Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6239-6246.doi: 10.12307/2026.172
Previous Articles Next Articles
Methylation alterations of Fbln1 gene in the hippocampus of PSEN1/PSEN2 double knockout and #br# APP/PS1 transgenic mice#br#
Ruan Sibei1, Li Li2, Jian Yue3, Ling Feng1, Tang Mingxi1, 4
- 1Department of Pathology, The Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China; 2Department of Pathology, Luzhou Second People’s Hospital, Luzhou 646000, Sichuan Province, China; 3School of Basic Medicine, Southwest Medical University, Luzhou 646000, Sichuan Province, China; 4Department of Pathology, Precision Medicine Center, Yaan People’s Hospital (Yaan Hospital of West China Hospital, Sichuan University), Yaan 625000, Sichuan Province, China
-
Received:2025-06-16Revised:2025-09-11Online:2026-08-28Published:2026-01-30 -
Contact:Tang Mingxi, MD, Professor, Master’s supervisor, Department of Pathology, The Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China; Department of Pathology, Precision Medicine Center, Yaan People’s Hospital (Yaan Hospital of West China Hospital, Sichuan University), Yaan 625000, Sichuan Province, China -
About author:Ruan Sibei, MS, Attending Physician, Department of Pathology, The Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China -
Supported by:Southwest Medical University (Natural Science Foundation for the Youth), No. 2019ZQN093 (to RSB)
CLC Number:
Cite this article
Ruan Sibei, Li Li, Jian Yue, Ling Feng, Tang Mingxi. Methylation alterations of Fbln1 gene in the hippocampus of PSEN1/PSEN2 double knockout and #br# APP/PS1 transgenic mice#br#[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6239-6246.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
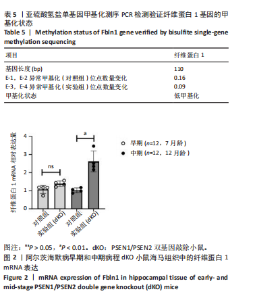
2.1 实验动物数量分析 分别使用9只7月龄和9只12月龄双基因敲除小鼠进行测序检测,其中每3只小鼠的海马组织作为一个检测样本,共3个检测结果综合分析。转录和翻译水平检测分为3组:双基因敲除小鼠组、双转基因小鼠组和对照组,每组9只小鼠,每3只小鼠作为一个检测值,每组3个值取均数进行组间比较。 2.2 dKO小鼠海马组织表观亚硫酸盐测序甲基化测序 9只雌性7月龄(早期阿尔茨海默病)、12月龄(中期阿尔茨海默病)dKO小鼠及野生型小鼠海马组织DNA质量符合表观亚硫酸盐测序甲基化测序要求:质量浓度为101.0-171.0 ng/μL,A260/280≥1.8。表观亚硫酸盐测序检测dKO小鼠海马组织全基因组甲基化水平,利用Bismark(v0.14.0)软件同小鼠参考基因序列进行对比分析,检测出位于15号外显子区域的纤维蛋白1基因在早期阿尔茨海默病动物模型中表现出低甲基化(P=0.190),但与野生型小鼠无显著差异;中期时表现出明显低于野生型小鼠的低甲基化状态(P=0.002),见表4。提示纤维蛋白1基因异常低甲基化主要发生在dKO小鼠的中期病程,这对dKO小鼠病程发生发展及药物靶标研究有重要意义。"
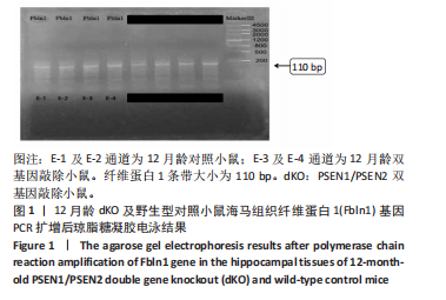
