Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (29): 7581-7591.doi: 10.12307/2026.187
Previous Articles Next Articles
Proteomic analysis of the mechanism of moxibustion intervention in a rat model of atopic dermatitis
Yang Yunhong, Guo Lihua, Tang Han, Lin Lvping, Kuang Hongjun, Zhao Hong
- Shenzhen Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shenzhen 518001, Guangdong Province, China
-
Received:2025-06-15Revised:2025-09-20Online:2026-10-18Published:2026-03-04 -
Contact:Zhao Hong, PhD, Chief physician, Shenzhen Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shenzhen 518001, Guangdong Province, China -
About author:Yang Yunhong, PhD candidate, Physician, Shenzhen Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shenzhen 518001, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China (General Program), No. 82474644 (to ZH); Shenzhen Science and Technology Program, No. JCYJ20210324120804012 (to ZH)
CLC Number:
Cite this article
Yang Yunhong, Guo Lihua, Tang Han, Lin Lvping, Kuang Hongjun, Zhao Hong. Proteomic analysis of the mechanism of moxibustion intervention in a rat model of atopic dermatitis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7581-7591.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
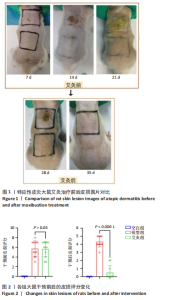
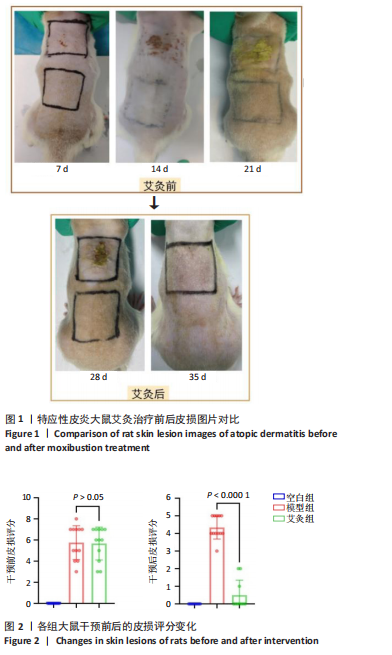
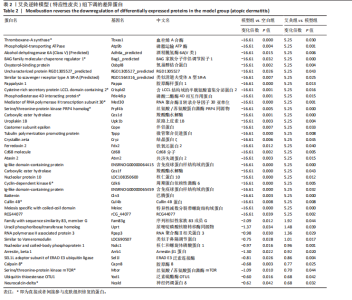
2.1 实验动物数量分析 实验过程中3组SD大鼠共34只均无脱失。 2.2 特应性皮炎大鼠模型建立及皮损变化 建立特应性皮炎大鼠模型后,大鼠精神状态差,易受惊和躁动不安;致敏区及其附近出现明显抓痕或咬痕,与空白组对比,用2,4-二硝基氯苯过敏源使大鼠的背部皮肤敏感,成功诱导出特应性皮炎的临床特征,如大体可见皮损呈红斑、结痂、苔藓样等病理改变。而经艾灸治疗后,可见病理变化明显改善,见图1。 根据各组大鼠皮损区的湿疹面积及严重度指数可见,干预之前,与空白组比较,模型组和艾灸组大鼠皮肤损伤评分明显升高,两组间的皮损变化无统计学差异(P > 0.05)。艾灸组干预后,模型组皮损评分较干预前稍有下降,但是不明显;而与模型组对比,艾灸组的皮肤损伤评分明显降低(P < 0.05),差异有显著性意义,见图2。 2.3 特应性皮炎及艾灸效应相关的差异表达蛋白分析 将获得的数据进行蛋白搜库和差异蛋白筛选,结果显示:所有样本中共检测出蛋白质4 969个,利用Persues软件 (|Fold Change| > 1.2,"
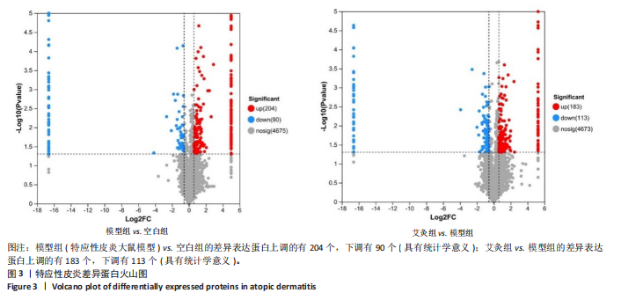
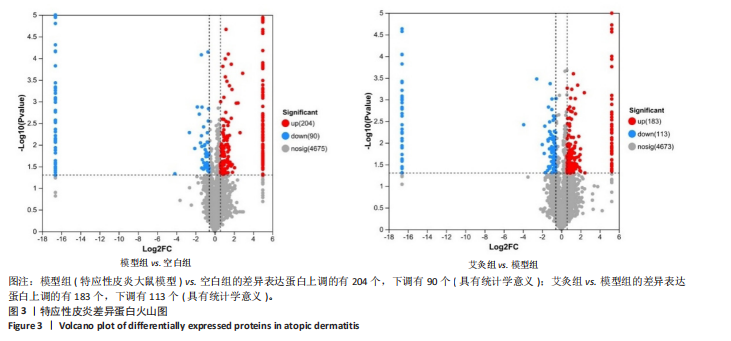
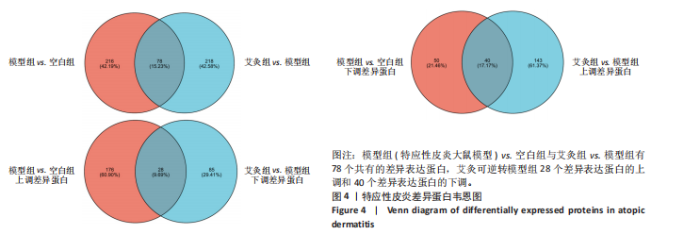
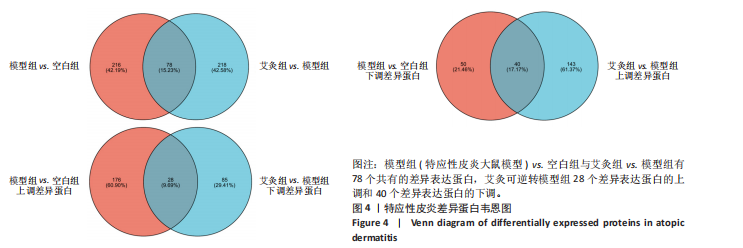
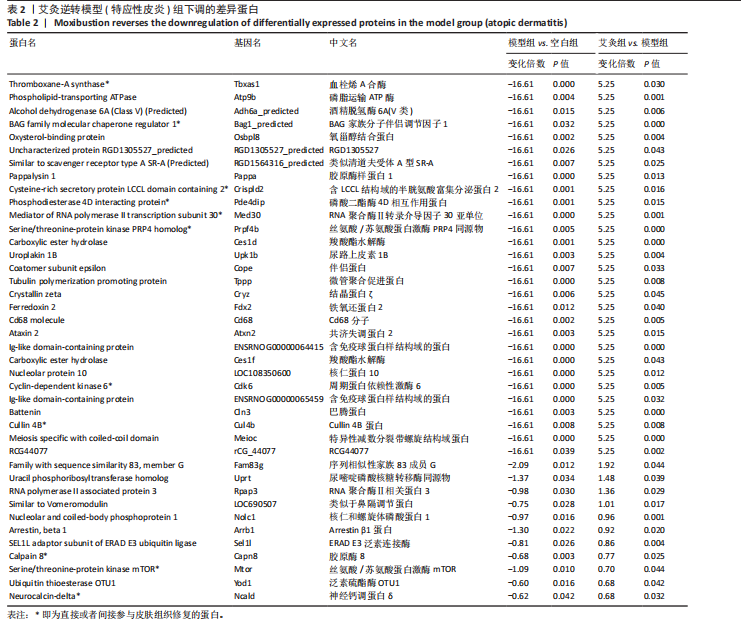
