Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (34): 5517-5522.doi: 10.12307/2021.249
Previous Articles Next Articles
Biological advantages of biodegradable microspheres in bone regeneration
Ma Shiqing, Wang Jie, Gao Ping, Liu Zihao
- Dental Hospital, Tianjin Medical University, Tianjin 300070, China
-
Received:2020-12-29Revised:2020-12-30Accepted:2021-02-05Online:2021-12-08Published:2021-07-28 -
Contact:Liu Zihao, PhD, Lecturer, Dental Hospital, Tianjin Medical University, Tianjin 300070, China -
About author:Ma Shiqing, PhD, Attending physician, Dental Hospital, Tianjin Medical University, Tianjin 300070, China -
Supported by:the National Natural Science Foundation of China, No. 81701019 (to MSQ); General Project of Tianjin Education Committee, No. 2019KJ173 (to LZH)
CLC Number:
Cite this article
Ma Shiqing, Wang Jie, Gao Ping, Liu Zihao. Biological advantages of biodegradable microspheres in bone regeneration[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5517-5522.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
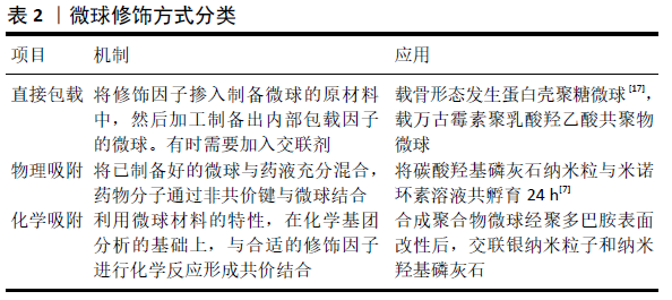
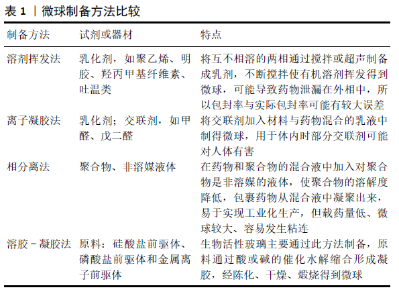
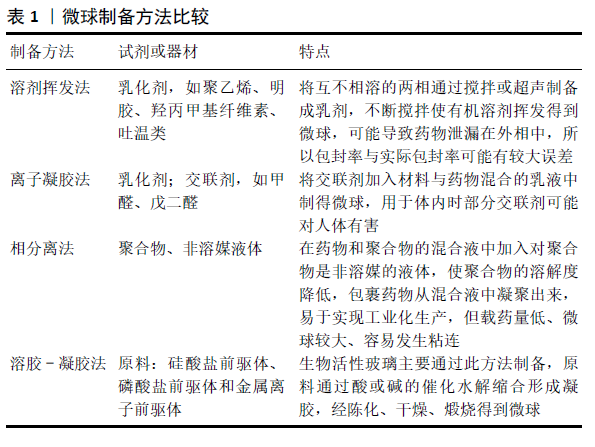
2.1.1 无机材料 生物活性陶瓷包括磷酸盐和含硅类材料。在磷酸盐材料中被研究最多的有羟基磷灰石、磷酸三钙及它们的混合物双相磷酸钙等。虽然磷酸钙陶瓷表现出的骨诱导性较弱,但近年研究发现纳米表面结构尤其是具有微纳米孔隙的复合结构,能够赋予其调控细胞骨再生行为的生物活性[6]。生物活性陶瓷虽机械强度优异,但明显的缺点是脆性大,与聚乙酸内酯或壳聚糖等聚合物混合能明显提升材料的弹性模量、抗张强度、亲水性及生物活性[7]。近年来,无定型磷酸钙微球在骨缺损修复领域中得到越来越多的关注,其骨传导及细胞黏附性能要优于羟基磷灰石,生物降解速率比可降解的磷酸三钙还要高,具备pH值响应的降解特性,对蛋白质的吸附能力良好,在骨缺损修复领域具有良好的发展前景[8]。 生物活性玻璃是含硅类生物活性陶瓷,包括二氧化硅和硅酸盐类材料,具有良好的生物相容性,在体内降解后可以释放钠离子、钙离子、硅酸根和磷酸根离子等,并通过矿化沉积纳米羟基磷灰石与骨组织发生化学键合[9]。生物活性玻璃的制备工艺由最初的熔融法发展为避免了热处理的溶胶-凝胶法。随着溶胶-凝胶法的出现,介孔材料也相应被合成出来[10]。介孔材料是孔径介于 2-50 nm 之间的一类分子筛材料,二氧化硅介孔材料由于具有丰富的孔隙结构常被用作药物运输载体。陈雪颖[11]将溶胶-凝胶法与模板法结合,通过改变反应条件、调节原料配比制备出放射状介孔生物活性玻璃微球,低浓度微球具有较好的生物相容性,而在较高浓度情况下则会抑制细胞增殖。钙含量高的微球有效加快了钙磷酸盐的沉积速度,具有良好的生物矿化活性。 由于骨组织本身含有羟基磷灰石这种无机成分,因此生物活性陶瓷、生物活性玻璃等无机材料与天然骨组织具有较高的适配性,拥有有机材料不可替代的机械性能及骨传导性能。近年此领域延伸出许多结构、组分富有特色的无机材料,在有机材料、骨再生因子的修饰下无机材料将具有十分广阔的发展前景,有望广泛应用于临床手术中。 2.1.2 天然高分子材料 天然高分子材料大多来源广泛,具有良好的生物可降解性及生物相容性等优点,常用于制作缓释微球的材料包括壳聚糖、明胶、海藻酸钠等。 壳聚糖是自然界中存在的唯一带正电的碱性氨基多糖,除了具备上述天然高分子材料的优良特性外,还具有抑菌的功能,在癌症治疗、局部炎症治疗、组织再生药物装载等方面得到了广泛应用[12]。XIA等[13]利用异位成骨模型证明了壳聚糖微球给药系统可显著增强重组骨形态发生蛋白异位成骨的诱导和促进作用,为壳聚糖/重组骨形态发生蛋白微球给药系统的临床应用提供了研究数据。此外,由于壳聚糖具有阳离子性质及较高的氨基反应活性,可以很容易地修饰成各种物理或化学衍生物以改善其低水溶性的性质[14]。 明胶是胶原部分水解得到的一类蛋白质,具有比胶原更低的抗原性,并且保留了其氨基酸序列,增强了细胞的黏附活性。ANNAMALAI等[15]研发了明胶载骨形态发生蛋白微球,研究发现炎性细胞的存在能够提高此微球降解速率,表明与炎症反应同步的控释系统可以更好地实现对生长因子的控制释放,在骨折修复的炎症阶段有良好的应用前景。 