Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (25): 4093-4100.doi: 10.12307/2023.551
Role and advantages of carbon nanotubes for tissue engineering
Lyu Jiayi1, Yao Qingqiang2, Zhu Yishen1
- 1College of Biotechnology and Pharmaceutical Engineering, Nanjing Tech University, Nanjing 211816, Jiangsu Province, China; 2Department of Orthopedic Surgery, Nanjing First Hospital, Nanjing Medical University, Nanjing 210006, Jiangsu Province, China
-
Received:2022-07-14Accepted:2022-08-22Online:2023-09-08Published:2023-01-18 -
Contact:Zhu Yishen, Professor, College of Biotechnology and Pharmaceutical Engineering, Nanjing Tech University, Nanjing 211816, Jiangsu Province, China -
About author:Lyu Jiayi, Master candidate, College of Biotechnology and Pharmaceutical Engineering, Nanjing Tech University, Nanjing 211816, Jiangsu Province, China -
Supported by:Jiangsu Provincial Key Social Development Project, No. BE2019736 (to ZYS)
CLC Number:
Cite this article
Lyu Jiayi, Yao Qingqiang, Zhu Yishen. Role and advantages of carbon nanotubes for tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 4093-4100.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
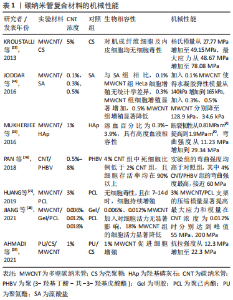
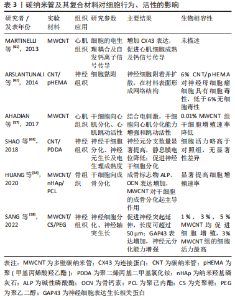
2.1 碳纳米管的组织工程材料特性 碳纳米管是管状结构的碳基纳米材料,其具有许多特性,包括良好的机械特性、良好的导热性、从金属到半导体的电学性能、低密度及化学稳定性等。针对其组织工程材料的相关特性,文章简述了碳纳米管基本形态结构及导电性,详细探讨了碳纳米管的机械性能。 2.1.1 碳纳米管的形态结构 碳纳米管作为碳的同素异形体,由通过sp2键相互键合的碳原子组成[16]。碳纳米管呈无缝圆柱形,具有纳米级直径、较大的长径比及高比表面积,有利于蛋白质吸附及细胞黏附生长[11-12],其纳米网络结构及适当的孔隙率有利于细胞外基质的物质交换[13-14]。碳纳米管通常分为2种:由单层管状石墨烯形成的单壁碳纳米管(single-walled carbon nanotubes,SWCNT)和由多个同心管状石墨烯层组成的多壁碳纳米管(multi-walled carbon nanotubes,MWCNT)。SWCNT通常存在于紧密排列的六边形束中,直径约为1 nm,长度可达1 mm或更长。MWCNT的结构更类似于中空石墨纤维,直径2-100 nm [17]。 2.1.2 碳纳米管的导电性 已有综述详细介绍了碳纳米管的导电性能,包括不同结构碳纳米管呈现电性不同,通过尺寸、层数及手性等参数调控导电性[13]。碳纳米管加入其他组织工程材料中有助于调控复合材料的导电性,例如:5% MWCNT加入壳聚糖/聚乙二醇复合材料后使其电导率提高50%[18];聚己内酯中加入质量分数0.3% MWCNT后导电性至少可增加3倍 [19];将质量分数1.6% 碳纳米管/聚乙二醇/丙烯酸酯加入可降解水凝胶后,复合水凝胶的电导率从0.001 S/m增加到0.005 S/m,并且与电刺激结合后促进了电传导,从而促进了细胞增殖[20]。 2.1.3 碳纳米管的机械性能 碳纳米管具有优良的力学性质,杨氏模量高达1.8 TPa,强度约为钢的100倍[21]。弹性应变可达5%-18%,约为钢的60倍;同时碳纳米管还具有极高的韧性,对于直径为30 nm的纳米管,存储应变能的临界点为100 KeV,这个下限是相同直径碳化硅纳米棒的5-10倍[22]。 碳纳米管可以相互重叠,相邻纳米管之间强烈的π-π键效应和范德华力导致纳米管之间的交织,从而形成网络状结构,该结构具有出色的应力转移效果,使得碳纳米管可以有效提高组织工程复合材料的强度,包括压缩强度、弯曲强度、杨氏模量等[23-29],见表1。"
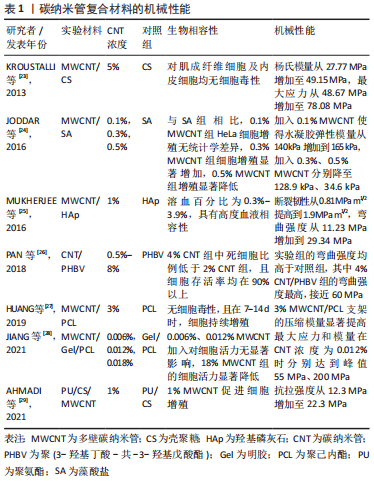
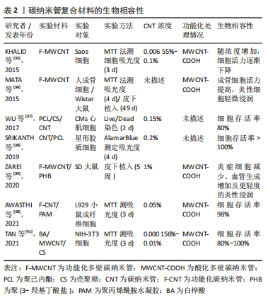
KROUSTALLI等[23]向机械性能较差的壳聚糖中加入质量分数5% MWCNT后,MWCNT和壳聚糖形成相互作用,MWCNT在复合凝胶中分散均一,复合凝胶弹性模量增加77%,符合心血管组织修复的要求。MUKHERJEE等[25]发现加入质量分数1% MWCNT使得羟基磷灰石的断裂韧性提高一倍以上。AHMADI等[29]在聚氨酯/壳聚糖中加入质量分数1% MWCNT进行交联,抗拉强度提高约一倍。MIKAEL等[30]将质量分数3% MWCNT加入到聚乳酸-乙醇酸共聚物中,复合材料的压缩强度提高约一倍,压缩模量增加近2倍。 但当碳纳米管在复合材料中含量过高时,会形成集群结构,从而削弱碳纳米管的增强作用。JODDAR等[24]研究发现加入1 mg/mL的MWCNT可以增加海藻酸钠水凝胶的刚度,当碳纳米管质量浓度达到3 mg/mL和5 mg/mL时,弹性模量逐渐降低。JIANG等[28]将MWCNT加入聚己内酯/明胶中,随碳纳米管浓度增加,最大应力和模量随之增加至峰值,而后随之降低;同时由于碳纳米管会限制弹性聚合物基质的塑性形变,最大应变随碳纳米管浓度增加而下降。 因此,可以通过改变碳纳米管浓度调控复合材料的机械性能,包括弹性模量、断裂韧性、抗拉强度、压缩强度及压缩模量等。结合损伤组织部位的生物力学特性及碳纳米管与其他材料的相互作用情况,有利于设计应用于相应组织修复的复合材料。 2.2 碳纳米管的细胞毒性和生物相容性 目前,关于碳纳米管是否存在毒性这一问题仍有争议,其毒性机制包括碳纳米管通过氧化蛋白质核酸等细胞成分、形成活性氧及阻断跨膜电子转移等破坏细胞膜从而造成细胞损伤或死亡[31-33]。