Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (29): 4663-4670.doi: 10.12307/2024.523
Previous Articles Next Articles
Antibacterial magnesium oxide-calcium phosphate composite coating prepared by combining electrodeposition and sol-gel impregnation
Tan Junjie, Du Jiaheng, Wen Zhenyu, Yan Jiyuan, He Kui, Duan Ke, Yin Yiran, Li Zhong
- Sichuan Provincial Laboratory of Orthopedic Implant Device Research and Development and Application Technology Engineering, Department of Orthopedics, Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China
-
Received:2023-07-18Accepted:2023-10-20Online:2024-10-18Published:2024-03-22 -
Contact:Duan Ke, MD, Professor, Sichuan Provincial Laboratory of Orthopedic Implant Device Research and Development and Application Technology Engineering, Department of Orthopedics, Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China Yin Yiran, Master, Associate professor, Sichuan Provincial Laboratory of Orthopedic Implant Device Research and Development and Application Technology Engineering, Department of Orthopedics, Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China -
About author:Tan Junjie, Master candidate, Sichuan Provincial Laboratory of Orthopedic Implant Device Research and Development and Application Technology Engineering, Department of Orthopedics, Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China -
Supported by:Science and Technology Project of Sichuan Province, No. 2020YFS0455 (to YYR); Science and Technology Project of Sichuan Province, No. 2022YFS0628 (to YJY); Joint Project of Luzhou and Southwest Medical University, No. 2020LZXNYDZ08 (to DK); Production, Teaching and Research Project of Southwest Medical University, No. 2022CXY03 (to DK)
CLC Number:
Cite this article
Tan Junjie, Du Jiaheng, Wen Zhenyu, Yan Jiyuan, He Kui, Duan Ke, Yin Yiran, Li Zhong. Antibacterial magnesium oxide-calcium phosphate composite coating prepared by combining electrodeposition and sol-gel impregnation[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(29): 4663-4670.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
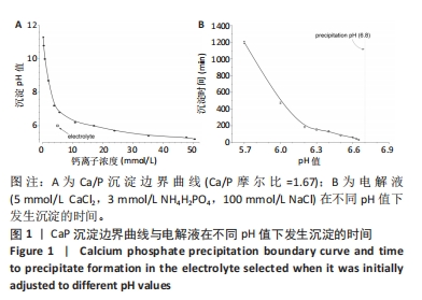
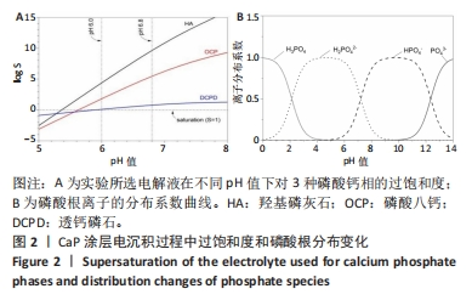
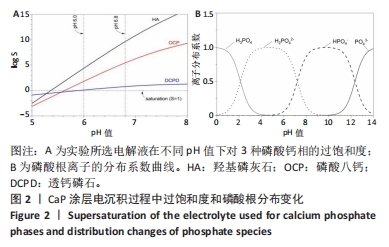
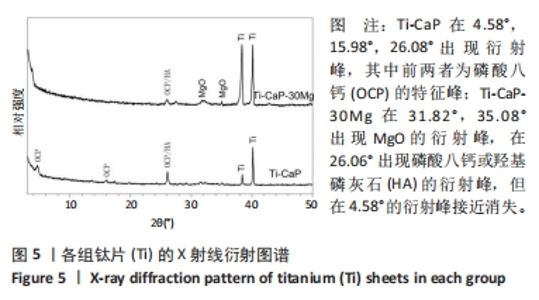
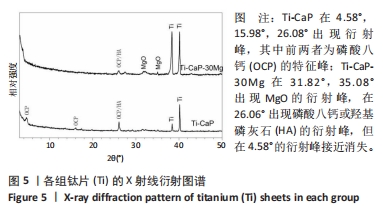
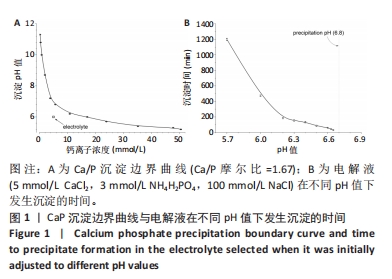
2.1 CaP沉积边界曲线 CaP沉淀边界曲线(图1A)代表在不同Ca2+浓度下发生沉淀时的pH值,该边界pH值随Ca2+浓度上升而下降;当Ca2+< 2 mmol/L时曲线陡峭,但斜率随Ca2+浓度上升而逐渐下降;当Ca2+ > 30 mmol/L,曲线平缓。该边界曲线代表电解液的临界过饱和度,它将溶液条件( pH值、Ca2+浓度)分为2个区域:当电解液条件位于曲线上方,电解液立刻发生沉淀;当电解液条件位于曲线以下,但充分接近该曲线(即离子积高于溶度积但低于临界过饱和度),电解液在较短时间内发生沉淀;当电解液pH值远低于沉淀边界时(如离子积低于溶度积),电解液在较长时间不发生沉淀。 根据该曲线,合理的电沉积电解液条件应位于沉淀边界以下,但距离边界适当距离使电解液不自发沉淀,但允许阴极反应产生的局部pH值上升使溶液条件跨越该边界。此外,当Ca2+浓度< 2 mmol/L,沉淀边界斜率大,表明微小Ca2+浓度变化会引起溶液中CaP沉淀pH值的较大变化,对电沉积的稳定性有不利影响。因此,实验选择电解液浓度为5 mmol/L CaCl2、3 mmol/L NH4H2PO4。实验结果发现当该电解液pH值距沉淀边界< 0.20时,电解液会在100 min内出现沉淀(图1B);当电解液pH远低于沉淀边界时(如pH值距边界> 1.00),电解液在较长时间(> 20 h)不发生沉淀。因此,电解液pH 值选择为6.00(图1A箭头示)。"
