Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (24): 6231-6238.doi: 10.12307/2026.168
Previous Articles Next Articles
Constructing an in vitro model of ulcerative colitis in mice based on organoid technology
Zhou Li1, Li Rui2, Chen Hao2, 3, Chen Jiaqi4, Liu Yuhong2, Wu Na4, 5
- 1School of Computing, 2School of Pharmacy, 4School of Clinical Medicine, Jiangxi University of Chinese Medicine, Nanchang 330004, Jiangxi Province, China; 3Department of Pharmacy, 5Department of Gastroenterology, Affiliated Hospital of Jiangxi University of Chinese Medicine, Nanchang 330006, Jiangxi Province, China
-
Received:2025-04-03Revised:2025-08-05Online:2026-08-28Published:2026-01-30 -
Contact:Wu Na, PhD, Associate chief physician, Associate professor, School of Clinical Medicine, Jiangxi University of Chinese Medicine, Nanchang 330004, Jiangxi Province, China; Department of Gastroenterology, Affiliated Hospital of Jiangxi University of Chinese Medicine, Nanchang 330006, Jiangxi Province, China -
About author:Zhou Li, MS, Lecturer, School of Computing, Jiangxi University of Chinese Medicine, Nanchang 330004, Jiangxi Province, China -
Supported by:National Natural Science Foundation of China, Nos. 82405409 and 82160903 (both to WN); Jiangxi Provincial Natural Science Foundation (General Project), No. 20242BAB25556 (to WN); Jiangxi Provincial Traditional Chinese Medicine Mid-Career and Young Talent Cultivation Program (Fourth Batch), No. [2022]7 (to WN); Jiangxi University of Chinese Medicine Key Discipline Construction Fund-Supported Project, No. 2024jzzdxk007 (to ZL); Jiangxi Provincial Department of Science and Technology Supported Key Laboratory Autonomous Project, No. 2023QGZDSYS001 (to CH [project participant])
CLC Number:
Cite this article
Zhou Li, Li Rui, Chen Hao, Chen Jiaqi, Liu Yuhong, Wu Na. Constructing an in vitro model of ulcerative colitis in mice based on organoid technology[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6231-6238.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
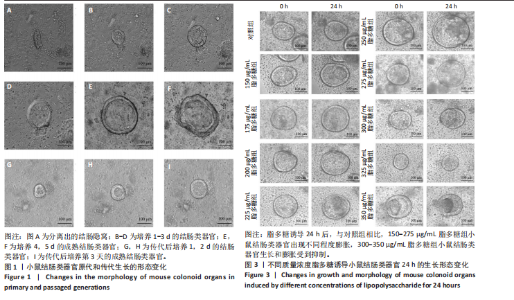
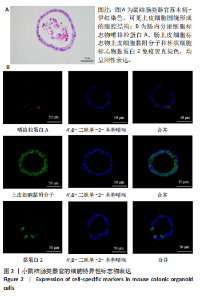
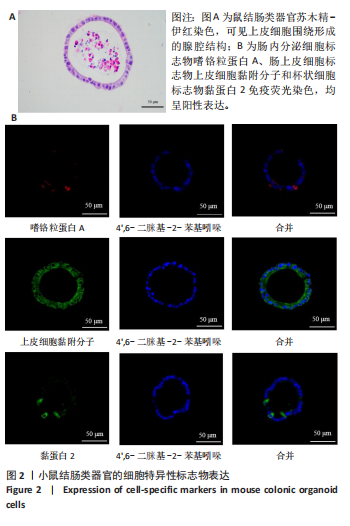
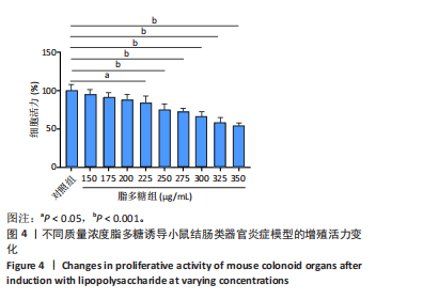
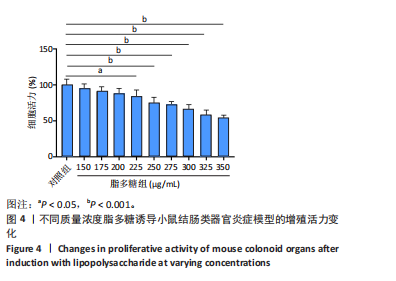
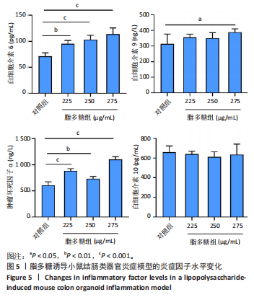
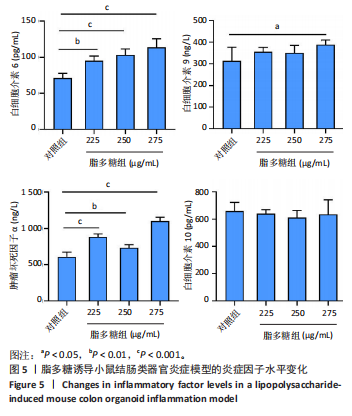
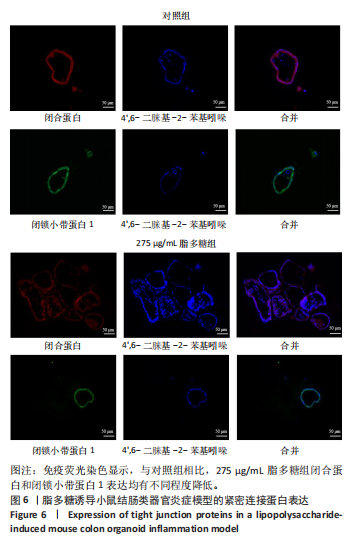
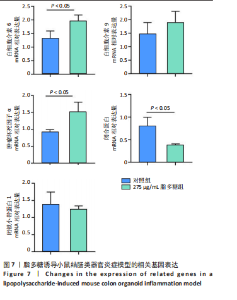
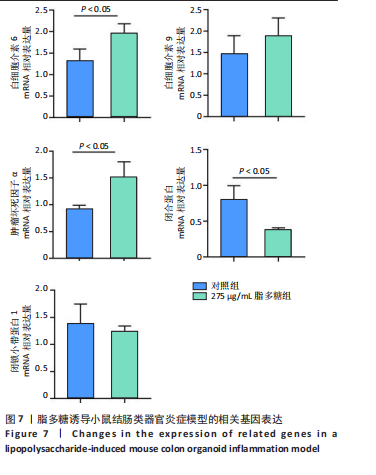
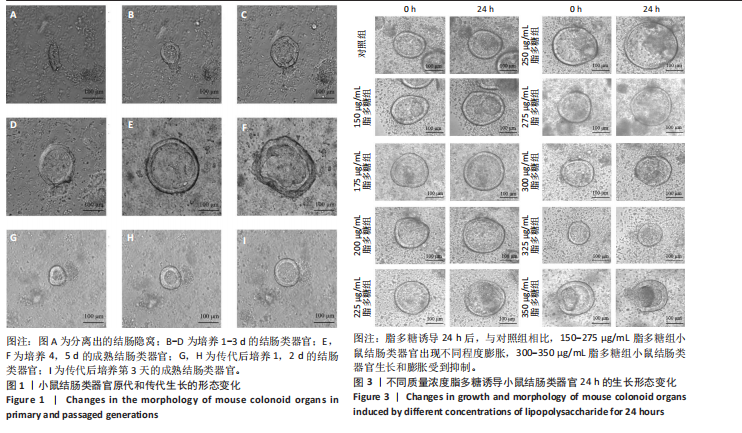