研究发现,可以通过改变碳纳米管浓度及表面功能化处理等方法降低或消除碳纳米管复合材料的细胞毒性,使其具有良好的生物相容性[34-41]。 功能化处理的CNT通过改善其水中的分散性,显著减少碳纳米管在溶液中的聚集行为,从而降低细胞毒性。OLIVEIRA 等[42]首次证明0.01% 以下的MWCNT-COOH对人去角质乳牙干细胞具有细胞相容性。TURK等[43]发现0.2% MWCNT-COOH/胶原/壳聚糖复合支架组的MG63人骨肉瘤细胞存活率比壳聚糖支架组增加9%,但功能化碳纳米管浓度过高,聚集行为增加,仍会增加细胞死亡。KHALID等[35]分析硫酸及硝酸3∶1体积混合处理的MWCNT对成骨细胞的影响,酸化MWCNT的质量浓度从 65.5 μg/mL提高到1 000 μg/mL,细胞存活率从98% 降至86%。另外,低浓度碳纳米管复合材料在动物体内的长期试验也表明其良好的生物相容性,1% SWCNT/聚(乳酸-乙醇酸)复合支架、0.1% MWCNT/壳聚糖水凝胶支架植入SD大鼠体内均无急性炎性反应[44-45]。此外,碳纳米管在复合材料中的暴露情况也会影响其细胞毒性,TAN等[41]发现,细胞存活率在MWCNT/白桦酸复合材料中时为40%,经壳聚糖包被后提高到80% 以上,以上研究成果见表2。"
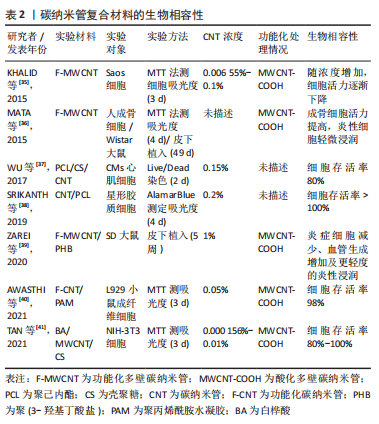
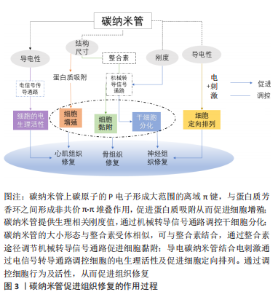
基于以上研究显示,碳纳米管备受关注的细胞毒性问题能够通过降低浓度、功能化处理、减少碳纳米管暴露等方法得到有效解决。另外,有研究发现碳纳米管的尺寸会影响其毒性,短MWCNT的细胞毒性高于长MWCNT[46],但目前对于碳纳米管的尺寸效应对其毒性的影响尚存在争议,仍需要进行更深入研究。 2.3 碳纳米管的降解性能 碳纳米管的降解性能是影响碳纳米管是否能够体内应用的要素之一。细胞或机体对于碳纳米管的吸收、保留及排泄等与碳纳米管的体内毒性相联系。显微成像证实了碳纳米管能够被细胞吸收,PORTER等[47]首次发现SWCNT可以被细胞摄取进入细胞质内,引起细胞产生剂量依赖性死亡。随后,CHENG等[48]观察到未纯化的MWCNT可以穿过质膜进入细胞质及细胞核。AL-JAMAL等[49]研究MWCNT在小鼠器官中的保留情况,发现碳纳米管的功能化化学改性修饰可以增加其肾脏清除率,而降低功能化比例有利于碳纳米管在网状内皮系统积累。因此探究并调控碳纳米管的生物降解尤其重要。 RUSSIER等[50]发现溶酶体模拟液中的氧化SWCNT结构发生断裂及分解,30 d后被完全降解;而氧化MWCNT 60 d内未能完全降解。KAGAN等[51]研究表明人嗜中性粒细胞过氧化物酶的次氯酸盐和反应性自由基中间体在体外催化SWCNT的生物降解,生物降解后的SWCNT吸入小鼠肺部不会引起炎性反应,而非降解的SWCNT诱导组织肉芽肿的形成引起急性炎性反应。HOU等[52]比较了SWCNT、氧化SWCNT和SWCNT-OH的降解性能,发现SWCNT几乎没有缺陷位点因此降解率低,而SWCNT-OH的缺陷位点多,提供了更多的氧化攻击位点,从而其生物降解速率高于氧化SWCNT。此外,XU等[53]研究MWCNT复合支架在溶菌酶溶液中的体外降解,第4周时,0.3% MWCNT的复合材料的降解速率可达75% 以上,但随着MWCNT含量的增加,支架的降解速率降低。 碳纳米管的生物降解特性对于调节碳纳米管生物相容性有重要意义。与碳纳米管相比,功能化碳纳米管能够更快地通过酶生物降解,可以通过碳纳米管功能化调控碳纳米管的生物降解速率,从而保证碳纳米管的体内安全性。 2.4 碳纳米管在组织工程修复中的应用 碳纳米管的独特结构、导电性及机械性能有助于在组织工程修复中应用。碳纳米管组织修复的作用过程如图3所示:碳纳米管与蛋白质芳香环之间的非共价-堆叠相互作用,可以促进蛋白质吸附[54],从而促进细胞增殖[55];碳纳米管提供生理相关刚度值通过机械转导信号通路或分子途径调控干细胞分化[56-57];碳纳米管的大小形态与整合素受体相似,通过整合素途径调节机械转导信号通路促进细胞黏附[57];碳纳米管的导电性结合电刺激通过电信号传导途径改善细胞的电生理活性[58];导电碳纳米管结合电刺激使得碳纳米管复合材料排列对齐从而促进细胞定向排列。"
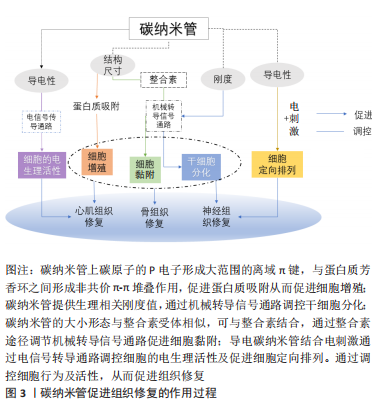
碳纳米管复合材料可以调控细胞行为及活性,主要包括:细胞增殖分化黏附、电生理活性及定向排列等。PAN等[55]发现在聚己内酯加入MWCNT,促进了大鼠骨髓间充质干细胞增殖及成骨向分化,且干细胞黏附能力强,可完全覆盖复合支架表面。MARTINELLI等[59]首次报告了在MWCNT表面处理平台上的心肌细胞生长情况:增殖能力提高,3 d后细胞数量加倍;加速心肌细胞成熟,诱导更低的静息膜电位。WANG等[60]将碳纳米管加入丝素蛋白/聚己内酯中制备导电对齐纳米纤维,PC12神经细胞完全附着在复合材料表面且有60% 的PC12神经细胞沿纳米纤维方向生长;同时神经突延伸能力增强。这些体外细胞实验为碳纳米管在组织修复的体内应用提供了数据支撑[61-63],见 表3。"
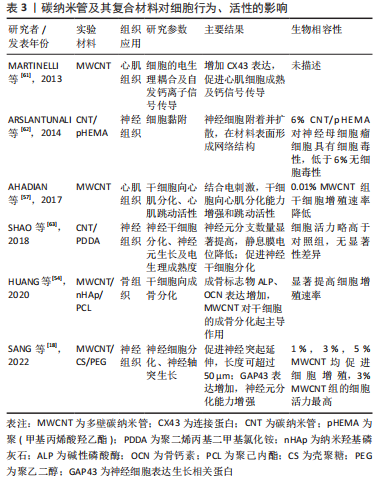

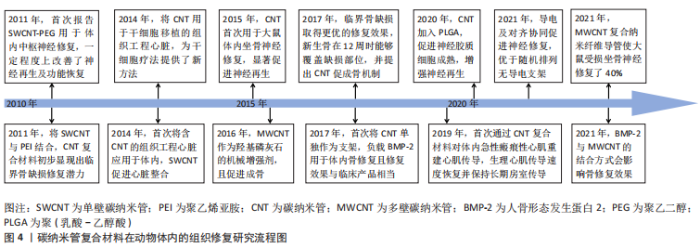
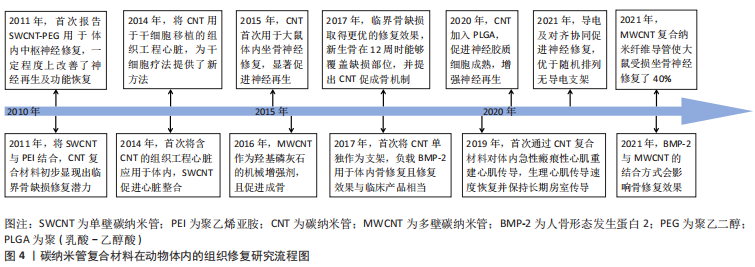
碳纳米管在骨组织工程中主要用于增强支架的硬度和成骨性。MUKHERJEE等[25]通过添加MWCNT改善了羟基磷灰石的不良机械性能,提高其作为承重可植入材料的可行性,且与羟基磷灰石对照组相比,MWCNT通过减少破骨细胞数量抑制骨吸收,同时促进羟基磷灰石的矿化提高新生骨沉积。而多离子配合物水凝胶本身具有优良的机械性能,但其骨诱导能力差,CUI等[64]首次将MWCNT与多离子配合物水凝胶结合形成多孔支架,复合支架保持了多离子配合物水凝胶优异的机械性能,同时加入MWCNT有效促进了骨髓间充质干细胞增殖及成骨分化,体内骨缺损修复速率提高近一倍。 碳纳米管复合材料支架显现出修复临界尺寸骨缺损的潜力。BHATTACHARYA等[56]在大鼠颅骨1 cm临界缺损植入SWCNT-聚醚酰亚胺复合支架,4周时碳纳米管支架一侧形成新骨,而对照组无新生骨,但由于实验时间较短,6周内缺损未能完全愈合。JING等[65]为期12周的8 mm临界缺损修复则取得了更好的修复效果,MWCNT/胶原/羟基磷灰石复合支架组中新生骨和结缔组织完全覆盖缺损部位,并且骨裂蛋白和骨钙素表达显著提高,他们提出MWCNT增强成骨的机制为通过吸附蛋白与细胞接触的高亲和力将更多的刺激从细胞外基质传递到细胞核中,从而促进细胞黏附并产生高细胞间应激反应,进一步导致成骨生物标志物在基因和蛋白质水平上的表达变化,促进了成骨修复。 骨组织工程应用中的碳纳米管材料多是复合支架,TANAKA等[66]首次将MWCNT单独作为修复支架负载骨形态发生蛋白2促进了异位骨的生成,且MWCNT作为骨形成支架的有效性与MedGel?相当。