P < 0.05) 筛选,得到各组间的差异表达蛋白,模型组与空白组相比共有294个差异表达蛋白,其中上调的蛋白有204个,下调的蛋白有90个;艾灸组与模型组相比共有296个差异表达蛋白,其中上调的蛋白有183个,下调的蛋白有113个,见图3。通过进一步探究不同组间差异蛋白的重叠关系,发现有78个差异蛋白与艾灸治疗特应性皮炎的效应机制密切相关。 艾灸可逆转模型组28个差异表达蛋白的上调和40个差异表达蛋白的下调,见图4,其中艾灸可逆转模型组蛋白的上调主要包括3个与细胞代谢相关蛋白(糖代谢:Aldoc、Glb3;药物代谢:Ugt1a1),3个与细胞结构和运动相关的蛋白(Actb、Bicd2、 Emilin1),1个与免疫和防御相关的蛋白(Ncf2)等,见表1。艾灸可逆转模型组蛋白的下调主要包括5个代谢相关蛋白(Tbxas1、Atp9b、Fdx2、Ces1d、Ces1f),5个细胞结构和功能相关蛋白(Tppp、Crispld2、Cryz、Upk1b、Osmbp18),4个免疫和炎症相关蛋白"
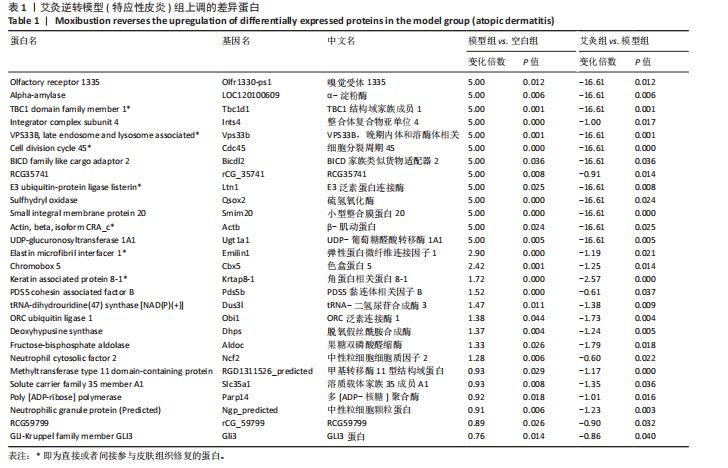
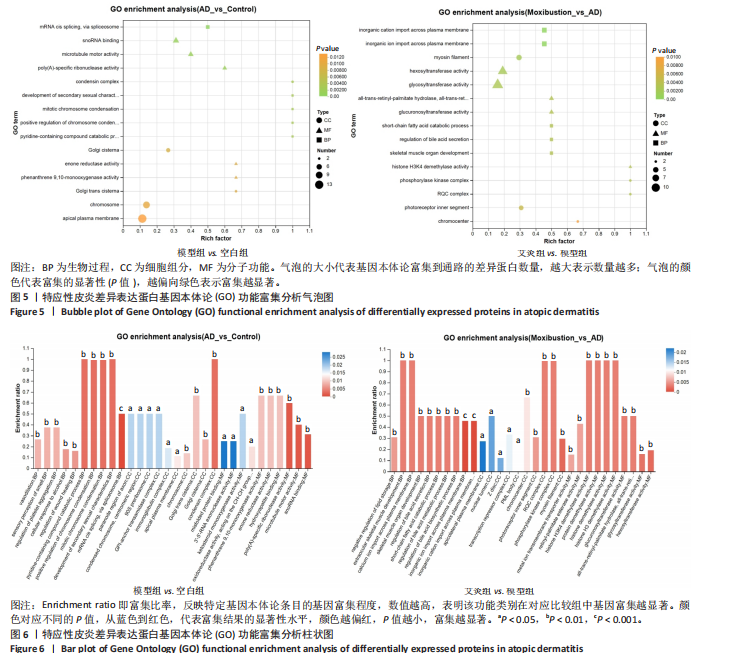
(Cd68、Arrb1、Sell1、Pde4dip),3个神经系统相关蛋白(Atxn2、Cln3、Ncald)和2个信号转导相关(G 蛋白偶联受体信号通路相关:Arrb1;mTOR信号通路相关:Mtor)等,见表2。说明艾灸对特应性皮炎的调控效应主要与细胞代谢、细胞结构与功能、免疫和炎症、神经系统、信号转导等多种生物学过程相关。 2.4 差异表达蛋白基因本体论功能富集分析 为了进一步探索艾灸的效应机制,进行了基因本体论功能富集分析。模型组相对于空白组,细胞组分主要涉及浓缩染色体、高尔基体池、高尔基体反式池、质膜和染色体等;分子功能主要涉及微管马达活性、多聚腺苷酸特异性核糖核酸酶活性、烯酮还原酶活性和作用于CH-CH基团的氧化还原酶活性等;生物过程主要涉及伤口愈合的调节、第二性征的发育、染色体凝聚的正向调节和有丝分裂染色体凝聚等。艾灸组相对于模型组,主要涉及的细胞组分包括核膜、Z 盘、光感受器外段、染色中心和线粒体内膜等;分子"

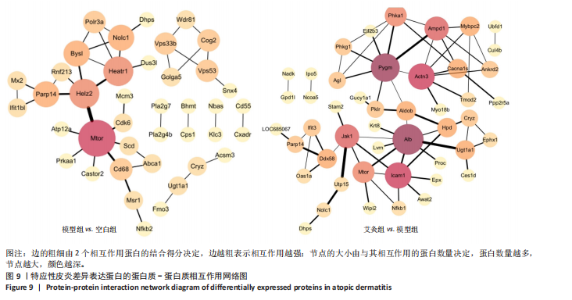
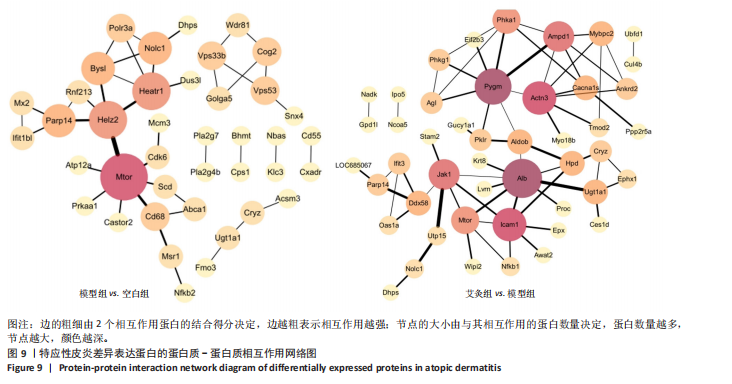
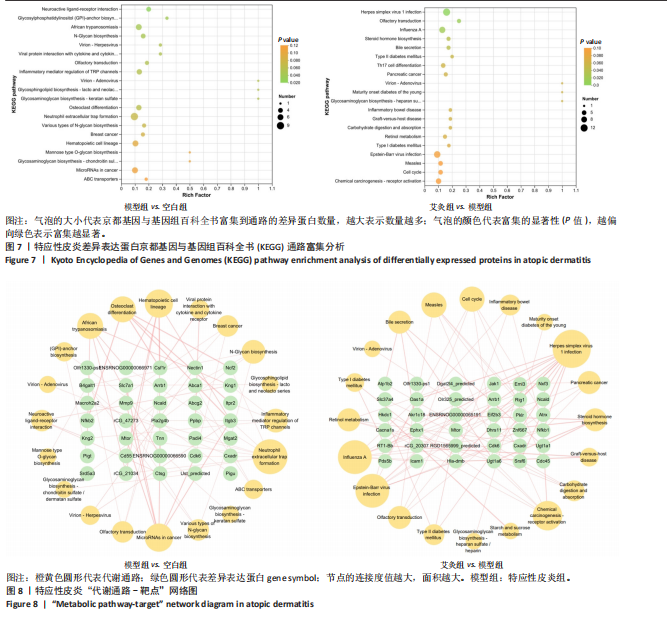
2.5 特应性皮炎及艾灸效应相关的代谢通路及靶点分析 对组间的差异蛋白进行京都基因与基因组百科全书代谢通路分析,结果显示模型组 vs. 空白组的差异蛋白共涉及信号通路263条,富集显著性排名前20的条目见图7,主要涉及神经活性配体-受体相互作用通路、糖基磷脂酰肌醇锚定生物合成通路、非洲锥虫病通路、N-聚糖生物合成通路、病毒蛋白与细胞因子及细胞因子受体相互作用通路、炎症介质对瞬时受体电位通道的调节通路、中性粒细胞胞外陷阱形成通路和癌症有关信号通路等。利用Cytoscape软件进行可视化分析,构建“代谢通路-靶点”网络图,见图8。拓扑分析显示,该网络共有55个节点、64条边。度值排名前3位的蛋白(包括并列关系)为Itgb3、B4galt1、Csf1r、Mtor、Kng1、ENSRNOG00000066971、rCG_21034、ENSRNOG00000066590、Kng2、Mgat2、rCG_47273、Nfkb2、Cdk6、Itpr2、Ctsg和Ncf2,其中度值最大的为Itgb3和B4galt1,代表其参与的代谢通路最多,其表达调控可能与特应性皮炎致病机制有关。 艾灸组与模型组相比,差异蛋白总共涉及261条信号通路,主要涉及单纯疱疹病毒1型感染、嗅觉转导、甲型流感、类固醇激素生物合成、胆汁分泌、Th17 细胞分化和其他与感染和免疫相关的信号通路。利用Cytoscape软件进行可视化分析,构建“代谢通路-靶点”网络图。拓扑分析显示,该网络共有56个节点、83条边,度值排名前3的蛋白为Nfkb1、Hla-dmb、RT1-Bb、 Jak1和Cdk6;其中度值最大的为Nfkb1。核因子κB 是一类重要的转录因子,在先天免疫、适应性免疫及炎症反应中起关键作用。由此可见,Nfkb1的表达调控可能与艾灸治疗特应性皮炎的效应机制有关。 2.6 艾灸对特应性皮炎的效应机制与免疫和炎症相关 为了探究差异蛋白之间进一步的相互作用,利用STRING数据库和Cytoscape软件构建蛋白质-蛋白质相互作用网络图。模型组"
