Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (25): 6632-6642.doi: 10.12307/2026.287
Previous Articles Next Articles
Action mechanisms of Bushen Qiangjin capsule in regulating inflammatory signaling pathways and improving knee osteoarthritis
He Wenyang1, 2, Chen Weijian2, Ran Qingzhi3, Huang Yidie1, 2, Lin Xiaodong2, Xu Xuemeng2, Guo Meirong2, Liu Wengang1, 2
1Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; 2Guangdong Second Traditional Chinese Medicine Hospital (Guangdong Province Engineering Technology Research Institute of TCM), Guangzhou 510095, Guangdong Province, China; 3Guang'anmen Hospital of China Academy of Chinese Medical Sciences, Beijing 100053, China
-
Received:2025-10-15Revised:2025-12-26Online:2026-09-08Published:2026-04-23 -
Contact:Liu Wengang, Chief physician, Doctoral supervisor, Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; Guangdong Second Traditional Chinese Medicine Hospital (Guangdong Province Engineering Technology Research Institute of TCM), Guangzhou 510095, Guangdong Province, China -
About author:He Wenyang, MS candidate, Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; Guangdong Second Traditional Chinese Medicine Hospital (Guangdong Province Engineering Technology Research Institute of TCM), Guangzhou 510095, Guangdong Province, China -
Supported by:Guangdong Provincial Basic and Applied Basic Research Foundation - Natural Science Foundation Project, No. 2023A1515012615 (to LWG); Guangdong Provincial Basic and Applied Basic Research Foundation - Provincial Enterprise Joint Fund - General Project, No. 2022A1515220157 (to LWG); Guangdong Provincial Second Affiliated Hospital of Traditional Chinese Medicine Scientific Research Innovation Fund - Excellent Team Project, No. SEZYY2023B16 (to LWG); Guangdong Provincial Medical Science and Technology Research Foundation Project, No. B2023352 (to LXD); Guangdong Provincial Administration of Traditional Chinese Medicine Project, No. 20221033 (to GMR)
CLC Number:
Cite this article
He Wenyang, Chen Weijian, Ran Qingzhi, Huang Yidie, Lin Xiaodong, Xu Xuemeng, Guo Meirong, Liu Wengang. Action mechanisms of Bushen Qiangjin capsule in regulating inflammatory signaling pathways and improving knee osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6632-6642.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
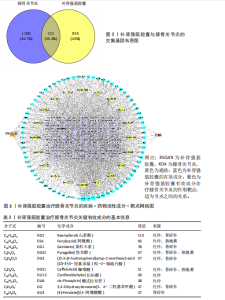
