Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (37): 6548-6554.doi: 10.3969/j.issn.2095-4344.2013.37.002
Previous Articles Next Articles
Bone remodeling of rabbit tibia diaphysis cortical bone under bending force
Wang Yu, Li Cheng-mei, Li Yu-sheng, Zhang Zhi-gang
- The 208th Hospital of PLA, Changchun 130062, Jilin Province, China
-
Received:2013-06-14Revised:2013-07-05Online:2013-09-10Published:2013-09-10 -
Contact:Zhang Zhi-gang, the 208th Hospital of PLA, Changchun 130062, Jilin Province, China -
About author:Wang Yu☆, M.D., Associate chief physician, the 208th Hospital of PLA, Changchun 130062, Jilin Province, China 208wangyu@sina.com -
Supported by:Natural Science Foundation of Jilin Province, No. 201015123*
CLC Number:
Cite this article
Wang Yu, Li Cheng-mei, Li Yu-sheng, Zhang Zhi-gang. Bone remodeling of rabbit tibia diaphysis cortical bone under bending force[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(37): 6548-6554.
share this article
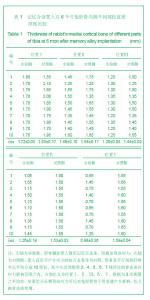
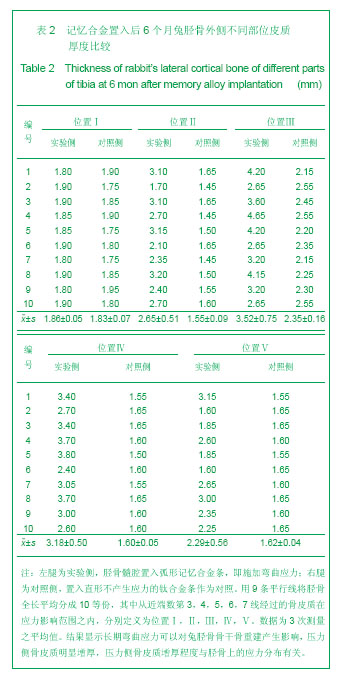
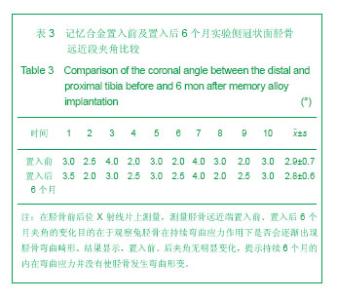
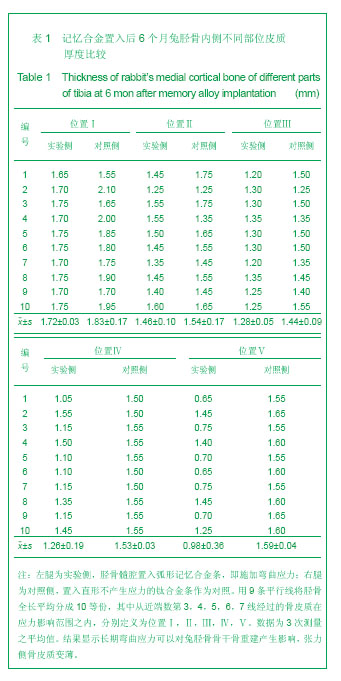
2.1 实验动物数量分析 实验纳入日本大耳白兔10只,全部进入结果分析,无脱失。 2.2 兔胫骨骨皮质厚度测定 见表1,2。 2.2.1 实验侧与对照侧胫骨内侧骨皮质厚度比较 实验侧胫骨内侧骨皮质与对照侧相比主要差异表现在:实验侧胫骨内侧骨皮质承受由记忆合金条所产生的非生理性张力,而对照侧不承受非生理性张力。位置Ⅰ实验侧与对照侧胫骨内侧骨皮质厚度差异无显著性意义,位置Ⅱ、Ⅲ、Ⅳ、Ⅴ4个平面骨皮质厚度差异有显著性意义(P < 0.05),实验侧胫骨内侧骨皮质薄于对照侧胫骨内侧骨皮质。 2.2.2 实验侧与对照侧胫骨外侧骨皮质厚度比较 实验侧胫骨外侧骨皮质与对照侧相比主要差异表现在:实验侧胫骨外侧骨皮质承受由记忆合金条所产生的非生理性压应力,而对照侧不承受非生理性压应力。位置Ⅰ实验侧与对照侧胫骨外侧骨皮质厚度差异无显著性意义,位置Ⅱ、Ⅲ、Ⅳ、Ⅴ4个平面骨皮质厚度差异有显著性意义(P < 0.05),实验侧胫骨外侧骨皮质厚于对照侧胫骨外侧骨皮质。实验侧胫骨外侧骨皮质增厚以记忆合金片中点处最明显,向两端增厚程度逐渐减低。 2.2.3 实验侧自身胫骨内外侧骨皮质厚度比较 实验侧胫骨外侧骨皮质承受由记忆合金条所产生的非生理性压应力,实验侧胫骨内侧骨皮质承受由记忆合金条所产生的非生理性张力,压应力侧骨皮质增厚,张应力侧骨皮质变薄。位置Ⅰ、Ⅱ、Ⅲ、Ⅳ、Ⅴ实验侧胫骨内外侧骨皮质厚度差异均有显著性(P < 0.05),5个平面胫骨外侧骨皮质均厚于内侧骨皮质,见图3。 2.2.4 对照侧自身胫骨内外侧骨皮质厚度比较 位置Ⅰ、Ⅱ、Ⅴ对照侧胫骨内外侧骨皮质厚度无明显差异。位置Ⅲ、Ⅳ 2个平面内外侧骨皮质厚度差异有显著性意义(P < 0.05),外侧骨皮质厚。这主要是由于兔胫骨结构的特殊性决定的,该部位恰好为胫腓骨融合处,故外侧骨皮质略厚。 2.2.5 实验侧胫骨5个位置外侧骨皮质厚度比较 位置Ⅰ、Ⅴ外侧骨皮质厚度无明显差异。位置Ⅱ、Ⅲ、Ⅳ之间以及位置Ⅱ、Ⅲ、Ⅳ与位置Ⅰ、Ⅴ之间外侧骨皮质厚度差异有显著性意义(P < 0.05)。实验侧外侧骨皮质厚度排序为Ⅲ>Ⅳ>Ⅱ>Ⅰ/Ⅴ。说明实验侧胫骨外侧骨皮质5个平面骨皮质厚度变化并不一致,骨皮质厚度排序间接反映不同平面承受弯曲应力大小不同。"
| [1] Wolff J, Das Gesetz der. Transformation. der Knochen, Hirschwald, Berlin,1892: 110-157. [2] Wolff J. The law of bone remodeling. Springer, Berlin, 1896: 57-123. [3] Hsieh YF, Turner CH. Effects of loading frequency on mechanically induced bone formation. J Bone Miner Res. 2001;16(5) : 918-924. [4] Burr DB, Turner CH, Naick P, et al. Does microdamage accumulation affect the mechanical properties of bone? J Biomech. 1998;31(4): 337-345. [5] Saxon LK, Robling AG, Alam I, et al. Mechanosensitivity of the Rat skeleton decreases after a long period of loading, but is improved with time off. Bone. 2005;36(3): 454-464. [6] 刘迎曦,赵文志,张军,等.不同应力环境下生长期大鼠股骨的生物力学试验研究[J]. 生物医学工程学杂志, 2005,22(3):472-475. [7] Globus RK, Bikle DD, Morey HE. The temporal response of bone to unloading. Endocrinology. 1986;118:733. [8] Bloomfield SA, Allen MR, Hogan HA,et al. Site-and Compartment-specific changes in bone with hindlimb unloading in mature adult rats. J Bone. 2002;31(1)∶149. [9] Kwon JY, Naito H, Matsumoto T, et al. Estimation of change of bone structures after total hip replacement using bone remodeling simulation. Clin Biomech (Bristol, Avon). 