Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 5082-5092.doi: 10.12307/2026.789
Exosomes and neuropathic pain: visualization analysis on literature
Ma Jing1, Han Jing2, Shi Linyu1, Wu Yuwei3, Yuan Haiguang1, 4, Li Yongfeng1, 3
- 1College of Acupuncture-Moxibustion and Tuina, Shaanxi University of Chinese Medicine, Xianyang 712046, Shaanxi Province, China; 2College of Education, Shaanxi Normal University, Xi’an 710062, Shaanxi Province, China; 3Institute of Frontier Cross-Science and Technology of Chinese Medicine, Xianyang 712046, Shaanxi Province, China; 4Affiliated Hospital of Shaanxi University of Chinese Medicine, Xianyang 712000, Shaanxi Province, China
-
Received:2025-09-11Accepted:2025-11-21Online:2026-07-08Published:2026-02-24 -
Contact:Li Yongfeng, PhD, Associate professor, Master’s supervisor, College of Acupuncture-Moxibustion and Tuina, Shaanxi University of Chinese Medicine, Xianyang 712046, Shaanxi Province, China; Institute of Frontier Cross-Science and Technology of Chinese Medicine, Xianyang 712046, Shaanxi Province, China -
About author:Ma Jing, MS candidate, College of Acupuncture-Moxibustion and Tuina, Shaanxi University of Chinese Medicine, Xianyang 712046, Shaanxi Province, China -
Supported by:Shaanxi Province Young Science and Technology Rising Star Project, No. 2025ZC-KJXX-70 (to LYF); National Natural Science Foundation of China, No. 82004489 (to LYF); Qinchuangyuan Traditional Chinese Medicine Industry Innovation Aggregation Zone Project (University of Traditional Chinese Medicine, Medical Institutions), No. L2024-QCY-ZYYJJQ-X154 (to LYF); Xianyang Key Research and Development Program Project, No. L2014-ZDYF-ZDYF-SF-0036 (to LYF); National Natural Science Foundation of China, No. 82205263 (to WYW); Shaanxi Province Natural Science Basic Research Program Project, No. 2024JC-YBMS-796 (to YHG)
CLC Number:
Cite this article
Ma Jing, Han Jing, Shi Linyu, Wu Yuwei, Yuan Haiguang, Li Yongfeng. Exosomes and neuropathic pain: visualization analysis on literature[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5082-5092.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
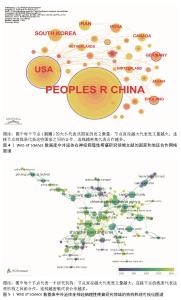
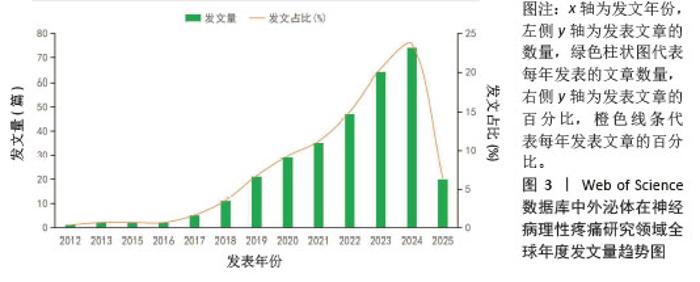
2.2 国家/地区和机构分析 在国家/地区合作网络中,节点外围出现紫色圆圈,表示中心度> 0.1,中心度较强,与其他国家/地区合作更密切(图4)。共有42个国家/地区参与相关研究,其中发文量排名前3位的国家分别为中国(189篇)、美国(70篇)和韩国(13篇);中心度排名前3位的国家分别为美国(1.00)、英国(0.19)和德国(0.15)。 在机构合作图谱中(图5),共有202个机构开展了相关研究,发文量排名前10位的机构均来自中国,其中发文量前3位的机构分别为上海交通大学(18篇)、南通大学(13篇)和中国人民解放军总医院(12篇);中心度排名前3位的机构分别为上海交通大学(0.08)、南通大学(0.06)和海军医科大学(0.05)。上海交通大学是发文量和中心度均为最高的机构。"


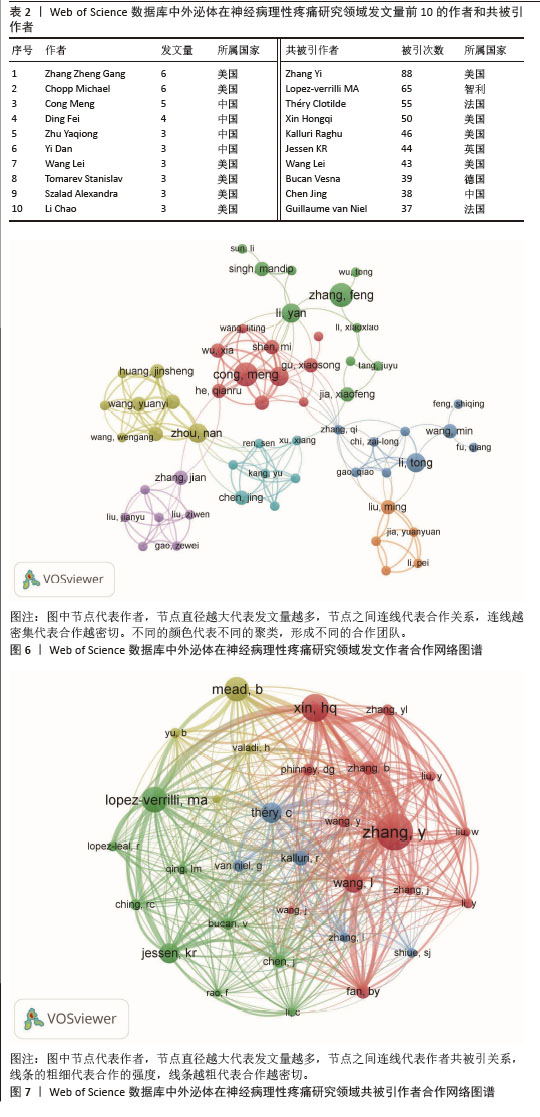