2.1 小鼠结肠类器官稳定培养体系的建立 种板后在显微镜下观察到成活的结肠隐窝,培养1-3 d能看到长条发夹样的隐窝变为闭合逐渐增大的圆球状,轮廓逐渐清晰,四五天内部类似肠腔的中空结构也逐渐增大并形成成熟的结肠类器官,随着结肠类器官的不断生长,中间肠腔会逐渐出现脱落坏死的细胞并开始走向死亡趋势,颜色也会逐渐变黑;传代后的结肠类器官在第3天会重新长成成熟的结肠类器官,此时可用于后续实验,见图1。 2.2 小鼠结肠类器官的鉴定 苏木精-伊红染色结果显示,小鼠结肠类器官中可见上皮细胞围绕形成的腺腔结构,表明已成功构建由单层极化柱状上皮细胞构成的结肠类器官,见图2A。免疫荧光染色结果显示,小鼠结肠类器官表达肠内分泌细胞标志物嗜铬粒蛋白A、肠上皮细胞标志物上皮细胞黏附分子和杯状细胞标志物黏蛋白2,见图2B。 2.3 脂多糖诱导小鼠结肠类器官炎症模型的生长形态变化 显微镜下可见,与对照组相比,150-275 μg/mL 脂多糖组小鼠结肠类器官出现不同程度膨胀,300-350 μg/mL脂多糖组小鼠结肠类器官生长和膨胀受到抑制,见图3。 2.4 脂多糖诱导小鼠结肠类器官炎症模型的增殖活力 CCK-8检测结果显示,与对照组相比,150-350 μg/mL 脂多糖组小鼠结肠类器官增殖活力出现不同程度降低,其中225-350 μg/mL脂多糖组小鼠结肠类器官增殖活力降低有显著性意义(P < 0.05,P < 0.001),见图4。 2.5 脂多糖诱导结肠类器官炎症模型的炎症因子水平检测结果 与对照组相比,225-275 μg/mL 脂多糖组促炎因子白细胞介素6、肿瘤坏死因子α和白细胞介素9水平均有不同程度升高,抗炎因子白细胞介素10水平有不同程度降低趋势,见图5。 与对照组相比,225-275 μg/mL脂多糖组白细胞介素6和肿瘤坏死因子α水平升高(P < 0.01,P < 0.001),275 μg/mL脂多糖组白细胞介素9水平升高(P < 0.05),225,250 μg/mL 脂多糖组白细胞介素9水平无明显变化(P > 0.05),4组间白细胞介素10水平比较差异无显著性意义(P > 0.05)。 2.6 脂多糖诱导结肠类器官炎症模型的上皮屏障功能检测结果 免疫荧光染色结果显示,与对照组相比,275 μg/mL 脂多糖组闭合蛋白和闭锁小带蛋白1表达均有不同程度降低,说明小鼠结肠上皮屏障的结构和功能有不同程度损坏,见图6。 2.7 脂多糖诱导结肠类器官炎症模型的相关基因表达检测结果 qPCR检测结果显示,与对照组相比,275 μg/mL脂多糖组白细胞介素6和肿瘤坏死因子α mRNA表达升高(P < 0.05),闭合蛋白mRNA表达降低(P < 0.05),白细胞介素9与闭锁小带蛋白1mRNA表达无明显变化(P > 0.05),见图7。"
| [1] MARSOOL MDM, VORA N, MARSOOL ADM, et al. Ulcerative colitis: Addressing the manifestations, the role of fecal microbiota transplantation as a novel treatment option and other therapeutic updates. Dis Mon. 2023;69(11):101606. [2] LE BERRE C, HONAP S, PEYRIN-BIROULET L. Ulcerative colitis. Lancet. 2023;402(10401):571-584. [3] ZHANG C, ZHANG J, ZHANG Y, et al. Identifying neutrophil-associated subtypes in ulcerative colitis and confirming neutrophils promote colitis-associated colorectal cancer. Front Immunol. 2023;14:1095098. [4] DU L, HA C. Epidemiology and Pathogenesis of Ulcerative Colitis. Gastroenterol Clin North Am. 2020;49(4):643-654. [5] KRUGLIAK CLEVELAND N, TORRES J, RUBIN DT. What Does Disease Progression Look Like in Ulcerative Colitis, and How Might It Be Prevented? Gastroenterology. 2022;162(5):1396-1408. [6] GAJENDRAN M, LOGANATHAN P, JIMENEZ G, et al. A comprehensive review and update on ulcerative colitis. Dis Mon. 2019;65(12):100851. [7] NAKASE H, SATO N, MIZUNO N, et al. The influence of cytokines on the complex pathology of ulcerative colitis. Autoimmun Rev. 2022; 21(3):103017. [8] WEI J, CHEN C, FENG J, et al. MUC2 mucin O-glycosylation interacts with enteropathogenic Escherichia coli to influence the development of ulcerative colitis based on the NF-kB signaling pathway. J Transl Med. 2023;21(1):793. [9] KOBAYASHI T, SIEGMUND B, LE BERRE C, et al. Ulcerative colitis. Nat Rev Dis Primers. 