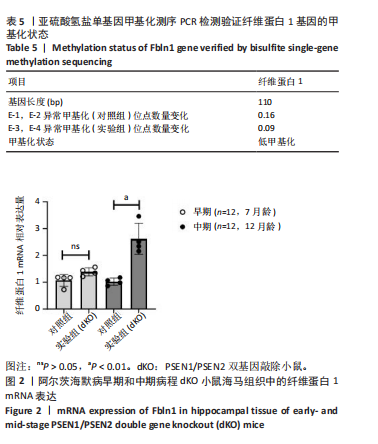
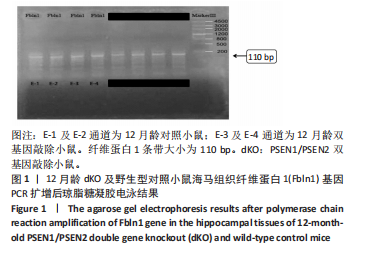
2.3 dKO小鼠海马组织亚硫酸氢盐单基因甲基化测序PCR检测单基因纤维蛋白1基因甲基化测序验证结果 对9只12月龄dKO及野生型小鼠海马组织DNA的纤维蛋白1基因PCR产物进行1%琼脂糖凝胶电泳,条带显示清晰,对照组及dKO小鼠的纤维蛋白1基因分布扩增出相应110 bp条带,可用于亚硫酸氢盐单基因甲基化测序PCR检测分析,见图1;对PCR产物进行亚硫酸氢盐单基因甲基化测序PCR检测,结果显示纤维蛋白1基因甲基化位点率为9%,与野生型小鼠相比呈现低甲基化状态(表5),与表观亚硫酸盐测序检测结果一致,验证了dKO小鼠阿尔茨海默病中期纤维蛋白1基因的低甲基化状态。 2.4 RT-PCR检测纤维蛋白1基因在7月龄(早期阿尔茨海默病)及12月龄(中期阿尔茨海默病)的dKO小鼠及DTG小鼠海马组织中mRNA表达情况 如图2所示,7月龄(早期阿尔茨海默病)dKO小鼠海马组织中纤维蛋白1基因的mRNA水平与同龄野生型小鼠间无差异(t=2.407,P > 0.05),12月龄(中期阿尔茨海默病)dKO小鼠海马组织中纤维蛋白1基因的mRNA水平升高,且显著高于同龄野生型小鼠 (t=5.366,P < 0.01 ),这与甲基化测序方法检测早、中期dKO小鼠纤维蛋白1基因甲基化结果相一"
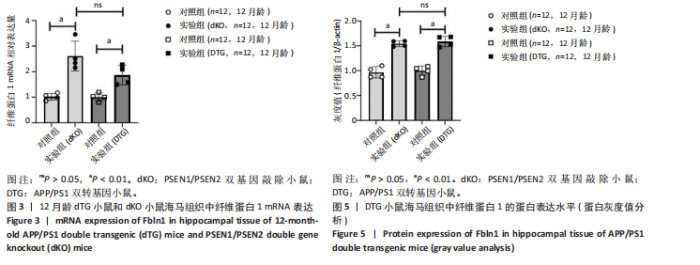
致。另外,为了对比具有β-淀粉样蛋白异常沉积特点的阿尔茨海默病动物模型,使用了DTG小鼠,该小鼠并没有年龄依赖性神经退行性病变的特点,因此检测其12月龄时海马组织中纤维蛋白1基因 mRNA水平,结果显示DTG小鼠阿尔茨海默病中期的纤维蛋白1 mRNA水平均显著高于同龄野生型小鼠 (t=4.151,P < 0.01),见图3,这也说明在DTG小鼠的海马组织中纤维蛋白1基因表达同样是升高的状态。然而,dKO与DTG两种阿尔茨海默病模型小鼠纤维蛋白1 mRNA的水平相比差异无显著性意义(t=2.164,P > 0.05)。说明纤维蛋白1基因在有/无β-淀粉样蛋白异常沉积的两种阿尔茨海默病动物模型中均发挥了作用,提示纤维蛋白1基因在阿尔茨海默病的发生发展中可能还存在非β-淀粉样蛋白相关的作用机制。"
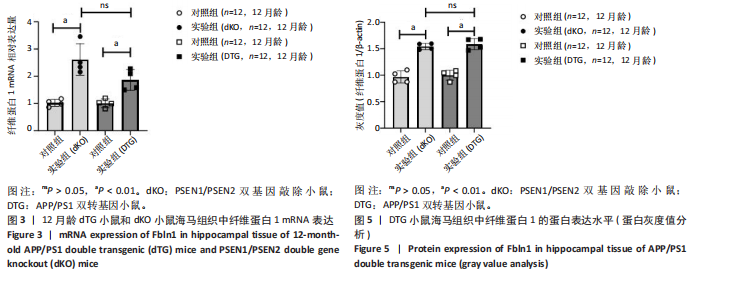
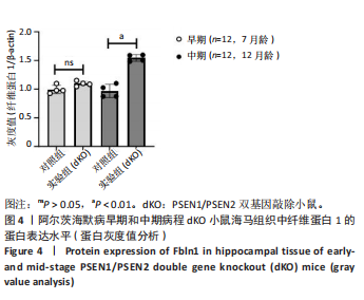
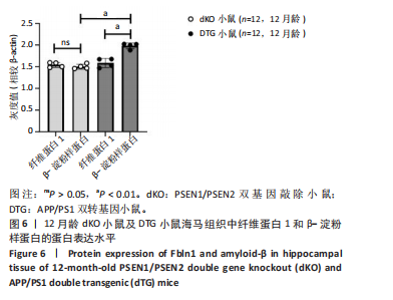
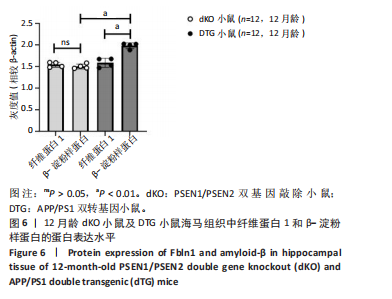
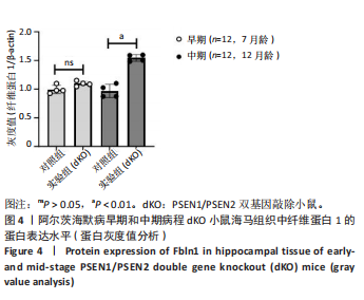
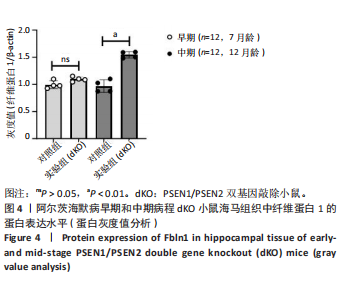
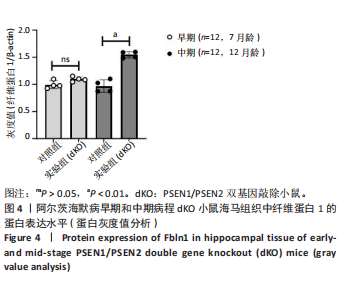
2.5 Western blot检测纤维蛋白1基因在7月龄(早期阿尔茨海默病)及12月龄(中期阿尔茨海默病)dKO小鼠及DTG小鼠海马组织中的蛋白表达情况 7月龄dKO小鼠海马组织中纤维蛋白1蛋白水平与同龄野生型小鼠相比差异无显著性意义(t=2.380,P > 0.05 ),12月龄dKO小鼠海马组织中纤维蛋白1蛋白水平升高,显著高于同龄野生型小鼠(t=8.985,P < 0.01),见图4。12月龄DTG小鼠表现出纤维蛋白1蛋白水平显著高于对照组(t=8.392,P < 0.01),见图5。以上均与mRNA表达水平相对应。最后,比较了12月龄dKO与DTG两种阿尔茨海默病模型小鼠纤维蛋白1蛋白的表达水平,差异无显著性意义(t=0.736,P > 0.05)。同样提示纤维蛋白1蛋白水平在有/无β-淀粉样蛋白异常沉积的两种阿尔茨海默病动物模型中也发挥了作用。"
| [1] SCHELTENS P, DE STROOPER B, KIVIPELTO M, et al. Alzheimer’s disease. Lancet. 2021;397(10284):1577-1590. [2] GRAHAM WV, BONITO-OLIVA A, SAKMAR TP. Update on Alzheimer’s Disease Therapy and Prevention Strategies. Annu Rev Med. 2017;68:413-430. [3] ZHANG F, ZHONG RJ, CHENG C, et al. New therapeutics beyond amyloid-β and tau for the treatment of Alzheimer’s disease. Acta Pharmacol Sin. 2021;42(9):1382-1389. [4] PLEEN J, TOWNLEY R. Alzheimer’s disease clinical trial update 2019-2021. J Neurol. 2022;269(2):1038-1051. [5] MIRNICS K, NORSTROM EM, GARBETT K, et al. Molecular signatures of neurodegeneration in the cortex of PSEN1/PSEN2 double knockout mice. Mol Neurodegener. 