Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (1): 114-120.doi: 10.12307/2022.971
Previous Articles Next Articles
Cellular microenvironment in nerve repair after spinal cord injury
Zhu Zhenghuan, Zou Hongjun, Song Zhiwen, Liu Jinbo
- The Third Affiliated Hospital of Soochow University, Changzhou 213003, Jiangsu Province, China
-
Received:2022-01-24Accepted:2022-02-15Online:2023-01-08Published:2022-06-06 -
Contact:Liu Jinbo, MD, Chief physician, Professor, The Third Affiliated Hospital of Soochow University, Changzhou 213003, Jiangsu Province, China Song Zhiwen, Master, Physician, The Third Affiliated Hospital of Soochow University, Changzhou 213003, Jiangsu Province, China -
About author:Zhu Zhenghuan, Master candidate, The Third Affiliated Hospital of Soochow University, Changzhou 213003, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China (General Program), No. 81972048 (to LJB); National Natural Science Foundation of China (Youth Project), No. 81901247 (to SZW)
CLC Number:
Cite this article
Zhu Zhenghuan, Zou Hongjun, Song Zhiwen, Liu Jinbo. Cellular microenvironment in nerve repair after spinal cord injury[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 114-120.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
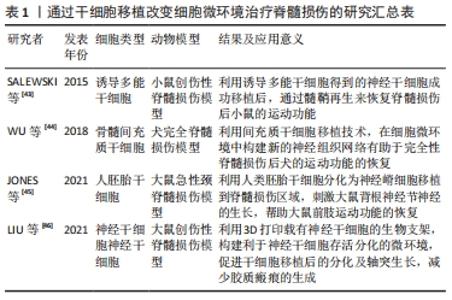
2.1 脊髓损伤后各细胞及细胞间相互调控对神经修复的影响 2.1.1 脊髓损伤后细胞微环境中胶质细胞对神经修复的作用 星形胶质细胞在构成脊髓的胶质细胞中所占比例最高,但是最初仅被认为起到神经元的结构支持与保护作用。随着对星形胶质细胞研究的深入,证实星形胶质细胞具有神经营养支持、促进突触发生、成熟和维持等多种功能[8]。星形胶质细胞还可以表达多种通道、神经递质受体,能够释放大量因子,保持同神经元的双向通讯。JAUDON等[12]就发现星形胶质细胞能够表达Kidins220,一种神经营养因子通路中的关键效应因子,参与神经元-星形胶质细胞间的相互调控。而最近,KITCHEN等[13]发现脊髓损伤后由钙调素(calmodulin,CaM)介导调控星形胶质细胞上的水通道蛋白4(aquaporin 4,AQP4)表位变化,从而促进脊髓损伤区域微环境内水肿的发生。而钙调素的抑制剂三氟拉嗪(trifluoperazine,TFP)能够通过调控星形胶质细胞内水通道蛋白4的质膜转移缓解细胞微环境内的水肿,从而减少神经细胞的水肿[13]。脊髓损伤发生后,损伤区域内的微环境发生剧烈变化,星形胶质细胞发生活化,ZAMANIAN等[14]通过基因芯片技术证实,脊髓损伤后损伤区域主要存在2种类型的反应性星形胶质细胞,分别称为A1和A2型,它们的作用可能完全相反,研究发现A1型星形胶质细胞失去了支持神经元存活、协助突触形成、生长以及吞噬的功能,能够诱导神经元和少突胶质细胞死亡,而A2型星形胶质细胞具有抗炎保护作用,能够促进脊髓缺血损伤后的神经修复。目前对于反应性星形胶质细胞的分型及其对神经元生长的作用目前的观点尚未统一,一般认为星形胶质细胞最终的命运是形成瘢痕,而瘢痕对轴突生长通常是不利的,但最近研究发现星形胶质细胞的缺失不利于瘢痕的形成,同时更不利于脊髓损伤后神经元轴突伸长[15]。故星形胶质细胞在脊髓损伤后微环境中对神经元的具体修复机制及作用值得进一步探索。 小胶质细胞在中枢神经系统中具有类似巨噬细胞的作用,并拥有类似星形胶质细胞的分型,在生理条件下处于静息态(M0),起到“免疫监视”的作用。近期LI等[16]报道了新生小鼠脊髓损伤后的无瘢痕修复,这其中的主要参与者就是新生小胶质细胞,这揭示了小胶质细胞在参与神经修复中的巨大潜力。脊髓损伤后,小胶质细胞的形态、基因表达和功能行为的变化,即发生“小胶质细胞激活”[9]。在脊髓损伤发生后的急性期,损伤中心区域的小胶质细胞迅速减少,而共表达促炎及抗炎分子的血源性巨噬细胞被招募到损伤区域占据主导地位。而在边缘区域,小胶质细胞增殖相对活跃,且主要表现为M1型促炎表型小胶质细胞[17]。M1型小胶质细胞能够释放促炎因子等杀灭病原体,但这也是一把“双刃剑”,这些释放的炎症递质、氧自由基、细胞毒性物质也会抑制髓鞘再生。鞘氨醇-1-磷酸(sphingosine-1-phospate,S1P)被认为是调节小胶质细胞向M1极化的重要介质。WANG等[18]发现鞘氨醇激酶1(sphingosine kinases 1,Sphk1)在脊髓损伤时在损伤局部微环境中的表达和活性升高,并能通过细胞内外的S1P与S1PR3结合,通过Sphk1/S1P/S1PR3轴调节小胶质细胞向促炎型M1极化,并产生促炎细胞因子。而M2小胶质细胞主要释放抗炎因子,同时发挥其吞噬功能,吞噬掉损伤的神经细胞碎片,促进组织修复和再生,支持和保护中枢神经系统的结构和功能的完整性[19]。 少突胶质细胞是由少突胶质前体细胞分化而来,是形成髓鞘的主要细胞。在McTigue等[20]的研究中,大鼠脊髓损伤后7 d,损伤中心的少突胶质细胞减少了93%。由此可见,少突胶质细胞受微环境变化影响很大,这可能与其对局部炎症递质、细胞毒性物质及活性氧等敏感有一定关系[21]。由于少突胶质细胞是构成髓鞘的主要细胞,而髓鞘可以促进轴突的快速信号传导,因此轴突的功能和健康依赖于少突胶质细胞。