影响因素与调控机制 2.2.1 线粒体转移具有一定方向性 Plotnikov等[6]研究了大鼠心肌细胞和人骨髓间充质干细胞间的相互交流,发现胞质可以通过连接管道或者紧密连接无方向性地在细胞间转移,而线粒体的转移则只能从骨髓间充质干细胞到心肌细胞而不可逆,提出线粒体转移可能具有一定方向性。他们在连接管道结构内的微泡中观察到线粒体经典的亚显微结构,在一些直径更小的微泡中发现了细胞骨架样结构,提出细胞骨架微丝可能是引导微泡单向转运的重要元素。 Ahmad等[4]在骨髓间充质干细胞与气道上皮细胞的培养中,也只观察到了从骨髓间充质干细胞到气道上皮细胞的线粒体转移,而没有从气道上皮细胞到骨髓间充质干细胞的转移,进一步说明了线粒体转移的方向性。此外,当上皮细胞被特殊线粒体抑制剂或者炎症损伤后,骨髓间充质干细胞源性线粒体的转移会增加,而当骨髓间充质干细胞损伤时,这种转移会减少,骨髓间充质干细胞介导的受损上皮细胞的修复功能也消失。另一个研究也提出,在躯体细胞损伤的情况下,骨髓间充质干细胞的线粒体通过连接管道频繁和几乎单向的转移到受体细胞中,提示受体细胞损伤可能促进从供体细胞到受体细胞的线粒体转移[5]。近期研究表明,受损细胞表面表达的一些损伤或凋亡因子(如磷脂酰丝氨酸),可以选择性聚集和引导连接管道形成[28]。这个过程与坏死细胞的吞噬作用呈现出相似性[5],被认为是细胞凋亡早期的一种保护性机制。当这些因子被膜联蛋白V(Annexin V)遮盖时,连接管道的形成被抑制,从反面证实了细胞损伤可以促进线粒体转移,其具体参与信号有待进一步研究。 在几个报道中,线粒体的转移仅仅在靶细胞含有无功能性线粒体或者处于细菌内毒素作用下时才发 生[2-3,7,29-30],而不能在携带致病性线粒体DNA突变(如A3243G突变或者4 977 bp缺失)的细胞中被观察到[7],提示线粒体转移可能仅发生在线粒体功能近乎缺失的情况下。 2.2.2 含有连接蛋白43(Cx43)的间隙连接通道的形成是线粒体转移的必备条件,这个过程依赖Ca2+的浓集 Islam等[3]对已知的连接蛋白进行分析发现,小鼠骨髓间充质干细胞只表达Cx43。同时在小鼠肺损伤模型中,经气道灌注骨髓间充质干细胞倾向于聚集在Cx43高表达的肺泡上皮细胞区域,通过形成以Cx43为基础的间隙连接通道黏附于损伤细胞,随后在细胞间以Ca2+依赖的方式,形成表达Cx43的连接管道并释放含有线粒体的微泡,进行线粒体转移。对照试验中,灌注Cx43缺乏或者含有突变的Cx43的骨髓间充质干细胞不能将线粒体转移到受损肺泡细胞中,也不增加这些细胞的存活。此外,在没有Ca2+浓集的情况下,细胞之间可以黏附但是不能形成连接管道,也没有观察到骨髓间充质干细胞释放微泡。当使用特异性连接管道阻断剂或者Cx43抑制剂阻断连接管道形成时[3,31],细胞间染色剂交换消失,从反面证实了含有Cx43的连接管道形成对线粒体成功转移是必不可少的。随后Otsu等[32]在骨髓间充质干细胞与大鼠肺微血管内皮细胞的共同培养中也观察到了细胞间以Cx43为基础的连接管道的形成及随后发生的线粒体转移现象。另外,Islam等[3]还发现,细胞渗透性发动蛋白抑制剂Dynasore可以阻断线粒体转移,提示线粒体转移是一个主动过程。 2.2.3 Miro1对线粒体在连接管道内的移动具有引导与加速作用(图2) 在最近一些针对神经元细胞的广泛研究中,有学者发现Miro1(一种使线粒体附着于KIF5肌动蛋白的钙敏感性衔接蛋白),可以在一系列辅助蛋白如Miro2,TRAK1,TRAK2和Myo19的帮助下,协助线粒体沿着细胞内的微管移动[33-36]。 Ahmad等[4]观察了鱼藤酮处理前后骨髓间充质干细胞中一些已经报道过的胞间线粒体转运蛋白(如:Miro1,Miro2,TRAK1,KHC,Myo19)的表达图谱,发现鱼藤酮损伤的治疗只与Miro1相关。实验还发现骨髓间充质干细胞中Miro1的过度表达(MSCmiroHi)可以增加线粒体转移和对受损细胞的修复功能,而Miro1含量极低的骨髓间充质干细胞(MSCmiroLo)的治疗效能明显降低。在小鼠的气道损伤和过敏性炎症模型中,与没有经过生物工程改造的骨髓间充质干细胞相比,MSCmiroHi具有更好的线粒体供给能力及治疗效能。研究还发现,除了一些非特异性旁分泌效应外,Miro1修复受损上皮细胞线粒体功能障碍时并不影响骨髓间充质干细胞来源抗炎产物(如:一氧化氮、转化生长因子β,白细胞介素10和前列腺素E2)的分泌和功能发挥,排除了分泌功能增加引起骨髓间充质干细胞疗效提升的可能性。进一步的研究表明,Miro1的缺失并不阻止连接管道形成,但是在很大程度上减缓了线粒体通过这些管道的移动。Chang等[35]在针对神经元轴突的研究中发现,线粒体Ca2+含量与线粒体移动速度相关,Miro1主要通过调节线粒体对Ca2+的摄取影响线粒体移动速度。 几乎所有的组织均含有干/祖细胞,研究发现,其他间质源性细胞也能将线粒体转移到损伤上皮细胞[4],但这种转移仅相当于骨髓间充质干细胞源性线粒体转移的30%。这些细胞作为供体细胞产生的效能很低,达到的修复功能也很轻微。随后实验对骨髓间充质干细胞、肺上皮细胞、成纤维细胞中Miro1的表达进行了研究,发现相对于受体细胞(肺上皮细胞),供体细胞(骨髓间充质干细胞、成纤维细胞)具有更高的Miro1水平,支持细胞间线粒体转移是一个由Miro1驱动的过程。Miro1的发现使其他间质源性细胞成为线粒体转移合格供体细胞的设想成为可能[4],怎样通过分子工程学改造这些细胞仍有待研究。 2.2.4 线粒体的成功转移可能伴随受体细胞内损伤线粒体的清除 线粒体是活性氧产生的源泉,也是其作用目标[37]。如果细胞中损伤线粒体没有清除,则可能释放大量的活性氧类物质,诱导更多线粒体源性活性氧类物质的释放[5],造成更大损伤。然而,在上述模型中,这种情况并未发生,因此,连接管道的形成可能与损伤线粒体的清除相关[5]。线粒体自噬作用是目前已知的细胞内线粒体降解的惟一机制,为溶酶体依赖的细胞降解过程。研究表明,真核细胞中溶酶体的pH可以调节线粒体功能和生长周期[38]。Yasuda等[28]发现,内皮祖细胞可以通过连接管道介导溶酶体交换,阻止过早衰老的内皮细胞凋亡。因此,有学者提出,骨髓间充质干细胞可能通过以下两种方式调节损伤细胞中线粒体的质量:①通过连接管道补充功能性线粒体。②通过连接管道调节溶酶体转移消灭毁损线粒体[5]。 2.3 受体细胞对供体细胞的影响 干细胞与损伤细胞的相互作用激活了干细胞对损伤细胞的保护功能,这个相互作用的过程对干细胞的生长与存在也具有一定调节作用。