Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 4949-4964.doi: 10.12307/2026.224
Previous Articles Next Articles
Single-cell sequencing data identifies differentially expressed genes and immune cell subtypes in periodontitis patients
Qiu Xuedi1, 2, Guo Chao1, He Jiayue1, 2, Zhou Zheng1
- 1Department of Stomatology, The First Affiliated Hospital of Shihezi University, Shihezi 832000, Xinjiang Uyghur Autonomous Region, China; 2Medical College of Shihezi University, Shihezi 832000, Xinjiang Uyghur Autonomous Region, China
-
Received:2025-08-01Accepted:2025-11-09Online:2026-07-08Published:2026-02-14 -
Contact:Zhou Zheng, Chief physician, Professor, Master’s supervisor, Department of Stomatology, The First Affiliated Hospital of Shihezi University, Shihezi 832000, Xinjiang Uyghur Autonomous Region, China -
About author:Qiu Xuedi, MS, Department of Stomatology, The First Affiliated Hospital of Shihezi University, Shihezi 832000, Xinjiang Uyghur Autonomous Region, China; Medical College of Shihezi University, Shihezi 832000, Xinjiang Uyghur Autonomous Region, China -
Supported by:Xinjiang Production and Construction Corps Science and Technology Program, No. 2023ZD062 (to ZZ); Hospital-level Science and Technology Program of The First Affiliated Hospital of Shihezi University, No. ZP2024004 (to QXD)
CLC Number:
Cite this article
Qiu Xuedi, Guo Chao, He Jiayue, Zhou Zheng. Single-cell sequencing data identifies differentially expressed genes and immune cell subtypes in periodontitis patients[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4949-4964.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
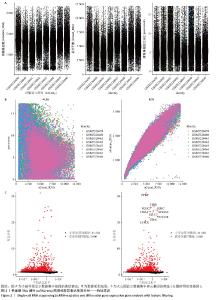
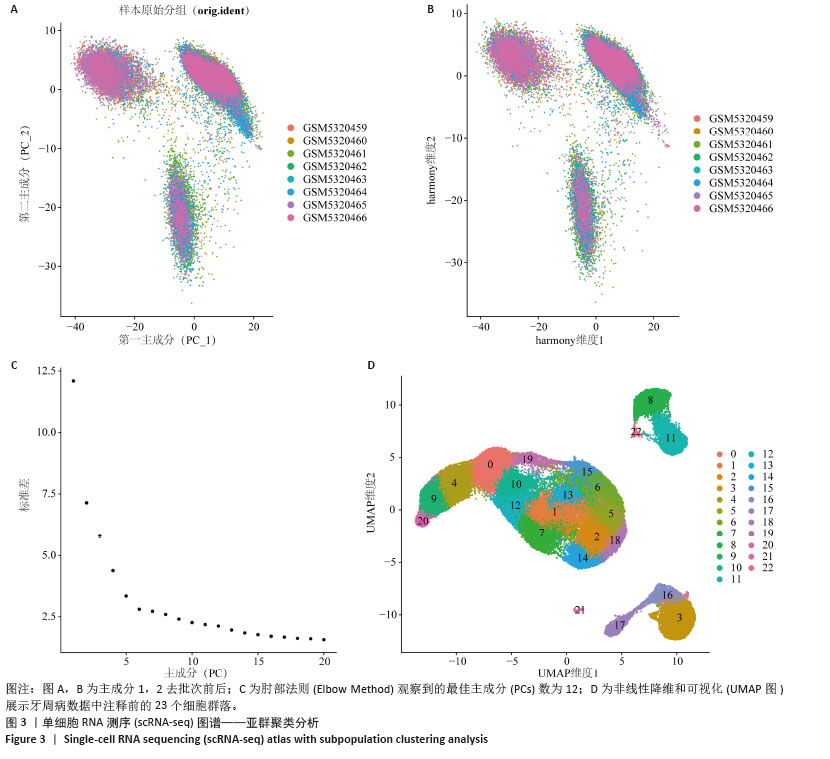
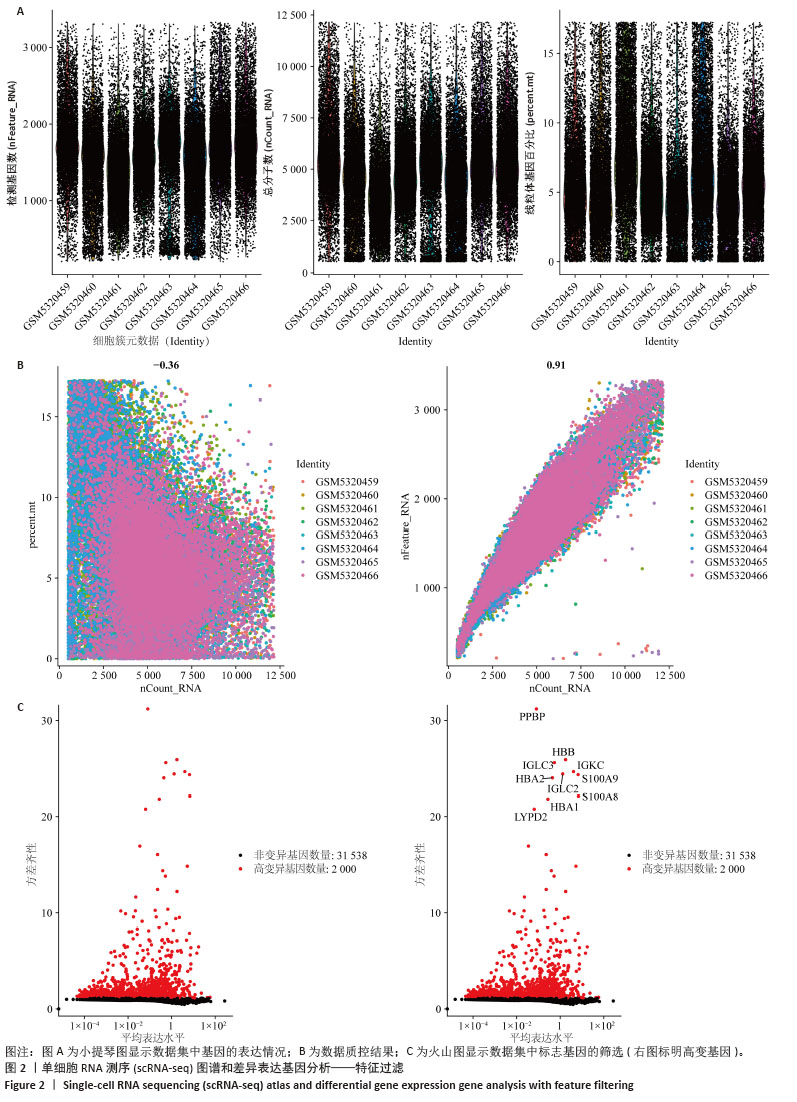