在脊髓损伤后微环境发生剧烈变化,导致少突胶质细胞死亡和之后的脱髓鞘的发生,而脱髓鞘会沉默或减缓动作电位的传递并最终损害神经元的功能。此时,少突胶质细胞的再生就显得尤为重要,而其主要贡献者就是少突胶质前体细胞[22]。在脊髓损伤后2周,幸存的少突胶质前体细胞持续分化,使得少突胶质细胞数量持续增加[20],此过程也帮助髓鞘重新形成,从而恢复对轴突的营养支持作用以及快速传导功能。 随着胶质细胞在脊髓损伤后的研究的深入,同一种胶质细胞在损伤不同阶段呈现不同状态和功能,这是同细胞微环境相互影响的结果。同时,脊髓损伤后存在胶质细胞之间相互调控作用,而这种调控作用也值得深入研究。 2.1.2 脊髓损伤后细胞微环境中胶质细胞间的相互作用及其影响 星形胶质细胞与小胶质细胞间的交互方式是通过分泌多种细胞因子和炎症递质来完成的。小胶质细胞能够产生白细胞介素1α、肿瘤坏死因子α、补体C1q共3种促炎因子,可诱导反应性星形胶质细胞的极化。LIDDELOW等[23]认为这3种因子在脂多糖诱导的体外星形胶质细胞极化中都是必不可少的,当小胶质细胞中缺失其中任何一种促炎因子的基因,就不具有诱导星形胶质细胞极化功能。另外,由于M1型与M2型小胶质细胞在功能上存在一定程度的相反作用,因此,SONN等[24]通过脂多糖-干扰素γ和白细胞介素4将M0型的小胶质细胞激活为M1、M2型后,再分别与星形胶质细胞共培养,实验结果表明同M0和M1型小胶质细胞相比,M2型诱导星形胶质细胞的趋化作用明显增强,且M2型小胶质细胞能够通过Wnt/β-catenin信号通路诱导星形胶质细胞的极化。此外,受损的小胶质细胞中的细胞色素C可被释放到细胞外,并以Toll样受体4(Toll-like receptor 4,TLR4)依赖的方式引起星形胶质细胞的免疫激活,促进其释放白细胞介素1β及粒-巨噬细胞集落刺激因子(Granulocyte- macrophage colony stimulating factor,GM-CSF)等[25]。由此可见,小胶质细胞在与星形胶质细胞交互中占有主导地位,但二者间的联系并非单向的。KIM等[26]在混合胶质细胞培养过程中发现,星形胶质细胞能够刺激小胶质细胞的增殖,并向M2型极化。星形胶质细胞分泌的白细胞介素6和GM-CSF等炎症递质也能够调节小胶质细胞的转录状态[27-28]。 星形胶质细胞与少突胶质细胞间的相互作用方式较为独特。脊髓损伤后,激活的A1型促炎型星形胶质细胞能够杀伤少突胶质细胞外,二者之间也存在“友好交流”的方式,甚至在一定条件下可以发生“逆分化”。MOORE等[29]通过对金属蛋白酶组织抑制因子1( tissue inhibitor of matrix metalloproteinase 1,TIMP-1)的基因敲除小鼠中枢神经系统祖细胞的培养发现,TIMP-1对星形胶质细胞及小胶质细胞生长激活具有调控作用,星形胶质细胞分泌的TIMP-1能够促进少突胶质细胞生长分化,进而帮助髓鞘再生。值得注意的是,DING等[30]利用反应性星形胶质细胞本身的干细胞潜能,通过使用神经调节蛋白1(neuroregulin1,Nrg1)激活并上调PI3K-AKT-mTOR信号通路,将原本有害的反应性星形胶质细胞转化为构成髓鞘的少突胶质细胞系细胞,用于脊髓损伤后的脱髓鞘治疗。而少突胶质细胞在一定条件下也能转化为星形胶质细胞,研究发现在NG2+的少突胶质前体细胞细胞中敲除Olig2后,能够使向星形胶质细胞发生转化,且这种转化能力随新生鼠生长时间的推移逐渐变弱[31]。 研究发现小胶质细胞在与少突胶质细胞的相互作用中占主导地位。激活的小胶质细胞在少突胶质细胞及其前体细胞分化以及髓鞘修复中起积极作用。MIRON等[32]研究发现M2型的小胶质细胞在中枢神经系统髓鞘再分化过程中,通过细胞衍生激活素A(cell-derived activin-A)促进少突胶质细胞的分化。有研究利用小胶质细胞来源的细胞外囊泡,能够维持巨噬细胞/小胶质细胞本身的活性,并促进表达G蛋白偶联受体17(G protein-coupled receptors 17,GPR17)的少突胶质前体细胞的成熟。这说明小胶质细胞能够通过细胞外囊泡这一细胞间交流的媒介,如携带跨膜肿瘤坏死因子,帮助少突胶质前体细胞分化,来对抗中枢神经系统损伤后的脱髓鞘作用。激活的巨噬细胞/小胶质细胞可以通过分泌一些有害的细胞因子如白细胞介素1β或活性氧,引起少突胶质细胞分化紊乱[33]。 胶质细胞是细胞微环境中占比最高的一类细胞,胶质细胞间存在着广泛的相互作用,这种相互的调控对它们的表型、功能变化都有着很大的影响,最终影响到脊髓损伤后的神经修复。在后续的研究中,研究者们应当重视细胞微环境中的这种相互调控关系。 2.1.3 脊髓损伤后细胞微环境中免疫细胞的作用 脊髓损伤后,除了引起神经胶质细胞的激活,同时原本不存在于正常细胞微环境的免疫细胞也从外周被招募到损伤区域。主要分为固有免疫细胞,如中性粒细胞以及单核细胞。在ZRZAVY等[17]对人脊髓损伤的研究中发现,脊髓损伤区域的中性粒细胞在损伤后3 d内达到高峰,而有些病例在损失区域边缘也可检出中性粒细胞。在脊髓损伤发生的3周后,中性粒细胞的数量有所下降,并最终消失。中性粒细胞通常在损伤区域发挥吞噬作用,并且释放炎症递质起到及时防御的作用。而SAS等[34]却在视神经损伤微环境中发现了一种独特的粒细胞亚群Ly6Glo,具有未成熟中性粒细胞的特征,拥有独特的神经保护性。他们将这种粒细胞亚群引入脊髓损伤后的小鼠,人为改变了脊髓细胞微环境,发现能够诱导背根神经节神经元的生长。KUCHAROVA等[35]提出脊髓损伤后NG2+的少突胶质前体细胞也能通过募集髓系细胞,帮助完成髓鞘的修复,他们发现敲除NG2的少突胶质前体细胞减少了对髓系细胞的募集,少突胶质前体细胞的增殖明显减少,而后导致成熟少突胶质细胞数量变少以及轴突的“再髓鞘化”水平减低。这是因为尽管巨噬细胞数量的减少使得初始损伤面积减少,但也会导致对神经损伤修复的减少,这种修复能力的下降可能与巨噬细胞缺乏后,髓鞘碎片清除减少有关。脊髓损伤后,先天免疫细胞还会激活另一类免疫细胞,即适应性免疫细胞,如T淋巴细胞和 B淋巴细胞,但是它们发挥的作用至今仍不明确。目前已知存在一种保护性自身免疫,一种对中枢神经系统损伤后的一种T细胞依赖性的免疫反应,促进神经保护因子的分泌从而发挥抗炎和神经保护的作用[36]。但在ZRZAVY等[17]的研究中,大多数脊髓损伤病例的免疫组织化学染色切片中,B细胞几乎不存在,而T 细胞则以CD8+ T 细胞占优势,并没有直接接触神经元或少突胶质细胞,而是弥漫性分布的。因此,到目前为止适应性免疫细胞在脊髓损伤中是否扮演积极的角色,还是只是被动招募到损伤区域仍是后续需要探究的难题。 2.1.4 脊髓损伤后细胞微环境中神经元对胶质细胞的调控作用 脊髓损伤后细胞微环境发生剧烈变化,神经元在物理因素和生物因素的双重作用下被大量破坏而死亡。因此在大多数报道中,神经元常是以“受害者”身份出现的。