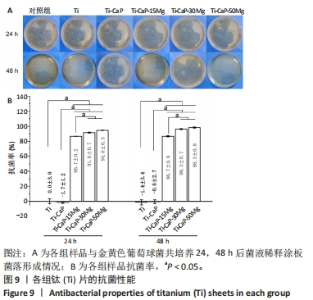
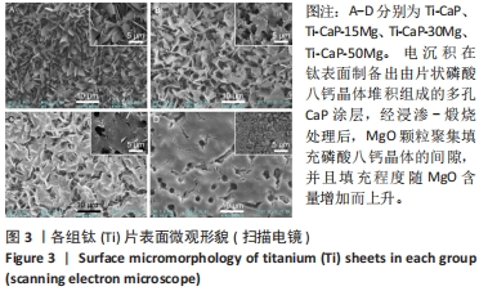
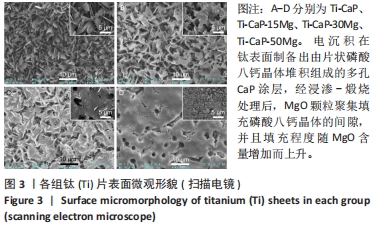
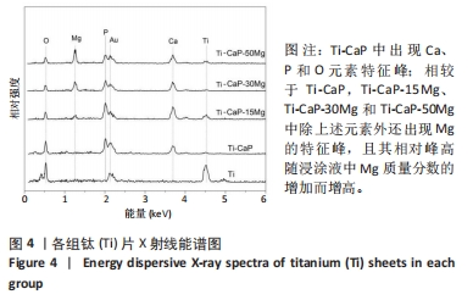
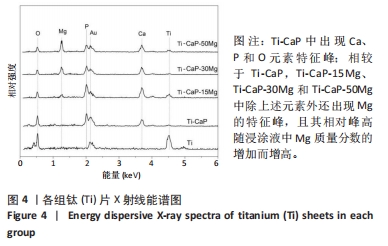
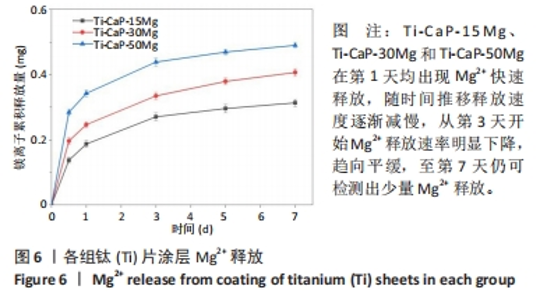
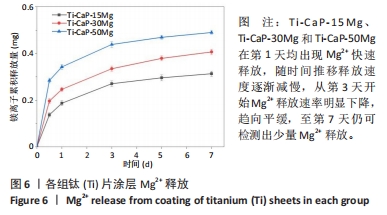
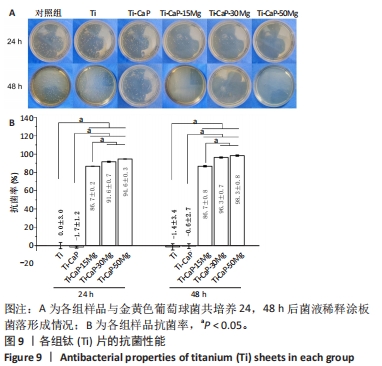
2.4 涂层体外抗菌性能评价结果 培养不同时间后各组平板上形成的菌落数,见图9A。培养24 h后平板上形成的菌落数由多到少为:Ti≈Ti-CaP > Ti-CaP-15Mg > Ti-CaP-30Mg > Ti-CaP-50Mg。纯钛组的抗菌率接近0,Ti-CaP组的抗菌率为(1.7±1.2)%,说明无抗菌性能;而3个复合涂层组的抗菌率均> 0.85,且随浸涂液Mg质量分数的增加而上升,见图9B。Ti-CaP-15Mg组、Ti-CaP-30Mg组、Ti-CaP-50Mg组抗菌率大于纯钛组、Ti-CaP(P均< 0.05);相比Ti-CaP-15Mg组,Ti-CaP-30Mg组抗菌率上升5.7%(P=0.01);相比Ti-CaP-30Mg组,Ti-CaP-50Mg组抗菌率上升3.3%,但差异无显著性意义(P=0.10)。 培养48 h后,纯钛、Ti-CaP仍然无抗菌性能,而3个复合涂层组的抗菌率仍均> 0.85,且随浸涂液Mg质量分数的增加而上升,见图9B。Ti-CaP-15Mg组、Ti-CaP-30Mg组、Ti-CaP-50Mg组抗菌率大于纯钛组、Ti-CaP(P均< 0.05);相比Ti-CaP-15Mg组,Ti-CaP-30Mg组抗菌率上升11.1%(P < 0.01);相比Ti-CaP-30Mg组,Ti-CaP-50Mg组抗菌率上升2.1%,但差异无显著性意义(P=0.63)。 "
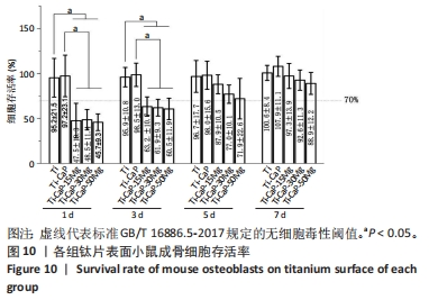
2.5 涂层细胞毒性实验结果 各组样品与小鼠成骨细胞培养后的CCK-8检测结果,见图10。共培养1 d,纯钛组和Ti-CaP组细胞存活率> 0.95,3个复合涂层组细胞存活率为0.46-0.49,且随浸涂液Mg质量分数的增加而下降,Ti-CaP-15Mg组、Ti-CaP-30Mg组和Ti-CaP-50Mg组细胞存活率低于纯钛组、Ti-CaP涂层钛片组(P均< 0.05)。共培养3 d,总体趋势与共培养1 d一致,但3个复合涂层组相比共培养1 d的细胞存活率略有上升。共培养5 d,3个复合涂层组细胞存活率明显上升,其中Ti-CaP-15Mg组达0.88,Ti-CaP-50Mg组达0.72,5组间细胞存活率比较差异无显著性意义(P > 0.05)。共培养7 d,3个复合涂层组细胞存活率均升至0.89-0.97,5组间细胞存活率比较差异无显著性意义(P > 0.05)。"
| [1] KUMAR ST, DEVI SP, KRITHIKA C, et al. Review of metallic biomaterials in dental applications. J Pharm Bioallied Sci. 2020;12(Suppl 1):S14. [2] ALVAND A, REZAPOOR M, PARVIZI J. The role of biomarkers for the diagnosis of implant-related infections in orthopaedics and trauma. Adv Exp Med Biol. 2017;971:69-79. [3] NANA A, NELSON SB, MCLAREN A, et al. What’s new in musculoskeletal infection: update on biofilms. J Bone Joint Surg Am. 2016;98(14): 1226-1234. [4] DONLAN RM. Biofilms: microbial life on surfaces. Emerg Infect Dis. 2002;8(9):881. [5] NARAYANAN R, SESHADRI SK, KWON TY, et al.Calcium phosphate-based coatings on titanium and its alloys. J Biomed Mater Res B Appl Biomater. 