但结合方式可能对骨修复性能产生影响,最近,有研究利用不同官能团将骨形态发生蛋白2与MWCNT共价或非共价结合,发现共价结合有效提高骨形态发生蛋白2的稳定性,作用更持久[67];并且相比于结合MWCNT-COOH的骨修复效果,结合MWCNT-NH2的更优,可能是由于氨基官能团所带电荷不同导致。 综上,初步阐明碳纳米管促进成骨的机制是通过与细胞的高亲和力将刺激传导进入细胞核,促进细胞黏附和细胞间应激反应,提高成骨生物标志物表达水平。碳纳米管的机械增强能力及成骨诱导性在骨组织修复中得到广泛关注,并且在大块骨缺损修复中取得良好效果。 2.4.2 神经组织修复 神经损伤主要包括中枢神经损伤及周围神经损伤,研究发现碳纳米管的导电性可以模拟天然神经组织的电生理特性,促进神经细胞分化、改善神经轴突生长和调节突触强度[18],促进神经生长从而可以更快地进行神经组织修复[63]。 ROMAN等[68]认为不可降解的碳纳米管能够持续作用于受伤的神经组织,在此基础上首次报告了SWCNT-聚乙二醇促进体内受损中枢神经修复的情况:SWCNT-聚乙二醇改善了脊髓损伤神经的轴突修复和再生,并诱导适度的功能恢复。AHN等[69]首次报告了碳纳米管复合神经导管在大鼠坐骨神经再生的体内应用,植入大鼠16周后,碳纳米管复合神经导管组的再生轴突数量显著高于无导电对照组,再生神经支配肌肉的横截面积显著增加。类似地,FALLAH-DARREHCHI等[70]制备的MWCNT复合纳米纤维导管使得大鼠受损坐骨神经修复了40%。HUANG等[71]发现导电MWCNT加入聚乙丙交酯中,细胞沿导电纤维方向拉伸,促进了神经胶质细胞成熟,且神经功能恢复速度加快。 除导电性之外,支架材料的排列对齐也是影响神经修复的一大因素,二者协同促进神经再生。ZHANG等[72]研究发现定向排列的导电碳纳米管/聚己内酯组中出现有序的组织结构、更多的新生血管,神经修复效果优于随机排列的纤维支架组,接近自体移植组。 基于以上研究,碳纳米管在模拟电生理特性及引导细胞定向排列方面发挥关键作用,含碳纳米管复合材料的神经修复效果可接近自体移植,显示出碳纳米管在神经修复应用方面的巨大潜力。 2.4.3 心肌组织修复 心脏组织工程亟需开发可以模仿心肌、促进细胞生长及组织修复的新型生物材料。在心肌组织修复中,碳纳米管的优势在于其电学及机械性质,通过促进心肌细胞成熟、自发跳动及同步收缩等,有效改善心脏功能,促进梗死心肌修复。 鉴于体外实验已证明导电碳纳米管促心肌细胞收缩及电生理活性,ZHOU等[73]首次在SWCNT/明胶/甲基丙烯酯支架上构建组织工程心脏,加入SWCNT的组织工程支架组第2天产生自发跳动,第8天同步收缩;4周后,SWCNT通过迁移到梗死心脏的瘢痕区域与相邻细胞连接,促进组织工程心脏的整合并有效改善了心肌功能。类似地,利用碳纳米管的高导电性,MCCAULEY等[74]首次报道了碳纳米管复合材料促进体内急性瘢痕心肌重建心肌传导,其生理心肌传导速度几乎完全恢复。 细胞黏附是影响缺血性心肌干细胞移植的关键因素,LI等[75]制备了SWCNT/N-异丙基丙烯酰胺聚合物水凝胶,碳纳米管的加入显著提高了复合材料的纳米粗糙度,为干细胞在活性氧条件下的黏附和存活提供了更好的环境,修复后的心肌梗死面积由空白对照组的51% 减小至24.3%。 碳纳米管相关体外研究为其体内心肌修复应用奠定了基础,利用碳纳米管的导电性及提高细胞黏附、改善生物活性等进行心肌修复,已证实其体内修复有效性。但对于心肌修复而言,如何促进新生血管形成及生长尤其重要,后续研究中可以通过将碳纳米管与促血管生成相关生物活性因子结合,进一步修复心肌组织。"

| [1] 姚梦竹,盛晓霞,林军,等.碳纳米管在骨组织工程支架中的研究进展[J].浙江大学学报(医学版),2016,45(2):161-169. [2] TROJANI C, BALAGUER T, BOUKHECHBA F, et al. Cellular strategies in bone tissue engineering: a review. Rev Chir Orthop Reparatrice Appar Mot. 2008;94(1):1-11. [3] ZHAO X, WANG L, GAO J, et al. Hyaluronic acid/lysozyme self-assembled coacervate to promote cutaneous wound healing. Biomater Sci. 2020; 8(6):1702-1710. [4] LEI Z Y, LIU T, LI WJ, et al. Biofunctionalization of silicone rubber with microgroove-patterned surface and carbon-ion implantation to enhance biocompatibility and reduce capsule formation. Int J Nanomed. 2016;11:5563-5572. [5] WUBNEH A, TSEKOURA E K, AYRANCI C, et al. Current state of fabrication technologies and materials for bone tissue engineering. Acta Biomater. 2018;80:1-30. [6] SANTORO M, SHAH SR, WALKER JL, et al. Poly (lactic acid) nanofibrous scaffolds for tissue engineering. Adv Drug Deliv Rev. 2016;107:206-212. [7] SONG H, ZHANG Y, ZHANG Z, et al. Hydroxyapatite/NELL-1 nanoparticles electrospun fibers for osteoinduction in bone tissue engineering application. Int J Nanomedicine. 2021;16:4321-4332. [8] IIJIMA S. Helical microtules of grahitic carbon. Nature. 1991;354(6348): 56-58. [9] 申锴泉,李化毅,王垚.气相原位聚合制备碳纳米管/乙丙橡胶复合材料及其性能表征[J].新型炭材料,2020,35(1):73-79. [10] MENEZES BRC, RODRIGUES KF, FONSECA B, et al. Recent advances in the use of carbon nanotubes as smart biomaterials. J Mater Chem B. 2019;7(9):1343-1360. [11] KHALID P, SUMAN VB. Carbon nanotube-hydroxyapatite composite for bone tissue engineering and their interaction with mouse fibroblast L929 in vitro. J Bionanosci. 