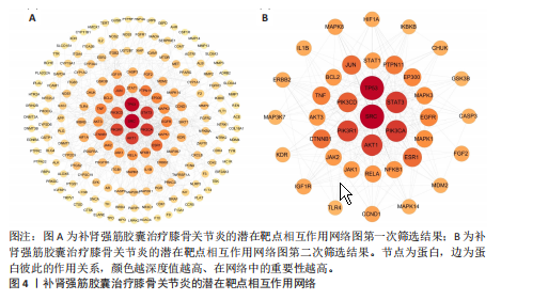
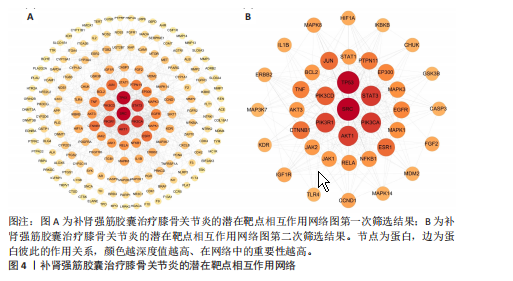
2.1 补肾强筋胶囊活性成分及靶标筛选结果 通过TCMSP与Herb数据库进行检索及筛选,共获得补肾强筋胶囊相关的活性成分85个,其中补骨脂含17个、杜仲含43个、骨碎补含16个、全蝎含1个、熟地黄含8个,血竭未筛选到活性成分;有7个活性成分同时存在于2种以上药材中。共获得补肾强筋胶囊的1 147个药物靶标基因。 2.2 补肾强筋胶囊干预膝骨关节炎的疾病靶标与交集靶标分析结果 借助Drugbank、GeneCards及OMIM等数据库,输入关键词“Knee Osteoarthritis”进行靶点检索,共筛选得到膝骨关节炎相关疾病靶标1 226个。将上述1 226个疾病靶标与补肾强筋胶囊的1 147个药物靶标基因,通过Venny 2.1.0系统进行交集分析,获得共同靶标222个,如图2所示。 2.3 补肾强筋胶囊治疗膝骨关节炎的“疾病-通路-药物成分-靶标”网络图构建 利用 Cytoscape 3.7.2软件对补肾强筋胶囊活性成分、潜在靶点与膝骨关节炎相关靶基因进行整合与可视化,构建了“疾病–通路–药物成分–靶标”网络图(图3)。网络包含化合物节点76个(去除无靶标或无通路关联的化合物后)、靶蛋白节点222个、通路节点20个。拓扑学分析结果显示,活性成分中度值排名前列的包括 Kaempferol(山柰酚)、Ferulic acid(阿魏酸)、Genistein(染料木素) 等,基本信息见表2,这些核心成分与多个信号通路密切相关,如磷脂酰肌醇3-激酶-丝氨酸/苏氨酸蛋白激酶、丝裂原活化蛋白激酶、核因子κB通路。 网络药理学分析提示,MAPK8(JNK1)为潜在关键基因。在网络中,MAPK8处于多条信号通路的交汇点,既是炎症因子(如肿瘤坏死因子α、白细胞介素1β)下游的核心调控分子,也直接参与软骨细胞凋亡及基质降解过程。已有研究表明,MAPK8的持续激活可促进基质金属蛋白酶13表达、NLRP3炎症小体激活,从而加重软骨损伤;而抑制MAPK8可有效缓解关节炎症反应并延缓软骨退变[22-24]。因此,此次研究提示补肾强筋胶囊可能通过活性成分(如山柰酚、染料木素)作用于MAPK8并调控其相关信号通路,从而实现抗炎与保护关节软骨的作用。 2.4 蛋白质互作网络图构建及核心靶标筛选结果 将上述获得的222个药物-疾病交集靶标导入STRING数据库(物种设置为“Homo sapiens”,蛋白互作置信度≥0.9),再利用Cytoscape 3.7.2软件构建蛋白质互作网络图。以介数中心性、紧密中心性、度中心性均大于中位数为条件(即度中心性> 5、紧密中心性> 0.076 441、介数中心性> 43.674 743),按照度值排序,初次筛选出的蛋白质互作网络见图4A。考虑初次筛选靶点较多,缺乏精确度,以介数中心性、紧密中心性、度中心性均大于中位数为条件(即度中心性> 14、紧密中心性> 0.077 872、介数中心性> 332.335 86),按照度值排序再次筛选,图4B为最终确定的核心靶标网络图。最终筛选出SRC(酪氨酸激酶C)、TP53(肿瘤蛋白p53)、STAT3(信号转导及转录"
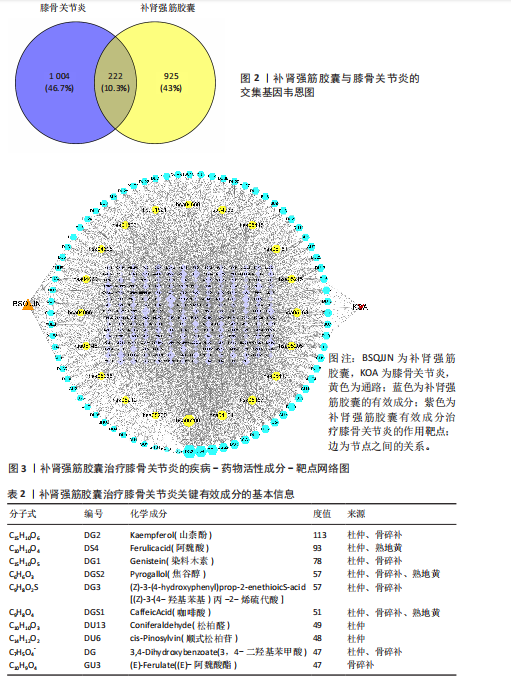
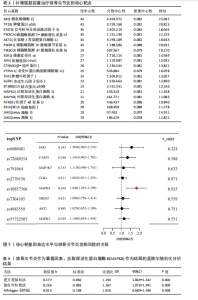
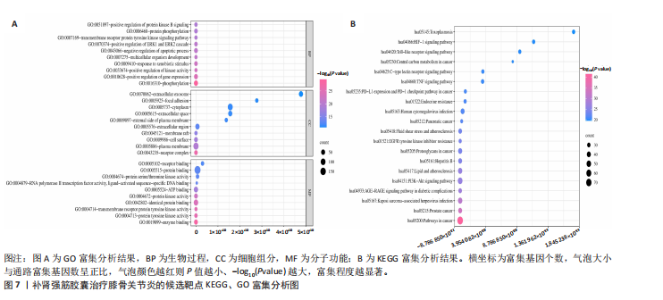
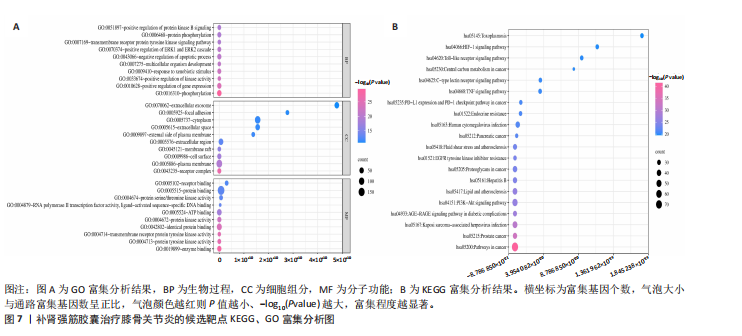
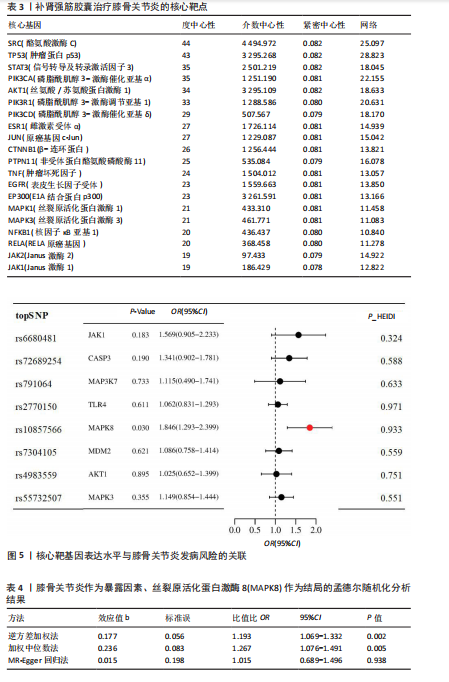
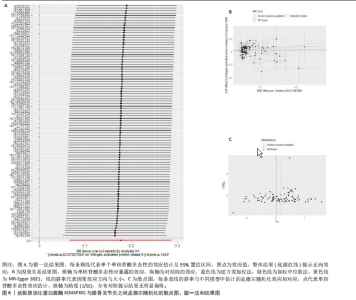
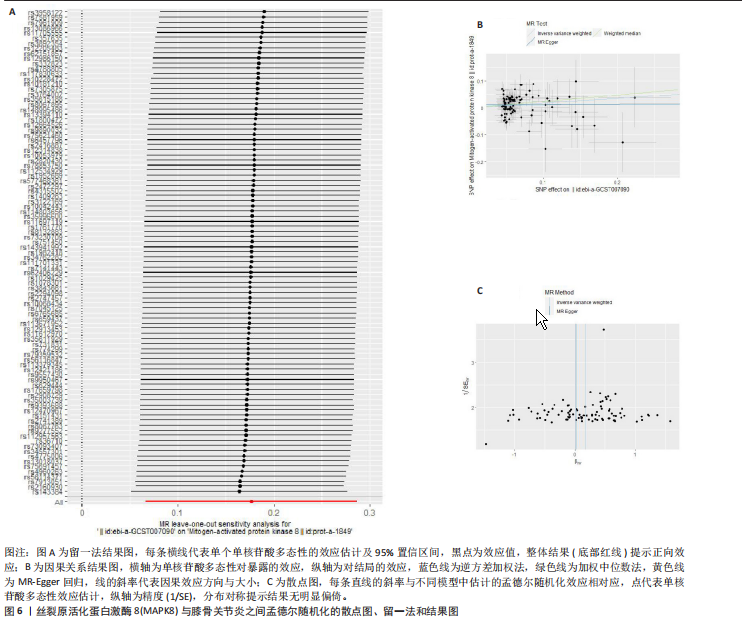
激活因子3)、PIK3CA(磷脂酰肌醇3-激酶催化亚基α)、AKT1(丝氨酸/苏氨酸蛋白激酶1)等39个核心靶标,列出前20个(表3),提示上述靶标可能在补肾强筋胶囊治疗膝骨关节炎过程中发挥关键作用。 2.5 基于汇总数据的孟德尔随机化分析与HEIDI检验结果 二次筛选的39个蛋白质互作核心靶点基因与膝骨关节炎的基于汇总数据的孟德尔随机化分析结果显示,JAK1(Janus激酶1)、CASP3(caspase-3)、MAP3K7(丝裂原活化蛋白激酶激酶激酶7)、TLR4(Toll样受体4)、MAPK8、MDM2(鼠双微体基因2蛋白)、AKT1、MAPK3 (丝裂原活化蛋白激酶3)查找到eQTL顺式,余靶点未查找到。血液中MAPK8基因的表达与膝骨关节炎显著相关(OR=1.846,95%CI:1.293-2.399,P=0.03),见图5。HEIDI检验提示,MAPK8基因表达与膝骨关节炎的关联是直接的因果关系(PHEIDI=0.933),其他基因所观察到的关联都是由于连锁不平衡造成的。综上分析,推测MAPK8可能是膝骨关节炎的潜在治疗靶点。 2.6 两样本孟德尔随机化分析结果 在以MAPK8表达作为暴露、膝骨关节炎风险作为结局的因果路径分析中,未观察到显著关联(P > 0.05)。然而,当以膝骨关节炎作为暴露、MAPK8表达作为结局进行反向分析时,逆方差加权法分析显示膝骨关节炎与MAPK8风险之间存在显著因果关联(OR=1.193,95%CI:1.069-1.332,P=0.002),加权中位数法分析同样呈现膝骨关节炎对MAPK8的促发趋势(OR=1.267,95%CI:1.076-1.491,P=0.005),见表4,结果提示膝骨关节炎可能导致MAPK8表达水平升高,表明MAPK8的异常上调更可能是膝骨关节炎发生和发展的反应性改变,而非膝骨关节炎的直接遗传易感因素。