而在健康的中枢神经系统内部,神经元能够介导分泌多种信号分子或神经营养素,参与调控胶质细胞的功能。例如神经元表达的CD200(OX-2膜糖蛋白)可以识别小胶质细胞上的CD200R并能够调控小胶质细胞的过度激活[37]。LEHRMAN等[38]则发现另一在神经元中表达的CD47可以通过CD47-Sirpα信号通路,防止小胶质细胞的过度吞噬作用。而在中枢神经系统发生缺血或创伤后,神经元对小胶质细胞的制约作用减弱,进而引发了小胶质细胞的大量激活。神经元也可以通过神经细胞黏附分子(neural cell adhesion molecule,NCAM),一种参与神经系统发育的重要表面糖蛋白,抑制星形胶质细胞的增殖[39]。此外,GAUTIER等[40]还报道了在脊髓损伤后的脱髓鞘发生时,神经元能通过突触释放谷氨酸作用于表达谷氨酸受体的少突胶质细胞前体细胞,促进其向少突胶质细胞转化,从而调节重新髓鞘的形成,帮助髓鞘完成修复。 2.1.5 干细胞在脊髓损伤后细胞微环境中的作用及在治疗脊髓损伤中的应用 室管膜细胞是一群起源于脊髓中央管壁的细胞,由于目前尚不能完全区分各细胞的组成,在研究中常视作为一个整体进行研究。在这组细胞群中有脊髓来源的神经干细胞,它们表达Sox2、巢蛋白以及波形蛋白,在健康的中枢神经系统中处于静息状态,而在脊髓损伤后可以分化并替换死亡的神经元以及胶质细胞,这有助于确保神经传导通路的完整性,从而重建神经功能[41]。但是由于存在内源性神经干细胞数量较少及诱导激活方向不明确的限制,一些研究者尝试利用生物活性分子或是药物结合生物材料诱导激活的内源性的神经干细胞更多地向神经元,而不是星形胶质细胞转化[42]。而更多尝试还是倾向于对体外不同来源的干细胞移植,如间充质干细胞和神经干细胞等,并常借助具有良好生物相容性的生物材料作为载体,取得了令人欣喜的成效[43-46],尤其是对于间充质干细胞方面的应用尤为广泛,见表1。 "


间充质干细胞易于获取、培养和储存,并具有由多种炎症因子介导的向损伤部位迁移的特性,因此不但能够直接移植到受损的脊髓中,还可以通过静脉注射进行输注,简单易行且效果也令人满意[47]。干细胞还能同脊髓损伤后微环境内其他组分发生相互作用,产生多种神经营养因子,这有利于重塑损伤部位的微环境,加速轴突的生长。另外,由干细胞分化而来的中间神经元可以引起轴突萌发,脊髓的近端和远端与损伤部位相连,诱导新突触的形成。由此可见,通过应用干细胞人为改变脊髓损伤后微环境内的细胞组成,在治疗脊髓损伤中的前景广阔,但也存在干细胞生存率不高、免疫排斥、肿瘤形成以及伦理问题等需要解决。 因此,在以往研究中单纯探讨脊髓损伤后损伤区域的单一细胞功能已经不能完全解释复杂脊髓微环境在脊髓损伤后作用,而越来越多的研究也证实细胞之间调控作用是脊髓损伤微环境的重要组成之一,其在脊髓损伤后信号传导、免疫调控、炎症释放等方面具有重要作用。 2.2 脊髓损伤后细胞微环境中细胞外组分对脊髓损伤后神经修复的影响 脊髓损伤后,损伤区域内的细胞微环境发生剧烈变化,微环境中的细胞外组分也在神经修复中发挥着重要作用。在这里,作者主要以作为“脚手架”的细胞外基质、参与细胞间交流的细胞外调节因子,起到“沟通桥梁”作用的细胞外囊泡为例,综述细胞外组分在脊髓损伤后对神经修复的影响。 2.2.1 细胞外基质 细胞外基质是脊髓微环境中的重要非细胞组分。细胞外基质可以调节神经组织的活动,同时细胞外基质通过形成交织的网络起到了支持与锚定神经元及胶质细胞的作用。富含大量细胞外基质的基底膜与彼此连接紧密的内皮细胞共同形成血脊髓屏障[48]。当脊髓损伤后,细胞外基质在损伤区域轴突生长、神经胶质增生、血运重建和可塑性等方面都发挥着重要影响。随着损伤区域微环境发生改变,细胞外基质成分变得失调,结构也发生改变,变得僵硬,在重塑过程中细胞外基质各组分发生沉积交联,最终形成胶质瘢痕[11]。胶质瘢痕的形成同时还上调了硫酸软骨素蛋白聚糖和其他细胞外基质分子的表达[49],可以有效抑制受伤轴突再进入损伤病灶。脊髓损伤后,硫酸软骨素蛋白聚糖从星形胶质瘢痕中心向外辐射呈梯度的上调,是限制轴突生长和功能恢复的关键细胞外基质抑制分子。当然,细胞外基质在脊髓损伤后并非完全不利于神经的修复。在TSATA等[50]的研究中发现,斑马鱼脊髓损伤部位的细胞外基质是轴突再生所必需的,损伤部位周围的PDGFRb细胞能够通过上调轴突生长促进的细胞外基质基因ctrc1a和col12a1a/b的表达,并减少不利于神经修复的基质分子lum和mfap2的合成。此外,非胶原糖蛋白,包括纤维连接蛋白和层粘连蛋白[51-52],在轴突生长和神经修复中起到了良好的支持作用,并能促进细胞的迁移。LI等[16]报道了新生小胶质细胞来源的纤维连接蛋白,在介导脊髓损伤后创面无瘢痕愈合连接两端的过程中,起着至关重要的作用。已有大量的研究开始利用这些特性,使用具有良好生物相容性的水凝胶或是其他生物纳米材料模仿天然细胞外基质[53-54],有效模拟和重建损伤区域内的细胞微环境,促进轴突伸长。 2.2.2 细胞外调节因子 细胞外调节因子是一组细胞分泌的小分子或中等分子质量的可溶性蛋白质与糖蛋白,主要包括细胞因子和趋化因子2大类,作用于特异的靶细胞受体,具有广泛的生物学效应。脊髓损伤诱导了损伤区域微环境的炎症风暴,激活免疫细胞及非免疫细胞分泌大量的炎症细胞因子如白细胞介素1β和活性氧等,以及多种趋化因子均参与诱发了脊髓损伤后继发性炎症反应,而这些细胞外调节因子又能加强对反应性胶质细胞激活,从而形成级联效应。当然损伤区域也存在一些抗炎细胞因子,一部分研究者通过提高损伤区域微环境内抗炎因子的表达量,控制炎症治疗脊髓损伤,但大多数研究的重点放在应用促炎因子的受体抑制剂或是阻断相关通路,下调细胞微环境内不利的细胞外调节因子的表达水平[55-57],见表2。 "

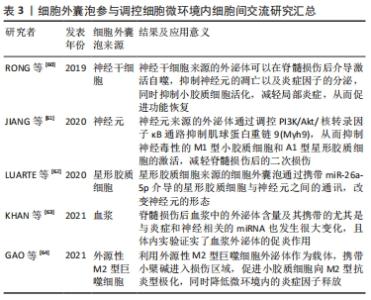
最近,LEIBINGER等[58]引入原本不存在于脊髓细胞微环境中的超级白细胞介素6这种细胞因子,帮助脊髓损伤后神经功能的修复。这种人为改变原本的脊髓细胞微环境,抑制促炎因子降低微环境中的炎症水平,引入有利于神经生长的细胞因子,这将是后续对脊髓损伤后微环境研究的全新方向。 2.2.3 细胞外囊泡 值得关注的是,目前一种参与细胞间信号交流的物质——细胞外囊泡正被广泛研究。由于几乎所有类型的细胞都能分泌细胞外囊泡,研究者们曾一度以为细胞外囊泡仅仅是细胞代谢的废物而被忽视。而最近的十多年里,人们逐步发现细胞外囊泡能够作为多种生物活性物质,如各种蛋白质(包括细胞因子及膜蛋白等)、核酸以及脂质、糖类等生物活性物质的载体,可以对外界刺激作出反应,是一种脊髓损伤等中枢神经系统损伤后的关键病理介质[59]。因此,应用细胞外囊泡作为载体,参与改变细胞微环境中细胞间的交流,从而帮助脊髓损伤的修复也成为了研究的热点。 如前所述,神经干细胞在脊髓损伤后的应用前景很广,但也存在一系列问题,而神经干细胞外泌体的成功提取,可能是解决这些难题的有利方法。