Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (34): 5462-5468.doi: 10.12307/2023.747
Previous Articles Next Articles
Establishment of transient expression system of eggplant cotyledon by agrobacterium-mediated injection
Lang Yaoling1, 2, Wang Qian1, Chen Bin1, Bai Guohui1, Guan Xiaoyan1, Liu Jianguo1
- 1Guizhou Provincial Key Laboratory of Oral Disease Research in Colleges and Universities and Zunyi Key Laboratory of Oral Disease Research, School of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Guiyang Stomatological Hospital, Guiyang 550002, Guizhou Province, China
-
Received:2022-11-03Accepted:2022-12-13Online:2023-12-08Published:2023-04-20 -
Contact:Liu Jianguo, MD, Professor, Chief physician, Doctoral supervisor, Guizhou Provincial Key Laboratory of Oral Disease Research in Colleges and Universities and Zunyi Key Laboratory of Oral Disease Research, School of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Lang Yaoling, Master, Attending physician, Guizhou Provincial Key Laboratory of Oral Disease Research in Colleges and Universities and Zunyi Key Laboratory of Oral Disease Research, School of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China; Guiyang Stomatological Hospital, Guiyang 550002, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 81260164 (to LJG); Guizhou Provincial Party Committee Organization Department Guizhou Province Sixth Batch of Talent Base, No. RCJD2019-9 (to LJG); First Batch of Municipal Talent Bases in Zunyi, No. ZSKHSZ [2017]20 (to LJG); Youth Science and Technology Talent Growth Project of Guizhou Provincial Department of Education, No. QJHKYZ [2022]276 (to CB); Guizhou Provincial Health Commission Science and Technology Fund Project, No. gzwkj2021-351 (to CB)
CLC Number:
Cite this article
Lang Yaoling, Wang Qian, Chen Bin, Bai Guohui, Guan Xiaoyan, Liu Jianguo. Establishment of transient expression system of eggplant cotyledon by agrobacterium-mediated injection[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5462-5468.
share this article

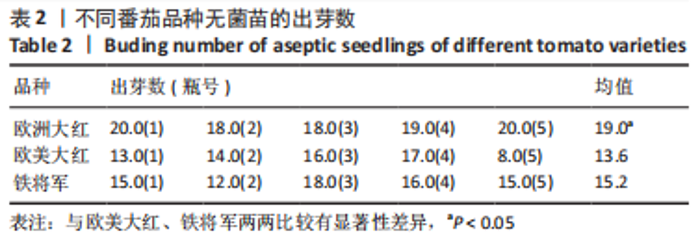
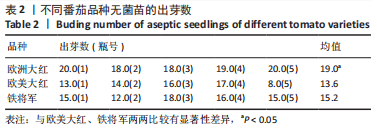
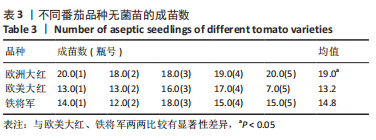
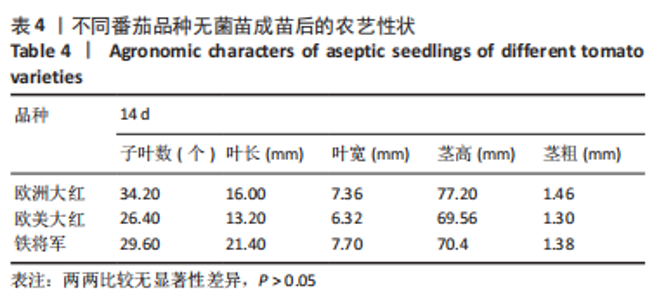
2.1 无菌苗的生长过程 正常番茄种子的形状为肾形或扁平卵圆形,颜色呈黄褐色或灰褐色,种子的表面覆盖有一层细绒毛,种子消毒时用小毛刷轻轻将种子上的绒毛搓掉,利于种子的吸水和发芽。将消毒后的番茄种子接种于含MS固体培养基的锥形瓶中,由于番茄种子的发芽具有好暗性,在25 ℃下暗培养3-5 d,番茄种子吸水膨大,胚根逐渐萌出,当胚根萌出至60%时,将种子转入光照下培养,7-12 d可见子叶逐渐萌出,舒展成双子叶,色绿,种子皮脱落,茎直立生长变粗。在幼苗生长前期,子叶是进行光合作用的主要器官;14 d真叶显露生长;18 d后无菌苗可长至瓶高。从种子萌动到子叶展开,第1片真叶显露,此期成为发芽期,其完成需要14 d左右。无菌苗生长过程见图2,无菌苗子叶及真叶见图3。"


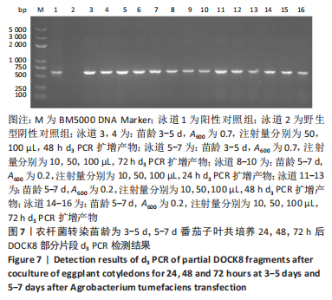
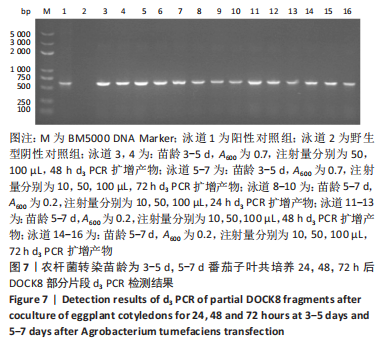
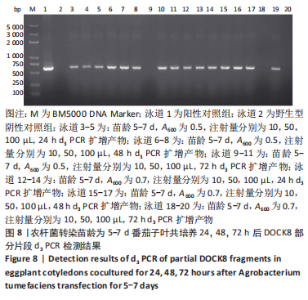
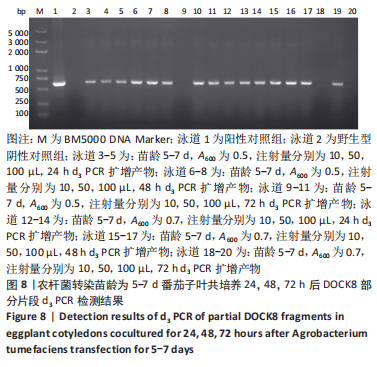
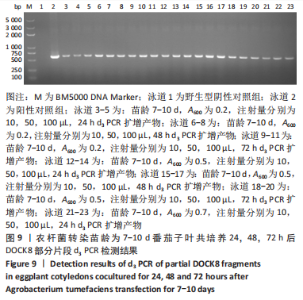
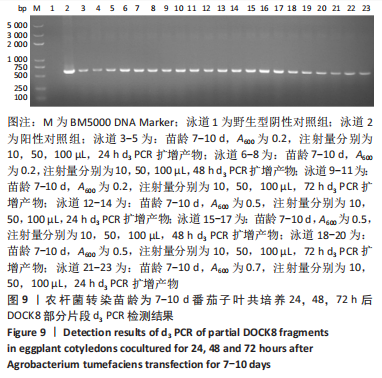