2020;6(1):74. [10] GROS B, KAPLAN GG. Ulcerative Colitis in Adults: A Review. JAMA. 2023; 330(10):951-965. [11] FUDMAN DI, MCCONNELL RA, HA C, et al. Modern Advanced Therapies for Inflammatory Bowel Diseases: Practical Considerations and Positioning. Clin Gastroenterol Hepatol. 2025;23(3):454-468. [12] YOO JH, DONOWITZ M. Intestinal enteroids/organoids: A novel platform for drug discovery in inflammatory bowel diseases. World J Gastroenterol. 2019;25(30):4125-4147. [13] LANNAGAN TR, JACKSTADT R, LEEDHAM SJ, et al. Advances in colon cancer research: in vitro and animal models. Curr Opin Genet Dev. 2021;66:50-56. [14] WATANABE S, KOBAYASHI S, OGASAWARA N, et al. Transplantation of intestinal organoids into a mouse model of colitis. Nat Protoc. 2022; 17(3):649-671. [15] BOBER Z, AEBISHER D, OLEK M, et al. Multiple Cell Cultures for MRI Analysis. Int J Mol Sci. 2022;23(17):10109. [16] XIANG T, WANG J, LI H. Current applications of intestinal organoids: a review. Stem Cell Res Ther. 2024;15(1):155. [17] JENSEN LH, ROGATTO SR, LINDEBJERG J, et al. Precision medicine applied to metastatic colorectal cancer using tumor-derived organoids and in-vitro sensitivity testing: a phase 2, single-center, open-label, and non-comparative study. J Exp Clin Cancer Res. 2023;42(1):115. [18] YUAN M, JIN T, WU J, et al. IAA-miR164a-NAC100L1 module mediates symbiotic incompatibility of cucumber/pumpkin grafted seedlings through regulating callose deposition. Hortic Res. 2023;11(2):uhad287. [19] XU T, LI X, ZHAO W, et al. SF3B3-regulated mTOR alternative splicing promotes colorectal cancer progression and metastasis. J Exp Clin Cancer Res. 2024;43(1):126. [20] 雷颖,徐恩伟,白中元,等.利用小鼠结肠类器官预测化疗药物肠道不良反应的研究[J].中国肿瘤临床,2024,51(9):447-453. [21] LUO H, GUO M, LI M, et al. Protective Effect of Rosavin Against Intestinal Epithelial Injury in Colitis Mice and Intestinal Organoids. J Inflamm Res. 2024;17:6023-6038. [22] SAYED IM, EI-HAFEEZ AAA, MAITY PP, et al. Modeling colorectal cancers using multidimensional organoids. Adv Cancer Res. 2021;151:345-383. [23] ALIPOURGIVI F, MOTOLANI A, QIU AY, et al. Genetic Alterations of NF-κB and Its Regulators: A Rich Platform to Advance Colorectal Cancer Diagnosis and Treatment. Int J Mol Sci. 2023;25(1):154. [24] DOU Y, PIZARRO T, ZHOU L. Organoids as a Model System for Studying Notch Signaling in Intestinal Epithelial Homeostasis and Intestinal Cancer. Am J Pathol. 