2017;11(3):233-240. [12] TUTAK W, CHHOWALLA M, SESTI F. The chemical and physical characteristics of single-walled carbon nanotube film impact on osteoblastic cell response. Nanotechnology. 2010;21(31):315102. [13] 任义行,黄若愚,王存阳,等.碳纳米管作为骨修复材料的优势和挑战[J].中国修复重建外科杂志,2021,35(3):271-277. [14] HOLMES B, FANG X, ZARATE A, et al. Enhanced human bone marrow mesenchymal stem cell chondrogenic differentiation in electrospun constructs with carbon nanomaterials. Carbon. 2016;97:1-13. [15] 李燕,刘惠亮.碳纳米管在心肌组织工程应用的研究进展[J].中华老年心脑血管病杂志,2016,18(9):993-995. [16] PEI B, WANG W, DUNNE N, et al. Applications of carbon nanotubes in bone tissue regeneration and engineering: superiority, concerns, current advancements, and prospects. Nanomaterials. 2019;9(10): 1501. [17] AHADIAN S, OBREGON R, RAMON-AZCON J, et al. Carbon nanotubes and graphene-based nanomaterials for stem cell differentiation and tissue regeneration. J Nanosci Nanotechnol. 2016;16(9):8862-8880. [18] SANG S, CHENG R, CAO Y, et al. Biocompatible chitosan/polyethylene glycol/multi-walled carbon nanotube composite scaffolds for neural tissue engineering. J Zhejiang Univ Sci B. 2022;23(1):58-73. [19] FLORES-CEDILLO ML, ALVARADO-ESTRADA KN, POZOS-GUILLEN AJ, et al. Multiwall carbon nanotubes/polycaprolactone scaffolds seeded with human dental pulp stem cells for bone tissue regeneration. J Mater Sci Mater Med. 2016;27(2):35. [20] LIU X, GEORGE MN, LI L, et al. Injectable electrical conductive and phosphate releasing gel with two-dimensional black phosphorus and carbon nanotubes for bone tissue engineering. ACS Biomater Sci Eng. 2020;6(8):4653-4665. [21] TREACY MMJ, EBBESEN TW, GIBSON JM. Exceptionally high Young’s modulus observed for individual carbon nanotubes. Nature. 1996; 381(6584):678-680. [22] WONG EW, SHEEHAN PE, LIEBER CM. Nanobeam mechanics: elasticity, strength, and toughness of nanorods and nanotubes. Science. 1997; 277(5334):1971-1975. [23] KROUSTALLI A, ZISIMOPOULOU AE, KOCH S, et al. Carbon nanotubes reinforced chitosan films: mechanical properties and cell response of a novel biomaterial for cardiovascular tissue engineering. J Mater Sci Mater Med. 2013;24(12):2889-2896. [24] JODDAR B, GARCIA E, CASAS A, et al. Development of functionalized multi-walled carbon-nanotube-based alginate hydrogels for enabling biomimetic technologies. Sci Rep. 2016;6:32456. [25] MUKHERJEE S, NANDI SK, KUNDU B, et al. Enhanced bone regeneration with carbon nanotube reinforced hydroxyapatite in animal model. J Mech Behav Mater. 2016;60:243-255. [26] PAN W, XIAO X, LI J, et al. The comparison of biocompatibility and osteoinductivity between multi-walled and single-walled carbon nanotube/PHBV composites. J Mater Sci Mater Med. 2018;29(12):189. [27] HUANG B, VYAS C, ROBERTS I, et al. Fabrication and characterisation of 3D printed MWCNT composite porous scaffolds for bone regeneration. Mater Sci Eng C Mater Biol Appl. 2019;98:266-278. [28] JIANG C, WANG K, LIU Y, et al. Using wet electrospun PCL/Gelatin/CNT yarns to fabricate textile-based scaffolds for vascular tissue engineering. ACS Biomater Sci Eng. 2021;7(6):2627-2637. [29] AHMADI P, NAZERI N, DERAKHSHAN MA, et al. Preparation and characterization of polyurethane/chitosan/CNT nanofibrous scaffold for cardiac tissue engineering. Int J Biol Macromol. 