| [1] MULLER S, WITTE F, STANDER S. Pruritus in atopic dermatitis-comparative evaluation of novel treatment approaches. Dermatologie (Heidelb). 2022;73(7): 538-549. [2] GATMAITAN JG, LEE JH. Challenges and Future Trends in Atopic Dermatitis. Int J Mol Sci. 2023;24(14):11380. [3] XUE Y, BAO W, ZHOU J, et al. Global Burden, Incidence and Disability-Adjusted Life-Years for Dermatitis: A Systematic Analysis Combined With Socioeconomic Development Status, 1990-2019. Front Cell Infect Microbiol. 2022;12:861053. [4] GUTTMAN-YASSK E, RENERT-YUVAL Y, BRUNNER PM. Atopic dermatitis. Lancet. 2025;405(10478):583-596. [5] SON SW, LEE JH, AHN J, et al. Assessment of Disease Severity and Quality of Life in Patients with Atopic Dermatitis from South Korea. Ann Dermatol. 2022; 34(6):419-430. [6] 张国山,刘密,章海凤,等.艾灸温通、温补效应之间的关系[J]. 时珍国医国药,2013,24(10):2468-2469. [7] 鲁熹. 艾灸疗法改善慢性湿疹瘙痒症状的临床疗效观察[D]. 成都:成都中医药大学,2022. [8] 伍娅欣. 电针结合艾灸治疗慢性湿疹的临床观察及其对血清IgE的影响[D]. 泸州:西南医科大学,2019. [9] 毛钟莹. 毫火针结合艾灸治疗脾虚湿蕴型慢性湿疹的临床观察[D]. 广州:广州中医药大学,2021. [10] AIERKEN K, LUO Y, MAIWULANJIANG M, et al. The Suppressive Effect of Mamiran Cream on Atopic Dermatitis-Like Skin Lesions In Vivo. Evid Based Complement Alternat Med. 2021;2021:2854238. [11] 林建红, 陈海军, 朱思颐. EGCG调节TLR4/MyD88/NF-κB信号通路对特应性皮炎大鼠Th1/Th2平衡的影响[J]. 西部医学,2024,36(2):211-216. [12] 苗明三, 田硕, 白明. 湿疹动物模型制备规范(草案)[J]. 中国实验方剂学杂志, 2017,23(24):6-10. [13] 赵辨. 湿疹面积及严重度指数评分法[J]. 中华皮肤科杂志,2004(1):7-8. [14] CHOPRE R, SILVERBERG JI. Assessing the severity of atopic dermatitis in clinical trials and practice. Clin Dermatol. 2018;36(5):606-615. [15] 汪秀梅. 针灸对湿热蕴肤证、血虚风燥证、脾虚湿蕴证三种湿疹模型表皮通透屏障功能影响的研究[D]. 乌鲁木齐:新疆医科大学, 2021. [16] 余曙光,徐斌. 实验针灸学[M]. 北京:人民卫生出版社, 2016. [17] 于燕艳,刘娟,杨越,等.艾灸抑制NLRP3/Caspase-1通路介导的细胞焦亡减轻脑缺血再灌注损伤[J].中国组织工程研究,2024,28(34):5473-5479. [18] PATRICK GJ, ARCHER NK, MILLER LS. Which Way Do We Go? Complex Interactions in Atopic Dermatitis Pathogenesis. J Invest Dermatol. 2021;141(2):274-284. [19] BIEBER T. Atopic dermatitis: an expanding therapeutic pipeline for a complex disease. Nat Rev Drug Discov. 2022;21(1):21-40. [20] MOYLE M, CEVIKBAS F, HARDEN JL, et al. Understanding the immune landscape in atopic dermatitis: The era of biologics and emerging therapeutic approaches. Exp Dermatol. 