该结果与炎症相关信号通路的调控机制相符,即膝骨关节炎作为一种炎症性和退行性疾病,可能通过促进MAPK8上调参与细胞凋亡、炎症反应以及软骨破坏过程。 在对MAPK8与膝骨关节炎的因果关系进行两样本孟德尔随机化分析中,异质性测试结果显示,MR-Egger回归法的Q值为102.4、自由度为93、P=0.24;逆方差加权法的Q值为103.2、自由度为94、P=0.24,由于Q检验的P值均> 0.05,表明各工具变量之间不存在显著的异质性,分析结果的一致性较强。在多效性检验中,MR-Egger回归法的截距(Intercept)为0.009 98、标准误为0.011 73、P=0.4,说明不存在显著的水平多效性,即工具变量未通过其他路径影响结局变量,满足孟德尔随机化分析假设。MR-PRESSO整体多效性检验结果亦显示P=0.262,未检出离群效应,进一步支持因果推断的可靠性和稳健性。此外,采用“留一法”逐个排除单核苷酸多态性并重新估计整体效应后发现,任何单个单核苷酸多态性的剔除均未显著影响膝骨关节炎与MAPK8之间的潜在因果关系(图6)。 2.7 GO及KEGG富集分析结果 利用David数据库对潜在靶标进行富集分析,共获得生物过程(BP)条目976项、细胞组分条目106项、分"
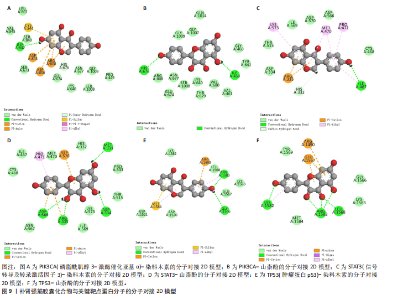
子功能条目190项、KEGG通路184条,以P值由小到大排序,分别选取生物过程、细胞组分、分子功能前10项和KEGG通路前20项(主要涉及炎性反应、氧化应激等机制,具体通路见图7。非类固醇类抗炎药主要通过抑制环氧合酶(环氧合酶1/环氧合酶2)、减少前列腺素合成从而发挥镇痛及抗炎作用,然而,该类药物的作用靶点相对集中,多为炎症反应下游环节,对软骨退变及结构损伤的保护作用有限;关节腔注射治疗(如透明质酸、糖皮质激素)主要改善关节润滑和局部炎症状态,但作用靶点单一且疗效维持时间较短。相比之下,补肾强筋胶囊不仅可作用于经典炎症通路(如核因子κB、肿瘤坏死因子α、白细胞介素1β信号轴),还具有调控Wnt/β-catenin-基质金属蛋白酶通路、抑制NLRP3炎症小体活化等多重机制,兼具抗炎、抗细胞焦亡及软骨保护作用。由此可见,补肾强筋胶囊在多通路、多靶点的综合调控方面展现出独特优势,更能体现中药复方多组分协同干预复杂疾病的整体观念。 2.8 分子对接验证结果 为进一步明确补肾强筋胶囊关键活性成分与核心靶标蛋白之间的相互作用,选取排名前5的活性成分,包括山柰酚(Kaempferol)、阿魏酸(Ferulic acid)、染料木"
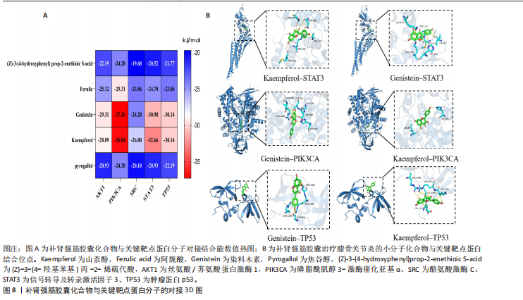
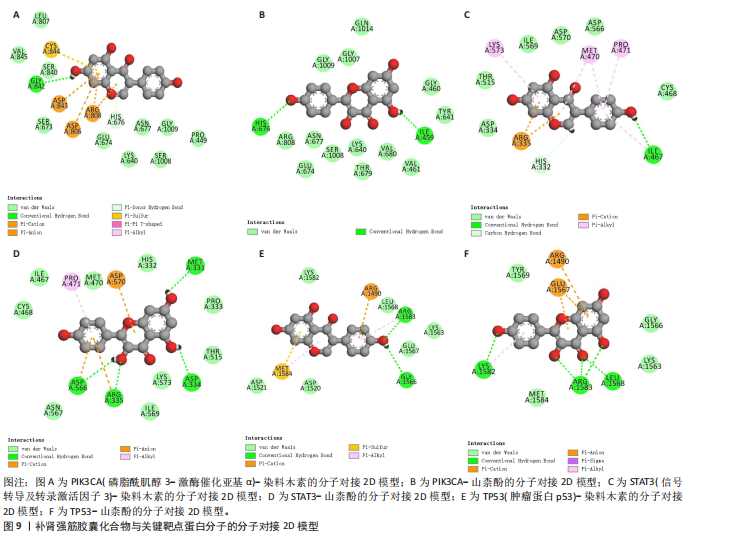
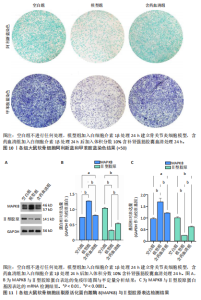
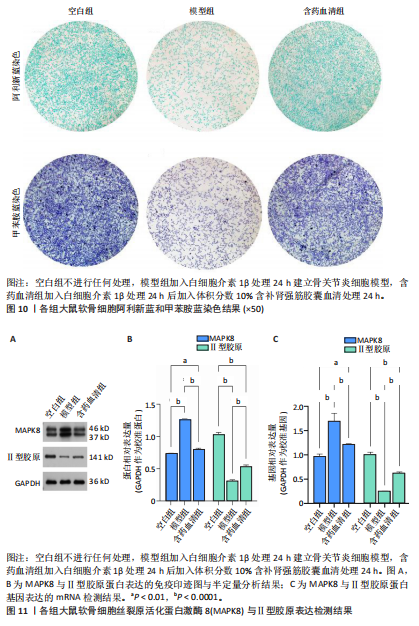
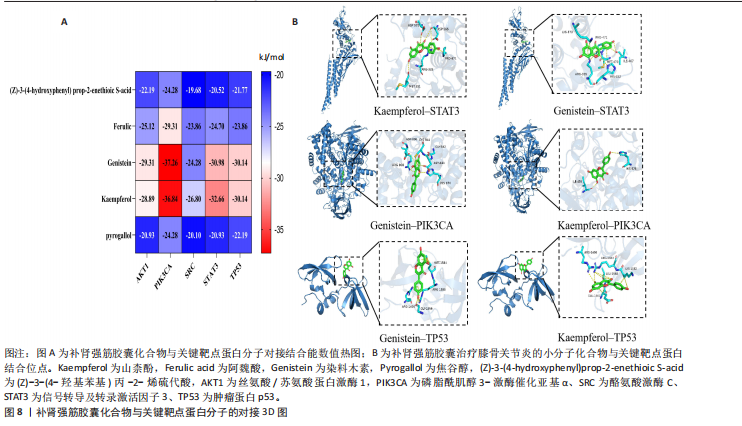
素(Genistein)、焦谷醇(Pyrogallol)及(Z)-3-(4-羟基苯基)丙-2-烯硫代酸[(Z)-3-(4-hydroxyphenyl)prop-2-enethioic S-acid],核心靶标蛋白选取AKT1、PIK3CA、SRC、STAT3、TP53,采用MOE 2019软件进行分子对接模拟分析。结果显示,分子对接结合能范围为-19.68至-37.26 kJ/mol,见图8A。结合能 < –17.79 kJ/mol提示存在一定结合活性, < –20.93 kJ/mol提示较好的结合活性, < –29.31 kJ/mol则表明强烈的结合活性[25]。具体而言,染料木素-PIK3CA(-37.26 kJ/mol)、山柰酚-PIK3CA(-36.84 kJ/mol)表现出显著较低的结合能,提示二者具有极佳的结合稳定性与活性,化合物与关键靶点蛋白分子对接结合能具体数值。此次研究进一步选择结合能最低且最稳定的前6组配体-受体组合,利用PyMOL 2.6.0及Discovery Studio 2019软件对结合模式进行可视化,代表性对接模型见图8B,2D结构见图9。 分子对接结果显示,染料木素与山柰酚均可与PIK3CA、STAT3和TP53蛋白形成稳定结合。对于PIK3CA,染料木素与Gly842形成氢键,并与Asp843、Asp806、Arg808发生静电相互作用,同时His676、Cys844参与碳氢及Pi-Sulfur作用;山柰酚在同一受体上则通过His676、Ile459形成氢键。对于STAT3,染料木素与Ile467形成氢键,并与 Lys573、Met470、Pro471 等残基产生疏水及静电作用;山柰酚则在Met331、Asp334、Arg335、Asp566 等位点形成氢键,并与 Pro471、Asp570产生疏水与静电作用。对于TP53,染料木素与Arg1583、Gly1566形成氢键,并在Arg1490、Met1584位点发生静电与Pi-Sulfur作用;山柰酚则与Lys1582、Arg1583、Leu1568形成氢键,并且与Glu1567、Arg1490发生静电作用。总体来看,两种化合物均在关键残基(如Arg335、Arg1583、His676等)处表现出氢键和疏水相互作用,提示其结合位点具有较高一致性和稳定性。 