2021;180:590-598. [30] MIKAEL PE, AMINI AR, BASU J, et al. Functionalized carbon nanotube reinforced scaffolds for bone regenerative engineering: fabrication, in vitro and in vivo evaluation. Biomed Mater. 2014;9(3):035001. [31] GUO L, MORRIS D G, LIU X, et al. Iron bioavailability and redox activity in diverse carbon nanotube samples. Chem Mater. 2007;19(14):3472-3478. [32] OBERDORSTER G, OBERDORSTER E, OBERDORSTER J. Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ Health Perspect. 2005;113(7):823-839. [33] NEL A, XIA T, MADLER L, et al. Toxic potential of materials at the nanolevel. science. 2006;311(5761):622-627. [34] LIANG X, CHENG Q. Synergistic reinforcing effect from graphene and carbon nanotubes. Compos Commun. 2018;10:122-128. [35] KHALID P, HUSSAIN MA, REKHA PD, et al. Carbon nanotube-reinforced hydroxyapatite composite and their interaction with human osteoblast in vitro. Hum Exp Toxicol. 2015;34(5):548-856. [36] MATA D, AMARAL M, FERNANDES A J, et al. Diels-Alder functionalized carbon nanotubes for bone tissue engineering: in vitro/in vivo biocompatibility and biodegradability. Nanoscale. 2015;7(20):9238-9251. [37] WU Y, WANG L, GUO B, et al. Interwoven aligned conductive nanofiber yarn/hydrogel composite scaffolds for engineered 3D cardiac anisotropy. ACS Nano. 2017;11(6):5646-5659. [38] SRIKANTH M, ASMATULU R, CLUFF K, et al. Material characterization and bioanalysis of hybrid scaffolds of carbon nanomaterial and polymer nanofibers. ACS Omega. 2019;4(3):5044-5051. [39] ZAREI M, KARBASI S, SARI ASLANI F, et al. In Vitro and in vivo evaluation of Poly (3-hydroxybutyrate)/carbon nanotubes electrospun scaffolds for periodontal ligament tissue engineering. J Dent (Shiraz). 2020;21(1): 18-30. [40] AWASTHI S, GAUR JK, PANDEY SK, et al. High-strength, strongly bonded nanocomposite hydrogels for cartilage repair. ACS Appl Mater Interfaces. 2021;13(21):24505-24523. [41] TAN JM, BULLO S, FAKURAZI S, et al. Characterization of betulinic acid-multiwalled carbon nanotubes modified with hydrophilic biopolymer for improved biocompatibility on NIH/3T3 cell line. Polymers (Basel). 2021;13(9):1362. [42] OLIVEIRA ER, FAYER L, ZANETTER SS, et al. Cytocompatibility of carboxylated multi-wall carbon nanotubes in stem cells from human exfoliated deciduous teeth. Nanotechnology. 2021;33(6):065101. [43] TURK S, ALTINSOY I, CELEBIEFE G, et al. 3D porous collagen/functionalized multiwalled carbon nanotube/chitosan/hydroxyapatite composite scaffolds for bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2018;92:757-768. [44] GUPTA A, LIBERATI TA, VERHULST SJ, et al. Biocompatibility of single-walled carbon nanotube composites for bone regeneration. Bone Joint Res. 2015;4(5):70-77. [45] NAWROTEK K, TYLMAN M, DECHERCHI P, et al. Assessment of degradation and biocompatibility of electrodeposited chitosan and chitosan-carbon nanotube tubular implants. J Biomed Mater Res A. 2016;104(11):2701-2711. [46] REZAZADEH AZARI M, MOHAMMADIAN Y. Comparing in vitro cytotoxicity of graphite, short multi-walled carbon nanotubes, and long multi-walled carbon nanotubes. Environ Sci Pollut Res Int. 2020; 27(13):15401-15406. [47] PORTER AE, GASS M, MULLER K, et al. Direct imaging of single-walled carbon nanotubes in cells. Nat Nanotechnol. 