2008;85(1):279-299. [6] SUN L, BERNDT CC, GROSS KA, et al. Material fundamentals and clinical performance of plasma-sprayed hydroxyapatite coatings: a review. J Biomed Mater Res. 2001;58(5):570-592. [7] HAMAGAMI J, ATO Y, KANAMURA K. Fabrication of highly ordered macroporous apatite coating onto titanium by electrophoretic deposition method. Solid State Ion. 2004;172(1-4):331-334. [8] LOPEZ-HEREDIA MA, WEISS P, LAYROLLE P. An electrodeposition method of calcium phosphate coatings on titanium alloy. J Mater Sci Mater Med. 2007;18:381-390. [9] KNUDSEN MB, THILLEMANN JK, JØRGENSEN PB, et al. Electrochemically applied hydroxyapatite on the cementless porous surface of Bi-Metric stems reduces early migration and has a lasting effect: an efficacy trial of a randomized five-year follow-up radiostereometric study. Bone Joint J. 2022;104(6):647-656. [10] KARLOV AV, KHLUSOV IA, PONTAK VA, et al. Adhesion of Staphylococcus aureus to implants with different physicochemical characteristics. Bull Exp Biol Med. 2002;134:277-280. [11] VOGELY HC, OOSTERBOS CJM, PUTS EWA, et al. Effects of hydroxyapatite coating on Ti‐6 Al‐4V implant‐site infection in a rabbit tibial model. J Orthop Res. 2000;18(3):485-493. [12] LAZARINIS S, MÄKELÄ KT, ESKELINEN A, et al. Does hydroxyapatite coating of uncemented cups improve long-term survival? An analysis of 28,605 primary total hip arthroplasty procedures from the Nordic Arthroplasty Register Association (NARA). Osteoarthritis Cartilage. 2017;25(12):1980-1987. [13] ALTOMARE L, VISAI L, BLOISE N, et al. Electrochemically deposited gentamicin-loaded calcium phosphate coatings for bone tissue integration. Int J Artif Organs. 2012;35(10):876-883. [14] KOSE N, OTUZBIR A, PEKŞEN C, et al. A silver ion-doped calcium phosphate-based ceramic nanopowder-coated prosthesis increased infection resistance. Clin Orthop Relat Res. 2013;471:2532-2539. [15] PIERRE C, BERTRAND G, PAVY I, et al. Antibacterial Electrodeposited Copper-Doped Calcium Phosphate Coatings for Dental Implants. J Funct Biomater. 2022;14(1):20. [16] CHEN Z, MENG H, XING G, et al. Acute toxicological effects of copper nanoparticles in vivo. Toxicol Lett. 2006;163(2):109-120. [17] VORMANN J. Magnesium: nutrition and metabolism. Mol Aspects Med. 2003;24(1-3):27-37. [18] ARANCIBIA-HERNÁNDEZA YL, HERNÁNDEZ-CRUZA EY, PEDRAZA-CHAVERRIA J. Magnesium (Mg2+) Deficiency, Not Well-Recognized Non-Infectious Pandemic: Origin and Consequence of Chronic Inflammatory and Oxidative Stress-Associated Diseases. Cell Physiol Biochem. 2023;57(S1):1-23. [19] 中国居民膳食常量元素参考摄入量[C]//中国营养学会微量元素营养分会.中国营养学会微量元素营养第十二次学术会议暨第六届微量元素营养分会会员大会论文集.四川西昌,2014:164. [20] PATEL MK, ZAFARYAB M, RIZVI M, et al. Antibacterial and cytotoxic effect of magnesium oxide nanoparticles on bacterial and human cells. J Nanoeng Nanomfg. 2013;3(2):162-166. [21] SAWAI J, KOJIMA H, IGARASHI H, et al. Antibacterial characteristics of magnesium oxide powder. World J Microbiol Biotechnol. 