2.9 细胞实验验证结果 2.9.1 阿利新蓝和甲苯胺蓝染色观察大鼠软骨细胞形态变化 阿利新蓝和甲苯胺蓝细胞染色显示,空白组细胞多为圆/卵圆形,排列致密均匀,细胞核清晰,基质染色均匀;含药血清组细胞总体形态正常,但较空白组分布略稀疏,排列和密度稍有变化,基质染色较均匀;模型组细胞呈纺锤或形态不规则,排列松散,基质染色减弱,提示结构/功能受损,见图10。结果说明模型组软骨细胞细胞外基质合成受抑制,并且糖胺聚糖和蛋白聚糖表达受限,含药血清干预后软骨细胞着色显著变深,说明补肾强筋胶囊能够显著促进软骨外基质的合成。 2.9.2 补肾强筋胶囊对软骨细胞内丝裂原活化蛋白激酶8与Ⅱ型胶原表达的影响 Ⅱ型胶原作为软骨基质的主要成分,在维持软骨组织的完整性、刺激软骨细胞的生长和再分化、促进骨骼整体健康等方面起着重要作用。RT-qPCR、Western blot检测显示,与空白组比较,模型组细胞内丝裂原活化蛋白激酶8蛋白与mRNA表达升高(P < 0.000 1),Ⅱ型胶原蛋白与mRNA表达降低(P < 0.000 1);与模型组比较,含药血清组细胞内丝裂原活化蛋白激酶8蛋白与mRNA表达降低(P < 0.000 1),Ⅱ型胶原蛋白与mRNA表达升高(P < 0.000 1),见图11。"
| [1] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182):1745-1759. [2] BROPHY RH, FILLINGHAM YA. AAOS Clinical Practice Guideline Summary: Management of Osteoarthritis of the Knee (Nonarthroplasty), Third Edition. J Am Acad Orthop Surg. 2022; 30(9):e721-e729. [3] WANG K, LEI G, HU Y. Chinese guideline for diagnosis and treatment of osteoarthritis (2021 edition). Chin J Orthop. 2021;41(18):1291-1314. [4] LV Y, SUI L, LV H, et al. Burden of knee osteoarthritis in China and globally from 1992 to 2021, and projections to 2030: a systematic analysis from the Global Burden of Disease Study 2021. Front Public Health. 2025;13:1543180. [5] 王毅,姜涛,刘文刚,等.补肾强筋胶囊通过调控NLRP3炎症小体介导的细胞焦亡改善KOA滑膜炎症和纤维化的实验研究[J].世界科学技术-中医药现代化,2024,26(6):1471-1480. [6] YAO N, CHEN GC, LU YY, et al. Bushen Qiangjin capsule inhibits the Wnt/α-catenin pathway to ameliorate papain-induced knee osteoarthritis in rat. J Tradit Chin Med. 2021;41(6):935-942. [7] 胡子旋,姚楠,黄丹娥,等.补肾强筋胶囊含药血清对膝骨关节炎软骨细胞模型自噬影响的实验研究 [J].中医正骨,2025,37(1):38-44. [8] 章家皓,田佳庆,王帅,等.补肾强筋胶囊对膝骨关节炎大鼠膝关节软骨下骨质的影响及其作用机制的实验研究[J].中医正骨,2024,36(8): 19-26. [9] ZHU Z, ZHANG F, HU H, et al. Integration of summary data from GWAS and eQTL studies predicts complex trait gene targets. Nat Genet. 2016;48(5):481-487. [10] CHAUQUET S, ZHU Z, O’DONOVAN MC, et al. Association of antihypertensive drug target genes with psychiatric disorders: a Mendelian randomization study. JAMA Psychiatry. 2021; 78(6):623-631. [11] SUN X, CHEN B, QI Y, et al. Multi-omics Mendelian randomization integrating GWAS, eQTL and pQTL data revealed GSTM4 as a potential drug target for migraine. J Headache Pain. 2024;25(1):117. [12] VÕSA U, CLARINGBOULD A, WESTRA HJ, et al. Large-scale cis-and trans-eQTL analyses identify thousands of genetic loci and polygenic scores that regulate blood gene expression. Nat Genet. 