2007;2(11):713-717. [48] CHENG C, MULLER KH, KOZIOL KK, et al. Toxicity and imaging of multi-walled carbon nanotubes in human macrophage cells. Biomaterials. 2009;30(25):4152-4160. [49] AL-JAMAL KT, NUNES A, METHVEN L, et al. Degree of chemical functionalization of carbon nanotubes determines tissue distribution and excretion profile. Angew Chem Int Ed Engl. 2012;51(26):6389-6393. [50] RUSSIER J, MENARD-MOYON C, VENTURELLI E, et al. Oxidative biodegradation of single- and multi-walled carbon nanotubes. Nanoscale. 2011;3(3):893-896. [51] KAGAN VE, KONDURU NV, FENG W, et al. Carbon nanotubes degraded by neutrophil myeloperoxidase induce less pulmonary inflammation. Nat Nanotechnol. 2010;5(5):354-359. [52] HOU J, WAN B, YANG Y, et al. Biodegradation of single-walled carbon nanotubes in macrophages through respiratory burst modulation. Int J Mol Sci. 2016;17(3):409. [53] XU J, HU X, JIANG S, et al. The application of multi-walled carbon nanotubes in bone tissue repair hybrid scaffolds and the effect on cell growth in vitro. Polymers (Basel). 2019;11(2):230. [54] HUANG B, VYAS C, BYUN JJ, et al. Aligned multi-walled carbon nanotubes with nanohydroxyapatite in a 3D printed polycaprolactone scaffold stimulates osteogenic differentiation. Mater Sci Eng C Mater Biol Appl. 2020;108:110374. [55] PAN L, PEI X, HE R, et al. Multiwall carbon nanotubes/polycaprolactone composites for bone tissue engineering application. Colloids Surf B. 2012;93:226-234. [56] BHATTACHARYA M, WUTTICHAROENMONGKOL-THITIWONGSAWET P, HAMAMOTO DT, et al. Bone formation on carbon nanotube composite. J Biomed Mater Res A. 2011;96(1):75-82. [57] AHADIAN S, YAMADA S, ESTILI M, et al. Carbon nanotubes embedded in embryoid bodies direct cardiac differentiation. Biomed Microdevices. 2017;19(3):57. [58] KINGHAM E, OREFFO RO. Embryonic and induced pluripotent stem cells:understanding, creating, and exploiting the nano-niche for regenerative medicine. ACS Nano. 2013;7(3):1867-1881. [59] MARTINELLI V, C WANG L, WU Y, et al. Carbon nanotubes promote growth and spontaneous electrical activity in cultured cardiac myocytes. Nano Lett. 2012;12(4):1831-1838. [60] WANG L, WU Y, HU T, et al. Aligned conductive core-shell biomimetic scaffolds based on nanofiber yarns/hydrogel for enhanced 3D neurite outgrowth alignment and elongation. Acta Biomater. 2019;96:175-187. [61] MARTINELLI V, CELLOT G, TOMA FM, et al. Carbon nanotubes instruct physiological growth and functionally mature syncytia:nongenetic engineering of cardiac myocytes. ACS Nano. 2013;7(7):5746-5756. [62] ARSLANTUNALI D, BUDAK G, HASIRCI V. Multiwalled CNT-pHEMA composite conduit for peripheral nerve repair. J Biomed Mater Res A. 2014;102(3):828-841. [63] SHAO H, LI T, ZHU R, et al. Carbon nanotube multilayered nanocomposites as multifunctional substrates for actuating neuronal differentiation and functions of neural stem cells. Biomaterials. 