2000;16:187-194. [22] BHATTACHARYA P, DEY A, NEOGI S. An insight into the mechanism of antibacterial activity by magnesium oxide nanoparticles. J Mater Chem B. 2021;9(26):5329-5339. [23] COELHO CC, ARAÚJO R, QUADROS PA, et al. Antibacterial bone substitute of hydroxyapatite and magnesium oxide to prevent dental and orthopaedic infections. Mater Sci Eng C Mater Biol Appl. 2019;97: 529-538. [24] 肖东琴,李文,段可,等.骨科植入金属材料表面电化学制备磷酸钙涂层方法及机理研究[J].西部医学,2023,35(8):1110-1116,1121. [25] 武汉大学.分析化学[M].6版.北京:高等教育出版社,2016. [26] RAMANUJAM K, SUNDRARAJAN M. Antibacterial effects of biosynthesized MgO nanoparticles using ethanolic fruit extract of Emblica officinalis. J Photochem Photobiol B. 2014;141:296-300. [27] 黄珂,杨伏良,陈力学,等.划痕法测定TiAlN涂层结合强度的研究[J].表面技术,2013,42(5):107-111. [28] 董自艳,戴翚,马仕洪,等.紫外-可见分光光度法快速确定细菌菌液的浓度[J].中国药品标准,2014(2):120-121. [29] YADAV P, SAXENA KK. Effect of heat-treatment on microstructure and mechanical properties of Ti alloys: An overview. Mater Today Proc. 2020;26:2546-2557. [30] DE ROOIJ JF, HEUGHEBAERT JC, NANCOLLAS GH. A pH study of calcium phosphate seeded precipitation. J Colloid Interface Sci. 1984;100(2): 350-358. [31] BROWN WE. Crystallographic and chemical relations between octacalcium phosphate and hydroxyapatite. Nature. 1962;196:1050-1055. [32] JEONG CH, KIM J, KIM HS, et al. Acceleration of bone formation by octacalcium phosphate composite in a rat tibia critical-sized defect. J Orthop Translat. 2022;37:100-112. [33] JIN H, JIA D, YANG Z, et al. Fabrication and properties of embedded dense coating on direct ink writing Si2N2O porous ceramics. Surf Coat Technol. 2020;394:125801. [34] SPIRANDELI BR, RIBAS RG, AMARAL SS, et al. Incorporation of 45S5 bioglass via sol-gel in β-TCP scaffolds: Bioactivity and antimicrobial activity evaluation. Mater Sci Eng C Mater Biol Appl. 2021;131:112453. [35] SUZUKI O, SHIWAKU Y, HAMAI R. Octacalcium phosphate bone substitute materials: Comparison between properties of biomaterials and other calcium phosphate materials. Dent Mater J. 2020;39(2): 187-199. [36] DONG C, HE G, ZHENG W, et al. Study on antibacterial mechanism of Mg (OH) 2 nanoparticles. Mater Lett. 2014;134:286-289. [37] Liu Y, Liu Y, Li X, et al. Fabrication and research of Mg (OH) 2/PCL/PVP nanofiber membranes loaded by antibacterial and biosafe Mg (OH) 2 nanoparticles. Polym Test. 2022;112:107635. [38] LORD MS, FOSS M, BESENBACHER F. Influence of nanoscale surface topography on protein adsorption and cellular response. Nano Today. 2010;5(1):66-78. [39] HICKEY DJ, ERCAN B, SUN L, et al. Adding MgO nanoparticles to hydroxyapatite–PLLA nanocomposites for improved bone tissue engineering applications. Acta Biomater. 