Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6937-6945.doi: 10.12307/2026.759
Previous Articles Next Articles
Main preparation methods of new fluorescent nanomaterial carbon quantum dots and their applications in tumor diagnosis and treatment
Guo Zhiyou1, Hu Rui2, Zhu Jinling1
- 1School of Basic Medicine, 2School of Pharmacy, Jiamusi University, Jiamusi 154100, Heilongjiang Province, China
-
Accepted:2025-09-25Online:2026-09-18Published:2026-03-16 -
Contact:Zhu Jinling, Professor, School of Basic Medicine, Jiamusi University, Jiamusi 154100, Heilongjiang Province, China -
About author:Guo Zhiyou, MS candidate, School of Basic Medicine, Jiamusi University, Jiamusi 154100, Heilongjiang Province, China
CLC Number:
Cite this article
Guo Zhiyou, Hu Rui, Zhu Jinling. Main preparation methods of new fluorescent nanomaterial carbon quantum dots and their applications in tumor diagnosis and treatment[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6937-6945.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
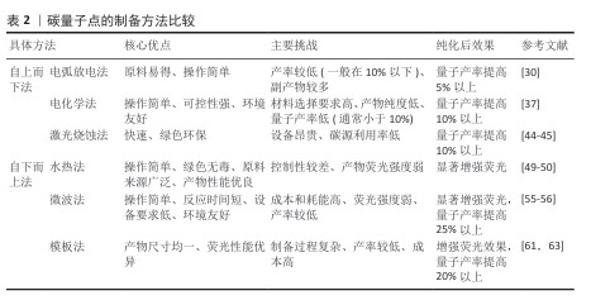
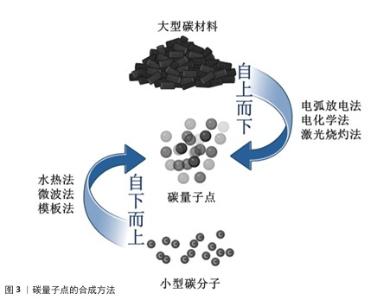
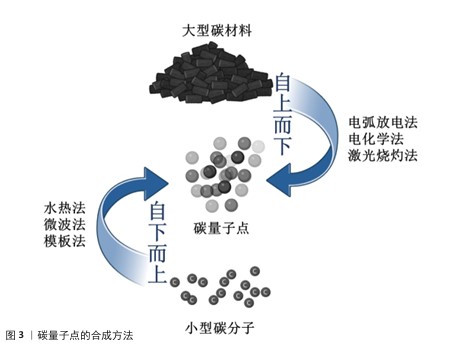
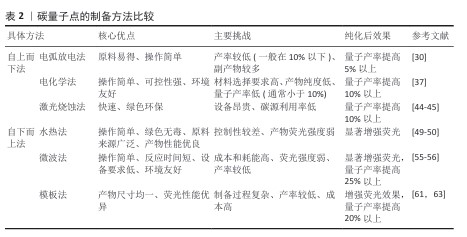
2.2.1 电弧放电法 人类首次发现的碳量子点由电弧放电法合成,这也是最早用于制备碳量子点的方法之一。2004年,XU等[15]在纯化电弧放电煤烟只准备单壁碳纳米管时分离出了一种未知的荧光碳材料,随后通过引入羧基官能团增强材料的亲水性,最终得到单壁碳纳米管、短管状碳结构和一个快速移动的高荧光材料,这种材料就是碳量子点。 在电弧放电过程中,先将石墨等大分子碳源在惰性气体(如氩气)特定条件下进行放电处理,使碳源电极尖端的碳原子受热蒸发形成等离子体,随后在溶液(如水或有机溶剂)中冷却凝结,最后通过氧化处理和分离纯化得到纳米级碳量子点[30]。 电弧放电法以石墨为直接碳源,前驱体制备工艺简单,可通过调节电极间距、电流强度等参数调控碳量子点粒径与表面性质[31]。但电弧过程中的碳源利用率不高,易生成大量副产物(如碳纳米管、石墨烯碎片)且量子产率较低,还需进行额外的纯化处理[32];另外,电弧放电法需在耐高温、耐高压的反应腔及惰性气体保护系统中进行,生产难度大[33]。LI等[30]在氩气的密闭反应室中,以高结晶度石墨电极(碳含量> 94%)为前体合成碳量子点,产生了15%-10%的碳纳米管、10%-15%的石墨烯碎片、5%-10%的无定形碳颗粒和小于5%的金属杂质4种副产物,质量占30%-40%,随后通过梯度离心、硝酸氧化回流、透析纯化3步法进行纯化处理,使量子产率从6.4%提升至16.2%。 2.2.2 电化学法 电化学法制作过程比较简单,对实验环境要求较低,一般在常温常压下进行即可[34-35]。电化学法通常选择高导电性、化学稳定性好的材料(如石墨、金、铂等)作为电极,采用含有碳源的电解质溶液,在电极间施加适当的直流或脉冲电压,促使碳源在电极表面发生氧化还原反应,进而经过裂解、聚合、碳化等一系列过程,最终形成碳量子点[36]。 电化学法可用碳源种类丰富且常见(如葡萄糖、果糖等),操作过程简便,相对电弧放电法而言更绿色环保,减少了有机溶剂的使用[37]。但电化学法对电极材料的导电性和化学稳定性要求较高,该法的产物中可能含有杂质,并且产物存在粒径分布宽、成分异质性高、荧光量子产率偏低(通常< 10%[38-39])等问题,需进行另外纯化处理来提高产率[40]。 2007年,DING课题组首次开创性提出采用电化学氧化多壁碳纳米管法制备碳量子点,以多壁碳纳米管为工作电极、铂丝为对电极、Ag/AgClO4为参比电极、四丁基高氯酸铵的乙腈溶液(0.1 mol/L)作为电解液,经分离、纯化等步骤得到了粒径为(2.8±0.5) nm、荧光量子产率为6.4%的碳量子点[16]。YAT等[37]将石墨电极浸入电解质溶液中在特定电压下进行电解,产生二氧化碳纳米气泡,剥离石墨表层形成碳量子点,再通过梯度离心、微孔膜过滤、透析纯化三步法进行纯化,最终形成尺寸在2-5 nm之间碳量子点,量子产率也从8.3%提升至26.17%。 2.2.3 激光烧蚀法 激光烧蚀法合成碳量子点原理是利用高能激光束照射碳靶,使碳靶表面的碳原子瞬间熔化、快速挥发形成等离子体,随后在冷却过程中结晶成纳米颗粒[41]。 激光烧蚀法能在短时间内高效完成碳量子点的制备,通过调节激光参数可以实现对碳量子点尺寸、形态和光学性质的精确调控,该方法无需使用复杂的化学试剂,绿色环保[42]。但激光烧蚀过程中产生的高温可能导致部分碳材料不均匀剥离,影响产物的纯度和尺寸分布;制备过程中未完全剥离的碳靶材料会作为杂质存在,需进一步纯化;需要高精度的激光设备和严格的操作环境,对设备及操作环境要求较高[43]。KANG等[44]通过一种简单、低成本、环保的液体脉冲激光烧蚀方法,以丰富且低成本的煤炭资源为碳源转化为碳量子点,产生柠檬酸残渣和煤中无机金属离子副产物,通过透析、离心与过滤、冷冻干燥三步纯化最终得到尺寸在2-5 nm的碳量子点,量子产率也从2.1%提升至14.6%。TORRISI等[45]通过特定激光波长烧蚀催化碳靶合成碳量子点,尺寸控制在2-8 nm,但产生了未完全碳化的非晶碳颗粒和金属离子副产物,通过离心、透析、凝胶过滤色谱三步法进行纯化,使量子产率从5%-8%提升至15%-25%。 2.2.4 水热法 水热法通过在高温高压条件下将有机前驱体(如化学试剂、生物质等)碳化,发生脱水、缩合、环化等反应形成碳量子点[46]。水热法合成的碳量子点表面富含氧官能团,水溶性优异,产物的结构和性质也可通过调整原料种类及制备条件(如温度、时间、化学试剂)来调控[47]。水热法操作条件温和、无需昂贵设备,也避免了有毒有机溶剂的使用,碳源材料前驱体常见且易获取,如葡萄糖、柠檬酸等,但产物荧光强度较弱,需纯化以进一步提高产物荧光强度[48]。MOHAPATRA课题组报道了以橘子汁为前驱体,通过一步水热反应制备碳量子点的方法,量子产率高达26%,这些碳量子点尺寸范围为1.5-4.5 nm,分布较窄,但碳量子点的荧光强度较弱,通过离心、透析两步进行纯化,显著提升碳量子点荧光强度[49]。ZHENG等[50]利用废旧蚕丝和全血样品为原料,通过水热方法在超纯水中制备了一种能用于血液分析和细胞成像的碳量子点,此碳量子点尺寸分布窄,在1.5-4.5 nm之间,量子产率高达42%,通过离心、超滤浓缩、透析三步法纯化使碳量子点的荧光强度提升3.2倍。ZHANG等[51]以柠檬酸为碳源,在超纯水条件下通过水热法合成碳量子点,尺寸分布较窄,在1.5-5 nm之间,量子产率为15.1%-15.3%,通过离心、超滤浓缩、透析三步法纯化将荧光背景噪声降低80%以上。 2.2.5 微波法 微波法是将含碳的有机小分子聚合物(如葡萄糖、柠檬酸等)与溶剂混合后形成溶液,置于微波反应器内使分子快速振动、摩擦,产生“内加热” 效果,促使溶液中的含碳有机分子聚合物快速脱水、碳化,制备表面修饰有聚乙烯亚胺的碳量子点[52]。聚乙烯亚胺是一种由乙烯亚胺单体聚合而成的阳离子聚合物,具有丰富的氨基基团,这些氨基基团可以与带负电荷的DNA分子通过静电作用结合,形成稳定的复合物[53]。