海藻酸钠是一种天然多糖,是从藻类中提取碘和甘露醇之后的产物,经常用于制作核壳结构微球的外壳。CALASANS-MAIA等[16]使用碳酸羟基磷灰石纳米粒负载了米诺环素,并用海藻酸盐包裹形成微球,无海藻酸盐包裹的粉末降低了成骨细胞90%的活性,而由于海藻酸钠外壳的存在米诺环素对于成骨细胞活性的影响被消除,体内实验显示复合微球植入42 d后表现出明显的新骨形成,表明海藻酸钠用于构建复合结构微球的巨大潜力。 2.1.3 合成聚合物材料 常用于制备微球的合成聚合物材料包括聚乳酸、聚乳酸羟乙酸共聚物、聚己内酯等,其优点是有良好的力学性能、降解速度可调,不足之处在于不具有骨诱导性、降解速度通常较慢,同时其降解产物会导致组织局部环境的酸性升高,进而导致组织炎症反应。 聚乳酸由乳酸在特定条件下缩水聚合而成,分为左旋聚乳酸、右旋聚乳酸、外消旋聚乳酸等。为改善聚合物微球的上述缺陷,ZHAO等[17]采用共混挤出法将聚乳酸微球装载上能够中和其酸性降解产物的晶须状MgO和纳米粒子状MgO,通过对微球的体外形貌、降解率、吸水率、pH值变化进行长达12个月的观察,发现MgO的存在增加了材料的吸水率,从而加速了基体的分解,晶须状材料的高结晶度降低了材料的降解速率;体内组织学切片观察结果表明此支架生物活性良好,能够应用于骨缺损的修复。此外,将聚乳酸-碳酸三亚甲基共聚物与天然高分子壳聚糖结合被证明能够改善材料的压缩回复性,减少酸性副产物[18]。 聚乳酸羟乙酸共聚物由乳酸和乙醇酸聚合而来,具有降解速率可控的优势,其药物释放受共聚物中乳酸与乙醇酸的比例[19]、微球的大小和使用的稳定剂等因素的影响[20-21]。ZHOU等[22]采用双乳化溶剂蒸发法制备了载万古霉素聚乳酸羟乙酸共聚物微球,并借助聚多巴胺将其包裹吸附在3D打印的聚己内酯纤维支架上,聚多巴胺明显提升了支架对兔骨髓间充质干细胞的黏附和增殖作用,而万古霉素微球的加入对细胞增殖无显著抑制作用,其抑菌作用可持续4周以上,显示了支架良好的生物相容性和抑菌性能。 天然高分子材料和合成聚合物材料均属于有机材料,相对于无机材料来说,由于具有较多的活性基团结构更为复杂灵活,更容易对其表面进行化学成分的修饰,这也极大地扩展了有机材料微球的应用。有机材料除了单独作为药物载体外,还常用于修饰无机材料,以弥补其弹性、生物活性的不足。 2.2 微球在骨组织再生中的应用 目前使用的微球均已具备良好的生物相容性和骨传导性,但大部分材料的骨诱导性能仍不够理想。近年来,关于微球的功能性修饰主要通过负载促骨再生因子、抗感染因子及促血管再生因子等方法进行。修饰因子的加载方式又包括物理吸附、化学吸附、直接包载等,加载方式是修饰因子释放速率的决定性因素,其具体应用见表2。"
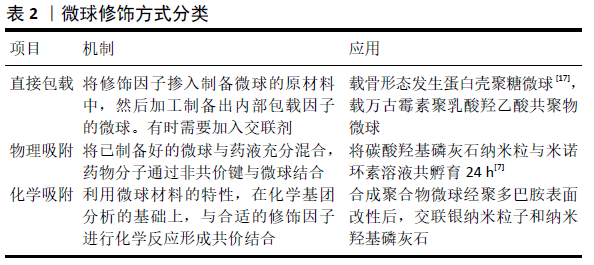

2.2.1 装载促骨再生因子 纳米碳材料:碳衍生物,比如碳点[23]、碳纳米管和石墨烯是生物材料领域新兴的十分有前景的材料[24-25],均被证明可以增强干细胞分化。碳点具有良好的生物相容性,并且在细胞内外都有强荧光性质,能够代替其他的荧光染料跟踪,并促进骨髓间充质干细胞的成骨分化[26]。KHAJURIA等[27]采用水热/共沉淀法制备了氮掺杂碳点与羟基磷灰石偶联纳米颗粒,与纯羟基磷灰石纳米颗粒相比,氮掺杂碳点的加入显著提高了斑马鱼颌骨再生能力,表明氮掺杂碳点/羟基磷灰石偶联纳米颗粒在骨再生和骨折愈合方面具有治疗潜力。 生长因子:生长因子种类繁多,主要分为转化生长因子、血小板衍生生长因子、成纤维细胞生长因子等。其中,骨形态发生蛋白属于转化生长因子β家族,其组成超过30多种蛋白质,是目前发现的唯一能独立诱导成骨的因子,在骨再生和骨吸收的过程中均发挥着重要作用[28],已被美国食品药品监督管理局批准应用于骨再生。由于骨形态发生蛋白的浓度与疗效的关系仍不明确,临床上为了取得良好的治疗效果一般采取较高浓度剂量,因此许多与浓度相关的不良反应逐渐引起人们的重视[29]。合适的微球载体材料被证明可有效解决这个问题,近年有研究以大豆卵磷脂为载体通过两步法制备了聚乳酸羟乙酸共聚物/大豆卵磷脂/骨形态发生蛋白微球,成功地将亲水蛋白捕获到疏水聚合物的微球核心中,与聚乳酸羟乙酸共聚物/骨形态发生蛋白微球相比,此微球具有更高的骨形态发生蛋白截留效率和受控的三相释放行为,在体内外更有利于基质的矿化及干细胞的附着、增殖、分化[30]。 磁性氧化铁纳米颗粒:氧化铁纳米颗粒具有优异的生物相容性和独特的磁性,它们在特定的外部磁场中具有可控的响应特性,在输送治疗剂、研究控制干细胞等方面有很大的应用潜力[31],但是由于降解过程中铁元素最终会进入到生物体的铁代谢过程中,有影响到特定脏器功能的风险[32]。LU等[33]利用已获美国食品药品监督管理局批准的药物SrFe12O19修饰介孔生物玻璃/壳聚糖多孔支架得到磁性壳聚糖微球,其产生的磁场除了促进了新骨的再生外,还改善了光热转换性能,具有优异的抗肿瘤效果。 镁离子(Mg2+):Mg2+具有生物活性,能促进骨组织再生,其促进效果与Mg2+浓度密切相关。YUAN等[34]通过调节聚乳酸羟乙酸共聚物微球中MgO/MgCO3的比例来控制Mg2+释放速率,发现当MgO/MgCO3为1∶1时,微球对骨髓间充质干细胞的矿物质沉积和成骨分化促进作用最强。LIN等[35]制备了以聚乳酸羟乙酸共聚物和Mg2+作为核心、海藻酸钠作为外壳的核壳结构微球,并利用微流控技术进行镁离子的精准控释,速率约50×10-6/d,此微球使形成骨的杨氏模量恢复到周围成熟骨的96%,证明其促进骨组织再生的良好效果。 促骨再生药物:可供选择的用于促进骨组织再生或治疗骨质疏松的药物种类繁多,然而其中很多药物水溶性较差,剂量不易控制,易产生严重的不良反应。