2021;53(9):1300-1310. [13] BUNIELLO A, MACARTHUR JAL, CEREZO M, et al. The NHGRI-EBI GWAS Catalog of published genome-wide association studies, targeted arrays and summary statistics 2019. Nucleic Acids Res. 2019;47(D1):D1005-D1012. [14] CAO Z, WU Y, LI Q, et al. A causal relationship between childhood obesity and risk of osteoarthritis: results from a two-sample Mendelian randomization analysis. Ann Med 2022;54(1):1636-1645. [15] YU F, YANG Y, WU D, et al. Deciphering genetic causality between inflammatory bowel disease and periodontitis through bi-directional two-sample Mendelian randomization. Sci Rep. 2023; 13(1):18620. [16] LI Y, LU J, WANG J, et al. Inflammatory Cytokines and Risk of Ischemic Stroke: A Mendelian Randomization Study. Front Pharmacol. 2021;12: 779899. [17] FANG P, LIU X, QIU Y, et al. Exploring causal correlations between inflammatory cytokines and ankylosing spondylitis: a bidirectional mendelian-randomization study. Front Immunol. 2023;14:1285106. [18] JIANG X, ZHOU R, HE Y, et al. Causal effect of serum 25-hydroxyvitamin D levels on low back pain: A two-sample mendelian randomization study. Front Genet. 2022;13:1001265. [19] DING Y, YANG S, FAN S, et al. Is vitamin D deficiency the cause or the effect of systemic lupus erythematosus: evidence from bidirectional mendelian randomization analysis. J Immunol Res. 2022;2022(1):8689777. [20] HEMANI G, BOWDEN J, DAVEY SMITH G. Evaluating the potential role of pleiotropy in Mendelian randomization studies. Hum Mol Genet. 2018;27(R2):R195-R208. [21] ZHU J, ZHOU D, WANG J, et al. Frailty and cardiometabolic diseases: a bidirectional Mendelian randomisation study. Age Ageing. 2022;51(11):afac256. [22] FAN Q, ZHAO M, ZHANG XD, et al. Research progress and prospect of MAPK signaling pathway in knee osteoarthritis. Eur J Orthop Surg Traumatol. 2025;35(1):134. [23] IMRAN M, RAUF A, SHAH ZA, et al. Chemo‐preventive and therapeutic effect of the dietary flavonoid kaempferol: A comprehensive review. Phytother Res. 2019;33(2):263-275. [24] ZHU L, BI Y, LIANG T, et al. Ginkgetin delays the progression of osteoarthritis by inhibiting the NF-κB and MAPK signaling pathways. J Orthop Surg Res. 2025;20(1):139. [25] 刘册家,刘羽康,张京华,等.北豆根配方颗粒的抗炎、抗肿瘤有效成分研究:基于UPLC-Q-TOF-MS/MS与网络药理学[J].西南大学学报(自然科学版),2024,46(11):227-240. [26] YAO YX, YU YJ, DAI S, et al. Kaempferol efficacy in metabolic diseases: Molecular mechanisms of action in diabetes mellitus, obesity, non-alcoholic fatty liver disease, steatohepatitis, and atherosclerosis. Biomed Pharmacother. 