微波法形成的碳量子点中含有聚乙烯亚胺共有2种作用,一是增强荧光,二是基因递送(将目的基因精准送至细胞内)[54]。 微波法反应迅速,能在数分钟内完成碳量子点的制备,极大缩短制备时间,并且该方法因加热均匀,生成的碳量子点尺寸大小几乎相同,聚乙烯亚胺能提升产物荧光强度与量子产率,原料和设备常见,实验条件友好[55]。但是,微波法能量消耗较大、成本较高,制备的碳量子点因结晶度欠佳影响性能稳定性,若反应参数控制不当易出现产物团聚现象[55]。 JIANG等[56]以柠檬酸为前驱体,运用微波法实现3.5 min内快速大规模合成碳量子点,产物尺寸在2.5-5.8 nm之间,通过离心、透析纯化后量子产量达42%,但在紫外照射6 h后稳定性降8.5%。ABD ELHALEEM等[55]以黑籽为碳源,运用微波法在6 min内快速制备碳量子点,产物尺寸较窄,在1.5-5 nm之间,但产量为仅12.1%且荧光强度较弱,通过离心、超滤浓缩、透析三步法纯化后量子产率提升至38.7%,荧光强度增强3.2倍。MA等[57]以废弃咖啡渣为原料,运用微波法在4 h内合成了一种尺寸分布在1.5-4.5 nm的碳量子点,通过离心、透析、冷冻干燥三步法纯化将产物荧光强度增强12.3倍、量子产率也从3.1%提升至38.1%。 2.2.6 模板法 模板法是利用具有特定的多孔结材料(如硅胶球、介孔氧化硅微球、沸石等)为模板,引导碳源进入其表面的微孔中,再通过碳化、缩聚等反应后形成碳纳米聚合物,通过酸碱刻蚀等方法去除模板,得到碳量子点[58]。 模板法中因所用模板孔径大小均一,所得碳量子点粒径分布均匀且纯度较高,可通过选择不同孔径的模板材料精确控制所产碳量子点的尺寸,并且所得产物产率较高[59]。但该方法中模板的去除步骤较为繁琐,操作不当极易影响产物的纯度和荧光性能,需进一步纯化提升产物荧光强度;并且模板材料的制备和去除过程可能涉及昂贵的试剂(如氢氟酸或强碱),增加了生产成本和操作中的危险性,不利于规模化生产[60]。 2009年,LIU等[17]第一次提出采用模板法合成碳量子点,他们利用二氧化硅球为模板,以可溶性酚醛树脂为碳源,经高温处理并用氢氧化钠溶液除去硅载体后,再通过酸处理和表面钝化得到尺寸为1.5-2.5 nm、量子产率为14.7%的碳量子点,经过离心、透析、冷冻干燥三步纯化将荧光强度增强4.5倍、终量子产率提升至42%。YANG等[61]利用有机模板六氧化硅,在300 ℃、真空条件下去除模板提高了操作的安全性和绿色性,制备出尺寸大小为2-5 nm的碳量子点,通过洗涤干燥进行纯化,使无荧光性能的碳量子点发射荧光。LI等[62]以二氧化硅球为模板,制备尺寸为2-6 nm、量子产率为22%的碳量子点,经过氢氟酸去除模板,再通过离心、透析、冷冻干燥三步法纯化,使得产物的荧光强度增强3倍。 2.2.7 小结 制备碳量子点的方法繁多,每一种方法制备出的产物都需要通过纯化来提高部分性能,关于每种方法的比较和纯化后的效果见表2。由于碳量子点制备方法的多样性和灵活性,需根据应用场景选择合适的方法,同时优化条件以克服现有方法的局限。碳量子点制备技术的关键在于平衡效率、成本和产品质量,未来应聚焦于开发绿色高效的复合制备技术,提升量子产率与可控性;拓展生物质前驱体(如农业废弃物)的应用,降低成本并契合可持续发展;加强制备过程与产物性能的构效关系研究,建立标准化生产流程。"
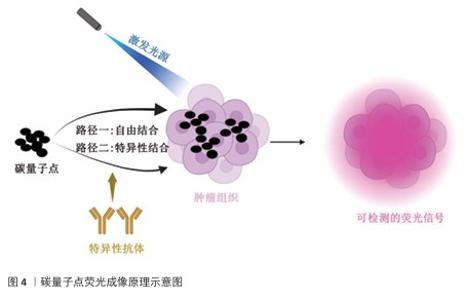
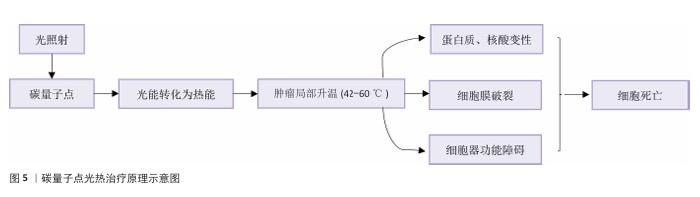
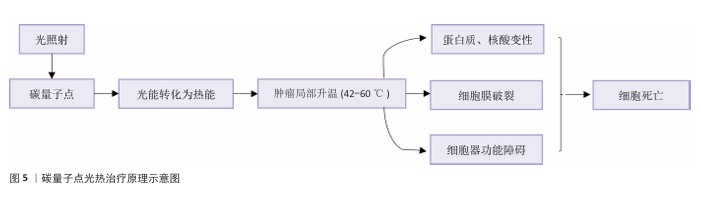
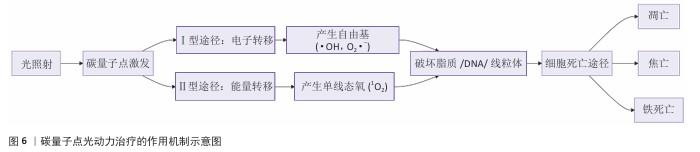
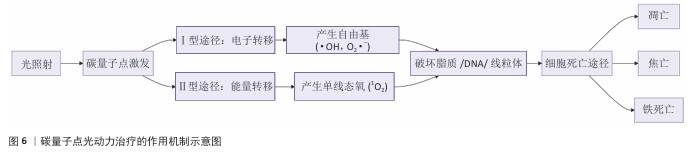
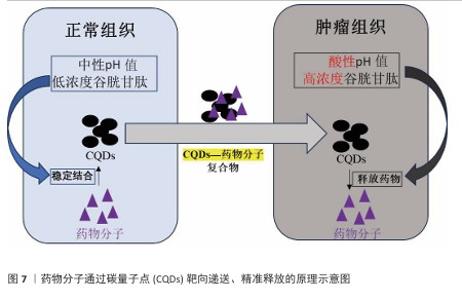
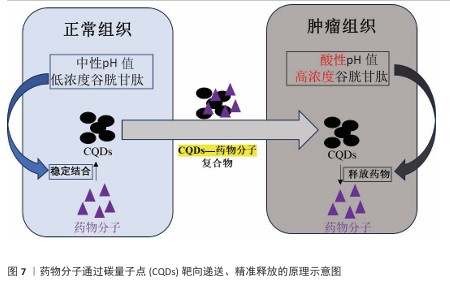
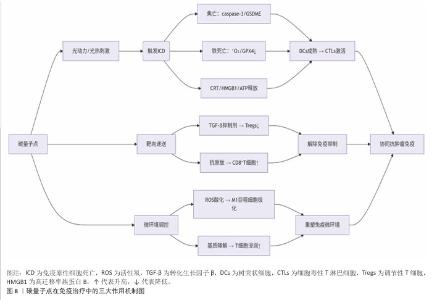
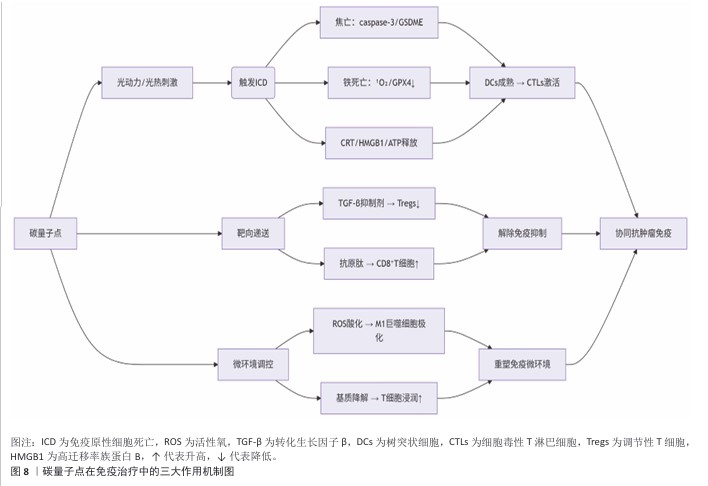
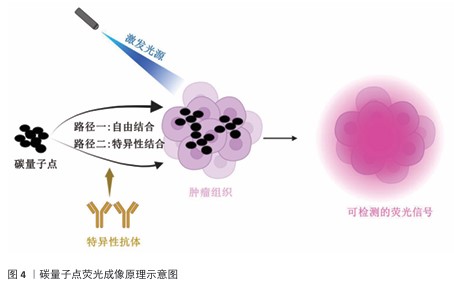
2.3 碳量子点在肿瘤诊疗中的主要应用方式 近年来,碳量子点凭借着优异的荧光特性、生物相容性和低毒性,在医学的领域展现出巨大潜力。碳量子点在肿瘤诊疗中的应用主要有荧光成像、光热治疗、光动力治疗、药物载体、免疫治疗等几种方式[63-64]。 2.3.1 荧光成像 荧光成像是碳量子点在肿瘤诊疗中的一项重要应用[65-66]。利用碳量子点的荧光特性,将它作为荧光探针标记肿瘤细胞或组织,实现对肿瘤的早期诊断和实时监测[67],或者通过碳量子点与肿瘤特异性抗体或配体结合可实现对肿瘤的靶向成像,用于肿瘤早期诊断[68],示意图见图4。 ALARFAJ等[18]合成的一种碳量子点携带CA19-9的特异性抗体,以检测胰腺癌的肿瘤标志物CA19-9,由于碳量子点的荧光特异性,在小鼠体内与CA19-9特异性结合以后能引发荧光信号的变化,通过检测荧光信号强弱便能实现对CA19-9的定量分析。HUANG等[20]合成了一种具有稳定荧光的碳量子点,将它与肝癌细胞Smmc-7721共同孵育,发现肝癌细胞Smmc-7721能将碳量子点有效摄取,在随后的小鼠实验中发现该碳量子点在3 h后出现在小鼠肝癌细胞中,在随后12,24 h的检测中几乎完全稳定富集在肝癌细胞中,检测到强烈的荧光信号,体现了碳量子点的特异性和低毒性。 2.3.2 光热治疗 当碳量子点受到特定波长的光照射时,它表面或内部的电子会从基态跃迁到激发态,随后电子通过非辐射复合将吸收的光能转换为热能,基于此特性,碳量子点可作为光热转换剂[69]。在光热治疗中,碳量子点作为光热剂将吸收的光能转化为热能,使局部温度升高(42-60 ℃)[70],由于肿瘤细胞对热的耐受性较差,蛋白质、核酸等生物大分子会发生变性、损伤,导致细胞膜破裂、细胞器功能障碍,最终导致死亡[71],见图5。由于碳量子点可进行表面修饰,使它具有肿瘤组织靶向能力,特异性地聚集在肿瘤组织中,实现对肿瘤的精准光热治疗,最大限度地减少对正常组织的损伤[72]。 BAO等[19]将合成的碳量子点与宫颈癌HeLa细胞共同孵育,发现碳量子点不影响HeLa细胞的活性,表现出低细胞毒性;将碳量子点注入肿瘤小鼠体内,在655 nm的激光照射下展现出59%的光热转换效率,肿瘤部位温度明显升高且肿瘤生长受到显著抑制,检测主要器官(心、肝、脾、肺和肾)均代谢良好,证明碳量子点的低毒性。LI等[73]制备的碳量子点能在808 nm激光照射下达到55.4%的光热转换率,以较低质量浓度(45 μg/mL)抑制小鼠体内的肿瘤。 2.3.3 光动力治疗 碳量子点能与光敏剂(能被光激活的分子或材料)结合,在特定波长的光照射下,光敏剂可通过电子转移或能量转移的方式产生自由基或单线态氧,进而产生活性氧簇[74],这些活性氧具有强氧化性,可以破坏肿瘤细胞的细胞膜、线粒体、DNA等生物大分子,导致细胞凋亡、焦亡或铁死亡。光动力治疗机制路线如图6所示[23,75-76]。 ZHU等[21]设计出携带有透明质酸、光敏剂Ce6的碳量子点,透明质酸的加入能增强了HeLa细胞中的Ⅰ型跨膜糖蛋白(CD44,增殖、分化、迁移、血管生成的关键蛋白)对碳量子点的摄取,Ce6的加入提高了活性氧产量,协同达到特异性抑制肿瘤细胞生长、促进凋亡的目的。WANG等[77]设计出单原子锰锚定的碳量子点,将它注入到人肝癌细胞HepG2和人脐静脉内皮细胞中,在488 nm激光照射下,单原子锰的锚定显著增强了碳量子点的荧光性,能增强光热和光动力治疗效果。 2.3.4 药物递送 碳量子点凭借优异的水溶性和生物相容性可以作为药物载体,它表面富含多种官能团,能更好地与药物分子结合[78],再将抗肿瘤药物靶向递送到肿瘤部位,减少药物在正常组织中的分布,降低不良反应[79]。 基于肿瘤微环境与正常组织的差异(如酸性pH值、高浓度谷胱甘肽等),碳量子点可通过静电相互作用实现药物的精准控释[80],见图7。 PAN等[22]将抗肿瘤药物奥沙利铂与碳量子点结合形成CQDs-Oxa缀合物,注入到携带肿瘤的小鼠体内后发现,CQDs-Oxa缀合物显著抑制了肿瘤生长,91.3%的肿瘤在6 d内得到治愈。RAEISPOUR等[81]制备了一种与抗肿瘤药物米托蒽醌结合的碳量子点形成CQD-MTX复合物,在pH=7.4的生理环境中,48 h内复合物中的米托蒽醌累积释放率为8%;但在pH=5.2的肿瘤微环境中,48 h内复合物中的米托蒽醌累积释放率增加到11%。 