2.1 单细胞表达谱数据的预处理 8个牙周组织样本按以下标准过滤出低表达细胞:每个细胞中检测到的基因数量(200 < nFeature_RNA≤ 3 MAD);细胞内线粒体基因占比(percent.mt)≤3 MAD;每个细胞的唯一分子标识符(UMIs)总数(nCount_RNA)≤3 MAD,并排除双重细胞后,共纳入66 209个细胞。其中8个样本数据的每个细胞的唯一分子标识符总数与细胞内线粒体基因占比之间的相关系数为-0.36;每个细胞的唯一分子标识符总数与每个细胞中检测到的基因数量之间的相关系数为0.91,数据质控显示质量可。鉴定出了标准偏差最高的基因:PPBP、HBB、IGKC、IGLC2、IGLC3、HBA2、HBA1、S100A9、S100A8和LYPD2。见图2。 "
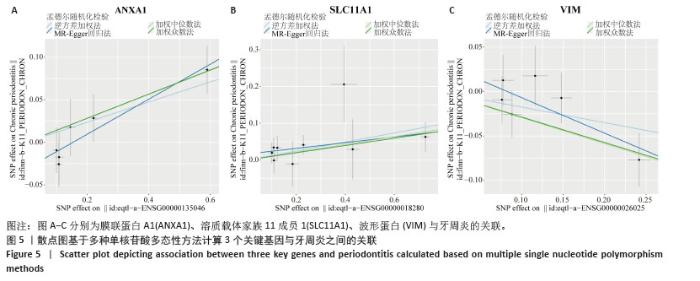
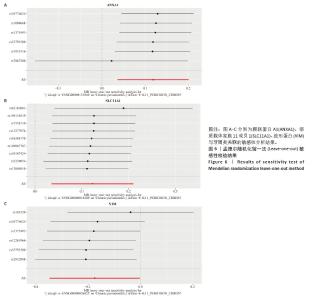
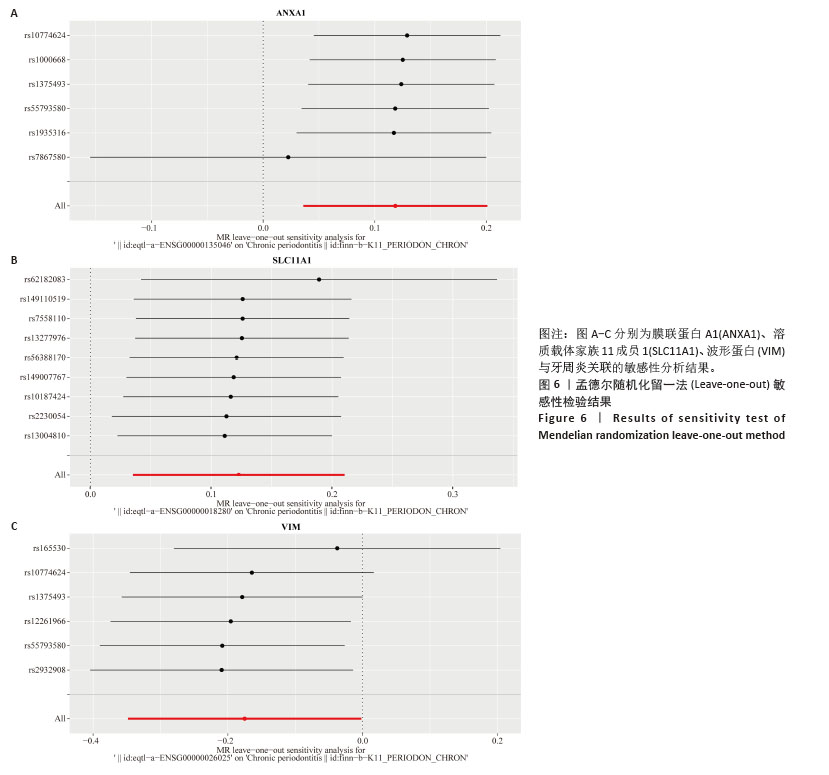
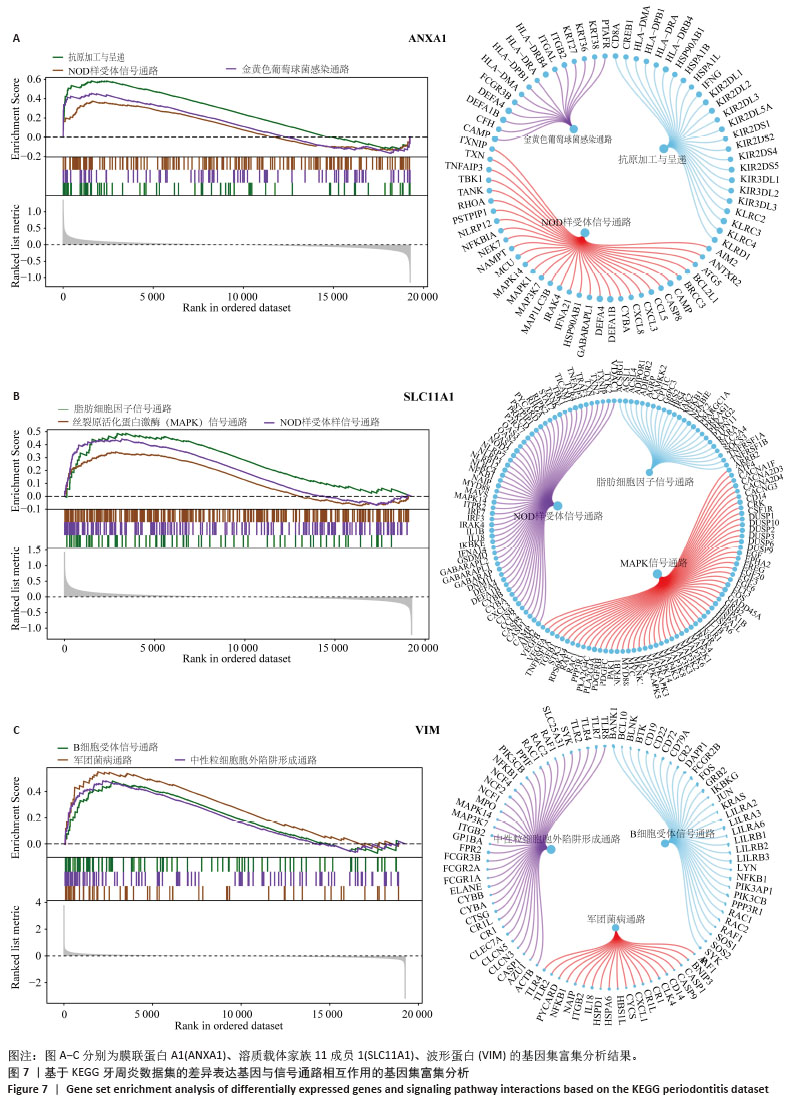
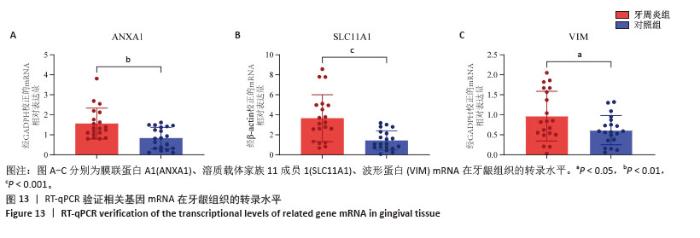
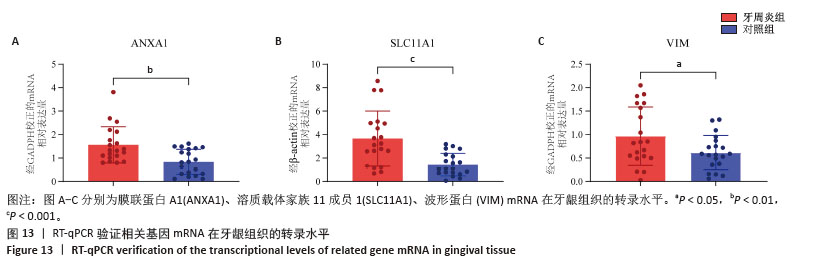
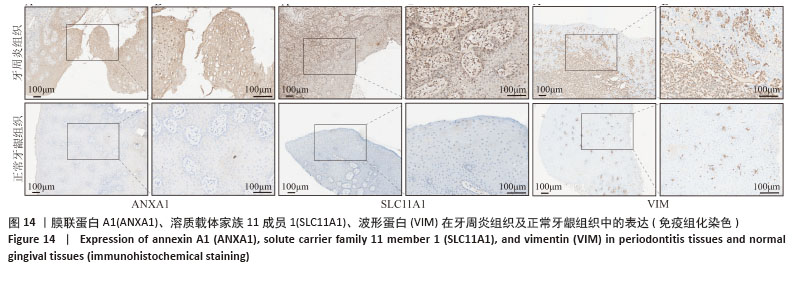
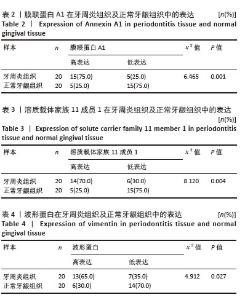
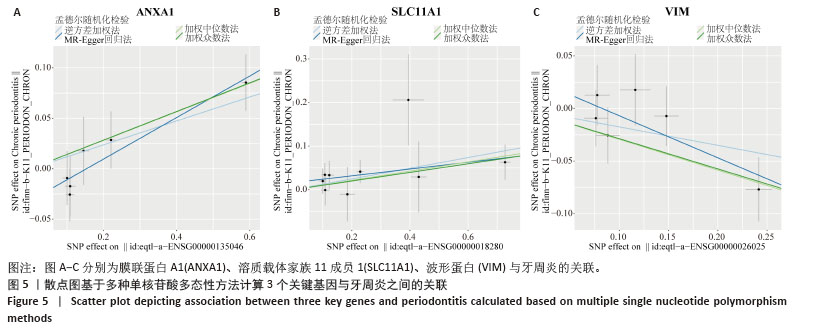
2.3 孟德尔随机化分析结果 逆方差加权法、MR-Egger、加权中位数和MR-presso 4种方法显示一致的效应估计,表明有可靠的关联。孟德尔随机化分析揭示出膜联蛋白A1和溶质载体家族11成员1与牙周炎有正向因果关系(图5A,B),波形蛋白与牙周炎有反向因果关系(图5C),使用逆方差加权法确定以上关联具有统计学意义(P < 0.05)并进行了统计显著性验证:膜联蛋白A1(OR=1.126;95%CI:1.037-1.222;P=0.005)、溶质载体家族11成员1(OR=1.131;95%CI:1.036-1.234;P=0.006)、波形蛋白(OR=0.839;95%CI:0.706-0.998;P=0.047),即膜联蛋白A1、溶质载体家族11成员1与牙周炎风险增加有关,波形蛋白可能与较低的牙周炎风险有关。敏感性分析结果显示,排除任意一个单核苷酸多态性后对整体误差条的影响并不明显,证实以上的因果关系是稳健的(图6A-C)。 "
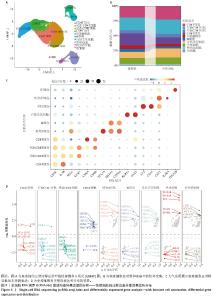
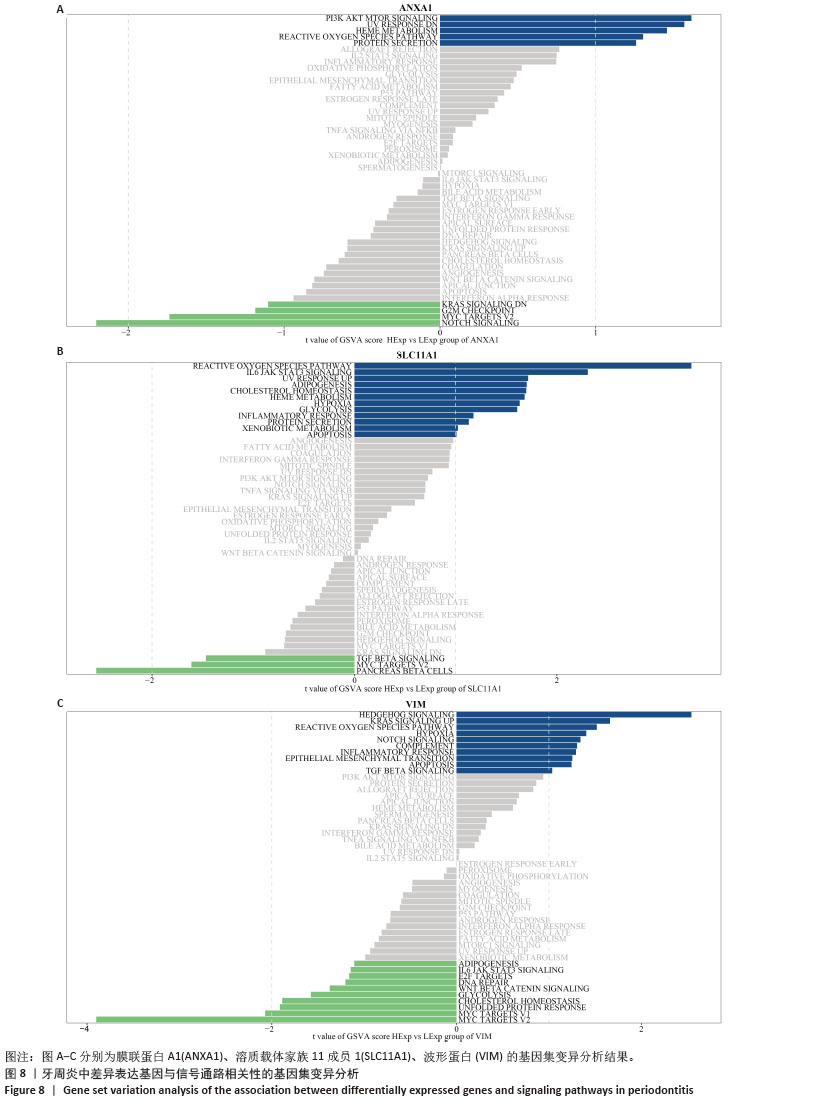
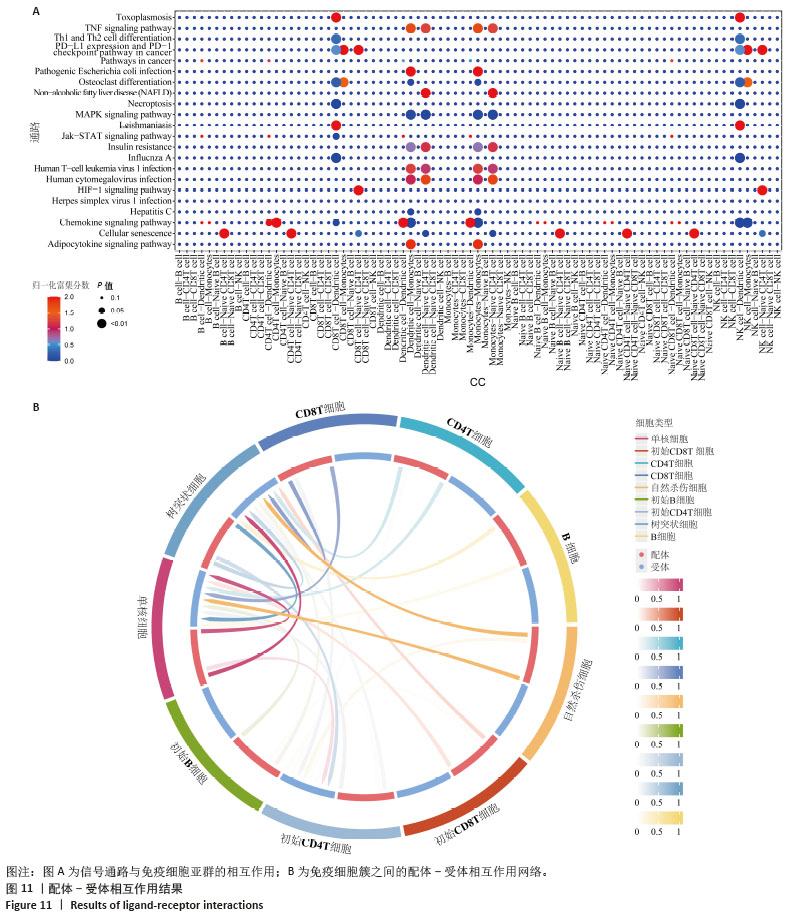
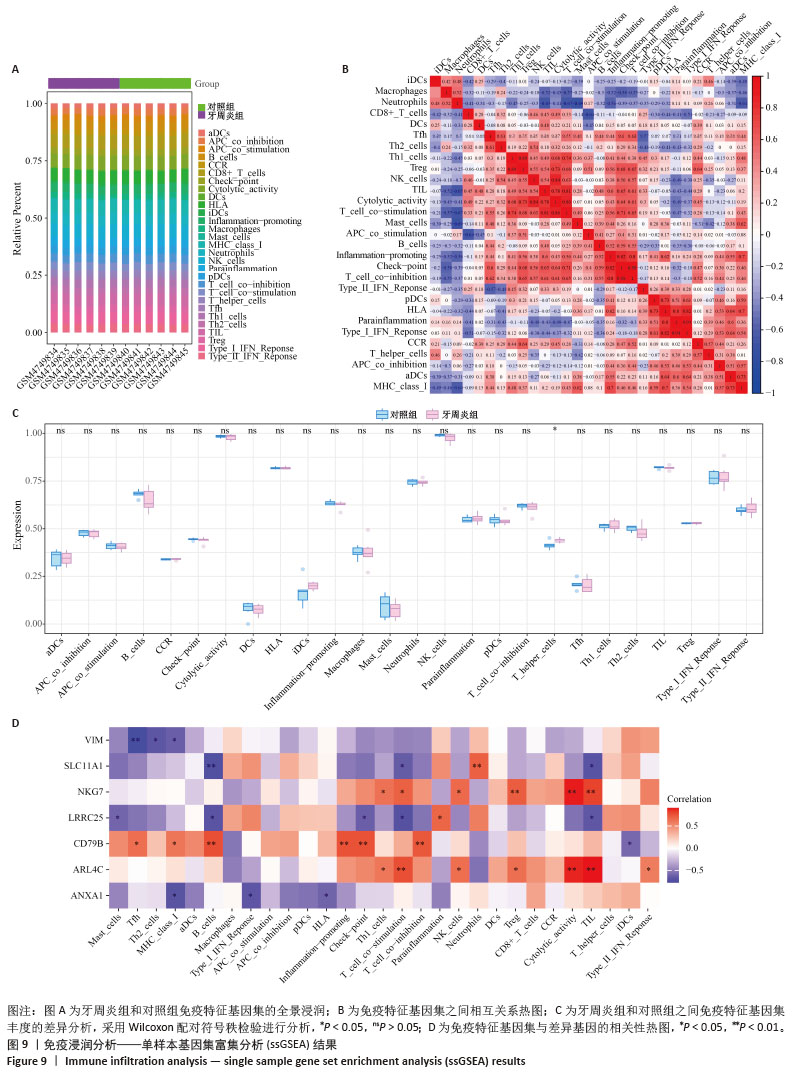
2.5 免疫浸润分析结果 通过分析牙周炎数据集中关键基因与免疫浸润的关系,进一步研究关键基因影响牙周炎进展的分子机制,量化每个患者中各种免疫细胞的比例,了解不同免疫细胞类型之间的相互作用(图9A),分析了29个免疫特征的基因集(16种免疫细胞和13种免疫功能评分)之间的相关性,通过CIBERSORT进行可视化。结果显示,检查点(Check-point)与T细胞共抑制(T_cell_co-inhibition)有显著正相关性(0.98);Ⅰ型干扰素(Type_I_IFN_Reponse)与副炎症(Parainflammation)有显著正相关性(0.94);调节性T细胞(Treg)与Th1辅助细胞(Th1_cells)有显著正相关性(0.89);细胞溶解活性(Cytolytic_activity)与自然杀伤细胞(NK_cells)及T细胞共刺激(T_cell_co-stimulation)有显著正相关性(0.84;0.86)(图9B)。与对照组相比,牙周炎患者中的B细胞、辅助T细胞、NK细胞更高(图9C)。 同时探索了关键基因与免疫细胞之间的相关性(图9D)。膜联蛋白A1与主要组织相容性复合体Ⅰ类分子(MHC_class_I)呈负相关(P < 0.05);溶质载体家族11成员1与中性粒细胞等呈显著正相关,与B细胞呈显著负相关(P < 0.01);波形蛋白与滤泡辅助性T细胞(Tfh)呈显著负相关(P < 0.01)。这些结果反映了所鉴定的细胞亚群的独特和合作作用,并表明牙周炎的免疫浸润具有关键作用。 "