RONG等[60]成功提取了神经干细胞来源的细胞外囊泡(NSC-sEVs),并发现该囊泡可以抑制脊髓损伤后神经元的凋亡及小胶质细胞活化,减轻局部炎症,从而促进功能恢复;此外,他们还发现该囊泡可以介导激活自噬,抑制细胞凋亡和炎性因子的分泌。JIANG等[61]应用miRNA微阵列筛选出神经元来源的外泌体中富集程度最高的miR-124-3p,并用一系列实验证明通过调控PI3K/Akt/核转录因子κB通路而抑制Myh9,可以在体内外抑制神经毒性的M1型小胶质细胞和A1型星形胶质细胞的激活,减轻脊髓损伤后的二次损伤。除了上文已经提及的小胶质细胞来源的细胞外囊泡外,也有研究者报道了星形胶质细胞来源的细胞外囊泡及其携带的miRNA-26a-5p介导的星形胶质细胞与神经元之间的通讯,改变神经元的形态[62]。由此可见,脊髓细胞微环境中不同细胞组分释放的细胞外囊泡是细胞间沟通的重要媒介[60-64],细胞外囊泡用于治疗脊髓损伤主要就是利用其携带的各种生物活性物质,具有广泛的研究前景,见表3。 "
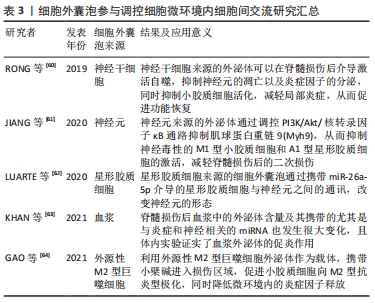
| [1] BEJARANO L, JORDĀO M, JOYCE J. Therapeutic targeting of the tumor microenvironment. Cancer Discov. 2021;11(4):933-959. [2] HUANG Y, HUANG J, WANG Y, et al. Progressive aggregation-induced emission strategy for imaging of aluminum ions in cellular microenvironment. Talanta. 2020;211:120699. [3] STUDENT S, MILEWSKA M, OSTROWSKI Z, et al. Microchamber microfluidics combined with thermogellable glycomicrogels - Platform for single cells study in an artificial cellular microenvironment. Mater Sci Eng C Mater Biol Appl. 2021; 119:111647. [4] HIRANO N, KUSUHARA H, SUEYOSHI Y, et al. Ethanol treatment of nanoPGA/PCL composite scaffolds enhances human chondrocyte development in the cellular microenvironment of tissue-engineered auricle constructs. PLoS One. 2021;16(7):e0253149. [5] ZHAO Y, XIAO Z, CHEN B, et al. The neuronal differentiation microenvironment is essential for spinal cord injury repair. Organogenesis. 2017;13(3):63-70. [6] SHU J, CHENG F, GONG Z, et al. Transplantation strategies for spinal cord injury based on microenvironment modulation. Curr Stem Cell Res Ther. 2020;15(6):522-530. [7] TRAN A, WARREN P, SILVER J. The biology of regeneration failure and success after spinal cord injury. Physiol Rev. 2018;98(2):881-917. [8] LIDDELOW S, BARRES B. SnapShot: astrocytes in health and disease. Cell. 2015; 162(5):1170-1170.e1. [9] KETTENMANN H, HANISCH UK, NODA M, et al. Physiology of microglia. Physiol Rev. 2011;91(2):461-553. [10] MELETIS K, BARNABE-HEIDER F, CARLEN M, et al. Spinal cord injury reveals multilineage differentiation of ependymal cells. PLoS Biol. 2008;6(7):e182. [11] BONNANS C, CHOU J, WERB Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol. 2014;15(12):786-801. [12] JAUDON F, CHIACCHIARETTA M, ALBINI M, et al. Kidins220/ARMS controls astrocyte calcium signaling and neuron-astrocyte communication. Cell Death Differ. 2020;27(5):1505-1519. [13] KITCHEN P, SALMAN MM, HALSEY AM, et al. Targeting aquaporin-4 subcellular localization to treat central nervous system edema. Cell. 2020;181(4):784-799.e19. [14] ZAMANIAN JL, XU L, FOO LC, et al. Genomic analysis of reactive astrogliosis. J Neurosci. 2012;32(18):6391-6410. [15] ANDERSON MA, BURDA JE, REN Y, et al. Astrocyte scar formation aids central nervous system axon regeneration. Nature. 2016;532(7598):195-200. [16] LI Y, HE X, KAWAGUCHI R, et al. Microglia-organized scar-free spinal cord repair in neonatal mice. Nature. 