Plotnikov等[6]观察了大鼠心肌细胞和人骨髓间充质干细胞的共同培养过程,他们在胞间延伸结构中发现了无方向性转移的胞质和从骨髓间充质干细胞到心肌细胞的线粒体转移,随后发现与大鼠心肌细胞共同培养的人骨髓间充质干细胞表达了特异性的人肌球蛋白。且随着时间的推移,表达人肌球蛋白的干细胞逐渐增多,而单独培养的骨髓间充质干细胞没有观察到这个现象。因此他们猜测:从心肌细胞到骨髓间充质干细胞的胞质转运可能决定干细胞的分化方向。 另一个报道提出:骨髓间充质干细胞与其他类型细胞间的相互作用可能激发骨髓间充质干细胞的增殖反应[9]。这种细胞增殖的上调依赖于骨髓间充质干细胞和血管平滑肌细胞的直接接触,且呈时间依赖性。鉴于血管平滑肌细胞的增殖没有改变,这种对细胞生长的影响是单向的。Vallabhaneni等[9]在实验中观察到了通过连接管道进行的线粒体交换,当使用细胞松弛素和拉春库林阻断连接管道后骨髓间充质干细胞增殖的上调被终止,由此得出,从血管平滑肌细胞到骨髓间充质干细胞的线粒体交换对共同培养骨髓间充质干细胞增生的调节可能是必须的,其中的复杂机制仍有待进一步研究。"
| [1] Tatum EL, Lederberg J.Gene Recombination in the Bacterium Escherichia coli.J Bacteriol. 1947;53(6):673-684. [2] Spees JL, Olson SD, Whitney MJ, et al.Mitochondrial transfer between cells can rescue aerobic respiration.Proc Natl Acad Sci U S A. 2006;103(5):1283-1288. [3] Islam MN, Das SR, Emin MT,et al.Mitochondrial transfer from bone-marrow-derived stromal cells to pulmonary alveoli protects against acute lung injury.Nat Med. 2012;18(5): 759-765. [4] Ahmad T, Mukherjee S, Pattnaik B,et al.Miro1 regulates intercellular mitochondrial transport & enhances mesenchymal stem cell rescue efficacy.EMBO J. 2014; 33(9):994-1010. [5] Liu K, Ji K, Guo L,et al.Mesenchymal stem cells rescue injured endothelial cells in an in vitro ischemia-reperfusion model via tunneling nanotube like structure-mediated mitochondrial transfer.Microvasc Res. 2014;92:10-18. [6] Plotnikov EY, Khryapenkova TG, Vasileva AK,et al.Cell-to-cell cross-talk between mesenchymal stem cells and cardiomyocytes in co-culture.J Cell Mol Med. 2008;12(5A): 1622-1631. [7] Cho YM, Kim JH, Kim M,et al.Mesenchymal stem cells transfer mitochondria to the cells with virtually no mitochondrial function but not with pathogenic mtDNA mutations.PLoS One. 2012;7(3):e32778. [8] Pasquier J, Guerrouahen BS, Al Thawadi H,et al.Preferential transfer of mitochondria from endothelial to cancer cells through tunneling nanotubes modulates chemoresistance.J Transl Med. 2013;11:94. [9] Vallabhaneni KC, Haller H, Dumler I.Vascular smooth muscle cells initiate proliferation of mesenchymal stem cells by mitochondrial transfer via tunneling nanotubes.Stem Cells Dev. 2012;21(17):3104-3113. [10] Krause DS, Theise ND, Collector MI,et al.Multi-organ, multi-lineage engraftment by a single bone marrow-derived stem cell.Cell. 2001;105(3):369-377. [11] Ortiz LA, Gambelli F, McBride C,et al.Mesenchymal stem cell engraftment in lung is enhanced in response to bleomycin exposure and ameliorates its fibrotic effects.Proc Natl Acad Sci U S A. 2003;100(14):8407-8411. [12] Liang X, Ding Y, Zhang Y,et al.Paracrine mechanisms of Mesenchymal Stem cell-based therapy: Current status and perspectives.Cell Transplant. 2013 May 14. [Epub ahead of print] [13] Maron-Gutierrez T, Laffey JG, Pelosi P, et al.Cell-based therapies for the acute respiratory distress syndrome.Curr Opin Crit Care. 2014;20(1):122-131. [14] Zhang Q, Raoof M, Chen Y,et al.Circulating mitochondrial DAMPs cause inflammatory responses to injury.Nature. 