2.3.5 免疫治疗 碳量子点在免疫治疗中主要通过触发免疫原性细胞死亡、逆转免疫抑制微环境和协同免疫调节3种机制发挥作用[82-83]。 触发免疫原性细胞死亡:碳量子点通过光动力治疗产生活性氧,激活细胞焦亡通路,导致细胞膜穿孔并释放白细胞介素1β、白细胞介素18等促炎因子,募集树突状细胞并促进其成熟,进而激活细胞毒性T淋巴细胞,启动自身免疫治疗肿瘤[84]。CHENG等[23]构建的碳量子点能在白光下产生大量羟基自由基,通过活性氧-线粒体-caspase-3/GSDME通路轴诱导肿瘤细胞焦亡。传统碳量子点在合成后单线态氧的产率较低,一般小于10%[85-87],但加入重原子(如溴Br)后能通过重原子效应促进单线态氧的量子产生,从而诱发细胞膜脂质过氧化,消耗谷胱甘肽,抑制谷胱甘肽过氧化物酶4活性,最终诱导肿瘤细胞铁死亡[88]。LI等[25]在合成的碳量子点中引入溴原子,使材料在光照下单线态氧的产率提高3.2倍,诱导肿瘤细胞膜脂质过氧化,发生铁死亡。碳量子点介导光动力-光热联合治疗可促使肿瘤细胞钙网蛋白膜暴露、高迁移率族蛋白B1释放及ATP分泌,三者协同激活T细胞发生免疫应答[89]。GUO 等[90]在设计的碳量子点中加入铂,该复合物在细胞中释放能促进自由基的产生,引发氧化应激,通过钙网蛋白膜-高迁移率族蛋白B1-ATP机制促进树突状细胞的成熟和T细胞活化。 逆转免疫抑制:碳量子点在光照下产生的活性氧能酸化肿瘤微环境,增强巨噬细胞极化,增强吞噬能力并分泌肿瘤坏死因子α、白细胞介素12等促炎因子[91]。此外,碳量子点还可通过靶向递送转化生长因子β1抑制剂,以逆转肿瘤免疫抑制微环境[92]。转化生长因子β1通过激活其关键转录因子(Smad2/3)磷酸化,抑制调节性T细胞分化,解除细胞毒性T淋巴细胞的功能抑制[93-94]。XIONG 等[26]利用碳量子点的增强渗透与滞留效应富集转化生长因子β抑制剂,使肿瘤局部药物浓度提升3倍,有效阻断调节性T细胞分化并解除细胞毒性T淋巴细胞抑制。碳量子点还可通过降解肿瘤基质(如透明质酸)和增加血管通透性增强T细胞在肿瘤组织中的浸润[95],或通过携带肿瘤抗原肽靶向树突状细胞,激活CD8? T细胞(携带CD8抗原的T细胞),增强抗原的呈递[23]。ZHANG等[96] 将透明质酸酶修饰的碳量子点用于治疗胰腺癌,使肿瘤核心细胞毒性T淋巴细胞密度从8%提升至35%,胰腺癌肿瘤体积缩小74%。WANG等[24]设计的碳量子点负载树突状细胞膜囊泡,表面修饰肿瘤特异性抗原肽,能在体内直接结合CD8? T细胞并将其激活增殖3.5倍。 协同免疫调节:碳量子点可通过负载并靶向递送免疫调节剂(如Smad2/3抑制剂)促进树突状细胞成熟并增强T细胞活性[97]。LIU等[98]设计的双功能碳量子点能同时递送Smad2/3抑制剂和肿瘤抗原肽,使CD8? T细胞增殖3.5倍。碳量子点介导的光热治疗能释放肿瘤抗原,而光动力治疗产生的活性氧能激活干扰素刺激因子分泌,促进抗原交叉呈递[99]。MA等[100]设计的碳量子点能在特定波长光照下提高温度至42-45 ℃,诱导肿瘤释放抗原;同时又能通过光动力治疗产生活性氧,促进中性粒细胞富集,激活干扰素刺激因子信号通路。 综上所述,在的免疫治疗中,碳量子点能通过光动力效应诱导焦亡(caspase-3/GSDME通路)或铁死亡(溴掺杂增强单线态氧),激活树突状细胞与调节性T细胞,触发免疫原性细胞死亡;能酸化微环境促进巨噬细胞极化,靶向递送转化生长因子β抑制剂阻断调节性T细胞分化,降解基质增强T细胞浸润,实现免疫抑制;能通过负载抗原肽/Smad抑制剂直接激活CD8? T细胞,光疗-免疫联合激活干扰素刺激因子通路促进抗原交叉呈递,实现协同免疫调节。碳量子点免疫治疗的三大作用机制图见图8。 2.3.6 多模态治疗 碳量子点的多模态治疗是基于其独特的光学、电学和化学性质,整合肿瘤诊疗传统手段(如化疗、放疗)或上述新兴手段中的两种或多种治疗模式相整合,实现多模态治疗[101]。 LIU等[27]将合成的碳量子点与抗肿瘤药物阿霉素和近红外光热剂靛青绿共同封装在脂质体中,形成能通过叶酸靶向肿瘤并释放阿霉素的复合物,该复合物在808 nm激光照射下展现出高达47.14%的光热转换效率,能进行药物递送与光热治疗的协同治疗。LI等[102]合成在表面修饰多重α-羧基与氨基对并负载阿霉素的碳量子点复合物,使它能被大多数肿瘤中高表达的标志物大中性氨基酸转运酶1特异性识别并摄取,精准释放药物;该复合物能在700 nm激光照射下使肿瘤局部升温,通过光热疗法提高药物效率;该复合物还能促进肿瘤相关巨噬细胞的活化,激活抗肿瘤免疫,进行免疫治疗。 2.3.7 小结 碳量子点在肿瘤诊疗中的5种主要应用方式为荧光成像、光热治疗、光动力治疗、药物递送和免疫治疗。在荧光成像中,碳量子点作为荧光探针标记肿瘤细胞,实现早期诊断和实时监测。光热治疗利用碳量子点的光热转换效率,将光能转化为热能杀伤肿瘤细胞。在光动力治疗中,碳量子点产生活性氧破坏肿瘤细胞生物大分子。作为药物载体,碳量子点能靶向递送抗肿瘤药物至肿瘤部位,减少不良反应。在免疫治疗方面,碳量子点可调节免疫细胞和增强抗原呈递。在多模态治疗中,碳量子点整合多种治疗模式提高疗效并减少耐药性。碳量子点在肿瘤诊疗中的主要应用方式见表3。 "
| [1] BOLITHO A, LIU H. Epigenetic Regulation in Wilms Tumor. Biomedicines. 2025;13(7):1678. [2] MAO X, PENG S, LU Y, et al. Regulatory Functions of microRNAs in Cancer Stem Cells: Mechanism, Facts, and Perspectives. Cells. 2025;14(14):1073. [3] HARIBABU M, GURUVIAH V, YOGARAJAH P. Recent Advancements in Multimodal Medical Image Fusion Techniques for Better Diagnosis: An Overview. Curr Med Imaging. 2023;19(7):673-694. [4] ZAFAR A, KHATOON S, KHAN MJ, et al. Advancements and Limitations in Traditional Anti-Cancer Therapies: A Comprehensive Review of Surgery, Chemotherapy, Radiation Therapy, and Hormonal Therapy. Discov Oncol. 2025;16(1):607. [5] DEBELA DT, MUZAZU SG, HERARO KD, et al. New Approaches and Procedures for Cancer Treatment: Current Perspectives. SAGE Open Med. 2021;9:20503121211034366. [6] SULAIMAN JMA, ALTALBAWY FMA, KUMAR A, et al. Recent Advances in Carbon Nanomaterials: Removal, Photodegradation and Electrochemical Detection of Tetracycline, A Review. Inorg Chem Commun. 2025. doi:10.1016/j.inoche.2025. 113897. [7] ESMAEILI Y, TOISERKANI F, QAZANFARZADEH Z, et al. Unlocking the potential of green-engineered carbon quantum dots for sustainable packaging biomedical applications and water purification. Adv Colloid Interface Sci. 2025;338:103414. [8] KONG J, WEI Y, ZHOU F, et al. Carbon Quantum Dots: Properties, Preparation, and Applications. Molecules. 2024;29(9):2002. [9] AZAM N, NAJABAT ALI M, JAVAID KHAN T. Carbon Quantum Dots for Biomedical Applications: Review and Analysis. Front Mater. 