2.3 作者与共被引作者分析 为了更好地反映神经病理性疼痛外泌体领域的核心作者身份和相关性,对313篇论文的作者协作图谱进行了可视化,VOSviewer设置的最低引用阈值为2,有56位作者达到了网络可视化分析的阈值(图6)。在图6中,节点大小表示作者发表的论文数量,连接线表示相互协作关系。从2012-2025年,全球共有382位作者参与了神经病理性疼痛外泌体领域相关的研究,表2列出了发表文章和被引次数最多的前10位作者。来自美国Henry Ford Health System神经内科的Zhang Zheng Gang (6篇)和Chopp Michael(6篇)发表论文最多,其次是中国Nantong University的Cong Meng (5篇)和Ding Fei (4篇)。 在13 464位共被引作者中,共被引50次以上的作者有4位。共被引最多的作者是Zhang Yi (n=88),其次是Lopez-verrilli MA (n=65)、Théry Clotilde (n=55)和Xin Hongqi (n=50)。筛选出最少引用次数为20次的作者,绘制共被引网络(图7)。如图7所示,不同的共引作者之间存在积极的合作,例如Zhang Yi,Lopez-verrilli MA和Xin Hongqi之间。 "

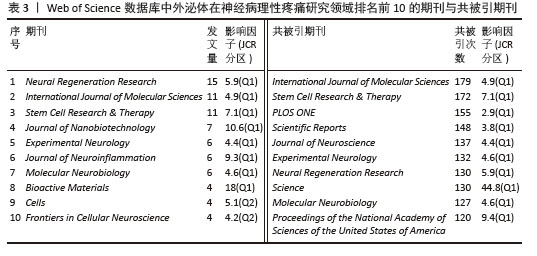
2.4 期刊与共被引期刊分析 此次研究检索到的313篇文献共发表在184种期刊上。发文量排在前10位的期刊见表3。排名前3的期刊分别是《Neural Regeneration Research》(15篇)、《International Journal of Molecular Sciences》(11篇)、《Stem Cell Research & Therapy》(11篇)。从影响因子来看,平均影响因子为7.41,80%的期刊排名Q1。 共被引频次排名前10的期刊见表3,被引频次前3位的期刊分别是《International Journal of Molecular Sciences》(179次),《Stem Cell Research & Therapy》(172次)、《PLOS ONE》(155次)。在这10种期刊中,均属于Q1区主流期刊,最高影响因子为《Science》(44.8)。 "

2.5 参考文献及共被引参考文献分析 此次研究分析了313篇文章的17 264次引用。选择最低引用阈值≥15的44篇共被引文章,并用VOSviewer 进行可视化分析(图8),图中每个节点代表一篇被引文章,这44篇共被引文章分为3个聚类,并通过3种颜色区分。此外,还列出了外泌体在神经病理性疼痛领域被引用次数最多的前10篇文献(表4),这些高被引文献主要聚焦于外泌体在神经损伤修复和疼痛管理中的作用,并探讨作为潜在治疗手段和生物标志物的价值,显示出广泛的应用前景和临床转化潜力。从图8的文献共被引网络可以看出,María Alejandra Lopez-Verrilli的“Schwann cell-derived exosomes enhance axonal regeneration in the peripheral nervous system”与Vesna Bucan的“Effect of Exosomes from Rat Adipose-Derived Mesenchymal Stem Cells on Neurite Outgrowth and Sciatic Nerve Regeneration After Crush Injury”存在显著共被引关系。此外还借助Citespace梳理了突现性引用强度最高的18篇文献(图9)。其中,Baglio SR发表于“Stem Cell Research & Therapy”的论文最早出现突现(2017年)。Zhang Yi团队发表的“Exosomes Derived from Mesenchymal Stromal Cells Promote Axonal Growth of Cortical Neurons”突现强度最高(7.67),突现时间范围为2018-2021年,显著揭示了该领域的研究趋势。 "
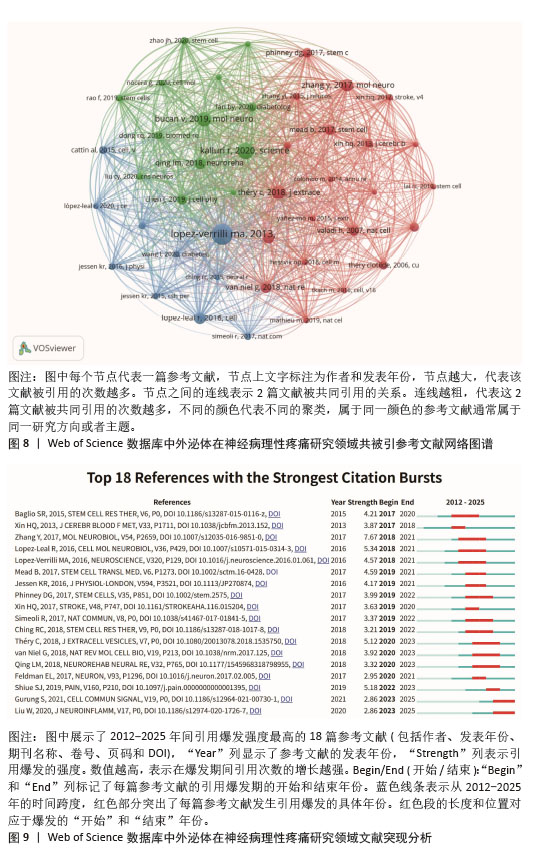

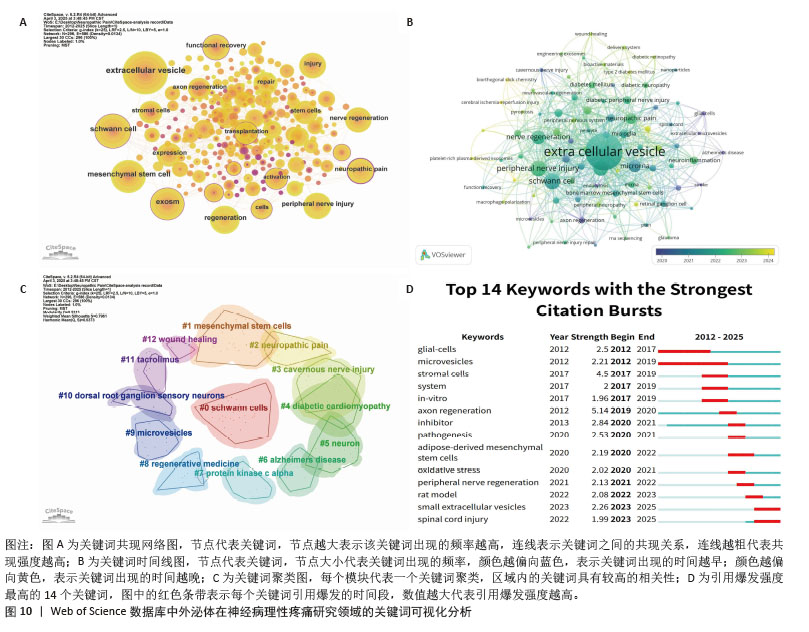
2.6 研究热点和趋势 关键词作为文献中所代表的各种主题之间相互关系的指标,概括了文章的本质。分析关键词有助于发掘该领域的热点。 2.6.1 关键词共现分析 通过关键词共现分析,可以快速了解某一领域的研究热点,图10A呈现了外泌体与神经病理性疼痛研究领域的关键词共现图谱,通过合并意义相近的关键词后,共获得296个节点、586条连线,网络密度为0.013 4,图中节点大小与关键词出现频次呈正比,连线粗细表示共现关系强度,节点外围包绕的紫色线条代表高中心度的关键词,表明节点在网络中的作用越大、影响力越高。排名前3的关键词为extracellular vesicle(109次)、schwann cell(64次)、mesenchymal stem cell(60次)。图10B展示了作者关键词的叠加可视化,之前出现的关键词用深蓝色表示,而浅黄色代表最近出现的关键词。“spinal cord”“microrna”“apoptosis and proliferation”“mesenchymal stromal cell”“cavernous nerve injury”等关键词是早期出现的主要话题。而近年来逐渐转向“diabetic neuropathy”“nerve regeneration”“neuroinflammation”“peripheral nerve injury”等关键词。 2.6.2 关键词聚类分析 关键词聚类分析有助于发现一个领域相关研究的分布情况,该研究利用Citespace软件中的LLR检验算法对关键词进行聚类分析。聚类编号越小,表明该聚类下的文献研究数量越多,研究热度越高。对关键词聚类分析以后共得到13个有意义的聚类,见图10C,关键词聚类的Modularity Q=0.531 3(> 0.3)表明聚类结构显著;Mean Sihouette=0.796 1(> 0.7),表明该聚类成员一致性高。表5列出了每个集群下包含的主要关键词及其轮廓值,根据聚类结果,大致可以概括出目前研究主要集中于以下几个方面:①细胞与组织修复机制,如#0、#1、#5、#10;②疾病模型与病理机制,如#2、#3、#4、#6;③分子调控与信号通路,如#7、#9、#11;④再生医学与治疗策略,如#8,#12。 2.6.3 关键词突发性检测 关键词突现是指在关键词较短的一段时间内出现频次显著增加,反映了这一时期内该领域研究者们共同关注的研究内容;可以看出近几年来热点领域的兴衰,可对未来发展方向进行预测,通过CiteSpace识别出了14个具有强被引爆发的关键词(图10D)。如图10D所示,爆发持续时间最长为7年。关键词“glial-cells”(2012-2017年)、“microvesicles”(2012-2019年)、“stromal cells”(2017-2019年)、“in vitro”(2017-2019年)在早期受到了较多的关注。而“rat model”(2022-2023年)、“small extracellular vesicles”(2023-2025年)、“spinal cord injury”(2023-2025年)等关键词使用较晚,表明这些关键词近年来受到了足够的关注,很可能代表了当前领域的研究热点。 "
