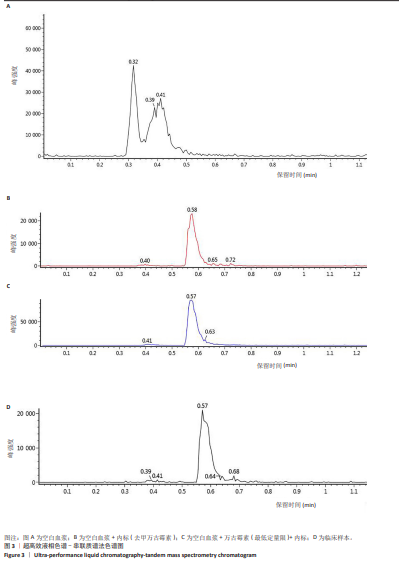
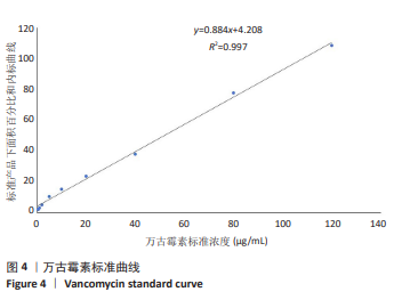
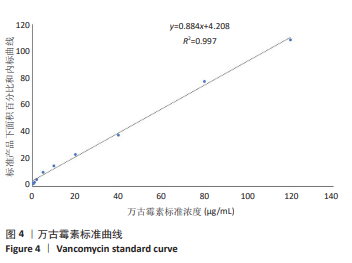
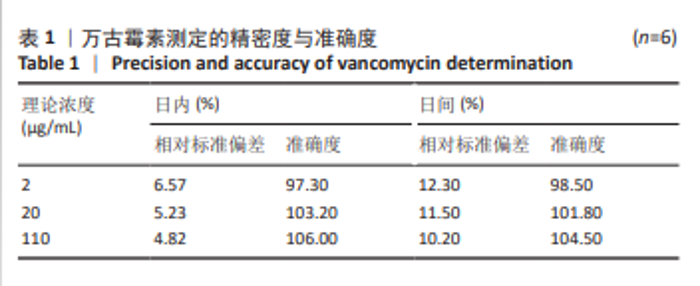
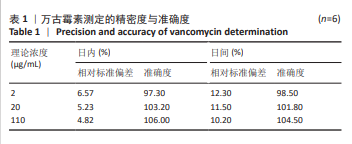
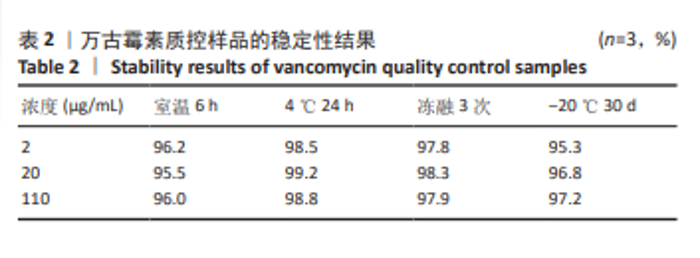
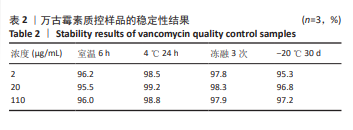
[1] 中国医药教育协会感染疾病专业委员会. 抗菌药物药代动力学/药效学理论临床应用专家共识[J]. 中华结核和呼吸杂志,2018,41(6): 409-446.
[2] 潘坤明, 李艳丽, 徐辰祺, 等. 1106例万古霉素血药浓度监测及影响因素分析[J]. 医药导报,2024,43(2):184-189.
[3] 刘东华, 全香花, 邢晓敏, 等. 骨科药物治疗临床路径管理模式的建立与实践[J]. 医药导报,2021,40(1):139-143.