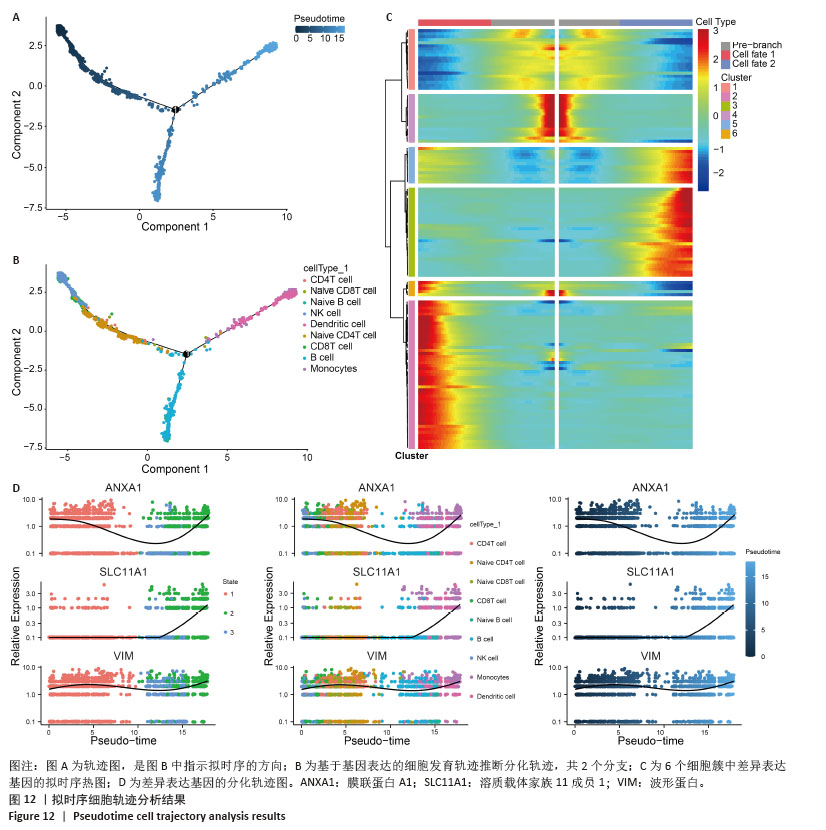
细胞通过伪时间值、细胞类型和状态(通过路径分支区分)着色,提供分化途径的全面视图(图12A,B)。对牙周炎细胞亚群进行拟时序分析,以“NK细胞、CD8+ T细胞”为起点,而“树突状细胞、单核细胞、B细胞、初始B细胞”处于2个不同分支的终点。细胞轨迹分析表明B细胞增殖分化促进了牙周炎发生发展。 通过分析沿发育轨迹在关键分支点上的差异表达基因,发现基因表达的显著变化。热图突出显示了分支点前后具有实质性表达差异的基因(图12C)。在整个伪时间过程中追踪了关键基因的表达变化,揭示了从细胞分化开始到完成的动态调控过程(图12D)。膜联蛋白A1在细胞分化早期和末期表达都有升高,溶质载体家族11成员1在细胞分化末期表达量升高,波形蛋白在细胞分化期表达稳定。 "
| [1] VILLORIA GEM, FISCHER RG, TINOCO EMB, et al. Periodontal disease: A systemic condition. Periodontol 2000. 2024;96(1):7-19. [2] LIU Y, ZHANG T, ZHANG C, et al. Aspirin Blocks Orthodontic Relapse via Inhibition of CD4+ T Lymphocytes. J Dent Res. 2017;96(5):586-594. [3] WU L, SU Y, LIN F, et al. MicroRNA-21 promotes orthodontic tooth movement by modulating the RANKL/OPG balance in T cells. Oral Dis. 2020;26(2):370-380. [4] LO RUSSO L, ZHURAKIVSKA K, MONTARULI G, et al. Effects of crown movement on periodontal biotype: a digital analysis. Odontology. 2018;106(4):414-421. [5] LI H, JIANG X, XIAO Y, et al. Combining single-cell RNA sequencing and population-based studies reveals hand osteoarthritis-associated chondrocyte subpopulations and pathways. Bone Res. 2023;11(1):58. [6] CORBI SCT, DE VASCONCELLOS JF, BASTOS AS, et al. Circulating lymphocytes and monocytes transcriptomic analysis of patients with type 2 diabetes mellitus, dyslipidemia and periodontitis. Sci Rep. 2020;10(1):8145. [7] BIRNEY E. Mendelian Randomization. Cold Spring Harb Perspect Med. 2022;12(4): a041302. [8] WANG J, YU Z, XU W, et al. Uncovering Molecular and Genetic Drivers of Dental Caries Via scRNA-seq and Mendelian Randomisation. Int Dent J. 2025;75(2):668-682. [9] LEE H, JOO JY, SOHN DH, et al. Single-cell RNA sequencing reveals rebalancing of immunological response in patients with periodontitis after non-surgical periodontal therapy. J Transl Med. 2022;20(1):504. [10] 孟焕新.2018年牙周病和植体周病国际新分类简介[J].