2022;192(10):1347-1357. [25] MARTINEZ-SILGADO A, YOUSEF YENGEJ FA, PUSCHHOF J, et al. Differentiation and CRISPR-Cas9-mediated genetic engineering of human intestinal organoids. STAR Protoc. 2022;3(3):101639. [26] SUGIMOTO S, KOBAYASHI E, KANAI T, et al. In Vivo Intestinal Research Using Organoid Transplantation. Keio J Med. 2022;71(4):73-81. [27] 陈嘉琪,史秀丽,黄雪云,等.结肠类器官培养体系的构建研究进展[J].实用医学杂志,2023,39(17):2159-2163. [28] IZADPARAST F, RIAHI-ZAJANI B, YARMOHAMMADI F, et al. Protective effect of berberine against LPS-induced injury in the intestine: a review. Cell Cycle. 2022;21(22):2365-2378. [29] LI Q, VON EHRLICH-TREUENSTATT V, SCHARDEY J, et al. Gut Barrier Dysfunction and Bacterial Lipopolysaccharides in Colorectal Cancer. J Gastrointest Surg. 2023;27(7):1466-1472. [30] ZHU H, TONG S, CUI Y, et al. Tanshinol alleviates ulcerative colitis by promoting the expression of VLDLR. Drug Dev Res. 2021;82(8): 1258-1268. [31] IZADPARAST F, RIAHI-ZAJAN B, YARMOHAMMADI F, et al. Protective effect of berberine against LPS-induced injury in the intestine: a review. Cell Cycle. 2022;21(22):2365-2378. [32] DENG L, HE S, LI Y, et al. Identification of Lipocalin 2 as a Potential Ferroptosis-related Gene in Ulcerative Colitis. Inflamm Bowel Dis. 2023; 29(9):1446-1457. [33] BROOKS P, ZUR BRUEGGE T, BOYLE EC, et al. CD14 and ALPK1 Affect Expression of Tight Junction Components and Proinflammatory Mediators upon Bacterial Stimulation in a Colonic 3D Organoid Model. Stem Cells Int. 2020;2020:4069354. [34] SOUZA RF, CAETANO MAF, MAGALHAES HIR, et al. Study of tumor necrosis factor receptor in the inflammatory bowel disease. World J Gastroenterol. 2023;29(18):2733-2746. [35] YAO D, DAI W, DONG M, et al. MUC2 and related bacterial factors: Therapeutic targets for ulcerative colitis. EBioMedicine. 2021;74:103751. [36] CANDELLI M, FRANZA L, PIGNATARO G, et al. Interaction between Lipopolysaccharide and Gut Microbiota in Inflammatory Bowel Diseases. Int J Mol Sci. 2021;22(12):6242. [37] SHAHINI A, SHAHINI A. Role of interleukin-6-mediated inflammation in the pathogenesis of inflammatory bowel disease: focus on the available therapeutic approaches and gut microbiome. J Cell Commun Signal. 2023;17(1):55-74. [38] ZHAO Y, LUAN H, JIANG H, et al. Gegen Qinlian decoction relieved DSS-induced ulcerative colitis in mice by modulating Th17/Treg cell homeostasis via suppressing IL-6/JAK2/STAT3 signaling. Phytomedicine. 