2019;28(7):756-768. [21] XIN P, XU X, DENG C, et al. The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int Immunopharmacol. 2020;80:106210. [22] AGASHE RP, LIPPMAN SM, KURZROCK R. JAK: Not Just Another Kinase. Mol Cancer Ther. 2022;21(12):1757-1764. [23] 李文婕, 周萌, 苏银妹, 等. 周萌从脾论治湿疹经验[J]. 江西中医药,2019, 50(10):31-32. [24] 刁庆春, 刘毅. 湿疹(湿疮)中医诊疗专家共识[J]. 中国中西医结合皮肤性病学杂志,2021,20(5):517-521. [25] 严格,王怡,杜欣冉,等.“血虚风燥证”皮肤疾病生物学基础研究进展[J].皮肤科学通报,2024,41(3):229-235. [26] HAMIDI H, IVASKA J. Every step of the way: integrins in cancer progression and metastasis. Nat Rev Cancer. 2018;18(9):533-548. [27] XU YH, LI ZL, QIU SF. IFN-gamma Induces Gastric Cancer Cell Proliferation and Metastasis Through Upregulation of Integrin beta3-Mediated NF-kappaB Signaling. Transl Oncol. 2018;11(1):182-192. [28] THOMPSON EE, PAN L, OSTROVNAYA I, et al. Integrin beta 3 genotype influences asthma and allergy phenotypes in the first 6 years of life. J Allergy Clin Immunol. 2007;119(6):1423-1429. [29] ARTEAGA-BADILLO DA, PORTILLO-REYES J, VARGAS-MENDOZA N, et al. Asthma: New Integrative Treatment Strategies for the Next Decades. Medicina (Kaunas), 2020,56(9). [30] MACIAG MC, PHIPATANAKUL W. Preventing the development of asthma: stopping the allergic march. Curr Opin Allergy Clin Immunol. 2019;19(2):161-168. [31] 李淑芬, 李希. β1,4半乳糖基转移酶的生物学功能研究进展[J]. 生命的化学, 2016,36(5):589-595. [32] ASANO M, NAKAE S, KOTANI N, et al. Impaired selectin-ligand biosynthesis and reduced inflammatory responses in beta-1,4-galactosyltransferase-I-deficient mice. Blood. 2003;102(5):1678-1685. [33] HAN Y, ZHOU X, JI Y, et al. Expression of beta-1,4-galactosyltransferase-I affects cellular adhesion in human peripheral blood CD4+ T cells. Cell Immunol. 2010; 262(1):11-17. [34] MOHAMMAD S, KARIM MR, IQBAL S, et al. Atopic dermatitis: Pathophysiology, microbiota, and metabolome-A comprehensive review. Microbiol Res. 2024; 281:127595. [35] LAMPSON BL, RAMΊREZ AS, BARO M, et al. Positive selection CRISPR screens reveal a druggable pocket in an oligosaccharyltransferase required for inflammatory signaling to NF-κB. Cell. 2024;187(9):2209-2223.e16. [36] LAWRENCE T, GILROY DW, COLVILLE-NASH