| [1] 杜成欣,董建群,颜流霞,等.神经病理性疼痛患者的自我管理:定义、要素与展望[J].中国慢性病预防与控制,2023,31(9):702-705. [2] SMITH PA. The Known Biology of Neuropathic Pain and Its Relevance to Pain Management. Can J Neurol Sci. 2024;51(1):32-39. [3] HANGE N, POUDEL S, OZAIR S, et al. Managing Chronic Neuropathic Pain: Recent Advances and New Challenges. Neurol Res Int. 2022; 2022:8336561. [4] MOGHASSEMI S, DADASHZADEH A, SOUSA MJ, et al. Extracellular vesicles in nanomedicine and regenerative medicine: A review over the last decade. Bioact Mater. 2024;36:126-156. [5] YANG S, SUN Y, YAN C. Recent advances in the use of extracellular vesicles from adipose-derived stem cells for regenerative medical therapeutics. J Nanobiotechnology. 2024;22(1):316. [6] ZHANG YU, YE G, ZHAO J, et al. Exosomes carried miR-181c-5p alleviates neuropathic pain in CCI rat models. An Acad Bras Cienc. 2022;94(3):e20210564. [7] GOLMAKANI H, AZIMIAN A, GOLMAKANI E. Newly discovered functions of miRNAs in neuropathic pain: Transitioning from recent discoveries to innovative underlying mechanisms. Mol Pain. 2024;20:17448069231225845. [8] CONG M, SHEN M, WU X, et al. Improvement of sensory neuron growth and survival via negatively regulating PTEN by miR-21-5p-contained small extracellular vesicles from skin precursor-derived Schwann cells. Stem Cell Res Ther. 2021;12(1):80. [9] HUA T, YANG M, SONG H, et al. Huc-MSCs-derived exosomes attenuate inflammatory pain by regulating microglia pyroptosis and autophagy via the miR-146a-5p/TRAF6 axis. J Nanobiotechnology. 2022;20(1):324. [10] WANG C, ZHANG Y, ZHANG Y, et al. A bibliometric analysis of gastric cancer liver metastases: advances in mechanisms of occurrence and treatment options. Int J Surg. 2024;110(4):2288-2299. [11] YIN T, CHEN Y, TANG L, et al. Relationship between modifiable lifestyle factors and chronic kidney disease: a bibliometric analysis of top-cited publications from 2011 to 2020. BMC Nephrol. 2022;23(1):120. [12] CHENG K, GUO Q, SHEN Z, et al. Frontiers of ferroptosis research: An analysis from the top 100 most influential articles in the field. Front Oncol. 2022;12:948389. [13] LI R, LIU H, ZHANG K, et al. Global tendency and research trends of minimally invasive surgery for glaucoma from 1992 to 2023: A visual bibliometric analysis. Heliyon. 2024; 10(16):e36591. [14] CHOPP M, ZHANG ZG. Emerging potential of exosomes and noncoding microRNAs for the treatment of neurological injury/diseases. Expert Opin Emerg Drugs. 2015;20(4): 523-526. [15] ZHANG Y, QIN Y, CHOPP M, et al. Ischemic Cerebral Endothelial Cell-Derived Exosomes Promote Axonal Growth. Stroke. 