中华口腔医学杂志, 2019,54(2):73-78. [11] GAO X, JIANG C, YAO S, et al. Identification of hub genes related to immune cell infiltration in periodontitis using integrated bioinformatic analysis. J Periodontal Res. 2022;57(2):392-401. [12] ANSARI J, SENCHENKOVA EY, VITAL SA, et al. Targeting the AnxA1/Fpr2/ALX pathway regulates neutrophil function, promoting thromboinflammation resolution in sickle cell disease. Blood. 2021;137(11):1538-1549. [13] SUGIMOTO MA, VAGO JP, TEIXEIRA MM, et al. Annexin A1 and the Resolution of Inflammation: Modulation of Neutrophil Recruitment, Apoptosis, and Clearance. J Immunol Res. 2016;2016:8239258. [14] KOHLGRUBER AC, DEZFULIAN MH, SIE BM, et al. High-throughput discovery of MHC class I- and II-restricted T cell epitopes using synthetic cellular circuits. Nat Biotechnol. 2025;43(4):623-634. [15] BECK JD, DIKEN M, SUCHAN M, et al. Long-lasting mRNA-encoded interleukin-2 restores CD8+ T cell neoantigen immunity in MHC class I-deficient cancers. Cancer Cell. 2024;42(4):568-582.e11. [16] GAVINS FN, HICKEY MJ. Annexin A1 and the regulation of innate and adaptive immunity. Front Immunol. 2012;3:354. [17] PERRETTI M, DALLI J. Exploiting the Annexin A1 pathway for the development of novel anti-inflammatory therapeutics. Br J Pharmacol. 2009;158(4):936-946. [18] WANG X, TANG Y, XIAO R. Chemerin contributes to inflammatory responses and suppresses osteogenic differentiation in chronic periodontitis. Oral Dis. 2023;29(4):1706-1714. [19] YANG YH, MORAND EF, GETTING SJ, et al. Modulation of inflammation and response to dexamethasone by Annexin 1 in antigen-induced arthritis. Arthritis Rheum. 