PR, et al. Possible new role for NF-kappaB in the resolution of inflammation. Nat Med. 2001;7(12):1291-1297. [37] POLADIAN N, ORUJYAN D, NARINYAN W, et al. Role of NF-κB during Mycobacterium tuberculosis Infection. Int J Mol Sci. 2023;24(2):1772. [38] LIU T, ZHANG L, JOO D, et al. NF-kappaB signaling in inflammation. Signal Transduct Target Ther. 2017;2:17023. [39] LONG H, ZHANG G, WANG L, et al. Eosinophilic Skin Diseases: A Comprehensive Review. Clin Rev Allergy Immunol. 2016;50(2):189-213. [40] DU X, SHI H, LIU X, et al. Genetic support for the causal association between 91 circulating inflammatory proteins and atopic dermatitis: A two-sample Mendelian randomization trial. Skin Res Technol. 2024;30(8):e13872. [41] KO KI, MERLET JJ, DERGARABEDIAN BP, et al. NF-kappaB perturbation reveals unique immunomodulatory functions in Prx1(+) fibroblasts that promote development of atopic dermatitis. Sci Transl Med. 2022;14(630):j324. [42] LIU W, SONG W, LUO Y, et al. Angelica Yinzi alleviates 1-chloro-2,4-dinitrobenzene-induced atopic dermatitis by inhibiting activation of NLRP3 inflammasome and down-regulating the MAPKs/NF-kB signaling pathway. Saudi Pharm J. 2022; 30(10):1426-1434. [43] LI X, LU C, DU W, et al. Development of new dehydrocostuslactone derivatives for treatment of atopic dermatitis via inhibition of the NF-kappaB signaling pathway. RSC Med Chem. 2024;15(8):2773-2784. [44] ROY T, BOATENG ST, UDDIN MB, et al. The PI3K-Akt-mTOR and Associated Signaling Pathways as Molecular Drivers of Immune-Mediated Inflammatory Skin Diseases: Update on Therapeutic Strategy Using Natural and Synthetic Compounds. Cells. 2023;12(12):1671. [45] BUERGER C. Epidermal mTORC1 Signaling Contributes to the Pathogenesis of Psoriasis and Could Serve as a Therapeutic Target. Front Immunol. 2018;9:2786. [46] HOU T, SUN X, ZHU J, et al. IL-37 Ameliorating Allergic Inflammation in Atopic Dermatitis Through Regulating Microbiota and AMPK-mTOR Signaling Pathway-Modulated Autophagy Mechanism. Front Immunol. 