2013; 28(5):514-518. [10] Boerckel JD, Kolambkar YM, Stevens HY, et al. Effects of in vivo mechanical loading on large bone defect regeneration. J Orthop Res. 2012;30(7):1067-1075. [11] Roshan-Ghias A, Lambers FM, Gholam-Rezaee M, et al. In vivo loading increases mechanical properties of scaffold by affecting bone formation and bone resorption rates. Bone. 2011;49(6):1357-1364. [12] Yokota H, Leong DJ, Sun HB. Mechanical loading: bone remodeling and cartilage maintenance. Curr Osteoporos Rep. 2011;9(4):237-242. [13] Lee CS, Szczesny SE, Soslowsky LJ. Remodeling and repair of orthopedic tissue: role of mechanical loading and biologics: part II: cartilage and bone. Am J Orthop (Belle Mead NJ). 2011;40(3):122-128. [14] Robling AG, Turner CH. Mechanical signaling for bone modeling and remodeling. Crit Rev Eukaryot Gene Expr. 2009;19(4):319-338. [15] Epari DR, Duda GN, Thompson MS. Mechanobiology of bone healing and regeneration: in vivo models. Proc Inst Mech Eng H. 2010;224(12):1543-1553. [16] Moukoko D, Pourquier D, Pithioux M, et al. Influence of cyclic bending loading on in vivo skeletal tissue regeneration from periosteal origin. Orthop Traumatol Surg Res. 2010;96(8): 833-839. [17] Papachroni KK, Karatzas DN, Papavassiliou KA, et al. Mechanotransduction in osteoblast regulation and bone disease. Trends Mol Med. 2009;15(5):208-216. [18] Drenjan?evi? I, Davidovi? Cvetko E. Influence of physical activity to bone metabolism. Med Glas (Zenica). 2013; 10(1):12-19. [19] Boccafoschi F, Mosca C, Ramella M, et al. The effect of mechanical strain on soft (cardiovascular) and hard (bone) tissues: common pathways for different biological outcomes. Cell Adh Migr. 2013;7(2):165-173. [20] Rasouli MR, Porat MD, Hozack WJ, et al. Proximal femoral replacement and allograft prosthesis composite in the treatment of periprosthetic fractures with significant proximal bone loss. Orthop Surg. 2012;4(4):203-210. [21] Buck DW 2nd, Dumanian GA. Bone biology and physiology: Part I. The fundamentals. Plast Reconstr Surg. 2012;129(6): 1314-1320. [22] Robling AG. The interaction of biological factors with mechanical signals in bone adaptation: recent developments. Curr Osteoporos Rep. 2012;10(2):126-131. [23] Jaimes C, Jimenez M, Shabshin N, et al. Taking the stress out of evaluating stress injuries in children. Radiographics. 2012; 32(2):537-555. [24] Zuo C, Huang Y, Bajis R, et al. Osteoblastogenesis regulation signals in bone remodeling. Osteoporos Int. 2012;23(6): 1653-1663. [25] Del Fattore A, Teti A, Rucci N. Bone cells and the mechanisms of bone remodelling. Front Biosci (Elite Ed). 2012;4:2302-2321. [26] Beck RT, Illingworth KD, Saleh KJ. Review of periprosthetic osteolysis in total joint arthroplasty: an emphasis on host factors and future directions. J Orthop Res. 2012;30(4): 541-546. [27] Al Nazer R, Lanovaz J, Kawalilak C, et al. Direct in vivo strain measurements in human bone-a systematic literature review. J Biomech. 