2004;50(3):976-984. [20] XU YZ, THURAISINGAM T, KANAGARATHAM C, et al. c-Src kinase is involved in the tyrosine phosphorylation and activity of SLC11A1 in differentiating macrophages. PLoS One. 2018;13(5):e0196230. [21] GÖLZ L, MEMMERT S, RATH-DESCHNER B, et al. LPS from P. gingivalis and hypoxia increases oxidative stress in periodontal ligament fibroblasts and contributes to periodontitis. Mediators Inflamm. 2014;2014:986264. [22] KLEBANOFF SJ, KETTLE AJ, ROSEN H, et al. Myeloperoxidase: a front-line defender against phagocytosed microorganisms. J Leukoc Biol. 2013;93(2):185-198. [23] LIU S, CHEN Y, JIANG Y, et al. The bidirectional effect of neutrophils on periodontitis model in mice: A systematic review. Oral Dis. 2024;30(5):2865-2875. [24] SILVA LM, DOYLE AD, GREENWELL-WILD T, et al. Fibrin is a critical regulator of neutrophil effector function at the oral mucosal barrier. Science. 2021;374(6575):eabl5450. [25] DESAI J, KUMAR SV, MULAY SR, et al. PMA and crystal-induced neutrophil extracellular trap formation involves RIPK1-RIPK3-MLKL signaling. Eur J Immunol. 2016;46(1):223-229. [26] KIM TS, SILVA LM, THEOFILOU VI, et al. Neutrophil extracellular traps and extracellular histones potentiate IL-17 inflammation in periodontitis. J Exp Med. 2023;220(9):e20221751. [27] BRATTON DL, HENSON PM. Neutrophil clearance: when the party is over, clean-up begins. Trends Immunol. 2011;32(8):350-357. [28] DUTZAN N, KAJIKAWA T, ABUSLEME L, et al. A dysbiotic microbiome triggers T<sub>H</sub>17 cells to mediate oral mucosal immunopathology in mice and humans. Sci Transl Med. 2018;10(463):eaat0797. [29] HAJISHENGALLIS G. New developments in neutrophil biology and periodontitis. Periodontol 2000. 2020;82(1):78-92. [30] STARK MA, HUO Y, BURCIN TL, et al. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity. 2005;22(3):285-294. [31] DAS V, BHATTACHARYA S, CHIKKAPUTTAIAH C, et al. The basics of epithelial-mesenchymal transition (EMT): A study from a structure, dynamics, and functional perspective. J Cell Physiol. 