2021;8:700403. [10] ZHANG J, ZHANG H, JIANG J, et al. Doxorubicin-Loaded Carbon Dots Lipid-Coated Calcium Phosphate Nanoparticles for Visual Targeted Delivery and Therapy of Tumor. Int J Nanomedicine. 2020;15:433-444. [11] LIU J, LI Q, TU H, et al. Bismuth/Manganese-Doped Carbon Dots in Calcium Phosphate Matrix: Dynamic Conformational Nanoparticles Enhancing Tumor Accumulation, Deep Penetration, and Radiosensitivity. Chem Eng J. 2024;487:150621. [12] DENG K, ZHANG L, WU M, et al. A functional carbon dots induce ferroptosis by suppressing PLPP4 activity to inhibit glioblastoma growth. Chem Eng J. 2023;472:144876. [13] WANG X, LU WM, JIA F, et al. Strategies to Improve Antitumor Drug Delivery by Increasing EPR Effect. Chin Pharm J. 2023;58(6):475-483. [14] GAO M, ZHANG S, ZHANG Z, et al. Comparison of Toxic Effects and Underlying Mechanisms of Carbon Quantum Dots and CdSe Quantum Dots on Chromochloris zofingiensis from the Chemical Composition Perspective. Chemosphere. 2024;363:142911. [15] XU XY, RAY R, LI Y, et al. Electrophoretic analysis and purification of fluorescent single-walled carbon nanotube fragments. J Am Chem Soc. 2004;126(40):12736-12737. [16] ZHOU JG, BOOKER C, LI RY, et al. An Electrochemical Avenue to Blue Luminescent Nanocrystals from Multiwalled Carbon Nanotubes (MWCNTs). J Am Chem Soc. 2007;129(4):744-745. [17] LIU RL, WU DQ, LIU SH, et al. An Aqueous Route to Multicolor Photoluminescent Carbon Dots Using Silica Spheres as Carriers. Angew Chem Int Ed. 2009;48(25):4598-4601. [18] ALARFAJ NA, EL-TOHAMY MF, ORABY HF. CA 19-9 Pancreatic Tumor Marker Fluorescence Immunosensing Detection via Immobilized Carbon Quantum Dots Conjugated Gold Nanocomposite. Int J Mol Sci. 2018;19(11):1162. [19] BAO X, YUAN Y, CHEN J, et al. In Vivo Theranostics with Near-Infrared-Emitting Carbon Dots—Highly Efficient Photothermal Therapy Based on Passive Targeting After Intravenous Administration. Light Sci Appl. 2018;7:91. [20] HUANG C, DONG H, SU Y, et al. Synthesis of Carbon Quantum Dot Nanoparticles Derived from Byproducts in Bio-Refinery Process for Cell Imaging and In Vivo Bioimaging. Nanomaterials. 2019;9(3):387. [21] ZHU J, WANG Y, HE Y, et al. Hyaluronic Acid Mediated Biomineralization of Multifunctional Ceria Nanocomposites as ROS Scavengers and Tumor Photodynamic Therapy Agents. J Mater Chem B. 2019;7(37):5814-5824. [22] PAN Q, XIE P, LI M, et al. Integrating Oxaliplatin with Highly Luminescent Carbon Dots: An Unprecedented Theranostic Agent for Personalized Medicine. Hepatology. 2023;81(2):774-790. [23] CHENG Q, CHEN H, ZHANG Y, et al. Photocatalytic Carbon Dots-Triggered Pyroptosis for Whole Cancer Cell Vaccines. Adv Mater. 2024;36(28): e2408685. [24] WANG T, HAN M, HAN Y, et al. Antigen Self-Presented Personalized Nanovaccines Boost the Immunotherapy of Highly Invasive and Metastatic Tumors. ACS Nano. 2024;18(8):6333-6347. [25] LI J, XUE X, CHEN T, et al. Heavy-Atom-Effect Enhanced Singlet Oxygen Generation of Carbon Dots for Lipid-Targeting Tumor Photodynamic Therapy Inducing Ferroptosis. Biomaterials. 2025; 325:124495. [26] XIONG Y, LI Y, CHEN L, et al. Enhancing the Therapeutic Efficacy of Gemcitabine in Bladder Cancer Through TGF-β1 Inhibition and Pluronic F-127-Based Microsphere Delivery. J Biol Eng. 2025;19(1):62. [27] LIU C, CHANG Q, FAN X, et al. Rational Construction of CQDs-Based Targeted Multifunctional Nanoplatform for Synergistic Chemo-Photothermal Tumor Therapy. J Colloid Interface Sci. 2025;677(Pt B):79-90. [28] YAO B, HUANG H, LIU Y, et al. Carbon Dots: A Small Conundrum. Trends Chem. 2019;1(2):235-246. [29] SHESHMANI S, MARDALI M, SHOKROLLAHZADEH S, et al. Synthesis, Optical, and Photocatalytic Properties of Cellulose-Derived Carbon Quantum Dots. Sci Rep. 2025;15(1):19027. [30] LI Y, WANG Q, ZHANG L, et al. Controllable Synthesis of Graphene Quantum Dots via Arc-Discharge Plasma: The Role of Carbon Precursor Crystallinity. Adv Mater Interfaces. 