2020;587(7835):613-618. [17] ZRZAVY T, SCHWAIGER C, WIMMER I, et al. Acute and non-resolving inflammation associate with oxidative injury after human spinal cord injury. Brain. 2021;144(1): 144-161. [18] WANG C, XU T, LACHANCE BB, et al. Critical roles of sphingosine kinase 1 in the regulation of neuroinflammation and neuronal injury after spinal cord injury. J Neuroinflammation. 2021;18(1):50. [19] BELLVER-LANDETE V, BRETHEAU F, MAILHOT B, et al. Microglia are an essential component of the neuroprotective scar that forms after spinal cord injury. Nat Commun. 2019;10(1):518. [20] MCTIGUE DM, WEI P, STOKES BT. Proliferation of NG2-positive cells and altered oligodendrocyte numbers in the contused rat spinal cord. J Neurosci. 2001;21(10):3392-3400. [21] GIACCI MK, BARTLETT CA, SMITH NM, et al. Oligodendroglia are particularly vulnerable to oxidative damage after neurotrauma in vivo. J Neurosci. 2018; 38(29):6491-6504. [22] ASSINCK P, DUNCAN GJ, PLEMEL JR, et al. Myelinogenic Plasticity of Oligodendrocyte Precursor Cells following Spinal Cord Contusion Injury. J Neurosci. 2017;37(36): 8635-8654. [23] LIDDELOW SA, GUTTENPLAN KA, CLARKE LE, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. 2017;541(7638):481-487. [24] SONN I, NAKAMURA M, RENAULT-MIHARA F, et al. Polarization of reactive astrocytes in response to spinal cord injury is enhanced by M2 macrophage-mediated activation of wnt/beta-catenin pathway. Mol Neurobiol. 2020;57(4): 1847-1862. [25] WENZEL TJ, BAJWA E, KLEGERIS A. Cytochrome c can be released into extracellular space and modulate functions of human astrocytes in a toll-like receptor 4-dependent manner. Biochim Biophys Acta Gen Subj. 2019;1863(11):129400. [26] KIM S, SON Y. Astrocytes stimulate microglial proliferation and m2 polarization in vitro through crosstalk between astrocytes and microglia. Int J Mol Sci. 2021; 22(16):8800. [27] TANG Y, LIU L, XU D, et al. Interaction between astrocytic colony stimulating factor and its receptor on microglia mediates central sensitization and behavioral hypersensitivity in chronic post ischemic pain model. Brain Behav Immun. 2018; 68:248-260. [28] TRAN AP, SUNDAR S, YU M, et al. Modulation of receptor protein tyrosine phosphatase sigma increases chondroitin sulfate proteoglycan degradation through cathepsin b secretion to enhance axon outgrowth. J Neurosci. 2018;38(23):5399-5414. [29] MOORE CS, MILNER R, NISHIYAMA A, et al. Astrocytic tissue inhibitor of metalloproteinase-1 (TIMP-1) promotes oligodendrocyte differentiation and enhances CNS myelination. J Neurosci. 2011;31(16):6247-6254. [30] DING Z, DAI C, ZHONG L, et al. Neuregulin-1 converts reactive astrocytes toward oligodendrocyte lineage cells via upregulating the PI3K-AKT-mTOR pathway to repair spinal cord injury. Biomed Pharmacother. 2021;134:111168. [31] ZUO H, WOOD WM, SHERAFAT A, et al. Age-dependent decline in fate switch from ng2 cells to astrocytes after olig2 deletion. J Neurosci. 