[4] 范淑盼, 丁楠, 陈美琪, 等. 万古霉素治疗药物监测的热点与进展[J]. 海军军医大学学报,2024,45(2):135-144.
[5] 赵暄, 郑婷婷, 李雪梅, 等. 万古霉素不良反应报告分析[J]. 中国药物警戒,2024,21(7):811-815.
[6] 陈金成, 张吟, 林丽梅, 等. 高效液相色谱法测定人血浆中万古霉素浓度[J]. 中国当代医药,2022,29(25):45-48.
[7] REZK MR, BADR KA, ABDEL-NABY NS, et al. A novel validated UPLC-MS/MS method for quantification of favipiravir in human plasma: Application to a bioequivalence study. J Chromatogr B. 2021;35(7): e5098.
[8] 陈曜曜, 史道华, 刘光华, 等. OCT2、MDR1基因多态性与婴幼儿万古霉素血药浓度及临床疗效的相关性研究[J]. 中国临床药理学与治疗学,2018,23(2):154-158.
[9] 桂明珠,李静,谢晓恬,等.伏立康唑治疗儿童侵袭性真菌感染血药浓度与疗效相关性研究[J].药学实践杂志,2022,40(4):359-363.
[10] KELLUM JA, LAMEIRE N, ASPELIN P, et al. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int Suppl. 2012;2(1):1-138.
[11] 王李赛, 李寿林, 王东亮, 等. HPLC 法同时测定儿童患者血清中万古霉素, 咖啡因和氨茶碱浓度及其临床应用[J]. 药物分析杂志, 2025,45(1):92-98.
[12] LU W, PAN M, KE H, et al. An LC-MS/MS method for the simultaneous determination of 18 antibacterial drugs in human plasma and its application in therapeutic drug monitoring. Front Pharmacol. 2022;13: 1044234.
[13] SUN H, XING H, TIAN X, et al. UPLC‐MS/MS method for simultaneous determination of 14 antimicrobials in human plasma and cerebrospinal fluid: application to therapeutic drug monitoring. J Anal Methods Chem. 2022;2022:7048605.
[14] 李燕,陈礼治,王学彬,等.Smart Dose对万古霉素血药浓度的预测效能评价[J].药学实践杂志,2021,39(2):164-168+177.
[15] YANG H, XIONG X, LIU C, et al. Analysis of influencing factors and prediction models of vancomycin AUC0-24h in postoperative orthopedic patients. China Pharmacist. 2024;27(1):66-75.
[16] 段晶晶, 纪国文, 郭芷君, 等. HPLC-MS/MS 测定人血清中丙戊酸钠和万古霉素的浓度[J]. 药学实践与服务,2022,40(4):350-353, 378.
[17] 李京峰, 时正媛, 张梦洁, 等. 丙泊酚血药浓度检测方法建立及在淋巴水肿患者中的应用[J]. 中国药房,2024,35(4):476-480.
[18] LI Y, YUE X, PAN Z, et al. Development and validation of an LC‐MS/MS method for quantifying nine antimicrobials in human serum and its application to study the exposure of Chinese pregnant women to antimicrobials. J Clin Lab Anal. 2021;35(3):e23658.
[19] OYAERT M, PEERSMAN N, KIEFFER D, et al. Novel LC–MS/MS method for plasma vancomycin: Comparison with immunoassays and clinical impact. Clinica Chimica Acta. 2015;441:63-70.
[20] 韩颖, 魏国兰, 米楠, 等. UPLC-MS/MS法快速、灵敏、直接测定人血浆中的门冬胰岛素[J]. 药学学报,2021,56(9):2510-2517.
[21] RYBAK MJ, LE J, LODISE TP, et al. Therapeutic monitoring of vancomycin for serious methicillin-resistant Staphylococcus aureus infections: A revised consensus guideline and review by the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. Am J Health Syst Pharm. 2020; 77(11):835-864.