2020;51(12):3701-3712. [16] THÉRY C, WITWER KW, AIKAWA E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. [17] ZHANG Y, CHOPP M, LIU XS, et al. Exosomes Derived from Mesenchymal Stromal Cells Promote Axonal Growth of Cortical Neurons. Mol Neurobiol. 2017;54(4):2659-2673. [18] BUCAN V, VASLAITIS D, PECK CT, et al. Effect of Exosomes from Rat Adipose-Derived Mesenchymal Stem Cells on Neurite Outgrowth and Sciatic Nerve Regeneration After Crush Injury. Mol Neurobiol. 2019; 56(3):1812-1824. [19] 李杰,刘世平.外泌体及其miRNA在神经系统常见疾病中的研究进展[J].四川医学,2022,43(11):1147-1151. [20] LU Y, ZHANG J, ZENG F, et al. Human PMSCs-derived small extracellular vesicles alleviate neuropathic pain through miR-26a-5p/Wnt5a in SNI mice model. J Neuroinflammation. 2022;19(1):221. [21] LI Q, ZHANG F, FU X, et al. Therapeutic Potential of Mesenchymal Stem Cell-Derived Exosomes as Nanomedicine for Peripheral Nerve Injury. Int J Mol Sci. 2024; 25(14):7882. [22] 方俊,魏伟,薛亚婷,等.M2型巨噬细胞衍生外泌体促进小胶质细胞M2型极化[J].中国组织工程研究,2025,29(25): 5320-5327. [23] LI S, HUANG C, TU C, et al. Bone marrow mesenchymal stem cell-derived exosomes shuttling miR-150-5p alleviates mechanical allodynia in rats by targeting NOTCH2 in microglia. Mol Med. 2022;28(1):133. [24] KARNAS E, DUDEK P, ZUBA-SURMA EK. Stem cell- derived extracellular vesicles as new tools in regenerative medicine - Immunomodulatory role and future perspectives. Front Immunol. 2023;14: 1120175. [25] GAO X, GAO LF, ZHANG YN, et al. Huc-MSCs-derived exosomes attenuate neuropathic pain by inhibiting activation of the TLR2/MyD88/NF-κB signaling pathway in the spinal microglia by targeting Rsad2. Int Immunopharmacol. 2023;114:109505. [26] MOU C, LI Z, LIU N, et al. Low level TGF-β1-treated Umbilical mesenchymal stem cells attenuates microgliosis and neuropathic pain in chronic constriction injury by exosomes/lncRNA UCA1/miR-96-5p/FOXO3a. Biochem Biophys Rep. 2023;34:101477. [27] SHI B, DENG W, LONG X, et al. miR-21 increases c-kit+ cardiac stem cell proliferation in vitro through PTEN/PI3K/Akt signaling. PeerJ. 2017;5:e2859. [28] LIU W, RONG Y, WANG J, et al. Exosome-shuttled miR-216a-5p from hypoxic preconditioned mesenchymal stem cells repair traumatic spinal cord injury by shifting microglial M1/M2 polarization. J Neuroinflammation. 2020;17(1):47. [29] ZHANG K, LI P, JIA Y, et al. Concise review: Current understanding of extracellular vesicles to treat neuropathic pain. Front Aging Neurosci. 2023;15:1131536. [30] YU X, YU C, HE W. Emerging trends and hot spots of NLRP3 inflammasome in neurological diseases: A bibliometric analysis. Front Pharmacol. 2022;13:952211. [31] SIM J, PARK J, MOON JS, et al. Dysregulation of inflammasome activation in glioma. Cell Commun Signal. 2023;21(1):239. [32] LI Y, KANG S, HALAWANI D, et al. Macrophages facilitate peripheral nerve regeneration by organizing regeneration tracks through Plexin-B2. Genes Dev. 2022; 36(3-4):133-148. [33] HU T, CHANG S, QI F, et al. Neural grafts containing exosomes derived from Schwann cell-like cells promote peripheral nerve regeneration in rats. Burns Trauma. 