2024;11:2300912. [31] SHARMA AK, KUAMRI N, CHAUHAN P, et al. Comprehensive Insights into Carbon Quantum Dots: Synthesis Strategies and Multidomain Applications. J Fluoresc. 2025. doi:10.1007/s10895-025-04432-6. [32] MAHTO B, MAHANTY B, HAIT S, et al. A Review of Coal-Based Carbon and Graphene Quantum Dots: Synthesis, Properties, and Applications. Mater Sci Eng B. 2024;304:117386. [33] DU BH, ZHANG SH, GE Q, et al. Study on flow field characteristics of inert gas-air hybrid arc. J Exp Fluid Mech. 2022;36(5):69-75. [34] YANG ZX, DING LC, YANG GH, et al. Electrochemical Sulfonylation/Cyclization of N-Alkenylacrylamides with Sodium Sulfinates or Sulfonyl Hydrazides. J Org Chem. 2024;89(15):10660-10677. [35] DOS SANTOS AJ, SHEN H, LANZA MRV, et al. Electrochemical oxidation of surfactants as an essential step to enable greywater reuse. Environ Technol Innov. 2024;34:103563. [36] SEAD FF, JADEJA Y, KUMAR A, et al. Carbon Quantum Dots for Sustainable Energy: Enhancing Electrocatalytic Reactions Through Structural Innovation. Nanoscale Adv. 2025;7(13):3961-3998. [37] YAT YD, FOO HCY, TAN IS, et al. Synthesis of Carbon Quantum Dots via Electrochemically-Induced Carbon Dioxide Nanobubbles Exfoliation of Graphite for Heavy Metal Detection in Wastewater. J Environ Chem Eng. 2024;12(3):112715. [38] YANG GX, WANG Y, LI Y, et al. Synthesis of Vitamin B1-Stabilized Gold Nanoclusters with High Quantum Yields for Application as Sensors. ACS Appl Nano Mater. 2022;5(11):17234-17242. [39] HAN J, BAI X, XU X, et al. Advances and Challenges in the Electrochemical Reduction of Carbon Dioxide. Chem Sci. 2024;15(22):7870-7907. [40] YU T, WANG D, LIU M, et al. Exploiting Carbon Quantum Dots Synthesized by Electrochemical Exfoliation for Flexible Resistance Switching. ACS Mater Lett. 2024;6(3):793-800. [41] HIRANO T, KIKUCHI K, URANO Y, et al. Highly Zinc-Selective Fluorescent Sensor Molecules Suitable for Biological Applications. J Am Chem Soc. 2000; 122(49):12399-12400. [42] CORTES FRU, FALOMIR E, LANCIS J, et al. Pulsed laser fragmentation synthesis of carbon quantum dots (CQDs) as fluorescent probes in non-enzymatic glucose detection. Appl Surf Sci. 2024;665:160326. [43] KIM S, PARK J, LEE H. Pulsed Laser Fragmentation Synthesis of Carbon Quantum Dots: Thermal Gradient Effects on Size Distribution and Defect Formation. ACS Appl Nano Mater. 2024;7: 8890-8901. [44] KANG S, KIM KM, JUNG K, et al. Graphene Oxide Quantum Dots Derived from Coal for Bioimaging: Facile and Green Approach. Sci Rep. 2019;9(1):4101. [45] TORRISI L, SILIPIGNI L, TORRISI A, et al. Luminescence in Laser-Generated Functionalized Carbon Dots. Opt Laser Technol. 2024;177:111089. [46] NAZIBUDIN NA, ZAINUDDIN MF, ABDULLAH CAC. Hydrothermal Synthesis of Carbon Quantum Dots: An Updated Review. J Adv Res Fluid Mech Therm Sci. 2023;101(1):192-206. [47] CUI T, FAN Y, LIU Y, et al. Synthesizing Carbon Quantum Dots via Hydrothermal Reaction to Produce Efficient Antibacterial and Antibiofilm Nanomaterials. Foods. 2023;13(1):58. [48] MA X, LI J. Preparation of Multicolor Carbon Quantum Dots by Hydrothermal Method and Their Functionalization Applications. J Lumin. 2024;266:120296. [49] SAHU S, BEHERA B, MAITI TK, et al. Simple One-Step Synthesis of Highly Luminescent Carbon Dots from Orange Juice: Application as Excellent Bio-Imaging Agents. Chem Commun (Camb). 