将此类药物包载于微球中可以显著提升其功效,近年的研究已覆盖地塞米松[36]、阿司匹林[37]、辛伐他汀[38]、阿托伐他汀[39]、二膦酸盐等骨形成药物的包载[40]。 2.2.2 装载抗感染因子 金属纳米粒子:银纳米粒子近年被广泛应用于提高材料的抗菌性能。多孔微球表面的聚多巴胺通过其邻苯二酚基团作为还原剂可以固定硝酸银水溶液中的银纳米粒子。WEI 等[41]先用W1/O/W2法合成了聚乳酸-聚乙二醇-聚乳酸微球,并借助聚多巴胺涂层利用上述方法装载了银纳米粒子和纳米羟基磷灰石,在体外和鼠颅骨模型中均显示出良好的生物相容性、抗菌活性和成骨性。PORRELLI等[42]以海藻酸盐、壳聚糖、羟基磷灰石和纳米银为原料制备了适配外科注射器挤压压力的可注射纳米复合微球材料,实验结果表明微球1周后释放银量不足6%,该微球能杀灭多种菌体,并能破坏预形成的细菌生物膜;另一方面,微球对成骨样细胞无任何细胞毒性作用,有望用于临床治疗骨缺损的手术中。 此外也有研究报道,适当大小和丰度的金纳米粒子可以调节巨噬细胞的极化状态,具有有益的抗炎活性[43-44]。LIANG等[45]为探究金纳米粒子对骨组织的影响,制备出物理吸附金纳米粒子的介孔二氧化硅微球,发现其可通过刺激抗炎反应和促进巨噬细胞分泌成骨细胞因子构建良好的免疫微环境,加速大鼠临界大小颅骨缺损部位的新骨形成。 多肽:LL37是含有37个氨基酸的多肽,是目前发现的唯一来源于人体的抗菌肽,已发现其具有多种调节活性,如抗菌和调节免疫应答,将其加载至金属纳米粒子、高分子材料和脂质体系等多种缓释给药系统中,可改善其易受蛋白酶降解、生物利用度差等不足[46]。 抗菌十肽KSL-W(KKVVFWVKFK-ConH2)能在唾液中保持稳定的抗菌活性,被广泛用于抑制牙菌斑的形成。LI等[47]采用电喷雾和交联-乳化相结合的方法制备了KSL-W/聚乳酸羟乙酸共聚物/壳聚糖复合微球,对不同处方的微球进行了表征,证明此微球在口腔感染性疾病治疗及牙槽骨骨增量技术中的应用潜力。 HE等[48]将抗菌肽Pac-525包载入聚乳酸羟乙酸共聚物微球并与纳米羟基磷灰石共同加载到明胶-壳聚糖联合的引导组织再生膜中,对S.aureus 和 E.coli两种细菌保持了1周的杀菌性和1个月的抑菌性,证明了此种抗菌肽在组织再生中的抗感染潜力。 抗感染药物:四环素族药物是广谱抑菌剂,其中米诺环素被公认为是抑菌效果最好的药物之一,其抑菌机制已被深入研究[49]。MA等[50]制作出载米诺环素的壳聚糖微球,装载在胶原/壳聚糖不对称引导骨组织再生膜上,在体内体外均显示出良好的促骨再生效果。不同抗生素在不同的浓度范围内对骨再生会造成或大或小的影响,具体表现为对成骨细胞数量和碱性磷酸酶活性的影响[51],此现象表明对抗生素药物进行更为精准的控释缓释,将会显著提升抗生素在骨再生中的应用效果。 阿司匹林作为经典非类固醇抗炎药,具有抗炎、调节免疫和促矿化作用。陈英等[52]制备了聚乳酸羟乙酸共聚物复合CaSiO3负载阿司匹林微球,通过与CaSiO3或聚乳酸羟乙酸共聚物单独负载阿司匹林微球比较,发现复合的支架材料能够协同提高微球载药量、延长药物缓释时间,表明此微球有望成为理想的成骨材料。 2.2.3 装载促血管生成因子 功能性骨再生支架的有效性主要取决于它们支持和诱导血管生长扩布的能力,原因在于血管中的血液能够清除废物并为细胞提供必要的营养物质,这在骨再生过程中起到了十分关键的作用。成血管因子包括血管内皮生长因子、血小板衍生生长因子、成纤维细胞生长因子和转化生长因子等,其中血管内皮生长因子应用最为广泛。然而,有学者认为单独使用血管内皮生长因子不足以促进骨再生,模仿自然骨形成的过程,按照成血管和成骨的顺序释放生长因子的策略被证明能够取得更好的效果[53]。DOU等[54]将重组骨形态发生蛋白包载入羧甲基壳聚糖微球,并以羟基磷灰石-胶原支架为微球载体,血管内皮生长因子直接吸附在支架上,成功地实现了双因子的有序释放,血管内皮生长因子和重组骨形态发生蛋白分别在第7天和第21天时达到释放峰值,大鼠皮下植入支架的组织学实验结果显示双因子支架较单因子支架能更有效促进血管的生成。 此外,锶、钴等元素的修饰也有助于骨组织的血管化。WEI等[55]制备了聚乳酸载万古霉素微球,锶元素的掺入增强了间充质基质细胞的血管生成和成骨表达,将微球皮下注射到兔背部,其异位成骨效果较未掺杂锶的微球显著,实验结果证明了诱导血管新生对于诱导骨再生的有效性。BOLDBAATARAB等[56]通过乳化法制备的掺钴硅酸盐微球可持续释放硅酸根离子和钴离子,被证明可协同上调血管生成的关键基因,离子的掺入促进了内皮细胞的极化、迁移、归巢和血管的生成。"

| [1] WANG W, YEUNG K. Bone grafts and biomaterials substitutes for bone defect repair: A review. Bioact Mater. 2017;2(4):224-247. [2] BALDWIN P, LI D, AUSTON D, et al. Autograft, Allograft, and Bone Graft Substitutes: Clinical Evidence and Indications for Use in the Setting of Orthopaedic Trauma Surgery. J Orthop Trauma. 2019;33(4):203-213. [3] 李晏乐,岳肖华,聂真,等.生物可吸收材料特点及在骨科中的应用[J].中国组织工程研究,2021,25(12):2612-2617. [4] HOSSAIN KMZ, PATEL U, AHMED I. Development of microspheres for biomedical applications: a review. Prog Biomater. 