2023;11:tkad013. [34] LIU YY, LI Y, WANG L, et al. Mesenchymal stem cell-derived exosomes regulate microglia phenotypes: a promising treatment for acute central nervous system injury. Neural Regen Res. 2023;18(8):1657-1665. [35] YANG J, WANG B, WANG Y, et al. Exosomes Derived from Adipose Mesenchymal Stem Cells Carrying miRNA-22-3p Promote Schwann Cells Proliferation and Migration through Downregulation of PTEN. Dis Markers. 2022; 2022:7071877. [36] WANG X, ZHANG Y, JIN T, et al. Adipose-Derived Mesenchymal Stem Cells Combined With Extracellular Vesicles May Improve Amyotrophic Lateral Sclerosis. Front Aging Neurosci. 2022;14:830346. [37] PAPADOPOULOS KS, PIPERI C, KORKOLOPOULOU P. Clinical Applications of Adipose-Derived Stem Cell (ADSC) Exosomes in Tissue Regeneration. Int J Mol Sci. 2024; 25(11):5916. [38] SALEHPOUR A, KARIMI Z, GHASEMI ZADEH M, et al. Therapeutic potential of mesenchymal stem cell-derived exosomes and miRNAs in neuronal regeneration and rejuvenation in neurological disorders: a mini review. Front Cell Neurosci. 2024;18: 1427525. [39] 袁博,谢利德,付秀美.许旺细胞源性外泌体促进损伤周围神经的修复与再生[J].中国组织工程研究,2023,27(6):935-940. [40] CHEN YF, LUH F, HO YS, et al. Exosomes: a review of biologic function, diagnostic and targeted therapy applications, and clinical trials. J Biomed Sci. 2024;31(1):67. [41] XU Y, HU Y, XU S, et al. Exosomal microRNAs as Potential Biomarkers and Therapeutic Agents for Acute Ischemic Stroke: New Expectations. Front Neurol. 2022;12:747380. [42] HUANG J, ZHANG G, LI S, et al. Endothelial cell-derived exosomes boost and maintain repair-related phenotypes of Schwann cells via miR199-5p to promote nerve regeneration. J Nanobiotechnology. 2023;21(1):10. [43] 郭佳,任亚锋,李冰,等.负载miRNA间充质干细胞源外泌体改善脊髓损伤的作用机制[J].中国组织工程研究,2025,29(36):7827-7838. [44] DOGNY C, ANDRÉ-LÉVIGNE D, KALBERMATTEN DF, et al. Therapeutic Potential and Challenges of Mesenchymal Stem Cell-Derived Exosomes for Peripheral Nerve Regeneration: A Systematic Review. Int J Mol Sci. 2024;25(12):6489. [45] IKEDA T, KAWABORI M, ZHENG Y, et al. Intranasal Administration of Mesenchymal Stem Cell-Derived Exosome Alleviates Hypoxic-Ischemic Brain Injury. Pharmaceutics. 2024;16(4):446. [46] NAMINI MS, DANESHIMEHR F, BEHESHTIZADEH N, et al. Cell-free therapy based on extracellular vesicles: a promising therapeutic strategy for peripheral nerve injury. Stem Cell Res Ther. 2023;14(1):254. [47] LI Q, FU X, KOU Y, et al. Engineering strategies and optimized delivery of exosomes for theranostic application in nerve tissue. Theranostics. 