2024; 175:116694. [27] 赵莹,王靖雷,王萌,等.乳源外泌体包载连翘酯苷A和山奈酚的制备、表征及体外抗炎效果评价[J].畜牧兽医学报,2025,56(5):2481-2495. [28] PERIFERAKIS A, PERIFERAKIS K, BADARAU I A, et al. Kaempferol: antimicrobial properties, sources, clinical, and traditional applications. Int J Mol Sci. 2022;23(23):15054. [29] LI Y, ZHAO W, SAIR AT, et al. Ferulic acid restores mitochondrial dynamics and autophagy via AMPK signaling pathway in a palmitate-induced hepatocyte model of metabolic syndrome. Sci Rep. 2024;14(1):18970. [30] WONG SK, CHIN KY, IMA-NIRWANA S. The osteoprotective effects of kaempferol: the evidence from in vivo and in vitro studies. Drug Des Devel Ther. 2019;13:3497-3514. [31] LIN C, SHAO Y, ZENG C, et al. Blocking PI3K/AKT signaling inhibits bone sclerosis in subchondral bone and attenuates post‐traumatic osteoarthritis. J Cell Physiol. 2018; 233(8):6135-6147. [32] ZHANG K, FU W. HIF-1α: linking subchondral bone and cartilage as a therapeutic target in osteoarthritis. Biomater Transl. 2024;5(1):89-91. [33] WEN ZH, TANG CC, LIN YY, et al. Effects of Etanercept on Experimental Osteoarthritis in Rats: Role of Histone Deacetylases. Cartilage. 2024: 19476035241264012. [34] 傅永升,王卫国,黄旭东,等.中药复方抗膝骨关节炎研究进展[J].中国中西医结合杂志, 2026,46(2):250-256. [35] FUKAI A, KAWAMURA N, SAITO T, et al. Akt1 in murine chondrocytes controls cartilage calcification during endochondral ossification under physiologic and pathologic conditions. Arthritis Rheum. 2010;62(3):826-836. [36] WANG K, CHU M, WANG F, et al. Putative functional variants of PI3K/AKT/mTOR pathway are associated with knee osteoarthritis susceptibility. J Clin Lab Anal. 2020;34(6):e23240. [37] ZHANG H, ZHENG C, CHEN W, et al. PP2 alleviates the progression of osteoarthritis by inhibiting Wnt/β-catenin and activating TGF-β/Smad signaling. Int Immunopharmacol. 2023;124(Pt B):110948. [38] ZHANG Z, LI B, WU S, et al. Bergenin protects against osteoarthritis by inhibiting STAT3, NF-κB and Jun pathways and suppressing osteoclastogenesis. Sci Rep. 2024;14(1):20292. [39] QIU M, CHENG L, XU J, et al. Liquiritin reduces chondrocyte apoptosis through P53/PUMA signaling pathway to alleviate osteoarthritis. Life Sci. 2024;343:122536. [40] KLUSKA M, JUSZCZAK M, ŻUCHOWSKI J, et al. Effect of Kaempferol and Its Glycoside Derivatives on Antioxidant Status of HL-60 Cells Treated with Etoposide. Molecules. 