2018; 175:93-109. [64] CUI H, YU Y, LI X, et al. Direct 3D printing of a tough hydrogel incorporated with carbon nanotubes for bone regeneration. J Mater Chem B. 2019;7(45):7207-7217. [65] JING Z, WU Y, SU W, et al. Carbon nanotube reinforced collagen/hydroxyapatite scaffolds improve bone tissue formation in vitro and in vivo. Ann Biomed Eng. 2017;45(9):2075-2087. [66] TANAKA M, SATO Y, HANIU H, et al. A three-dimensional block structure consisting exclusively of carbon nanotubes serving as bone regeneration scaffold and as bone defect filler. PLoS One. 2017;12(2):e0172601. [67] 邸运涛,王存阳,朱惠学,等. BMP-2多肽/功能化碳纳米管复合材料修复兔颅骨缺损的实验研究[J].中国修复重建外科杂志,2021,35(3):286-294. [68] ROMAN JA, NIEDZIELKO TL, HADDON RC, et al. Single-walled carbon nanotubes chemically functionalized with polyethylene glycol promote tissue repair in a rat model of spinal cord injury. J Neurotrauma. 2011; 28(11):2349-2362. [69] AHN HS, HWANG JY, KIM MS, et al. Carbon-nanotube-interfaced glass fiber scaffold for regeneration of transected sciatic nerve. Acta Biomater. 2015;13: 324-334. [70] FALLAH-DARREHCHI M, ZAHEDI P, SAFARIAN S, et al. Conductive conduit based on electrospun poly (l-lactide-co-D, l-lactide) nanofibers containing 4-aminopyridine-loaded molecularly imprinted poly (methacrylic acid) nanoparticles used for peripheral nerve regeneration. Int J Biol Macromol. 2021;190:499-507. [71] HUANG Z, MA Y, JING W, et al. Tracing carbon nanotubes (CNTs) in rat peripheral nerve regenerated with conductive conduits composed of poly(lactide-co-glycolide) and fluorescent CNTs. ACS Biomater Sci Eng. 2020;6(11):6344-6355. [72] ZHANG J, ZHANG X, WANG C, et al. Conductive composite fiber with optimized alignment guides neural regeneration under electrical stimulation. Adv Healthc Mater. 2021;10(3):e2000604. [73] ZHOU J, CHEN J, SUN H, et al. Engineering the heart: evaluation of conductive nanomaterials for improving implant integration and cardiac function. Sci Rep. 2014;4:3733. [74] MCCAULEY MD, VITALE F, YAN JS, et al. In vivo restoration of myocardial conduction with carbon nanotube fibers. Circ Arrhythm Electrophysiol. 2019; 12(8):e007256. [75] LI X, ZHOU J, LIU Z, et al. A PNIPAAm-based thermosensitive hydrogel containing SWCNTs for stem cell transplantation in myocardial repair. Biomaterials. 2014; 35(22):5679-5688. |
| [1] | Lai Pengyu, Liang Ran, Shen Shan. Tissue engineering technology for repairing temporomandibular joint: problems and challenges [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-9. |
| [2] | Cao Yue, Ye Xinjian, Li Biyao, Zhang Yining, Feng Jianying. Effect of extracellular vesicles for diagnosis and therapy of oral squamous cell carcinoma [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1523-1530. |
| [3] | Sun Yuting, Wu Jiayuan, Zhang Jian. Physical factors and action mechanisms affecting osteogenic/odontogenic differentiation of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1531-1540. |