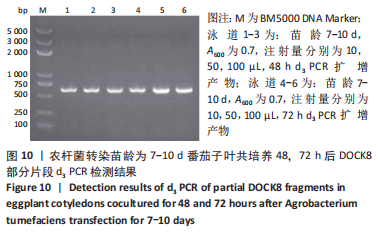
2.4 mRNA水平检测DOCK8在番茄中的表达 以生长至3-5 d,5-7 d,7-10 d的番茄子叶作为实验对象,用1 mL的无菌注射器吸取600 nm波长处的吸光度值分别为0.2,0.5,0.7的转化液,注射量分别为10,50,100 μL,共培养24,48,72 h。从番茄子叶中提取总mRNA,反转录成cDNA,以cDNA为模板,利用引物进行PCR扩增,反应产物进行1%的琼脂糖凝胶电泳检测。结果表明:在农杆菌介导下,在612 bp处出现目的条带,条带深浅不一,而未经农杆菌介导的野生型番茄子叶作为阴性对照组未出现目的条带,表明DOCK8 mRNA在番茄植株中成功表达,表达量之间可能存在差异。PCR电泳结果见图6-10。"


分别选取A600为0.2,0.5,0.7的农杆菌制成转化液,对生长3-10 d的番茄子叶进行注射,共培养24-72 h。为确保实验的准确性,以未经农杆菌注射的番茄子叶作为阴性对照组。结果显示:在A600为0.2-0.7时,报告基因均有表达。 分别注射10,50,100 μL的转化液,结果显示:当苗龄在5-7 d时,注射量为10 μL和100 μL,共培养至72 h未见报告基因的表达。因此,农杆菌注射量过多或过少,均会对实验结果产生影响。该实验最适转化液注射量为50 μL。 分别注射苗龄为3-5 d,5-7 d,7-10 d的番茄子叶。结果显示:苗龄为3-10 d的番茄子叶均有表达。考虑过于幼嫩的子叶对农杆菌的毒性抵抗力较弱,因此选择生长7-10 d的子叶比较适宜于此实验。 分别共培养24,48,72 h。结果显示:部分只在24,48 h表达,72 h未见表达,因此最适表达时间为24-48 h。"

| [1] HESSER A. Tomato Preserves. New York Times Magazine. 2008;8(24):49. [2] CUNNINGHAM C, PORTER JR. Recombinant proteins from plants:Production and Isolation of clinically useful compounds. J Nat Prod. 1998;61(11):1452. [3] METT V, LYONS J, MUSIYCHUK K, et al. A plant-produced plague vaccine candidate confers protection to monkeys. Vaccine. 2007;25(16):3014-3017. [4] RODE B, ENDRES C, RAN C, et al. Large-scale production and homogenous purification of long chain polysialic acids from E. coli K1. J Biotechnol. 2008; 135(2):202-209. [5] LEUZINGER K, DENT M, HURTADO J, et al. Efficient agroinfiltration of plants for high-level transient expression of recombinant proteins. J Vis Exp. 2013; (77):50521. [6] WROBLEWSKI T, TOMCZAK A, MICHELMORE R. Optimization of Agrobacterium-mediated transient assays of gene expression in lettuce, tomato and Arabidopsis. Plant Biotechnol J. 2005;3(2):259-273. [7] WYDRO M, KOZUBEK E, LEHMANN P. Optimization of transient Agrobacterium-mediated gene expression system in leaves of Nicotiana benthamiana. Acta Biochim Pol. 2006;53(2):289-298. [8] JOH LD, VANDERGHEYNST JS. Agroinfiltration of plant tissues for production of high-value recombinant proteins:an alternative to production in transgenic crops. J Sci Food Agric. 2006;86(13):2002-2004. [9] SONG GQ, YAMAGUCHI KI. Efficient Agroinfiltration-mediated transient GUS expression system for assaying different promoters in rice. Plant Biotechnology. 2003;20(3):235-239. [10] KATAYA AR, SULIMAN MN, KALANTIDIS K, et al. Cucurbit yellow stunting disorder virus p25 is a suppressor of post-transcriptional gene silencing. Virus Res. 2009;145(1):48-53. [11] ZHANG Y, CHEN M, SIEMIATKOWSKA B, et al. A Highly Efficient Agrobacterium- Mediated Method for Transient Gene Expression and Functional Studies in Multiple Plant Species. Plant Commun. 