2020;11:752. [47] KRISHNAMURTHY P, DA-SILVA-ARNOLD S, TURNER MJ, et al. Poly-ADP ribose polymerase-14 limits severity of allergic skin disease. Immunology. 2017;152(3): 451-461. [48] YANG X, MAO Z, HUANG Y, et al. Reductively modified albumin attenuates DSS-Induced mouse colitis through rebalancing systemic redox state. Redox Biol. 2021;41:101881. [49] INOUE M, NAKASHIMA R, ENOMOTO M, et al. Plasma redox imbalance caused by albumin oxidation promotes lung-predominant NETosis and pulmonary cancer metastasis. Nat Commun. 2018;9(1):5116. [50] SIMONETTI O, BACCHETTI T, FERRETTI G, et al. Oxidative Stress and Alterations of Paraoxonases in Atopic Dermatitis. Antioxidants (Basel). 2021;10(5):697. [51] WANG Y, LU H, CHENG L, et al. Targeting mitochondrial dysfunction in atopic dermatitis with trilinolein: A triacylglycerol from the medicinal plant Cannabis fructus. Phytomedicinel. 2024;132(2024):155856. [52] JI H, LI XK. Oxidative Stress in Atopic Dermatitis. Oxid Med Cell Longev. 2016; 2016:2721469. [53] SIVARANJANI N, RAO SV, RAJEEV G. Role of reactive oxygen species and antioxidants in atopic dermatitis. J Clin Diagn Res. 2013;7(12):2683-2685. [54] CHEN X, ZHANG Y, PEI J, et al. Phellopterin alleviates atopic dermatitis-like inflammation and suppresses IL-4-induced STAT3 activation in keratinocytes. Int Immunopharmacol. 2022;112:109270. [55] HAGIYAMA M, INOUE T, FURUNO T, et al. Increased expression of cell adhesion molecule 1 by mast cells as a cause of enhanced nerve-mast cell interaction in a hapten-induced mouse model of atopic dermatitis. Br J Dermatol. 2013; 168(4):771-778. [56] OH JE, KIM SN. Anti-Inflammatory Effects of Acupuncture at ST36 Point: A Literature Review in Animal Studies. Front Immunol. 2021;12:813748. [57] ZHAI L, WAN X, WU R, et al. Linc-RAM promotes muscle cell differentiation via regulating glycogen phosphorylase activity. Cell Regen. 2022;11(1):8. [58] ARRIZABALAGA O, LACERDA HM, ZUBIAGA AM, et al. Rac1 protein regulates glycogen phosphorylase activation and controls interleukin (IL)-2-dependent T cell proliferation. J Biol Chem. 2012;287(15):11878-11890. [59] SIERRA A, OLIVEIRA RA, SILVA ED, et al. Association Between Hematological Parameters and Iron Metabolism Response After Marathon Race and ACTN3 Genotype. Front Physiol. 2019;10:697. |