2012;48(70):8835-8837. [50] ZHENG XT, TAN YN. Development of Blood-Cell-Selective Fluorescent Biodots for Lysis-Free Leukocyte Imaging and Differential Counting in Whole Blood. Small. 2020;16(12):e1903328. [51] ZHANG M, WANG H, WANG B, et al. Maltase Decorated by Chiral Carbon Dots with Inhibited Enzyme Activity for Glucose Level Control. Small. 2019;15(48):e1901512. [52] WU J, CHEN T, GE S, et al. Synthesis and Applications of Carbon Quantum Dots Derived from Biomass Waste: A Review. Environ Chem Lett. 2023;21:3393-3424. [53] LI L, CHEN W, HU X, et al. Coupled Poly(ethylenimine) Coreactant to Enhance Electrochemiluminescence of Polymer Dots for Array Imaging of Protein Biomarkers. Anal Chem. 2024;96(10):4308-4313. [54] LIU C, ZHANG P, ZHAI X, et al. Nano-Carrier for Gene Delivery and Bioimaging Based on Carbon Dots with PEI-Passivation Enhanced Fluorescence. Biomaterials. 2012;33(13):3604-3613. [55] ABD ELHALEEM SM, BELAL F, EL-SHABRAWY Y, et al. Quality by Design-Aided Acid-Free Synthesis of Self P, N, S-Doped Black Seed-Derived Carbon Quantum Dots for Application as a Nanosensor for Eltrombopag Environmental and Bioanalysis and Pharmacokinetic Assay. Spectrochim Acta A Mol Biomol Spectrosc. 2024;319:124495. [56] JIANG XW, YE J, XU YT, et al. Nitrogen Doped Carbon Quantum Dots by Microwave Reaction Production Method and Optical Property. Chin J Lumin. 2018;39(8):1075. [57] MA CF, ZHOU YB, YAN W, et al. Green Synthesis of Nitrogen-Doped Carbon Dots from Coffee Waste for Sensitive Detection of Fe³⁺ and Cell Imaging. ACS Sustain Chem Eng. 2022;10(25):8162-8171. [58] WEI Z, YANFEI Z, JIAO W. Mesoporous Carbon Materials: Synthesis Methods, Properties, and Advanced Applications. Front Mater. 2025;12: 1548671. [59] ZHANG J, ABBASI F, CLAVERIE J. An Efficient Templating Approach for the Synthesis of Redispersible Size-Controllable Carbon Quantum Dots from Graphitic Polymeric Micelles. Chemistry. 2015;21(43):15142-15147. [60] SARASWAT SK, MUSTAFA MA, GHADIR GK, et al. Carbon Quantum Dots: A Comprehensive Review of Green Synthesis, Characterization and Investigation Their Applications in Bioimaging. Inorg Chem Commun. 2024;162:112279. [61] YANG P, ZHANG ZW, ZOU GD, et al. Template Thermolysis to Create a Carbon Dots-Embedded Mesoporous Titanium-Oxo Sulfate Framework for Visible-Light Photocatalytic Applications. Inorg Chem. 2020;59(3):2062-2069. [62] LI Y, ZHANG L, WANG H, et al. Carbon Quantum Dots: Synthesis, Properties, and Emerging Biomedical Applications in Cancer Therapy and Imaging. Adv Healthc Mater. 2022;11(18): e2200567. [63] ZONG J, ZHU Y, YANG X, et al. Synthesis of Photoluminescent Carbogenic Dots Using Mesoporous Silica Spheres as Nanoreactors. Chem Commun(Camb). 2011;47(2):764-766. [64] ZHAO J, LIU S, CHEN M, et al. Carbon Dots for Cancer Therapy: A Comprehensive Review of Recent Advances. Biomaterials Science. 2023; 11(15):5021-5045. [65] SALVE PL, BHINGE SD, BHUTKAR MA. Carbon Dots an Integrative Nanostructure for Fluorescent Bio-imaging, Targeted Delivery of Medication and Phototherapy in Malignancy: A Review. Nanoscale Research Letters. 2024;14(2):1-18. [66] BARVE K, SINGH U, YADAV P, et al. Carbon-Based Designer and Programmable Fluorescent Quantum Dots for Targeted Biological and Biomedical Applications. Mater Chem Front. 2023;7(9): 1781-1802. [67] NAIK K, CHAUDHARY S, YE L, et al. A Strategic Review on Carbon Quantum Dots for Cancer-Diagnostics and Treatment. Front Bioeng Biotechnol. 2022;10:882100. [68] VARGHESE S, ABRAHAM MK, INDONGO G, et al. In Vivo Imaging of Pancreatic Cancer in BALB/c Mice Using Erlotinib-Conjugated Carbon Quantum Dots Derived from 1,4,5,8-Tetraminoanthraquinone. Bioconjug Chem. 2025;36(6):1329-1339. [69] WANG Y, WU H, GUO Y, et al. Carbon Dot-Based Nanoparticles: A Promising Therapeutic Approach for Glioblastoma. Int J Nanomedicine. 