2008;3:14. [6] SAURA CA, CHOI SY, BEGLOPOULOS V, et al. Loss of presenilin function causes impairments of memory and synaptic plasticity followed by age-dependent neurodegeneration. Neuron. 2004;42(1):23-36. [7] GUÉNETTE SY, TANZI RE. Progress toward valid transgenic mouse models for Alzheimer’s disease. Neurobiol Aging. 1999;20(2):201-211. [8] FENG R, RAMPON C, TANG YP, et al. Deficient neurogenesis in forebrain-specific presenilin-1 knockout mice is associated with reduced clearance of hippocampal memory traces. Neuron. 2001;32(5):911-926. [9] WINES-SAMUELSON M, SCHULTE EC, SMITH MJ, et al. Characterization of age-dependent and progressive cortical neuronal degeneration in presenilin conditional mutant mice. PLoS One. 2010;5(4):e10195. [10] NIKOLAC PERKOVIC M, VIDETIC PASKA A, KONJEVOD M, et al. Epigenetics of Alzheimer’s Disease. Biomolecules. 2021;11(2):195. [11] LOK K, ZHAO H, SHEN H, et al. Characterization of the APP/PS1 mouse model of Alzheimer’s disease in senescence accelerated background. Neurosci Lett. 2013;557 Pt B:84-89. [12] ZHANG SS, ZHU L, PENG Y, et al. Long-term running exercise improves cognitive function and promotes microglial glucose metabolism and morphological plasticity in the hippocampus of APP/PS1 mice. J Neuroinflammation. 2022;19(1):34. [13] 唐晓琴,唐红,邓飞,等. 阿尔茨海默病小鼠模型海马组织Atp5a1基因甲基化改变[J]. 中国医药科学,2016,6(13):24-27. [14] VIÑA J, SANZ-ROS J. Alzheimer’s disease: Only prevention makes sense. Eur J Clin Invest. 2018;48(10):e13005. [15] QIU W, LIU H, LIU Y, et al. Regulation of beta-amyloid for the treatment of Alzheimer’s disease: Research progress of therapeutic strategies and bioactive compounds. Med Res Rev. 2023;43(4):1091-1140. [16] TREJO-LOPEZ JA, YACHNIS AT, PROKOP S. Neuropathology of Alzheimer’s Disease. Neurotherapeutics. 2022;19(1):173-185. [17] DE JAGER PL, SRIVASTAVA G, LUNNON K, et al. Alzheimer’s disease: early alterations in brain DNA methylation at ANK1, BIN1, RHBDF2 and other loci. Nat Neurosci. 2014;17:1156-1163. [18] ZHUANG B, MANCARCI BO, TOKER L, et al. Mega-Analysis of Gene Expression in Mouse Models of Alzheimer’s Disease. eNeuro. 2019;6(6): ENEURO.0226-19.2019. [19] GUAN Y, GAJEWSKA J, FLORYSZAK-WIECZOREK J, et al. Histone (de)acetylation in epigenetic regulation of Phytophthora pathobiology. Mol Plant Pathol. 