2010; 464(7285):104-107. [15] Alvarez-Dolado M, Pardal R, Garcia-Verdugo JM,et al.Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes.Nature. 2003;425(6961): 968-973. [16] Ishikawa F, Shimazu H, Shultz LD,et al.Purified human hematopoietic stem cells contribute to the generation of cardiomyocytes through cell fusion.FASEB J. 2006;20(7): 950-952. [17] Terada N, Hamazaki T, Oka M,et al.Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature. 2002;416(6880):542-545. [18] Ying QL, Nichols J, Evans EP,et al.Changing potency by spontaneous fusion.Nature. 2002;416(6880):545-548. [19] Ogle BM, Cascalho M, Platt JL.Biological implications of cell fusion.Nat Rev Mol Cell Biol. 2005;6(7):567-575. [20] Gurke S, Barroso JF, Gerdes HH.The art of cellular communication: tunneling nanotubes bridge the divide.Histochem Cell Biol. 2008;129(5):539-550. [21] Wang X, Veruki ML, Bukoreshtliev NV,et al.Animal cells connected by nanotubes can be electrically coupled through interposed gap-junction channels.Proc Natl Acad Sci U S A. 2010;107(40):17194-17199. [22] Acquistapace A, Bru T, Lesault PF,et al.Human mesenchymal stem cells reprogram adult cardiomyocytes toward a progenitor-like state through partial cell fusion and mitochondria transfer.Stem Cells. 2011;29(5):812-824. [23] Wang X, Gerdes HH.Long-distance electrical coupling via tunneling nanotubes.Biochim Biophys Acta. 2012;1818(8):2082-2086. [24] Lou E, Fujisawa S, Morozov A,et al.Tunneling nanotubes provide a unique conduit for intercellular transfer of cellular contents in human malignant pleural mesothelioma.PLoS One. 2012;7(3):e33093. [25] Ma Z, Yang H, Liu H,et al.Mesenchymal stem cell-cardiomyocyte interactions under defined contact modes on laser-patterned biochips.PLoS One. 2013;8(2):e56554. [26] Chauveau A, Aucher A, Eissmann P,et al.Membrane nanotubes facilitate long-distance interactions between natural killer cells and target cells.Proc Natl Acad Sci U S A. 2010;107(12):5545-5550. [27] Chhabra ES, Higgs HN.The many faces of actin: matching assembly factors with cellular structures.Nat Cell Biol. 2007; 9(10):1110-1121. [28] Yasuda K, Khandare A, Burianovskyy L,et al.Tunneling nanotubes mediate rescue of prematurely senescent endothelial cells by endothelial progenitors: exchange of lysosomal pool.Aging (Albany NY). 2011;3(6):597-608. [29] Prockop DJ, Oh JY.Medical therapies with adult stem/progenitor cells (MSCs): a backward journey from dramatic results in vivo to the cellular and molecular explanations.J Cell Biochem. 