2023;13(12):4266-4286. [48] JIANG M, WANG Y, WANG J, et al. The etiological roles of miRNAs, lncRNAs, and circRNAs in neuropathic pain: A narrative review. J Clin Lab Anal. 2022;36(8):e24592. [49] WEI Z, GUO C, ZHOU H, et al. Exosome-mediated miRNA delivery: a molecular switch for reshaping neuropathic pain therapy. Front Mol Neurosci. 2025;18:1625943. [50] WANG YH, TANG YR, GAO X, et al. Aspirin-triggered Resolvin D1 ameliorates activation of the NLRP3 inflammasome via induction of autophagy in a rat model of neuropathic pain. Front Pharmacol. 2023;14:971136. [51] ZHAO YY, WU ZJ, ZHU LJ, et al. Emerging roles of miRNAs in neuropathic pain: From new findings to novel mechanisms. Front Mol Neurosci. 2023;16:1110975. [52] XIN W, QIN Y, LEI P, et al. From cerebral ischemia towards myocardial, renal, and hepatic ischemia: Exosomal miRNAs as a general concept of intercellular communication in ischemia-reperfusion injury. Mol Ther Nucleic Acids. 2022;29:900-922. [53] ZEBOUDJ L, SIDERIS-LAMPRETSAS G, SILVA R, et al. Silencing miR-21-5p in sensory neurons reverses neuropathic allodynia via activation of TGF-β-related pathway in macrophages. J Clin Invest. 2023;133(11):e164472. [54] LIU YP, TIAN MY, YANG YD, et al. Schwann cells-derived exosomal miR-21 participates in high glucose regulation of neurite outgrowth. iScience. 2022;25(10):105141. [55] LAI Z, YE T, ZHANG M, et al. Exosomes as Vehicles for Noncoding RNA in Modulating Inflammation: A Promising Regulatory Approach for Ischemic Stroke and Myocardial Infarction. J Inflamm Res. 2024;17:7485-7501. [56] ZHANG L, MAO L, WANG H. The Neuroprotection Effects of Exosome in Central Nervous System Injuries: a New Target for Therapeutic Intervention. Mol Neurobiol. 2022;59(12):7152-7169. [57] SCHEIBER C, KLEIN HC, SCHNEIDER JM, et al. HSV-1 and Cellular miRNAs in CSF-Derived Exosomes as Diagnostically Relevant Biomarkers for Neuroinflammation. Cells. 2024;13(14):1208. [58] ZENG M, ZHANG T, LIN Y, et al. The Common LncRNAs of Neuroinflammation-Related Diseases. Mol Pharmacol. 2023;103(3):113-131. [59] LIU T, LI Y, CHEN S, et al. CircDDX17 enhances coxsackievirus B3 replication through regulating miR-1248/NOTCH receptor 2 axis. Front Microbiol. 2022;13: 1012124. [60] ZHANG YJ, ZHU WK, QI FY, et al. CircHIPK3 promotes neuroinflammation through regulation of the miR-124-3p/STAT3/NLRP3 signaling pathway in Parkinson’s disease. Adv Clin Exp Med. 2023;32(3):315-329. [61] ZHENGBIAO Z, LIANG C, ZHI Z, et al. Circular RNA_HIPK3-Targeting miR-93-5p Regulates KLF9 Expression Level to Control Acute Kidney Injury. Comput Math Methods Med. 2023;2023:1318817. [62] ZHOU C, ZHANG B, YANG Y, et al. Stem cell-derived exosomes: emerging therapeutic opportunities for wound healing. Stem Cell Res Ther. 2023;14(1):107. [63] KHAN MI, ALSAYED RKME, CHOUDHRY H, et al. Exosome-Mediated Response to Cancer Therapy: Modulation of Epigenetic Machinery. Int J Mol Sci. 2022;23(11):6222. [64] DAI W, QIAO X, FANG Y, et al. Epigenetics-targeted drugs: current paradigms and future challenges. Signal Transduct Target Ther. 2024;9(1):332. [65] YAO W, HU X, WANG X. Crossing epigenetic frontiers: the intersection of novel histone modifications and diseases. Signal Transduct Target Ther. 2024;9(1):232. [66] ZHANG C, GAO R, ZHOU R, et al. The emerging power and promise of non-coding RNAs in chronic pain. Front Mol Neurosci. 