2015;4(1):1-19. [5] 卢静一,杨军星,王雷.缓释载药微球在慢性牙周炎中的应用研究进展[J].现代口腔医学杂志,2018,32(3):176-180. [6] LIN K, XIA L, GAN J, et al. Tailoring the nanostructured surfaces of hydroxyapatite bioceramics to promote protein adsorption, osteoblast growth, and osteogenic differentiation. ACS Appl Mater Interfaces. 2013;5(16):8008-8017. [7] LEITE AJ, CARIDADE SG, MANO JF. Synthesis and characterization of bioactive biodegradable chitosan composite spheres with shape memory capability. J Non Cryst Solids. 2016;432:158-166. [8] 姚凯,何星.无定型磷酸钙微球的制备及在骨修复领域的应用[J].广东化工,2019,46(9):147-148. [9] HENCH LL, SPLINTER RJ, ALLEN WC, et al. Bonding mechanisms at the interface of ceramic prosthetic materials. J Biomed Mater Res. 1971; 5(6):117-141. [10] BAINO F, FIORILLI S, VITALE-BROVARONE C. Composite Biomaterials Based on Sol-Gel Mesoporous Silicate Glasses: A Review. Bioengineering (Basel). 2017;4(1):15. [11] 陈雪颖.新型放射状介孔生物活性玻璃的制备及其作为药物载体的研究[D].广州:华南理工大学,2019. [12] NASKAR S, SHARMA S, KUOTSU K. Chitosan-based nanoparticles: An overview of biomedical applications and its preparation. J Drug Deliv Sci Technol. 2019;49:66-81. [13] XIA Y, WEI W, XIA H, et al. Effect of recombinant human bone morphogenetic protein delivered by chitosan microspheres on ectopic osteogenesis in rats. Exp Ther Med. 2019;17(5):3891-3898. [14] AHMED T, ALJAEID B. Preparation, characterization, and potential application of chitosan, chitosan derivatives, and chitosan metal nanoparticles in pharmaceutical drug delivery. Drug Des Dev Ther. 2016;10:483-507. [15] ANNAMALAI R, TURNER P, CARSON W, et al. Harnessing macrophage-mediated degradation of gelatin microspheres for spatiotemporal control of BMP2 release. Biomaterials. 2018;161:216-227. [16] CALASANS-MAIA MD, BARBOZA JUNIOR CA, SORIANO-SOUZA CA, et al. Microspheres of alginate encapsulated minocycline-loaded nanocrystalline carbonated hydroxyapatite: therapeutic potential and effects on bone regeneration. Int J Nanomedicine. 2019;14:4559-4571. [17] ZHAO Y, LIANG H, ZHANG S, et al.Effects of Magnesium Oxide (MgO) Shapes on In Vitro and In Vivo Degradation Behaviors of PLA/MgO Composites in Long Term. Polymers. 2020;12(5):1074. [18] HU X, HE J, YONG X, et al. Biodegradable poly (lactic acid-co-trimethylene carbonate)/chitosan microsphere scaffold with shape-memory effect for bone tissue engineering. Colloids Surf B Biointerfaces. 