[22] 连霞, 林小凤, 李红娜, 等. 173例次万古霉素血药浓度监测与临床应用分析[J]. 中国医院药学杂志,2018,38(17):1823-1827.
[23] 张懿,杨中保,宋慧玲.某三甲医院2023年万古霉素血清药物浓度检测情况及合理用药分析[J].药物资讯,2025,14(1):58-64.
[24] 李燕, 王学彬, 陈礼治, 等. 万古霉素血药浓度与临床疗效及急性肾损伤的关系[J]. 第二军医大学学报,2021,42(3):281-286.
[25] 赵伟锋. 某院 223例骨科感染住院患者多重耐药菌的分布及耐药性分析[J]. 抗感染药学,2021,18(1):103-106.
[26] 占华松,陈跃平,章晓云.骨组织工程技术治疗感染性骨缺损:优势与问题[J].中国组织工程研究,2019,23(30):4848-4854.
[27] 盛长城, 谢娟, 张家兴, 等. 202例次万古霉素血药浓度监测与临床应用分析[J]. 药学与临床研究,2016,24(4):414-418.
[28] YE ZK, CHEN YL, CHEN K, et al. Therapeutic drug monitoring of vancomycin: a guideline of the Division of Therapeutic Drug Monitoring, Chinese Pharmacological Society. J Antimicrob Chemother. 2016;71(11):3020-3025.
[29] 唐冰颖, 伊茂礼, 高婷, 等. 利奈唑胺与万古霉素治疗肠球菌血流感染疗效和安全性的真实世界研究[J]. 药物流行病学杂志,2024, 33(7):770-777.
[30] JORGENSEN SCJ, MURRAY KP, LAGNF AM, et al. A multicenter evaluation of vancomycin-associated acute kidney injury in hospitalized patients with acute bacterial skin and skin structure infections. Infect Dis Ther. 2020;9(1):89-106.
[31] 袁世轩,吕子彦,杨勇,等.模型引导下的万古霉素血药浓度监测临床研究[J].中国临床药理学与治疗学,2024,29(12):1344-1352.
[32] 郭仙忠,林荣芳,林玮玮.基于群体药动学模型的万古霉素个体化给药软件研制及应用[J].中国临床药理学与治疗学,2021,26(1): 30-39.
[33] YALÇIN N, FLINT RB, VAN SCHAIK RHN, et al. The impact of pharmacogenetics on pharmacokinetics and pharmacodynamics in neonates and infants: a systematic review. Pharmgenomics Pers Med. 2022;15:675-696.
[34] KLAASSEN CD, ALEKSUNES LM. Xenobiotic, bile acid, and cholesterol transporters: function and regulation. Pharmacol Rev. 2010;62(1):1-96.
[35] 罗茜华,孟冬梅,周立成,等.基于TDM与基因检测指导1例患者应用他克莫司的案例分享[J].今日药学,2023,33(7):548-551.
[36] EHREN R, SCHIJVENS AM, HACKL A, et al. Therapeutic drug monitoring of mycophenolate mofetil in pediatric patients: novel techniques and current opinion. Expert Opin Drug Metab Toxicol. 2021;17(2):201-213.
[37] 徐文俊,陆康生,朱华,等.骨科术后患者万古霉素治疗药物监测分析[J].中国新药与临床杂志,2020,39(10):609-614.
[38] 范淑盼,丁楠,陈美琪,等.万古霉素治疗药物监测的热点与进展[J].海军军医大学学报,2024,45(2):135-144.
[39] 张明, 李娜, 王芳, 等. 2010-2015年北京医院外科病房万古霉素的使用情况分析[J]. 现代药物与临床,2016,31(9):1457-1462.
[40] ZHOU L, GAO Y, CAO W, et al. Retrospective analysis of relationships among the dose regimen, trough concentration, efficacy, and safety of teicoplanin in Chinese patients with moderate-severe Gram-positive infections. Infect Drug Resist. 2018;11:29-36. |