2020;1(5):100028. [12] JONES HD. Wheat transformation:current technology and applications to grain development and composition. J Cereal Sci. 2005;41(2):137-147. [13] CHEN Q, LAI H, HURTADO J, et al. Agroinfiltration as an Effective and Scalable Strategy of Gene Delivery for Production of Pharmaceutical Proteins. Adv Tech Biol Med. 2013;1(1):103. [14] HITZEROTH II, VAN ZYL AR. Transient Expression of Viral Proteins in Plants Using Agrobacterium tumefaciens. Methods Mol Biol. 2016;1404:581-595. [15] BIDARIGH FARD A, DEHGHAN NAYERI F, HABIBI ANBUHI M. Transient expression of etanercept therapeutic protein in tobacco (Nicotiana tabacum L.). Int J Biol Macromol. 2019;130:483-490. [16] PHOOLCHAROEN W, BHOO SH, LAI H, et al. Expression of an immunogenic Ebola immune complex in Nicotiana benthamiana. Plant Biotechnol J. 2011; 9(7):807-816. [17] 赵文婷,魏建和,刘晓东,等.植物瞬时表达技术的主要方法与应用进展[J].生物技术通讯,2013,24(2):294-300. [18] 李晓君,王绍梅,谢艳兰,等.农杆菌渗透法转化烟草条件的优化[J].江苏农业科学,2014,42(9):45-47. [19] 黎茵,张以顺.农杆菌注射渗透法转化因草实验研究[J].实验技术与管理,2010,27(11):50-52. [20] 邱扔,陶刚,李奇科,等.农杆菌渗入 法介导的基因瞬时表达技术及应用[J].分子植物育种,2009,7(5):1032-1039. [21] 刘兆明,刘宗旨,白庆武,等.Agroinfiltration在植物分子生物学研究中的应用[J].生物工程学报,2002,18(4):411-414. [22] DHADI SR, DESHPANDE A, RAMAKRISHNA W. A novel nonwounding transient expression assay for cereals mediated by Agrobacterium tumefaciens. Plant Molecular Biology Reporter. 2012;30(1):36-45. [23] KAPILA J, DE RYCKE R, VAN MONTAGU M, et al. An Agrobacterium-mediated transient gene expression system for intact leaves. Plant Science. 1997; 122(1):101-108. [24] 吴英杰,姜波,张岩,等.农杆菌介导的烟草瞬时表达试验条件优化[J].东北林业大学学报,2010,38(9):110-112. [25] 贾永芳,马玉坤,郭余龙,等.农杆菌介导的半夏GUS基因瞬时表达[J].华北农学报,2007,22(4):42-45. [26] MCINTOSH KB, HULM JL, YOUNG LW. A rapid Agrobacterium-mediated Arabidopsis thaliana transient assay system. Plant Molecular Biology Reporter. 2004;22(1): 53-61. [27] JELLY NS, VALAT L, WALTER B, et al. Transient expression assays in grapevine: a step towards genetic improvement. Plant Biotechnol J. 2014;12(9):1231-1245. [28] AHN YK, YOON MK, JEON JS. Development of an efficient Agrobacterium-mediated transformation system and production of herbicide-resistant transgenic plants in garlic (Allium sativum L.). Mol Cells. 2013;36(2):158-162. [29] 郎遥玲.PCAMBIA-E8-APBDOCK8融合基因表达质粒中标记基因的去除及瞬时转化番茄的研究[D].遵义:遵义医学院,2016. [30] MARILLONNET S, THOERINGER C, KANDZIA R, et al. Systemic Agrobacterium tumefaciens-mediated transfection of viral replicons for efficient transient expression in plants. Nat Biotechnol. 2005;23(6):718-723. [31] TZFIRA T, CITOVSKY V. From host recognition to T-DNA integration: the function of bacterial and plant genes in the Agrobacterium-plant cell interaction. Mol Plant Pathol. 2000;1(4):201-212. [32] DIAMOS AG, ROSENTHAL SH, MASON HS. 5’ and 3’ Untranslated Regions Strongly Enhance Performance of Geminiviral Replicons in Nicotiana benthamiana Leaves. Front Plant Sci. 2016;7:200. [33] 李静,陈敏,刘现伟,等.莴苣高效瞬时表达体系的建立[J].园艺学报, 2006,33(2):405-407. [34] 陶均,李玲.农杆菌转化的分子生物学[J].植物生理学通讯,2002,38(6): 639-644. [35] 孙春莲,王洪洋,田振东.