2020;195:111218. [19] YANG YY, CHUNG TS, NG NP. Morphology, drug distribution, and in vitro release profiles of biodegradable polymeric microspheres containing protein fabricated by double-emulsionsolvent extraction/evaporation method. Biomaterials. 2001;22(3):231-241. [20] SIEPMANN J, FAISANT N, AKIKI J, et al. Effect of the sizes of biodegradable microparticles of drug release: Experiment and theory. J Control Release. 2004;96(1):123-134. [21] BLANCOPRÍETO MJ, LEO E, DELIE F, et al. Study of the influence of several stabilizing agents on the entrapment and in vitro release of pBC 264 from poly(lactide-co-glycolide) microspheres prepared by a W/O/W solvent evaporation method. Pharm Res. 1996;13(7):1127-1129. [22] ZHOU Z, YAO Q, LI L, et al. Antimicrobial Activity of 3D-Printed Poly(ε-Caprolactone) (PCL) Composite Scaffolds Presenting Vancomycin-Loaded Polylactic Acid-Glycolic Acid (PLGA) Microspheres. Med Sci Monit. 2018;24:6934-6945. [23] CHEN H, WANG L, FU H, et al. Gadolinium Functionalized Carbon Dots for Fluorescence/Magnetic Resonance Dual-Modality Imaging of Mesenchymal Stem Cells. J Mater Chem B. 2016;4(46):7472-7480. [24] XU B, JU Y, CUI Y, et al. Carbon nanotube array inducing osteogenic differentiation of human mesenchymal stem cells. Mater Sci Eng C. 2015;51:182-188. [25] PARK J, KIM B, HAN J, et al. Graphene oxide flakes as a cellular adhesive: prevention of reactive oxygen species mediated death of implanted cells for cardiac repair. ACS Nano. 2015;9(5):4987-4999. [26] SHAO D, LU M, XU D, et al.Carbon dots for tracking and promoting the osteogenic differentiation of mesenchymal stem cells. Biomater Sci. 2017;5(9):1820-1827. [27] KHAJURIA D, KUMAR V, GIGI D, et al. Accelerated Bone Regeneration by Nitrogen-Doped Carbon Dots Functionalized with Hydroxyapatite Nanoparticles. ACS Appl Mater Interfaces. 2018;10(23):19373-19385. [28] 代鹏展,刘新.BMP-2骨吸收作用的研究进展[J].中国实验诊断学, 2020,24(4):687-690. [29] 陈良,王德飞,白涧飞,等.高浓度使用 BMP-2 的副作用及应对措施研究进展[J].中华老年口腔医学杂志,2018,16(6):358-361+377. [30] WEI D, QIAO R, DAO J, et al. Soybean Lecithin-Mediated Nanoporous PLGA Microspheres with Highly Entrapped and Controlled Released BMP-2 as a Stem Cell Platform. Small. 2018;14(22):e1800063. [31] LI Y, YE D, LI M, et al. Adaptive Materials Based on Iron Oxide Nanoparticles for Bone Regeneration. Chemphyschem. 2018;19(16):1965-1979. [32] 校搏.顺磁性纳米纤维复合材料诱导骨缺损修复的动物实验研究[D].