2015;14:175-184. [40] BARNES D, JOHNSON S, SNELL R, et al. Using scratch testing to measure the adhesion strength of calcium phosphate coatings applied to poly (carbonate urethane) substrates. J Mech Behav Biomed Mater. 2012;6: 128-138. [41] CAI Y, LI C, WU D, et al. Highly active MgO nanoparticles for simultaneous bacterial inactivation and heavy metal removal from aqueous solution. Chem Eng J. 2017;312:158-166. [42] HE Y, INGUDAM S, REED S, et al. Study on the mechanism of antibacterial action of magnesium oxide nanoparticles against foodborne pathogens. J Nanobiotechnol. 2016;14:1-9. [43] NAKAMURA T, NAGURO I, ICHIJO H. Iron homeostasis and iron-regulated ROS in cell death, senescence and human diseases. Biochim Biophys Acta Gen Subj. 2019;1863(9):1398-1409. [44] BUCKLEY RRE, MORAN CG, APIVATTHAKAKUL T.骨折治疗的AO原则[M].危杰,刘璠,吴新宝,等译.3版.上海:上海科学技术出版社,2019:14-15. [45] LIANG L, HUANG Q, WU H, et al. Stimulation of in vitro and in vivo osteogenesis by Ti-Mg alloys with the sustained-release function of magnesium ions. Colloids Surf B Biointerfaces. 2021;197:111360. [46] WANG Y, LIANG W, LIU X, et al. Osteogenesis and degradation behavior of magnesium alloy plate in vivo. Eur J Inflamm. 2021;19: 20587392211034078. [47] POURDANESH F, JEBALI A, HEKMATIMOGHADDAM S, et al. In vitro and in vivo evaluation of a new nanocomposite, containing high density polyethylene, tricalcium phosphate, hydroxyapatite, and magnesium oxide nanoparticles. Mater Sci Eng C Mater Biol Appl. 2014;40:382-388. [48] JANNING C, WILLBOLD E, VOGT C, et al. Magnesium hydroxide temporarily enhancing osteoblast activity and decreasing the osteoclast number in peri-implant bone remodelling. Acta Biomater. 2010;6(5): 1861-1868. [49] WITTE F, KAESE V, HAFERKAMP H, et al. In vivo corrosion of four magnesium alloys and the associated bone response. Biomaterials. 2005;26(17):3557-3563. |
| [1] | Li Zhifei, Yang Yin, Chen Hualong, Liang Qinqiu, Zhong Yuanming, Zhang Yisheng. Finite element analysis of the correlation between tilt angle of titanium cage and postoperative subsidence of titanium cage after anterior subtotal cervical corpectomy, decompression and fusion [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1313-1319. |
| [2] | Cheng Jinhui, Wu Quan, Peng Min, Huang Changli, Tian Huimin, Li Yang. Preparation and properties of selective laser melting of porous titanium at a low energy density [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 664-668. |
| [3] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [4] | Ren Hangling, Song Na, Xu Daxia, Li Zonghuan, Zhang Zhi, Zhang Jingtao. Effect of different fusion devices on cervical sagittal parameters after anterior cervical discectomy and fusion for cervical spondylotic myelopathy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(33): 5375-5382. |