农杆菌介导的pCB302-3载体在本氏烟中瞬时表达条件优化[J].华中农业大学学报,2015,34(3):8-12. [36] VICKERS CE, XUE G, GRESSHOFF PM. A novel cis-acting element, ESP, contributes to high-level endosperm-specific expression in an oat globulin promoter. Plant Mol Biol. 2006;62(1-2):195-214. [37] HAMILTON DA, ROY M, RUEDA J, et al. Dissection of a pollen-specific promoter from maize by transient transformation assays. Plant Mol Biol. 1992;18(2):211-218. [38] SCHEFFLER B, FRANKEN P, SCHÜTT E, et al. Molecular analysis of C1 alleles in Zea mays defines regions involved in the expression of this regulatory gene. Mol Gen Genet. 1994;242(1):40-48. [39] LI WJ, DAI LL, CHAI ZJ, et al. Evaluation of seed storage protein gene 3’-untranslated regions in enhancing gene expression in transgenic rice seed. Transgenic Res. 2012;21(3):545-553. [40] 彭昊,翟英,张芊,等.水稻高效RNA干涉体系的建立及其功能分析[J].中国农业科学,2006,9(9):1729-1735. [41] DE LANGE P, DE BOER GJ, MOL JN, et al. Conditional inhibition of beta-glucuronidase expression by antisense gene fragments in petunia protoplasts. Plant Mol Biol. 1993;23(1):45-55. [42] DI FIORE S, HOPPMANN V, FISCHER R, et al. Transient gene expression of recombinant terpenoid indole alkaloid enzymes in Catharanthus roseus leaves. Plant Molecular Biology Reporter. 2004;22(1):15-22. [43] 金建平,林左军,邓余,等.ɑ-CGRP融合基因的高效表达及载体蛋白长度对表达的影响[J].自然科学进展,1994,4(5):589-594. [44] NANDAKUMAR R, CHEN L, ROGERS SMD. Factors affecting the Agrobacterium-mediated transient transformation of the wetland monocot,Typha latifolia. Plant Cell Tissue Organ Cult. 2004;79(1):31-38. [45] JANI D, MEENA LS, RIZWAN-UL-HAQ QM, et al. Expression of cholera toxin B subunit in transgenic tomato plants. Transgenic Res. 2002;11(5):447-454. [46] JIANG XL, HE ZM, PENG ZQ, et al. Cholera toxin B protein in transgenic tomato fruit induces systemic immune response in mice. Transgenic Res. 2007;16(2):169-175. [47] SHCHELKUNOV SN, SALYAEV RK, REKOSLAVSKAYA NI, et al. The obtaining of transgenic tomato plant producing chimerical proteins TBI-HBsAg. Dokl Biochem Biophys. 2004;396:139-142. [48] CHEN HF, CHANG MH, CHIANG BL, et al. Oral immunization of mice using transgenic tomato fruit expressing VP1 protein from enterovirus 71. Vaccine. 2006;24(15):2944-2951. [49] MCGARVEY PB, HAMMOND J, DIENELT MM, et al. Expression of the rabies virus glycoprotein in transgenic tomatoes. Biotechnology (N Y). 1995;13(13): 1484-1487. [50] MCCORMICK AA, REDDY S, REINL SJ, et al. Plant-produced idiotype vaccines for the treatment of non-Hodgkin’s lymphoma: safety and immunogenicity in a phase I clinical study. Proc Natl Acad Sci U S A. 2008;105(29):10131-10136. [51] D’AOUST MA, COUTURE MM, CHARLAND N, et al. The production of hemagglutinin-based virus-like particles in plants: a rapid, efficient and safe response to pandemic influenza. Plant Biotechnol J. 2010;8(5):607-619. [52] CHEN Q. Expression and purification of pharmaceutical proteins in plants. Biological Engineering Transactions. 2008;1(4):291-321. [53] DORAN PM. Foreign protein degradation and instability in plants and plant tissue cultures. Trends Biotechnol. 2006;24(9):426-432. |