北京:北京协和医学院,2011. [33] LU J, YANG F, KE Q, et al. Magnetic nanoparticles modified-porous scaffolds for bone regeneration and photothermal therapy against tumors. Nanomedicine. 2018;14(3):811-822. [34] YUAN Z, WEI P, HUANG Y, et al. Injectable PLGA microspheres with tunable magnesium ion release for promoting bone regeneration. ACS Appl Mater. 2019;85:294-309. [35] LIN Z, WU J, QIAO W, et al. Precisely controlled delivery of magnesium ions thru sponge-like monodisperse PLGA/nano-MgO-alginate core-shell microsphere device to enable in-situ bone regeneration. Biomaterials. 2018;174:1-16. [36] ZHOU X, LIU P, NIE W, et al. Incorporation of dexamethasone-loaded mesoporous silica nanoparticles into mineralized porous biocomposite scaffolds for improving osteogenic activity. Int J Biol Macromol. 2020; 149:116-126. [37] ZHANG J, MA S, LIU Z, et al. Guided bone regeneration with asymmetric collagen-chitosan membranes containing aspirin-loaded chitosan nanoparticles. Int J Nanomedicine. 2017;12:8855-8866. [38] XUE Y, WU M, LIU Z, et al. In vitro and in vivo evaluation of chitosan scaffolds combined with simvastatin-loaded nanoparticles for guided bone regeneration. J Mater Sci Mater Med. 2019;30(4):47. [39] SHOKROLAHI F, KHODABAKHSHI K, SHOKROLLAHI P, et al. Atorvastatin loaded PLGA microspheres: Preparation, HAp coating, drug release and effect on osteogenic differentiation of ADMSCs. Int J Pharm. 2019;565: 95-107. [40] OSSIPOV D. Bisphosphonate-modified biomaterials for drug delivery and bone tissue engineering. Expert Opin Drug Deliv. 2015;12(9): 1443-1458. [41] WEI P, YUAN Z, CAI Q, et al. Bioresorbable Microspheres with Surface-Loaded Nanosilver and Apatite as Dual-Functional Injectable Cell Carriers for Bone Regeneration. Macromol Rapid Commun. 2018; 39(20):e1800062. [42] PORRELLI D, TRAVAN A, TURCO G, et al. Antibacterial-nanocomposite bone filler based on silver nanoparticles and polysaccharides. J Tissue Eng Regen Med. 2018;12(2):e747-e759. [43] CHEN X, GAO C. Influences of size and surface coating of gold nanoparticles on inflammatory activation of macrophages.Colloids Surf B Biointerfaces. 2017;160:372-380. [44] SUMBAYEV VV, YASINSKA IM, GARCIA CP, et al.Gold nanoparticles downregulate interleukin-1β-induced pro-inflammatory responses. Small. 2013;9(3):472-477. [45] LIANG H, JIN C, MA L, et al. Accelerated Bone Regeneration by Gold-Nanoparticle-Loaded Mesoporous Silica through Stimulating Immunomodulatio. ACS Appl Mater Interfaces. 