2021;84:153519. [39] HIRANO T. IL-6 in inflammation, autoimmunity and cancer. Int Immunol. 2021;33(3):127-148. [40] SCHREIBER S, ADEN K, BERNARDES JP, et al. Therapeutic Interleukin-6 Trans-signaling Inhibition by Olamkicept (sgp130Fc) in Patients With Active Inflammatory Bowel Disease. Gastroenterology. 2021;160(7):2354-2366.e11. [41] SON A, BARAL I, FALDUTO GH, et al . Locus of (IL-9) control: IL9 epigenetic regulation in cellular function and human disease. Exp Mol Med. 2024; 56(6):1331-1339. [42] BICK F, BLANCHETOT C, LAMBRECHT BN, et al. A reappraisal of IL-9 in inflammation and cancer. Mucosal Immunol. 2025;18(1):1-15. [43] TIAN L, LI Y, ZHANG J, et al. IL-9 promotes the pathogenesis of ulcerative colitis through STAT3/SOCS3 signaling. Biosci Rep. 2018;38(6):1-11. [44] MATUSIEWICZ M, NEUBAUER K, BEDNARZ-MISA I, et al. Systemic interleukin-9 in inflammatory bowel disease: Association with mucosal healing in ulcerative colitis. World J Gastroenterol. 2017;23(22):4039-4046. [45] KANG ZP, JIN J, JIANG QQ,et al. [Effect of Sishen Pills and its split prescriptions on Tfr/Tfh9/Tfh17 cells in colitis mice]. Zhongguo Zhong Yao Za Zhi. 2022;47(5):1300-1306. [46] TATIYA-APHIRADEE N, CHATUPHONPRASERT W, JARUKAMJORN K. Immune response and inflammatory pathway of ulcerative colitis. J Basic Clin Physiol Pharmacol. 2018;30(1):1-10. [47] NASCIMENTO RPD, MACHADO APDF, GALVEZ J, et al. Ulcerative colitis: Gut microbiota, immunopathogenesis and application of natural products in animal models. Life Sci. 2020;258:118129. [48] LIU C, YAN X, ZHANG Y, et al. Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for murine colitis therapy. J Nanobiotechnology. 2022;20(1):206. [49] CUI L, GUAN X, DING W, et al. Scutellaria baicalensis Georgi polysaccharide ameliorates DSS-induced ulcerative colitis by improving intestinal barrier function and modulating gut microbiota. Int J Biol Macromol. 2021;166:1035-1045. [50] WU J, NIU J, LI M, et al. Keratin 1 maintains the intestinal barrier in ulcerative colitis. Genes Genomics. 2021;43(12):1389-1402. [51] ZHOU W, ZHANG H, HUANG L, et al. Disulfiram with Cu2+ alleviates dextran sulfate sodium-induced ulcerative colitis in mice. Theranostics. 