2012;45(1):27-40. [28] Andrade AC, Chrysis D, Audi L, et al. Methods to study cartilage and bone development. Endocr Dev. 2011;21:52-66. [29] Liedert A, Kaspar D, Augat P, et al. Mechanobiology of Bone Tissue and Bone Cells. In: Kamkin A, Kiseleva I, editors. Mechanosensitivity in Cells and Tissues. Moscow: Academia, 2005. [30] Dodge T, Wanis M, Ayoub R, et al. Mechanical loading, damping, and load-driven bone formation in mouse tibiae. Bone. 2012;51(4):810-818. [31] Szczesny SE, Lee CS, Soslowsky LJ. Remodeling and repair of orthopedic tissue: role of mechanical loading and biologics. Am J Orthop (Belle Mead NJ). 2010;39(11):525-530. [32] Knothe Tate ML, Dolejs S. Multiscale mechanobiology of de novo bone generation, remodeling and adaptation of autograft in a common ovine femur model. J Mech Behav Biomed Mater. 2011;4(6):829-840. [33] Avrunin AS, Tikhilov RM. [Osteocytic bone remodeling: history of the problem, morphological markers]. Morfologiia. 2011;139(1):86-94. [34] Idhammad A, Abdali A, Alaa N. Computational simulation of the bone remodeling using the finite element method: an elastic-damage theory for small displacements. Theor Biol Med Model. 2013;10:32. [35] Waldorff EI, Christenson KB, Cooney LA, et al. Microdamage repair and remodeling requires mechanical loading. J Bone Miner Res. 2010;25(4):734-745. [36] Cheng HY, Chu KT, Shen FC, et al. Stress effect on bone remodeling and osseointegration on dental implant with novel nano/microporous surface functionalization. J Biomed Mater Res A. 2013;101(4):1158-1164. [37] Robling AG, Castillo AB, Turner CH. Biomechanical and molecular regulation of bone remodeling. Annu Rev Biomed Eng. 2006;8:455-498. [38] Bougherara H, Klika V, Marsík F, et al. New predictive model for monitoring bone remodeling. J Biomed Mater Res A. 2010; 95(1):9-24. [39] Wang C, Han J, Li Q, et al. Simulation of bone remodelling in orthodontic treatment. Comput Methods Biomech Biomed Engin. 2012. [Epub ahead of print] [40] Pérez MA, Fornells P, Doblaré M, et al. Comparative analysis of bone remodelling models with respect to computerised tomography-based finite element models of bone. Comput Methods Biomech Biomed Engin. 2010;13(1):71-80. [41] Mine Y, Makihira S, Nikawa H, et al. Impact of titanium ions on osteoblast-, osteoclast- and gingival epithelial-like cells. J Prosthodont Res. 2010;54(1):1-6. [42] Chen JH, Liu C, You L, et al. Boning up on Wolff's Law: mechanical regulation of the cells that make and maintain bone. J Biomech. 2010;43(1):108-118. |
| [1] | Kong Lingbao, Lü Xin. Effect of implant selection and approach on support in the operation of posterolateral tibial plateau fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 942-947. |
| [2] | Xie Chengxin, Wang Wei, Wang Chenglong, Li Qinglong, Yin Dong. Systematic review and meta-analysis of bone morphogenetic protein for the treatment of acute tibial fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 486-492. |
| [3] | Lin Wang, Wang Yingying, Guo Weizhong, Yuan Cuihua, Xu Shenggui, Zhang Shenshen, Lin Chengshou. Adopting expanded lateral approach to enhance the mechanical stability and knee function for treating posterolateral column fracture of tibial plateau [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3826-3827. |