2018;38(9):2359-2371. [32] MIRON VE, BOYD A, ZHAO JW, et al. M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat Neurosci. 2013; 16(9):1211-1218. [33] SAKAKIBARA S, NAKADATE K, OOKAWARA S, et al. Non-cell autonomous impairment of oligodendrocyte differentiation precedes CNS degeneration in the Zitter rat: implications of macrophage/microglial activation in the pathogenesis. BMC Neurosci. 2008;9:35. [34] SAS AR, CARBAJAL KS, JEROME AD, et al. A new neutrophil subset promotes CNS neuron survival and axon regeneration. Nat Immunol. 2020;21(12):1496-505. [35] KUCHAROVA K, STALLCUP WB. NG2-proteoglycan-dependent contributions of oligodendrocyte progenitors and myeloid cells to myelin damage and repair. J Neuroinflammation. 2015;12:161. [36] MARTIñóN S, GARCíA-VENCES E, TOSCANO-TEJEIDA D, et al. Long-term production of BDNF and NT-3 induced by A91-immunization after spinal cord injury. BMC Neurosci. 2016;17(1):42. [37] TIAN L, RAUVALA H, GAHMBERG CG. Neuronal regulation of immune responses in the central nervous system. Trends Immunol. 2009;30(2):91-99. [38] LEHRMAN EK, WILTON DK, LITVINA EY, et al. CD47 protects synapses from excess microglia-mediated pruning during development. Neuron. 2018;100(1):120-134.e6. [39] SAINI V, LOERS G, KAUR G, et al. Impact of neural cell adhesion molecule deletion on regeneration after mouse spinal cord injury. Eur J Neurosci. 2016;44(1):1734-1746. [40] GAUTIER HO, EVANS KA, VOLBRACHT K, et al. Neuronal activity regulates remyelination via glutamate signalling to oligodendrocyte progenitors. Nat Commun. 2015;6:8518. [41] CIZKOVA D, NAGYOVA M, SLOVINSKA L, et al. Response of ependymal progenitors to spinal cord injury or enhanced physical activity in adult rat. Cell Mol Neurobiol. 2009;29(6-7):999-1013. [42] LACROIX S, HAMILTON LK, VAUGEOIS A, et al. Central canal ependymal cells proliferate extensively in response to traumatic spinal cord injury but not demyelinating lesions. PLoS One. 2014;9(1):e85916. [43] SALEWSKI RP, MITCHELL RA, LI L, et al. Transplantation of induced pluripotent stem cell-derived neural stem cells mediate functional recovery following thoracic spinal cord injury through remyelination of axons. Stem Cells Transl Med. 2015;4(7):743-754. [44] WU G, SHI H, CHE M, et al. Recovery of paralyzed limb motor function in canine with complete spinal cord injury following implantation of MSC-derived neural network tissue. Biomaterials. 2018;181:15-34. [45] JONES I, NOVIKOVA L, WIBERG M, et al. Human embryonic stem cell-derived neural crest cells promote sprouting and motor recovery following spinal cord injury in adult rats. Cell transplantation. 2021;30:963689720988245. [46] LIU X, HAO M, CHEN Z, et al. 3D bioprinted neural tissue constructs for spinal cord injury repair. Biomaterials. 2021;272:120771. [47] COFANO F, BOIDO M, MONTICELLI M, et al. Mesenchymal stem cells for spinal cord injury: current options, limitations, and future of cell therapy. Int J Mol Sci. 2019;20(11):2689. [48] BARTANUSZ V, JEZOVA D, ALAJAJIAN B, et al. The blood-spinal cord barrier: morphology and clinical implications. Ann Neurol. 