2022;15:1037929. [67] HUANG X, LI A, XU P, et al. Current and prospective strategies for advancing the targeted delivery of CRISPR/Cas system via extracellular vesicles. J Nanobiotechnology. 2023;21(1):184. [68] ASHRAFIZADEH M, KUMAR AP, AREF AR, et al. Exosomes as Promising Nanostructures in Diabetes Mellitus: From Insulin Sensitivity to Ameliorating Diabetic Complications. Int J Nanomedicine. 2022;17:1229-1253. [69] TANG T, CHEN L, ZHANG M, et al. Exosomes derived from BMSCs enhance diabetic wound healing through circ-Snhg11 delivery. Diabetol Metab Syndr. 2024;16(1):37. [70] AHMED LA, AL-MASSRI KF. Exploring the Role of Mesenchymal Stem Cell-Derived Exosomes in Diabetic and Chemotherapy-Induced Peripheral Neuropathy. Mol Neurobiol. 2024; 61(8):5916-5927. [71] KALVALA AK, BAGDE A, ARTHUR P, et al. Cannabidiol-Loaded Extracellular Vesicles from Human Umbilical Cord Mesenchymal Stem Cells Alleviate Paclitaxel-Induced Peripheral Neuropathy. Pharmaceutics. 2023;15(2):554. [72] GE Y, WU J, ZHANG L, et al. A New Strategy for the Regulation of Neuroinflammation: Exosomes Derived from Mesenchymal Stem Cells. Cell Mol Neurobiol. 2024;44(1):24. [73] SHIUE SJ, RAU RH, SHIUE HS, et al. Mesenchymal stem cell exosomes as a cell-free therapy for nerve injury-induced pain in rats. Pain. 2019;160(1):210-223. [74] SHUEN TWH, ALUNNI-FABBRONI M, ÖCAL E, et al. Extracellular Vesicles May Predict Response to Radioembolization and Sorafenib Treatment in Advanced Hepatocellular Carcinoma: An Exploratory Analysis from the SORAMIC Trial. Clin Cancer Res. 2022; 28(17):3890-3901. [75] HU Y, ZHANG Q, WU Z, et al. Exosomal miR-200c and miR-141 as cerebrospinal fluid biopsy biomarkers for the response to chemotherapy in primary central nervous system lymphoma. Discov Oncol. 2023;14(1):205. [76] FAN B, CHOPP M, ZHANG ZG, et al. Emerging Roles of microRNAs as Biomarkers and Therapeutic Targets for Diabetic Neuropathy. Front Neurol. 2020;11:558758. |
| [1] | Xu Canli, He Wenxing, Wang Yuping, Ba Yinying, Chi Li, Wang Wenjuan, Wang Jiajia. Research context and trend of TBK1 in autoimmunity, signaling pathways, gene expression, tumor prevention and treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(在线): 1-11. |
| [2] | Zhu Xiaolong, Zhang Wei, Yang Yang. Visualization analysis of research hotspots and cutting-edge information in the field of intervertebral disc regeneration and repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2391-2402. |