2025;20:7061-7092. [70] ZHANG H, LIU Y, QU SN. Recent Advances in Photo-Responsive Carbon Dots for Tumor Therapy. Responsive Mater. 2024;2(2):e20240012. [71] BHATIA P, CHAIRA T, GUPTA LK. Therapeutic Applications of Carbon Quantum Dots (CQDs): A Review. J Inorg Organomet Polym. 2025;35: 3243-3259. [72] CHEN BB, LIUU ML, HUANG CZ.Recent Advances of Carbon Dots in Imaging-Guided Theranostics. Trends Anal Chem. 2021;134:116116. [73] LI D, HUANG K, SHE J, et al. Two-Photon Fluorescence-Guided Precise Photothermal Therapy Located in a Single Cancer Cell Utilizing Bifunctional N-Doped Carbon Quantum Dots. J Colloid Interface Sci. 2024;662:719-726. [74] JOVANOVIĆ S, MARKOVIĆ Z, BUDIMIR M, et al. Lights and Dots Toward Therapy-Carbon-Based Quantum Dots as New Agents for Photodynamic Therapy. Pharmaceutics. 2023;15(4):1170. [75] CHEN J, YIN S, YANG F, et al. A Single-Molecule Graphene Quantum Dot: A Novel Efficient Photosensitizer for Photodynamic Cancer Therapy. Chem Sci. 2025;16(30):13923-13934. [76] LI J, ZHANG Q, WANG X, et al. Creating Single Atomic Coordination for Hypoxia-Resistant Pyroptosis Nano-Inducer. Adv Mater. 2025;37(9): e2414697. [77] WANG S, MA M, LIANG Q, et al. Single-Atom Manganese Anchored on Carbon Dots for Promoting Mitochondrial Targeting and Photodynamic Effect in Cancer Treatment. ACS Appl Nano Mater. 2022;5(5):6679-6690. [78] KONDI SOUMYA K, NAMDEV MORE N, CHOPPADANDI M, et al. A Comprehensive Review on Carbon Quantum Dots as an Effective Photosensitizer and Drug Delivery System for Cancer Treatment. Biomed Technol. 2023;4:11-20. [79] SINGH H, DHAR D, DAS S, et al. Methotrexate-Loaded Manganese Nitrogen Dual-Doped Carbon Quantum Dots as Targeted Nano Drug-Delivery System for Potential Use in Cancer Theranostics. J Photochem Photobiol A. 2024;455(1):115692. [80] SAINI A, NANDI A, GUPTA GD, et al. Green Synthesis of Curcumin Loaded Carbon Dots as a Sustained Drug Delivery for Anticancer Therapy. Curr Pharm Des. 2025;31(28):2282-2289. [81] RAEISPOUR S, RAHMANDOUST M, KOUCHAKZADEH H. A Nanocarrier System Based on CQDs for Efficient Mitoxantrone Drug Delivery. Heliyon. 2024;10(11):e31674. [82] CAI H, ABBAS K, YANG Y, et al. The Application of Carbon Dots in Tumor Immunotherapy: Researches and Prospects. Appl Sci. 2023;13(3): 1678. [83] MIAO Y, WANG S, ZHANG B, et al. Carbon Dot-Based Nanomaterials: A Promising Future Nano-Platform for Targeting Tumor-Associated Macrophages. Front Immunol. 2023;14:1133238. [84] JIANG L, CAI H, QIN W, et al. Meticulously Designed Carbon Dots as Photo-Triggered RNA-Destroyer for Evoking Pyroptosis. Bioconjug Chem. 2023;34(8):1387-1397. [85] WANG S, MCCOY CP, LI P, et al. Carbon Dots in Photodynamic/Photothermal Antimicrobial Therapy. Nanomaterials. 2024;14(15):1250. [86] DING J, ZHOU P, SU B. Quantum Efficiency of Electrochemiluminescence Generation by Tris(2,2’‐bipyridine)ruthenium(II) and Tri‐n‐propylamine Revisited from a Kinetic Reaction Model. ChemElectroChem. 2022;9(12): e202200236. [87] GE J, CHEN X, YANG J, et al. Progress in Electrochemiluminescence of Nanoclusters: How to Improve the Quantum Yield of Nanoclusters. Analytical Methods. 2023;15(47):7787-7796. [88] YAO L, ZHAO MM, LUO QW, et al. Carbon Quantum Dots-Based Nanozyme from Coffee Induces Cancer Cell Ferroptosis to Activate Antitumor Immunity. ACS Nano. 2022;16(6):9228-9239. [89] TANG Y, BISOYI HK, CHEN XM, et al. Pyroptosis-Mediated Synergistic Photodynamic and Photothermal Immunotherapy Enabled by Tumor Membrane-Targeted Photosensitive Dimer. Adv Mater. 