| [4] | Li Shuai, Liu Hua, Shang Yonghui, Liu Yicong, Zhao Qihang, Liu Wen. Stress distribution on the maxilla when wearing the Twin-block appliance for Class II malocclusion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 881-887. |
| [5] | Ding Zhili, Huang Jie, Jiang Qiang, Li Tusheng, Liu Jiang, Ding Yu. Constructing rabbit intervertebral disc degeneration models by different methods under X-ray guidance: a comparative study [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(5): 995-1002. |
| [6] | Zhang Yu, Xu Ruian, Fang Lei, Li Longfei, Liu Shuyan, Ding Lingxue, Wang Yuexi, Guo Ziyan, Tian Feng, Xue Jiajia. Gradient artificial bone repair scaffold regulates skeletal system tissue repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 846-855. |
| [7] | Xiao Fang, Huang Lei, Wang Lin. Magnetic nanomaterials and magnetic field effects accelerate bone injury repair [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 827-838. |
| [8] | Yu Shuangqi, Ding Fan, Wan Song, Chen Wei, Zhang Xuejun, Chen Dong, Li Qiang, Lin Zuoli. Effects of polylactic acid-glycolic acid copolymer/lysine-grafted graphene oxide nanoparticle composite scaffolds on osteogenic differentiation of MC3T3 cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 707-712. |
| [9] | Zhao Shuai, Li Dongyao, Wei Suiyan, Cao Yijing, Xu Yan, Xu Guoqiang. Biocompatibility of poly(vinylidene fluoride) piezoelectric bionic periosteum prepared by electrospinning [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 730-737. |
| [10] | Dang Xiaowen, Huang Hailiang, Huang Lei, Wang Yajie . Research frontiers and hotspots of carbon nanomaterials in biomedical field over the past 10 years [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 752-760. |
| [11] | Wang Sifan, He Huiyu, Yang Quan, Han Xiangzhen. miRNA-378a overexpression of macrophage cell line composite collagen sponge: anti-inflammation and tissue repair promotion [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 789-799. |
| [12] | Li Mingzhe, Ye Xiangling, Wang Bing, Yu Xiang. Preparation and osteogenic properties of liquid crystal display light-cured polylactic acid scaffold loaded with nano-tantalum [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 670-677. |
| [13] | Tian Jiayu, Li Duohua, Zhang Feng, Feng Hu, Sun Wei. Construction and performance evaluation of polycaprolactone/nanodiamond-phospholipid composite materials [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(16): 3380-3387. |
| [14] | Xin Yuan, Wu Xixi, Quan Liang, Zhang Hengtong, Ao Qiang. REG-augmented decellularized porcine cornea/hydroxyethyl methacrylate in situ integrated composite artificial cornea [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(16): 3388-3399. |
| [15] | Li Yonghang, Li Wenming, Yan Caiping, Wang Xingkuan, Xiang Chao, Zhang Yuan, Jiang Ke, Chen Lu. Critical bone defect repaired with anti-fibrosis and “H”-type core-shell bionic scaffold [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(16): 3420-3431. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||