| [1] | Yan Chengbo, Luo Qiuchi, Fan Jiabing, Gu Yeting, Deng Qian, Zhang Junmei. Effect of type 2 diabetes mellitus on orthodontic tooth movement and bone microstructure parameters on the tension side in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 824-831. |
| [2] | Yong Qiao, Sun Xin, Wang Guoyou, Zhang Lei, Shen Huarui, Liu Huan, Guan Taiyuan. Shaoyang Shenggu Fang inhibits oxidative stress and delays cartilage aging in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7251-7259. |
| [3] | Tang Cen, Hu Wanqin. Establishing a diagnostic model for recurrent spontaneous abortion based on the levels of autophagy-related genes in the endometrium [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5728-5738. |
| [4] | Zhang Shujuan, Xu Qianqian, Wang Chao, Li Yunhui, Zhu Yanping. Transplantation of human umbilical cord mesenchymal stem cells to repair myelination disorders in neonatal rats with white matter injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 4890-4896. |
| [5] | Lu Anran, Wang Chenyu, Zhang Yan, Huang Huasheng. High-intensity interval training improves the function of exosomes derived from endothelial progenitor cells in spontaneously hypertensive rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4627-4637. |
| [6] | Fu Jingyue, Zhou Qinfeng, Li Muzhe, Ma Yong, Pan Yalan, Sun Jie, Huang Xiangyang, Guo Yang. Preparation and evaluation of an animal model of osteoporosis and osteoarthritis comorbidity in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4299-4308. |
| [7] | Peng Hao, Jiang Yang, Song Yanping, Wu Quan, Yao Na, Chen Qigang, Shen Zhen. H-type angiogenesis and its role in various skeletal disease animal models [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4154-4165. |
| [8] | Pan Li, Zhu Zhou, Yan Zhaobo, Zhang Ning, Yang Zhihong, Xiong Jiaojiao, Yang Xiaofang. Moxibustion improves endothelial function in atherosclerotic mice by regulating fatty acid oxidation through mediating mitochondrial autophagy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2764-2773. |