2023;35(23):e2300232. [90] GUO D, LEI H, RONG D, et al. Photocatalytic Pt(IV)-Coordinated Carbon Dots for Precision Tumor Therapy. Adv Sci. 2022;9(36):e2205106. [91] EFTEKHARIFAR M, HEIDARI R, MOHAGHEGH N, et al. Advances in Photoactivated Carbon-Based Nanostructured Materials for Targeted Cancer Therapy. Adv Drug Deliv Rev. 2025;222:115604. [92] ZHOU M, WANG J, PAN J, et al. Nanovesicles Loaded with a TGF-β Receptor 1 Inhibitor Overcome Immune Resistance to Potentiate Cancer Immunotherapy. Nat Commun. 2023; 14(1):3593. [93] YIN C, ZHANG C, WANG Y, et al. ALDOB/KAT2A Interactions Epigenetically Modulate TGF-β Expression and T Cell Functions in Hepatocellular Carcinogenesis. Hepatology. 2025;81(1):77-93. [94] ZHANG F, ZHENG Z, WANG L, et al. PKC-ζ Mediated Reduction of the Extracellular Vesicles-Associated TGF-β1 Overcomes Radiotherapy Resistance in Breast Cancer. Breast Cancer Res. 2023;25(1):38. [95] XIAO Z, TAN Y, CAI Y, et al. Nanodrug Removes Physical Barrier to Promote T-Cell Infiltration for Enhanced Cancer Immunotherapy. J Control Release. 2023; 356:360-372. [96] ZHANG L, WANG Y, DENG K, et al. Hyaluronidase-Conjugated Carbon Dots Degrade Extracellular Matrix to Potentiate T-Cell Infiltration in Pancreatic Cancer. ACS Nano. 2024;18(12):8912-8928. [97] LIU C, FENG Q, LIU Y, et al. Carbon Dots: Translating Versatile Physicochemistry into Multidisciplinary Application Frameworks. Nanoscale. 2025;17(31):17919-17946. [98] LIU Y, ZHANG R, WANG X, et al. Pluronic F-127/Carbon Dot Hybrid Micelles Co-Delivering TGF-β Inhibitor and Antigen Peptide Reverse Immunosuppression for Colorectal Cancer Immunotherapy. Nano Lett. 2025;25(3): 1120-1131. [99] LURI-REY C, TEIJEIRA Á, WCULEK SK, DE ANDREA C, et al. Cross-Priming in Cancer Immunology and Immunotherapy. Nat Rev Cancer. 2025;25(4): 249-273. [100] MA W, SUN R, TANG L, et al. Bioactivable STING Nanoagonists to Synergize NIR-II Mild Photothermal Therapy Primed Robust and Long-Term Anticancer Immunity. Adv Mater. 2023; 35(48):e2303149. [101] BHATTACHARYA T, PREETAM S, MUKHERJEE S, et al. Anticancer Activity of Quantum Size Carbon Dots: Opportunities and Challenges. Discov Nano. 2024;19(1):122. [102] LI S, SU W, WU H, et al. Targeted Tumour Theranostics in Mice via Carbon Quantum Dots Structurally Mimicking Large Amino Acids. Nat Biomed Eng. 2020;4(7):704-716. |
| [1] | Yang Xuetao, Zhu Menghan, Zhang Chenxi, Sun Yimin, Ye Ling. Applications and limitations of antioxidant nanomaterials in oral cavity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2044-2053. |
| [2] | Guo Yuchao, Ni Qianwei, Yin Chen, Jigeer·Saiyilihan, Gao Zhan . Quaternized chitosan hemostatic materials: synthesis, mechanism, and application [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2091-2100. |
| [3] | Liu Dawei, Cui Yingying, Wang Fanghui, Wang Zixuan, Chen Yuhan, Li Yourui, Zhang Ronghe. Epigallocatechin gallate-mediated bidirectional regulation of reactive oxygen species and its application in nanomaterials [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2101-2112. |
| [4] | Dilida·Bahetikelede, Zhou Xin, Wang Xinyi, Zeng Zhihan, Wang Liqiong, Hu Danrong. Research status and trends of nanotechnology in improving photodynamic therapy for hypoxic tumors [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6952-6960. |
| [5] | Feng Shuqi, Zhang Shiyong, Yao Keyi, Tang Yufei, Wang Kai, Zhou Xuemei, Xiang Lin. Application of photoresponsive nanomaterials in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(16): 3469-3475. |
| [6] | Feng Nan, Li Yunfeng. Antibacterial piezoelectric materials: no selective killing of bacteria and no bacterial resistance [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(10): 2105-2112. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||