| [3] | Wen Fayan, Li Yan, Qiang Tianming, Yang Chen, Shen Linming, Li Yadong, Liu Yongming. Unilateral biportal endoscopic technology for treatment of lumbar degenerative diseases: global research status and changing trends [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2380-2390. |
| [4] | Lai Yu, Chen Yueping, Zhang Xiaoyun. Research hotspots and frontier trends of bioactive materials in treating bone infections [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2132-2144. |
| [5] | Song Puzhen, Ma Hebin, Chen Hongguang, Zhang Yadong. Effect of bone marrow mesenchymal stem cell-derived exosomes combined with transforming growth factor beta 1 on macrophages [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1616-1623. |
| [6] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [7] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [8] | Chen Yulin, He Yingying, Hu Kai, Chen Zhifan, Nie Sha Meng Yanhui, Li Runzhen, Zhang Xiaoduo , Li Yuxi, Tang Yaoping. Effect and mechanism of exosome-like vesicles derived from Trichosanthes kirilowii Maxim. in preventing and treating atherosclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1768-1781. |
| [9] | Han Teng, Ma Hong, Yang Ruoyi, Luo Yi, Li Chao. Oral squamous cell carcinoma-derived exosomal delivery of angiopoietin-2 is involved in tumor angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1755-1767. |
| [10] | Huang Jiawen, Pan Zhiyi, Xue Wenjun, Lian Yuanpei, Xu Jianda. Plant-derived vesicles and malignant tumor therapy: cross-species communication and modulation of host cell responses [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1828-1838. |
| [11] | Wang Baiyan, Yang Shu, Wang Yiming, Wu Mengqing, Xiao Yu, Guo Zixuan, Zhang Boyi, Feng Shuying. Exosome-delivered CRISPR/Cas system enables gene editing in target cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1839-1849. |
| [12] | Wang Zhenze, Liu Fende, Zhang Rui, Li Wujun. Mesenchymal stem cells in treatment of arteriosclerosis obliterans of lower extremities: systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1869-1876. |
| [13] | Lai Jiaming, , Song Yuling, Chen Zixi, Wei Jinghuan, Cai Hao, , Li Guoquan, . Screening of diagnostic markers for endothelial cell Senescence in mice with radiation-induced heart disease and analysis of immune infiltration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1450-1463. |
| [14] | Huang Jie, Zeng Hao, Wang Wenchi, Lyu Zhucheng, Cui Wei. Visualization analysis of literature on the effect of lipid metabolism on osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1558-1568. |
| [15] | Zhang Haiwen, Zhang Xian, Xu Taichuan, Li Chao. Bibliometric and visual analysis of the research status and trends of senescence in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1580-1591. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||