| [5] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| [6] | Xu Yinghua, Liu Jing, You Quan, Wen Zhihao, Gao Lu. Effect of neodymium-doped:yttrium aluminum perovskite laser combined with two kinds of remineralizers on remineralization of early enamel caries [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 360-365. |
| [7] | Shen Yurong, Nai Rentong, Zhao Ling, Liu Feigang, Yin Caoyang, Gu Yuanping, Chen Tieyi. Influence of NIC X-FILE and DENCO Pre-Shaper nickel-titanium instruments on the shaping of simulated curved canals [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 387-391. |
| [8] | Zhu Zhiqi, Yuan Sijie, Zhang Zilin, Ji Shijie, Meng Mingsong, Yan Anming, Han Jing. Mechanism underlying the effect of Liuwei Dihuang Pill on osteolysis and osteogenesis induced by titanium particles [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 392-397. |
| [9] | Dong Bo, Li Xiaoyu, Li Birong, Li Zhen, Wang Zixuan, Yin Zhaoyi, Meng Weiyan. 3D-printed scaffolds repair infected bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(29): 4685-4690. |
| [10] | Yu Luxiang, Zhang Ruojin, Tan Fabing. Influence of heat treatment technology on mechanical properties of 3D printed titanium alloy specimens [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(29): 4741-4747. |
| [11] | Chu Fuchao, Wang Zhenxin, Zhang Dazhen, Yuan Feng. Osteogenic properties of polyacrylamide-modified gelatin methacryloyl grafted titanium alloy scaffold [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3472-3477. |
| [12] | Zheng Heishu, Zhang Yingjuan, Wei Yanhua, Huang Hui, Ma Xiangyu, Liao Hongbing. Antibacterial performance of cerium oxide nanoenzyme against Escherichia coli [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3496-3501. |
| [13] | Chen Mingxue, Niu Jianhua, Lin Haiyan, Wu Gang, Wan Ben. Physicochemical properties and cytocompatibility of biomimetically precipitated nanocrystalline calcium phosphate granules [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3502-3508. |
| [14] | Liu Yan, Zheng Xuexin. Performance of 3D-printed polylactic acid-nano-hydroxyapatite/chitosan/doxycycline antibacterial scaffold [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3532-3538. |
| [15] | Tang Ziniu, Chu Fengcheng, Wu Kang, Zhang Lin, Bai Yanjie, Lin Xiao, Yang Huilin, Zhou Huan, Liu Huiling, Yang Lei. Application of strontium polyphosphate with both radiopaque and osteogenic functions in calcium phosphate cement [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3539-3547. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||