2023;13(9):2879-2895. [52] YU X, LI X, XU Y, et al. Resveratrol ameliorates ulcerative colitis by upregulating Nrf2/HO1 pathway activity: Integrating animal experiments and network pharmacology. Mol Med Rep. 2024;29(5):77. [53] KUO WT, ODENWALD MA, TURNER JR, et al. Tight junction proteins occludin and ZO-1 as regulators of epithelial proliferation and survival. Ann N Y Acad Sci. 2022;1514(1):21-33. [54] CAPALDO CT. Claudin Barriers on the Brink: How Conflicting Tissue and Cellular Priorities Drive IBD Pathogenesis. Int J Mol Sci. 2023; 24(10):8562. [55] ABDULQADIR R, ENGERS J, AL-SADI R. Role of Bifidobacterium in Modulating the Intestinal Epithelial Tight Junction Barrier: Current Knowledge and Perspectives. Curr Dev Nutr. 2023;7(12):102026. [56] ARUMUGAM P, SAHA K, NIGHOT P. Intestinal Epithelial Tight Junction Barrier Regulation by Novel Pathways. Inflamm Bowel Dis. 2025;31(1):259-271. [57] LI H, YE XF, SU YS, et al. Mechanism of Acupuncture and Moxibustion on Promoting Mucosal Healing in Ulcerative Colitis. Chin J Integr Med. 2023;29(9):847-856. [58] HUANG S, ZHANG S, CHEN L, et al. Lipopolysaccharide induced intestinal epithelial injury: a novel organoids-based model for sepsis in vitro. Chin Med J (Engl). 2022;135(18):2232-2239. [59] KATSANDEGWAZA B, HORSNELL W, SMITH K. Inflammatory Bowel Disease: A Review of Pre-Clinical Murine Models of Human Disease. Int J Mol Sci. 2022;23(16):9344. [60] EICHELE DD, KHARBANDA KK. Dextran sodium sulfate colitis murine model: An indispensable tool for advancing our understanding of inflammatory bowel diseases pathogenesis. World J Gastroenterol. 2017;23(33):6016-6029. [61] WALTER J, ARMET AM, FINLAY BB, et al. Establishing or Exaggerating Causality for the Gut Microbiome: Lessons from Human Microbiota-Associated Rodents. Cell. 2020;180(2):221-232. [62] ALMEQDADI M, MANA MD, ROPER J, et al. Gut organoids: mini-tissues in culture to study intestinal physiology and disease. Am J Physiol Cell Physiol. 2019;317(3):C405-C419. |
| [1] | Cai Ziming, Yu Qinghe, Ma Pengfei, Zhang Xin, Zhou Longqian, Zhang Chongyang, Lin Wenping. Heme oxygenase-1 alleviates lipopolysaccharide-induced inflammatory response in nucleus pulposus mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1624-1631. |
| [2] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [3] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [4] | Chen Ju, Zheng Jinchang, Liang Zhen, Huang Chengshuo, Lin Hao, Zeng Li. Effect and mechanism of beta-caryophyllene in mice with osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1341-1347. |
| [5] | Zhu Kuicheng, Du Chunyan, Zhang Jintao. Mechanism by which hairless gene mutation promotes white adipose tissue browning in hairless mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1424-1430. |