| [1] | Chen Jingqiao, Li Ying, Meng Maohua, Xu Xingxing, Wang Qinying, Wang Huan, Lu Jing, Shu Jiayu, Dong Qiang. Research progress in platelet-rich fibrin in stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 441-446. |
| [2] | Liu Wenwen, Cui Zhanqin, Liu Yingqi. Systematic review of the research progress of nickel-titanium arch wires for orthodontics [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2556-2562. |
| [3] | Guan Weiwei, Gu Yu, Guan Xiaoyan, Wu Jiayuan, Bai Guohui, Tian Yuan, Liu Jianguo. Identification and expression of exogenous target genes in transgenic tomato [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 171-175. |
| [4] | Gu Yueguang, Shen Jianhuan, Ni Jieli, Guo Shuyu, Yan Zhongyi, Zhang Yang. Effect of osteogenesis in patients with alveolar cleft after bone grafting investigated by volume analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1501-1504. |
| [5] | Guan Weiwei, Gu Yu, Guan Xiaoyan, Wu Jiayuan, Bai Guohui, Tian Yuan, Liu Jianguo. Transgenic tomato vaccine expressing chimeric protein PAcA/CTB can immunize rats through gastrointestinal absorption [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1712-1716. |
| [6] |
Dong Xiling, Zhang Xiaoming, Liu Tongbin.
Recombinant human parathyroid hormone (1-34): pro-osteogenic action and application in oral field [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4231-4236. |
| [7] | Lu Lei, Yu Shibin, Shi Xiaowei, Ma Teng, Huang Jinghui, Yang Hongxu. Application of Dentistry-ProConsult software to assist plaster tooth modeling in stomatology teaching [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(30): 4811-4815. |
| [8] | Wang Haijiao, Huang Ruina, Wang Xiaojun, Zheng Bili, Chen Peina, Zeng Xiujuan. Analysis on the hotspot of the platelet-rich plasma based on VOSviewer [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(18): 2947-2952. |
| [9] | Zhou Jun, Hou Lu, Xu Shi-qian. Application of Functional Magnetic System in osseous deformity [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(50): 8741-8746. |
| [10] | Yu Hua, Zhang Xiao-dong, Wang Yi-jing, Wei Jing, Liu Jiao-jiao, Li Yu-jie. Light-cured composite resin materials for dental crown defective repair [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(47): 8269-8274. |
| Viewed | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
Full text 1462
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
Abstract 460
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||