| [4] | Jie Ke, Deng Peng, Zeng Yirong. Application and comparison of four commonly used methods for patellar height measurement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3875-3881. |
| [5] | Tian Lin, Shi Xiaoqing, Duan Zhenglan, Wang Kuan, Zhang Li, Wang Peimin. Efficacy and safety of transverse tibial bone transport technique in the treatment of diabetic foot:a meta-analysis#br# [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3275-3280. |
| [6] | Huang Zeling, Shi Shanni, He Junjun, Gao Hongjian, Ge Haiya, Hong Zhenqiang. Meta-analysis of safety and effectiveness of proximal fibular osteotomy and high tibial osteotomy in the treatment of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(18): 2945-2952. |
| [7] | Chen Tiangui, Gao Lei, Li Tianbo, Wang Jiangning. Effects of Mailuoning Injection on mitochondrial autophagy and PINK1/Parkin pathway in a pig model of crush injury syndrome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2676-2680. |
| [8] | Han Shichong, Li Chang, Xing Haiyang, Ge Wenlong, Wang Gang . Finite element analysis of two internal fixation methods for treating extra-articular proximal tibial fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2329-2333. |
| [9] | Wang Xiankang, Zhang Yuejing, Yang You, Liu Jun. Simulation analysis of wear performance for tibial insert of unicompartmental knee arthroplasty prosthesis under gait load [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1831-1835. |
| [10] | Ma Zhen, Liang Da, Wu Xiaolin, Zhong Wei . Safety evaluation of tranexamic acid in reducing perioperative blood loss in high tibial osteotomy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1847-1852. |
| [11] | Wang Lei, Qi Rong, Li Jia, Qiu Zhixue, Wang Kai, Suo Nanangxiu. Minimally invasive percutaneous plate osteosynthesis and elastic nail system in the treatment of distal tibiofibular fracture in adults [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1864-1868. |
| [12] | Pei Tianlong, Wang Yongkang, Wu Wenjie, Zhao Hong, Wang Longsheng . Anatomical and morphological characteristics of knee joint in hemophilic arthritis patients with three-dimensional CT and X-ray films [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1886-1890. |
| [13] | Wang Debin, Bi Zhenggang. Optimization application for internal fixation strategy and three-dimensional printing technique of posterolateral tibial plateau fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1904-1910. |
| [14] | Li Yang, Min Shengwei, Xie Feng, Zhang Mingyong. A meta-analysis of clinical efficacy of preoperative use of three-dimensional printing in the treatment of tibial plateau fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1959-1968. |
| [15] | Fan Xinchao, Qiu Changmao, Ren Zhongyuan, Li Xicheng. Arthroscopic treatment of Schatzker type III tibial plateau fractures with hydroxylapatite rods: 6-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1571-1576. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||