2019;234(9):14535-14555. [32] LAMOUILLE S, XU J, DERYNCK R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15(3):178-196. [33] YANG J, ANTIN P, BERX G, et al. Guidelines and definitions for research on epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2020;21(6):341-352. [34] POGODA K, BYFIELD F, DEPTUŁA P, et al. Unique Role of Vimentin Networks in Compression Stiffening of Cells and Protection of Nuclei from Compressive Stress. Nano Lett. 2022;22(12):4725-4732. [35] RIDGE KM, ERIKSSON JE, PEKNY M, et al. Roles of vimentin in health and disease. Genes Dev. 2022;36(7-8):391-407. [36] ALANBARI BF, AL-TAWEEL FB, COOPER PR, et al. Induction of Epithelial- Mesenchymal Transition in Periodontitis Rat Model. Eur J Dent. 2025;19(2):428-437. [37] SALIEM SS, BEDE SY, ABDULKAREEM AA, et al. Gingival tissue samples from periodontitis patients demonstrate epithelial- mesenchymal transition phenotype. J Periodontal Res. 2023;58(2):247-255. [38] WADIE KW, BASHIR MH, ABBASS MMS. Epithelial-mesenchymal transition in gingival tissues from chronic periodontitis patients: A case-control study. Dent Med Probl. 2021;58(3):311-319. [39] SONG K, FARZANEH M. Signaling pathways governing breast cancer stem cells behavior. Stem Cell Res Ther. 2021;12(1):245. [40] WANG X, SONG X, GAO J, et al. Hedgehog/Gli2 signaling triggers cell proliferation and metastasis via EMT and wnt/β-catenin pathways in oral squamous cell carcinoma. Heliyon. 2024;10(16):e36516. |
| [1] | Wu Zhilin, , He Qin, Wang Pingxi, Shi Xian, Yuan Song, Zhang Jun, Wang Hao . DYRK2: a novel therapeutic target for rheumatoid arthritis combined with osteoporosis based on East Asian and European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1569-1579. |