2022;27(2):333. [41] PARK JE, HAN JS. Improving the Effect of Ferulic Acid on Inflammation and Insulin Resistance by Regulating the JNK/ERK and NF-κB Pathways in TNF-α-Treated 3T3-L1 Adipocytes. Nutrients. 2024;16(2):294. [42] XU H, ZHANG T, HE L, et al. Exploring the mechanism of Danggui Buxue Decoction in regulating atherosclerotic disease network based on integrated pharmacological methods. Biosci Rep. 2021;41(10):BSR20211429. [43] DAVEY SMITH G, HEMANI G. Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum Mol Genet. 2014;23(R1):R89-98. [44] GILL D, GEORGAKIS MK, WALKER VM, et al. Mendelian randomization for studying the effects of perturbing drug targets. Wellcome Open Res. 2021;6:16. [45] LIU Y, WANG Y, YAN P, et al. NLRP3 Inflammasome-Mediated Osteoarthritis: The Role of Epigenetics. Biology (Basel). 2025;14(1):71. [46] ZHANG Y, ZHOU S, CAI W, et al. Hypoxia/reoxygenation activates the JNK pathway and accelerates synovial senescence. Mol Med Rep. 2020;22(1):265-276. [47] LAN CN, CAI WJ, SHI J, et al. MAPK inhibitors protect against early-stage osteoarthritis by activating autophagy. Mol Med Rep. 2021; 24(6):829. |
| [1] | Chen Yulin, He Yingying, Hu Kai, Chen Zhifan, Nie Sha Meng Yanhui, Li Runzhen, Zhang Xiaoduo , Li Yuxi, Tang Yaoping. Effect and mechanism of exosome-like vesicles derived from Trichosanthes kirilowii Maxim. in preventing and treating atherosclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1768-1781. |
| [2] | Zhou Sirui, Xu Yukun, Zhao Kewei. Ideas and methods of anti-melanogenesis of Angelica dahurica extracellular vesicles [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1747-1754. |
| [3] | Wu Zhilin, , He Qin, Wang Pingxi, Shi Xian, Yuan Song, Zhang Jun, Wang Hao . DYRK2: a novel therapeutic target for rheumatoid arthritis combined with osteoporosis based on East Asian and European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1569-1579. |
| [4] | Liu Hongtao, Wu Xin, Jiang Xinyu, Sha Fei, An Qi, Li Gaobiao. Causal relationship between age-related macular degeneration and deep vein thrombosis: analysis based on genome-wide association study data [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1602-1608. |
| [5] | Peng Zhiwei, Chen Lei, Tong Lei. Luteolin promotes wound healing in diabetic mice: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1398-1406. |