2012;113(5):1460-1469. [30] Prockop DJ.Mitochondria to the rescue.Nat Med. 2012;18(5): 653-654. [31] Bukoreshtliev NV, Wang X, Hodneland E,et al.Selective block of tunneling nanotube (TNT) formation inhibits intercellular organelle transfer between PC12 cells.FEBS Lett. 2009; 583(9): 1481-1488. [32] Otsu K, Das S, Houser SD,et al.Concentration-dependent inhibition of angiogenesis by mesenchymal stem cells.Blood. 2009;113(18):4197-4205. [33] Quintero OA, DiVito MM, Adikes RC,et al.Human Myo19 is a novel myosin that associates with mitochondria.Curr Biol. 2009; 19(23):2008-2013. [34] Brickley K, Stephenson FA.Trafficking kinesin protein (TRAK)-mediated transport of mitochondria in axons of hippocampal neurons.J Biol Chem. 2011;286(20):18079- 18092. [35] Chang KT, Niescier RF, Min KT.Mitochondrial matrix Ca2+ as an intrinsic signal regulating mitochondrial motility in axons.Proc Natl Acad Sci U S A. 2011;108(37):15456-15461. [36] Las G, Shirihai OS.Miro1: new wheels for transferring mitochondria. EMBO J. 2014;33(9):939-941. [37] Murphy MP.How mitochondria produce reactive oxygen species.Biochem J. 2009;417(1):1-13. [38] Hughes AL, Gottschling DE.An early age increase in vacuolar pH limits mitochondrial function and lifespan in yeast.Nature. 2012;492(7428):261-265. [39] McAnulty RJ.Fibroblasts and myofibroblasts: their source, function and role in disease.Int J Biochem Cell Biol. 2007; 39(4):666-671. [40] Schieke SM, Ma M, Cao L,et al.Mitochondrial metabolism modulates differentiation and teratoma formation capacity in mouse embryonic stem cells.J Biol Chem. 2008;283(42): 28506-28512. [41] Rebbeck CA, Leroi AM, Burt A.Mitochondrial capture by a transmissible cancer.Science. 2011;331(6015):303. [42] Prockop DJ, Brenner M, Fibbe WE, et al.Defining the risks of mesenchymal stromal cell therapy.Cytotherapy. 2010;12(5): 576-578. |
| [1] | Wang Jian-ji, Yang Long, Li Jing, Sun Qi, Zuo Wei-min, Ren Qi-feng, Sun Yu, Wu Zhan-yu, Zou Qiang, Ma Min-xian, Ye Chuan. Development and application of special-purpose grafter by femoral head decompression combined with bone marrow mesenchymal stem cells transplantation based on three-dimensional printing technology [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(44): 6636-6642. |
| [2] | He Wei, Li Yu-ming, Zhou Xin, Peng Peng, Qin Yong-sheng, Bo Hai. Effects of short-term high-intensity interval training on ventricular remodeling and mitochondrial content in rats after myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5986-5993. |
| [3] | Zhou Chang-yan, Zhou Qing-huan, Bian Jing, Chen Ke, Chen Wen. Bone marrow mesenchymal stem cells combined with calcium phosphate cement to repair articular cartilage defects in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1195-1199. |
| [4] | Xu Xiang, Yin He-ping. Platelet-rich plasma accelerates the proliferation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2144-2148. |