2011;70(2):194-206. [49] DAVID S, LACROIX S. Molecular approaches to spinal cord repair. Annu Rev Neurosci. 2003;26:411-440. [50] TSATA V, MOLLMERT S, SCHWEITZER C, et al. A switch in pdgfrb(+) cell-derived ECM composition prevents inhibitory scarring and promotes axon regeneration in the zebrafish spinal cord. Dev Cell. 2021;56(4):509-524.e9. [51] TOM VJ, DOLLER CM, MALOUF AT, et al. Astrocyte-associated fibronectin is critical for axonal regeneration in adult white matter. J Neurosci. 2004;24(42):9282-9290. [52] LI X, LIU X, JOSEY B, et al. Short laminin peptide for improved neural stem cell growth. Stem Cells Transl Med. 2014;3(5):662-670. [53] WISEMAN TM, BARON-HEERIS D, HOUWERS IGJ, et al. Peptide hydrogel scaffold for mesenchymal precursor cells implanted to injured adult rat spinal cord. Tissue Eng Part A. 2021;27(15-16):993-1007. [54] YANG Y, FAN Y, ZHANG H, et al. Small molecules combined with collagen hydrogel direct neurogenesis and migration of neural stem cells after spinal cord injury. Biomaterials. 2021;269:120479. [55] THOMPSON C, ZURKO J, HANNA B, et al. The therapeutic role of interleukin-10 after spinal cord injury. J Neurotrauma. 2013;30(15):1311-1324. [56] YATES A, JOGIA T, GILLESPIE E, et al. Acute IL-1RA treatment suppresses the peripheral and central inflammatory response to spinal cord injury. J Neuroinflamm. 2021;18(1):15. [57] SUN F, ZHANG H, HUANG T, et al. miRNA-221 Regulates spinal cord injury-induced inflammatory response through targeting tnf-α expression. Biomed Res Int. 2021; 2021:6687963. [58] LEIBINGER M, ZEITLER C, GOBRECHT P, et al. Transneuronal delivery of hyper-interleukin-6 enables functional recovery after severe spinal cord injury in mice. Nat Commun. 2021;12(1):391. [59] EL ANDALOUSSI S, MAGER I, BREAKEFIELD XO, et al. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12(5):347-357. [60] RONG Y, LIU W, WANG J, et al. Neural stem cell-derived small extracellular vesicles attenuate apoptosis and neuroinflammation after traumatic spinal cord injury by activating autophagy. Cell Death Dis. 2019;10(5):340. [61] JIANG D, GONG F, GE X, et al. Neuron-derived exosomes-transmitted miR-124-3p protect traumatically injured spinal cord by suppressing the activation of neurotoxic microglia and astrocytes. J Nanobiotechnol. 2020;18(1):105. [62] LUARTE A, HENZI R, FERNANDEZ A, et al. Astrocyte-derived small extracellular vesicles regulate dendritic complexity through mir-26a-5p activity. Cells. 2020; 9(4):930. [63] KHAN N, CAO T, HE J, et al. Spinal cord injury alters microRNA and CD81+ exosome levels in plasma extracellular nanoparticles with neuroinflammatory potential. Brain Behav Immun. 2021;92:165-183. [64] GAO Z, ZHANG C, XIA N, et al. Berberine-loaded M2 macrophage-derived exosomes for spinal cord injury therapy. Acta Biomateri. 2021;126:211-223. |