2024;25(7):e13497. [20] NANDHU MS, BEHERA P, BHASKARAN V, et al. Development of a function-blocking antibody against Fibulin-3 as a targeted reagent for glioblastoma. Clin Cancer Res. 2018;24(4):821-833. [21] SHEN H, ZHANG L, ZHOU J, et al. Epidermal growth factor-containing Fibulin-like extracellular matrix protein 1 (EFEMP1) acts as a potential diagnostic biomarker for prostate cancer. Medi Sci Moni. 2017;(23):216-222. [22] KANDA M, NOMOTO S, OKAMURA Y, et al. Promoter hypermethylation offibulin 1 gene is associated with tumor progression in hepatocellularcarcinoma. Mole Carcinogene. 2011;50(8):571. [23] BARDIN A, MOLL F, MARGUERON R, et al. Transcriptional and posttran-scriptional regulation of fibulin-1 by estrogens leads to differentialinduction of messenger ribonucleic acid variants in ovarian and breastcancer cells. Endocrinology. 2005;146(2):760-768. [24] KANDA M, NOMOTO S, OKAMURA Y, et al. Promoter hypermethylationof fibulin 1 gene is associated with tumor progression in hepatocellularcarcinoma. Mol Carcinog. 2011;50(8): 571-579. [25] WU BJ, ZHOU ZP, LI WP, et al. Abnormal hypermethylation andclinicopathological significance of FBLN1 gene in cutaneousmelanoma. Tumour Biol. 2014;35(1):123-127. [26] 王静,赵亮.Fibulin-1基因启动子区域甲基化与食管癌的关系研究[J].临床与病理杂志,2017,37(1):133-136. [27] OHSAWA I, TAKAMURA C, KOHSAKA S. Fibulin-1 binds the amino-terminal head of beta-amyloid precursor protein and modulates its physiological function. J Neurochem. 2001;76(5):1411-1420. [28] KATZEFF JS, BRIGHT F, LO K, et al. Altered serum protein levels in frontotemporal dementia and amyotrophic lateral sclerosis indicate calcium and immunity dysregulation. Sci Rep. 2020;10(1):13741. [29] SHEN L, LIAO L, CHEN C, et al. Proteomics Analysis of Blood Serums from Alzheimer’s Disease Patients Using iTRAQ Labeling Technology. J Alzheimers Dis. 2017;56(1):361-378. [30] COLLINS JM, KING AE, WOODHOUSE A, et al. Age Moderates the Effects of Traumatic Brain Injury on Beta-Amyloid Plaque Load in APP/PS1 Mice. J Neurotrauma. 