2019;11(44): 41758-41769. [46] LIN X, WANG R, MAI S. Advances in delivery systems for the therapeutic application of LL37. J Drug Deliv Sci Technol. 2020;60:102016. [47] LI Y, NA R, WANG X, et al. Fabrication of Antimicrobial Peptide-Loaded PLGA/Chitosan Composite Microspheres for Long-Acting Bacterial Resistance. Molecules. 2017;22(10):1637. [48] HE Y, JIN Y, WANG X, et al. An Antimicrobial Peptide-Loaded Gelatin/Chitosan Nanofibrous Membrane Fabricated by Sequential Layer-by-Layer Electrospinning and Electrospraying Techniques. Nanomaterials. 2018;8(5):327. [49] ASADI A, ABDI M, KOUHSARI E, et al. Minocycline, focused on mechanisms of resistance, antibacterial activity, and clinical effectiveness; Back to Future. J Glob Antimicrob Resist. 2020;22:161-174. [50] MA S, ADAYI A, LIU Z, et al. Asymmetric collagen/chitosan membrane containing minocycline-loaded chitosan nanoparticles for guided bone regeneration. Sci Rep. 2016;6:31822. [51] RATHBONE CR, CROSS JD, BROWN KV, et al. Effect of various concentrations of antibiotics on osteogenic cell viability and activity. J Orthop Res. 2011;29 (7):1070-1074. [52] 陈英,刘中宁,李波,姜婷.阿司匹林缓释微球的制备及体外缓释效果评估[J].北京大学学报(医学版),2019,51(5):907-912. [53] BAYER EA, GOTTARDI R, FEDORCHAK MV, et al. The scope and sequence of growth factor delivery for vascularized bone tissue regeneration. J Control Release. 2015;219 (Complete):129-140. [54] DOU D, ZHOU G, LIU H, et al. Sequential releasing of VEGF and BMP-2 in hydroxyapatite collagen scaffolds for bone tissue engineering: Design and characterization. Int J Biol Macromol. 2019;123:622-628. [55] WEI P, JING W, YUAN Z, et al. Vancomycin- and Strontium-Loaded Microspheres with Multifunctional Activities against Bacteria, in Angiogenesis, and in Osteogenesis for Enhancing Infected Bone Regeneration. ACS Appl Mater Interfaces. 2019;11(34):30596-30609. [56] BOLDBAATARAB K, DASHNYAMABC K, KNOWLES JC, et al. Dual-ion delivery for synergistic angiogenesis and bactericidal capacity with silica-based microsphere. Acta Biomater. 2019;83:322-333. |
| [1] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [2] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [3] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Luo Xuanxiang, Jing Li, Pan Bin, Feng Hu. Effect of mecobalamine combined with mouse nerve growth factor on nerve function recovery after cervical spondylotic myelopathy surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 719-722. |