| [9] | Jiao Taiqiang, Han Xingji, Li Xiangyang, Nan Yi, Yuan Ling, Li Jiaqing, Niu Yang. Mechanism by which Maxing Kugan Decoction intervenes in oleic acid-induced acute lung injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(10): 2430-2439. |
| [10] | Ye Xing, Liu Renyi. Effects of voluntary exercise on molecular expression profiles in the hippocampus of mice: a gene expression profile analysis based on the GEO database [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(24): 5237-5244. |
| [11] | Liang Zhou, Zhang Chi, Pan Chengzhen, Yang Bo, Pu Zhanglin, Liu Hua, Peng Jinhui, Wen Lichun, Ling Guanhan, Chen Feng. Anti-osteoporotic mechanisms of kaempferol based on gut microbiota and comprehensive targeted metabolomics [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(20): 4190-4204. |
| [12] | Ma Munan, Xie Jun, Sang Yuchao, Huang Lei, Zhang Guodong, Yang Xiaoli, Fu Songtao. Electroacupuncture combined with bone marrow mesenchymal stem cells in the treatment of chemotherapy-induced premature ovarian insufficiency in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 1-7. |
| [13] | Feng Jianbo, Li Chencheng, Liu Jinyue, Wang Xiaomin, Peng Jiachen. Implantation of Kirschner wire with Staphylococcus aureus biofilm establishes a traumatic osteomyelitis model in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 700-705. |
| [14] | Jia Qiyu, Huang Xiaoxia, Guo Jian, Huang Jinyong, Guo Xiaobin, Abdussalam·Alimujiang, Wu Tong, Ma Chuang. Integrin-targeted peptide promotes proliferation of bone marrow mesenchymal stem cells in SD rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4780-4786. |
| [15] | Zhang Wanxia, Nijiati·Nuermuhanmode, Maisituremu·Heilili, Liu Jianglong, Liu Di, Maimaitituxun·Tuerdi. An appropriate dose of intra-articular medical ozone injection in a rat model of temporomandibular joint osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3700-3705. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||