2019;36(11):1876-1889. [31] DONG Y, LI X, CHENG J, et al. Drug Development for Alzheimer’s Disease: Microglia Induced Neuroinflammation as a Target? Int J Mol Sci. 2019; 20(3):558. [32] SHIRAI M, NARA T, TAKAHASHI H, et al. Identification of aberrant transcription termination at specific gene loci with DNA hypomethylated transcription termination sites caused by DNA methyltransferase deficiency. Genes Genet Syst. 2022;97(3):139-152. [33] DIISATHAM J, BRENNAN L, JIAO X, et al. Changes in DNA methylation hallmark alterations in chromatin accessibility and gene expression for eye lens differentiation. Epigenetics Chromatin. 2022;15(1):8. [34] DAVIE JR, SATTARIFARD H, SUDHAKAR SRN, et al. Basic Epigenetic Mechanisms. Subcell Biochem. 2025;108:1-49. [35] PALSTRØM NB, ROJEK AM, MØLLER HEH, et al. Classification of Amyloidosis by Model-Assisted Mass Spectrometry-Based Proteomics. Int J Mol Sci. 2021;23(1):319. [36] STAROST MF, MURAKAMI T, VAUGHAN KL, et al. Identification of epidermal growth factor-containing fibulin-like extracellular matrix protein 1-derived amyloid deposition in a rhesus macaque. Amyloid. 2025;32(1):84-86. [37] KATAGIRI F, UEO D, OKUBO-GUNGE Y, et al. Fibulin-4 Accelerates Amyloid Formation by Binding with a Keratin 5 Peptide Fragment. JID Innov. 2022;2(3):100114. |
| [1] | Liu Huan, Zeng Shaopeng, Chen Jun, He Linqian, Yang Ying, Zhang Jing. Aging-related dysregulation of glucose metabolism: crossroads of cancer and neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1527-1538. |
| [2] | Hu Jing, Zhu Ling, Xie Juan, Kong Deying, Liu Doudou. Autophagy regulates early embryonic development in mice via affecting H3K4me3 modification [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1147-1155. |
| [3] | He Qiwang, , , Chen Bo, Liang Fuchao, Kang Zewei, Zhou Yuan, Ji Anxu, Tang Xialin, . Relationship between Alzheimer’s disease and sarcopenia and body mass index: analysis of GWAS datasets for European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1036-1046. |
| [4] | Xu Dongfang, Zhao Kun, Lu Changzhu, Wang Yuge, Bai Lianjie, Meng Fanmou, Wang Yang, , Yao Hongbo. m6A-related ferroptosis gene expression and its association with immune infiltration in Alzheimer’s disease: machine learning and molecular biology validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6421-6432. |
| [5] | Zhang Xiaomin, Du Pengyang, Zhang Xiuping, Xue Guofang. Potential targets of glucagon-like peptide 1 receptor agonist ticagrelor in the treatment of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6122-6133. |