| [6] | Guo Ying, Tian Feng, Wang Chunfang. Potential drug targets for the treatment of rheumatoid arthritis: large sample analysis from European databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1549-1557. |
| [7] | Gao Zengjie, , Pu Xiang, Li Lailai, Chai Yihui, Huang Hua, Qin Yu. Increased risk of osteoporotic pathological fractures associated with sterol esters: evidence from IEU-GWAS and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1302-1310. |
| [8] | Liu Fengzhi, Dong Yuna, Tian Wenyi, Wang Chunlei, Liang Xiaodong, Bao Lin. Gene-predicted associations between 731 immune cell phenotypes and rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1311-1319. |
| [9] | Zhang Cuicui, Chen Huanyu, Yu Qiao, Huang Yuxuan, Yao Gengzhen, Zou Xu. Relationship between plasma proteins and pulmonary arterial hypertension and potential therapeutic targets [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1331-1340. |
| [10] | Chen Yixian, Chen Chen, Lu Liheng, Tang Jinpeng, Yu Xiaowei. Triptolide in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 805-815. |
| [11] | Zeng Hao, Sun Pengcheng, Chai Yuan, Huang Yourong, Zhang Chi, Zhang Xiaoyun. Association between thyroid function and osteoporosis: genome-wide data analysis of European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1019-1027. |
| [12] | Rong Xiangbin, , Zheng Haibo, Mo Xueshen, Hou Kun, Zeng Ping, . Plasma metabolites, immune cells, and hip osteoarthritis: causal inference based on GWAS data from European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1028-1035. |
| [13] | He Qiwang, , , Chen Bo, Liang Fuchao, Kang Zewei, Zhou Yuan, Ji Anxu, Tang Xialin, . Relationship between Alzheimer’s disease and sarcopenia and body mass index: analysis of GWAS datasets for European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1036-1046. |
| [14] | Ding Yu, Chen Jingwen, Chen Xiuyan, Shi Huimin, Yang Yudie, Zhou Meiqi, Cui Shuai, . Circulating inflammatory proteins and myocardial hypertrophy: large sample analysis of European populations from GWAS Catalog and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1047-1057. |
| [15] | Wang Yanfei, Jin Lianhai, Li Qingya, Fu Yuanfei, Tan Huangsheng, Deng Pengwei, Gao Kun. Synovial fluid exosome-mediated crosstalk between synoviocytes and chondrocytes in development and progression of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9032-9040. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||