| [2] | Liu Hongtao, Wu Xin, Jiang Xinyu, Sha Fei, An Qi, Li Gaobiao. Causal relationship between age-related macular degeneration and deep vein thrombosis: analysis based on genome-wide association study data [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1602-1608. |
| [3] | Lai Jiaming, , Song Yuling, Chen Zixi, Wei Jinghuan, Cai Hao, , Li Guoquan, . Screening of diagnostic markers for endothelial cell Senescence in mice with radiation-induced heart disease and analysis of immune infiltration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1450-1463. |
| [4] | Guo Ying, Tian Feng, Wang Chunfang. Potential drug targets for the treatment of rheumatoid arthritis: large sample analysis from European databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1549-1557. |
| [5] | Gao Zengjie, , Pu Xiang, Li Lailai, Chai Yihui, Huang Hua, Qin Yu. Increased risk of osteoporotic pathological fractures associated with sterol esters: evidence from IEU-GWAS and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1302-1310. |
| [6] | Liu Fengzhi, Dong Yuna, Tian Wenyi, Wang Chunlei, Liang Xiaodong, Bao Lin. Gene-predicted associations between 731 immune cell phenotypes and rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1311-1319. |
| [7] | Zhang Cuicui, Chen Huanyu, Yu Qiao, Huang Yuxuan, Yao Gengzhen, Zou Xu. Relationship between plasma proteins and pulmonary arterial hypertension and potential therapeutic targets [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1331-1340. |
| [8] | Zeng Hao, Sun Pengcheng, Chai Yuan, Huang Yourong, Zhang Chi, Zhang Xiaoyun. Association between thyroid function and osteoporosis: genome-wide data analysis of European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1019-1027. |