| [6] | Hu Yalin, Huang Fengqin, Yang Boyin, Luo Xingmei. Transcription factor EB improves Alzheimer’s disease via the autophagy-lysosome pathway [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5844-5858. |
| [7] | Pei Xiaxia, Li Tian, Zhang Yanli, Gao Yanping, Su Qiang. A novel treadmill-based method for assessing learning and memory in mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4694-4701. |
| [8] | Liu Annan, Li Jianhui, Gao Wei, Li Xue, Song Jing, Xing Liping, Li Honglin. Bibliometric analysis of ferroptosis and Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4278-4288. |
| [9] | Xie Liugang, Cui Shuke, Guo Nannan, Li Aoyu, Zhang Jingrui. Research hotspots and frontiers of stem cells for Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1475-1485. |
| [10] |
Li Tian, Ren Yuhua, Gao Yanping, Su Qiang.
Mechanism of agomelatine alleviating anxiety- and depression-like behaviors in APP/PS1 transgenic mice #br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2025, 29(6): 1176-1182.
|
| [11] | He Yang, Tang Buyuan, Lu Changhuai. Molecular mechanisms of ligament flavum hypertrophy: analysis based on methylation sequencing and transcriptome integration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 1013-1020. |
| [12] | Su Qin, Jia Siwei, Guo Minfang, Meng Tao, Li Yanbing, Mu Bingtao, Song Lijuan, Ma Cungen, Yu Jiezhong. Lycium barbarum polysaccharide intervenes in SH-SY5Y cell injury induced by beta-amyloid protein 1-42: protective effect of mitochondrial autophagy [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(31): 6688-6696. |
| [13] | He Ningjuan, Li Li, Wang Su, Yang Jianshe, Lei Siyun, Wang Yang. Effects of aerobic or resistance exercise on hippocampal ras/Drebrin dendritic spine plasticity in a mouse model of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(26): 5528-5535. |
| [14] | Shen Zilong, Wu Mingjie, Chen Xiaojing, Zhou Xibin, Zhou Chunxiang. An experimental method for simultaneously extracting the dura mater and deep cervical lymph nodes [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(26): 5543-5548. |
| [15] | Nan Songhua, Peng Chaojie, Cui Yinglin. Mitochondrial dysfunction and brain aging: a bibliometrics analysis based on the Web of Science Core Collection database [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(26): 5642-5651. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||