| [5] | Gao Zhuo-yue, Liu Yong-qi, He Jian-xin, Wu Zhi-wei, Luo Ya-li, Su Yun, Zhang Li-ying, Zhang Qi, Wu You-ming, Zhou Ni-na. Regulatory effects of warming yang and invigorating qi treatment on the inflammatory balance and genetic stability of bone marrow mesenchymal stem cells under tumor microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2267-2272. |
| [6] | Han Xiang-zhen, He Hui-yu, Hu Yang, Ba Jiao-jiao, Wang Huan-huan, Mi Xue, Abulizi•Abudula. Recombinant lentiviral vector transfected sheep bone marrow mesenchymal stem cells and osteogenic gene expression changes [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 821-828. |
| [7] | Huang Jian-feng, Huang Ji-feng, Zhang Wei-cai. Bone marrow mesenchymal stem cells differentiate into neuron-like cells induced by combination of two cytokines [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 829-834. |
| [8] | Zou Bin, Zong Shao-hui, Zeng Gao-feng, Fang Ye, Gao Tai-hang. Effects of alpha-zearalanol on the osteogenic differentiation of mouse bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 835-840. |
| [9] | Yang Yi, Ding Wen-jing, Dong Wan-li. Autophagy-related gene Beclin-1 expression in neuron-like differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 841-846. |
| [10] | Su Xue-lian, Bao Guang-jie, Kang Hong, Liu Lin, Kong Nan-nan. Morphological changes of goat bone marrow mesenchymal stem cells differentiating into fibrochondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 860-865. |
| [11] | Nie De-zhi, Wan Ying, Ben Liang, Wang Ying-jun, Liu Xiang-zhu, Wang Li-hui, Li Chao, Zhang Shi-dong. Stem cell tumorigenicity in Balb/c nude mice [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 888-893. |
| [12] | Fan Yan, Wang Jian-jun, Wei Feng, Fan Xiao-hai, Ma Ai-qun. Effect of adipose-derived mesenchymal stem cell transplantation on inflammatory response and ventricular remodeling after myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 900-905. |
| [13] | Zhao Xiao-jian, Lu Cai-ping, Chu Wei-wei, Zhen Qiang, Tan Guo-liang, Zhang Ya-xiao, Wang Ren-feng, Zhang Bing, Liu Jia-bao. Bone marrow mesenchymal stem cell transplantation suppresses emphysema-induced inflammation and apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 906-911. |
| [14] | Yuan Shu-fang, Hu Lan-ying, Jiang Tao, Sun Li-hua, Zheng Rong-jiong, Zhao Jin-yan, Zhang Yue-xin . Effect of bone marrow mesenchymal stem cells transplantation on the expression of CD163 and interleukin-10 in rats with acute hepatic liver failure [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 919-925. |
| [15] | Wang Yan, Zhao Xin-li, Zhang Jun-yan, Tan Jun. Therapeutic applications of umbilical cord mesenchymal stem cells in Parkinson’s disease [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 932-937. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||