| [9] | Rong Xiangbin, , Zheng Haibo, Mo Xueshen, Hou Kun, Zeng Ping, . Plasma metabolites, immune cells, and hip osteoarthritis: causal inference based on GWAS data from European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1028-1035. |
| [10] | He Qiwang, , , Chen Bo, Liang Fuchao, Kang Zewei, Zhou Yuan, Ji Anxu, Tang Xialin, . Relationship between Alzheimer’s disease and sarcopenia and body mass index: analysis of GWAS datasets for European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1036-1046. |
| [11] | Ding Yu, Chen Jingwen, Chen Xiuyan, Shi Huimin, Yang Yudie, Zhou Meiqi, Cui Shuai, . Circulating inflammatory proteins and myocardial hypertrophy: large sample analysis of European populations from GWAS Catalog and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1047-1057. |
| [12] | Zhao Feifan, Cao Yujing. An artificial neural network model of ankylosing spondylitis and psoriasis shared genes and machine learning-based mining and validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 770-784. |
| [13] | Liu Chu, Qiu Boyuan, Tong Siwen, He Linyuwei, Chen Haobo, Ou Zhixue. A genetic perspective reveals the relationship between blood metabolites and osteonecrosis: an analysis of information from the FinnGen database in Finland [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 785-794. |
| [14] | Yin Xingxiao, Peng Hao, Song Yanping, Yao Na, Shen Zhen, Jiang Yang, Chen Hongbo, Huang Li, Song Yueyu, Li Yanqi, Chen Qigang. Sarcopenia and cognitive impairment: a data analysis based on European population databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7388-7395. |
| [15] | Chen Yongxi. Multi-omics approach unveils novel therapeutic targets for osteoporosis: integrated analysis of Asian and European gene-tissue expression consortium data [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6382-6389. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||