| [6] | Hu Jing, Zhu Ling, Xie Juan, Kong Deying, Liu Doudou. Autophagy regulates early embryonic development in mice via affecting H3K4me3 modification [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1147-1155. |
| [7] | Sun Yajie, Zhao Xinchen, Bo Shuangling. Spatiotemporal expression of bone morphologic protein 7 in mouse kidney development [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1156-1161. |
| [8] | Wang Wei, Chen Jun, Jia Shaohui, Xue Xinxuan, Dong Kunwei. Mechanisms by which exercise regulates gut microbiota in the prevention and treatment of non-alcoholic fatty liver disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6328-6336. |
| [9] | Wang Yan, Lyu Hao, Hu Zhimu, Zhou Yao, Liu Qiang, Yang Yuxiang, Yi Hairu, Wang Jiuxiang, Jiang Ting. Intervention with Compound Kidney-Invigorating Granules in a mouse model of osteoporosis: role of the TRIB3/beta-catenin axis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6142-6149. |
| [10] | Zhang Ye, An Zheqing, Xi Xin, Liu Xiaoyan, Hong Wei, Liao Jian. Zoledronic acid-loaded dissolvable microneedle patch inhibits lipopolysaccharide-induced osteoclast differentiation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5115-5124. |
| [11] | Chen Ling, Mao Qiuhua, Xu Pu, Zhang Wenbo. Effect of water-soluble matrix of nano-pearl powder on proliferation, migration and apoptosis of mouse fibroblasts#br# [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 338-344. |
| [12] | Zhang Jiahao, Liu Jidong, Qu Yi, Wang Jianbo, Li Yang, Xue Yanan. Electroacupuncture improves inflammatory pathology and intestinal integrity in mice with ulcerative colitis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4663-4674. |
| [13] | Li Feifan, Zhang Yibo, Wang Jing, Zhu Jinqiang, Zheng Wenke. Comparison and evaluation of three methods for preparing insomnia mouse models [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4685-4693. |
| [14] | Tan Yuhang, Li Bo, Tang Minghong, Sun Zeyu, Luo Xu. Isolation, cultivation, identification, and induction of M1/M2 polarization in bone marrow-derived macrophages from C57BL/6 mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3233-3241. |
| [15] | She Xu, Li Xiaojiang, Huang Haixia, Wan Lingling, Luo Qingqing. Lipopolysaccharides regulate the function of human umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3280-3287. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||