| [6] | Nie Huijuan, Huang Zhichun. The role of Hedgehog signaling pathway in transforming growth factor beta1-induced myofibroblast transdifferentiation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 754-760. |
| [7] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [8] | Zhang Qunhui, Li Yimei, Zhang Dejun. Hypoxia-inducible factor and coronary heart disease: antagonism and protection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5729-5734. |
| [9] | Xiao Deli, Sun Yinzhe, Cui Cheng, Liu Bo. Preparation and degradation properties of concentrated growth factor fibrin membrane [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5413-5419. |
| [10] | Zhang Zhiwen, Huang Yuliang, Zhang Lixuan, Wang Xiaofeng, Chen Ruixiong. Acellular bone matrix/chitosan scaffold combined with basic fibroblast growth factor for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5439-5444. |
| [11] | Yang Zhen, Li Hao, Fu Liwei, Gao Cangjian, Jiang Shuangpeng, Wang Fuxin, Yuan Zhiguo, Sun Zhiqiang, Zha Kangkang Tian Guangzhao, Cao Fuyang, Sui Xiang, Liu Shuyun, Guo Quanyi. Cartilage composite scaffold loaded with transforming growth factor beta 3 using three-dimensional bioprinting [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5445-5452. |
| [12] | Huang Minling, Lu Zhaoqi, Shen Zhen, Lin Haixiong, Feng Junjie, Huang Feng, Jiang Ziwei, Cai Qunbin. Effects of total flavone of Rhizoma Drynariae on the coupling of angiogenesis and osteogenesis in bone remodeling through notch signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(32): 5116-5122. |
| [13] |
Li Yinglian, Chang Qiong.
Changes in serum tumor necrosis factor alpha, transforming growth factor beta 1, and interleukin-6 levels in patients with chronic obstructive pulmonary disease combined with osteoporosis#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(32): 5085-5090.
|
| [14] | Wei Xiaoling, Hu Weiping, Liu Mingyue, Shi Xin, Liu Jie, Gao Li, Wang Siran, Duan Yajun. Dental pulp fibroblasts inducing and regulating dentin-pulp regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 5079-5084. |
| [15] | Tan Xiaowu, Yu Xiaofan, Jiang Huijiao, Xing Zhikun, Zhao Xueyuan, Gao Fengyi, Wu Xiangwei, Chen Xueling. Effect of xanthohumol on proliferation, migration, tube formation and apoptosis of endothelial progenitor cells and its mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4988-4994. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||