| [1] | Zhang Yujuan, Yuan Yitong, Du Ruochen, Tian Feng, Fu Yuan, Wang Chunfang. miR-31 promotes the proliferation and migration of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 66-71. |
| [2] | Liu Runyuan, Dong Ming, Han Wenqing, Dong Juhong, Niu Weidong. Application and progress of small extracellular vesicles in periodontal and pulp regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 83-90. |
| [3] | Liu Zhuoran, Jiang Ming, Li Yourui. Extracellular vesicles in chronic periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 99-104. |
| [4] | Shi Xu, Li Ruiyu, Zhang Bing, Chen Qi, Zuo Hua. Effect of inflammatory reaction mediated by microglia polarization in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 121-129. |
| [5] | Zhang Haobo, Zhao Yunan, Yang Xuejun. Role and therapeutic implications of pyroptosis in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1445-1451. |
| [6] | Kan Houming, Fan Lijun, Chen Xuetai, Shen Wen. Application of platelet-rich plasma in neuropathic pain [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1286-1292. |
| [7] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [8] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998. |
| [9] | Wen Xiaoyu, Sun Yuhao, Xia Meng. Effects of serum containing Wuzang Wenyang Huayu Decoction on phosphorylated-tau protein expression in Alzheimer’s disease cell model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1068-1073. |
| [10] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [11] | Huang Chuanjun, Zou Yu, Zhou Xiaoting, Zhu Yangqing, Qian Wei, Zhang Wei, Liu Xing. Transplantation of umbilical cord mesenchymal stem cells encapsulated in RADA16-BDNF hydrogel promotes neurological recovery in an intracerebral hemorrhage rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 510-515. |
| [12] | Liu Yapu, Su Yuanyuan, Liu Qi, Yang Zhou, Li Rong, Huang Zucheng, Huang Zhiping, Wu Xiaoliang, Zhu Qingan. Antioxidative stress of trihydroxyethyl rutin on cervical spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5868-5874. |
| [13] | Ma Jiang, Zhang Di, Zhao Tianyu, Liu Xiaoxiao, Wang Ju, Lu Li, Wang Ying, Jin Song. Mechanism and application prospects of motor imagery in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5897-5904. |
| [14] | Li Chuanhong, Yu Xing, Yang Yongdong, Yang Kaitan, Zhao He. Subarachnoid injection via the posterior atlanto-occipital interspace is an alternative way of administration in a rat model of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5376-5383. |
| [15] | Wang Kang, Zhi Xiaodong, Zhang Yuqiang, Gong Chao, Wang Chenliang, Wang Wei. Human amniotic epithelial cells regulate the proliferation, apoptosis and extracellular matrix synthesis of articular chondrocytes by activating the EGFR/ERK1 signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 4967-4974. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||