Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (25): 6522-6532.doi: 10.12307/2026.292
Previous Articles Next Articles
Establishment and validation of a Sprague-Dawley rat model of aging-related knee osteoarthritis
Wu Zugui1, Zhu Yue1, Li Jiao1, Yuan Rong1, Wu Zhiwei1, Li Junyi2, Li Congcong3, Shen Zhen1, Guo Ying1
- 1Third Clinical College/Third Affiliated Hospital of Yunnan University of Chinese Medicine, Kunming Municipal Hospital of Traditional Chinese Medicine, Kunming 650500, Yunnan Province, China; 2Henan Luoyang Orthopedic Hospital, Luoyang 471000, Henan Province, China; 3First Affiliated Hospital of Zhejiang Chinese Medical University, Hangzhou 310000, Zhejiang Province, China
-
Received:2025-07-04Revised:2025-12-10Online:2026-09-08Published:2026-04-21 -
Contact:Guo Ying, MS, Chief physician, Master’s supervisor, Third Clinical College/Third Affiliated Hospital of Yunnan University of Chinese Medicine, Kunming Municipal Hospital of Traditional Chinese Medicine, Kunming 650500, Yunnan Province, China Co-corresponding author: Shen Zhen, PhD, Attending physician, Master’s supervisor, Third Clinical College/Third Affiliated Hospital of Yunnan University of Chinese Medicine, Kunming Municipal Hospital of Traditional Chinese Medicine, Kunming 650500, Yunnan Province, China -
About author:Wu Zugui, MD, Attending physician, Master's supervisor, Third Clinical College/Third Affiliated Hospital of Yunnan University of Chinese Medicine, Kunming Municipal Hospital of Traditional Chinese Medicine, Kunming 650500, Yunnan Province, China -
Supported by:National Natural Science Foundation of China, No. 82360943 (to SZ); Yunnan Fundamental Research Project, No. 202501AU070167 (to WZG); Joint Project of Traditional Chinese Medicine of Yunnan Provincial Department of Science and Technology, No. 202301AZ070001-094 (to GY); 202101AZ070001-170 (to SZ); 202301AZ070001-15 (to LJ); Yunnan Provincial Clinical Medical Center Research Project, No. 2024YNLCYXZX0293 (to WZG)
CLC Number:
Cite this article
Wu Zugui, Zhu Yue, Li Jiao, Yuan Rong, Wu Zhiwei, Li Junyi, Li Congcong, Shen Zhen, Guo Ying. Establishment and validation of a Sprague-Dawley rat model of aging-related knee osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6522-6532.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
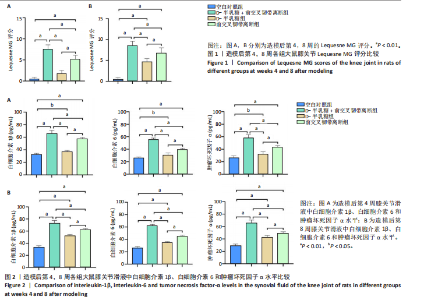
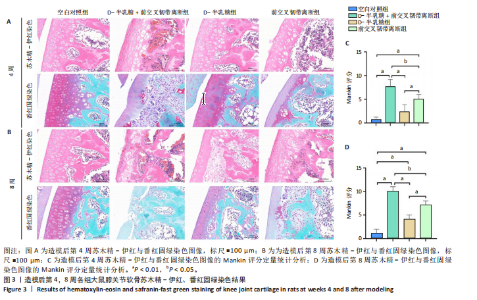
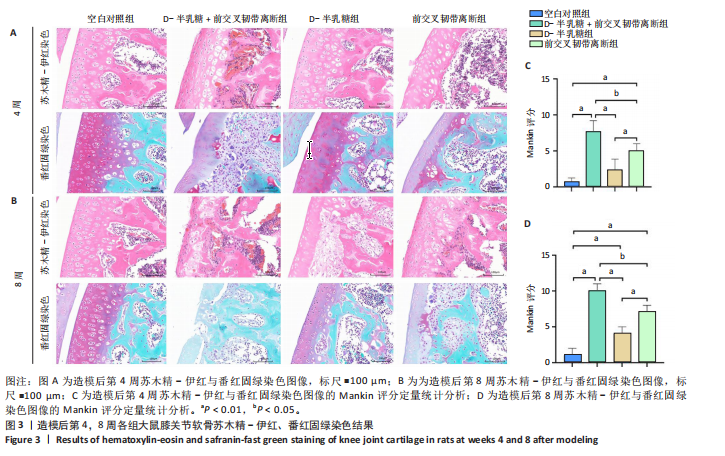
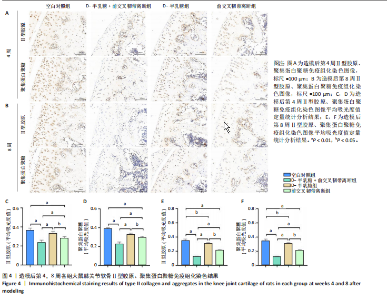
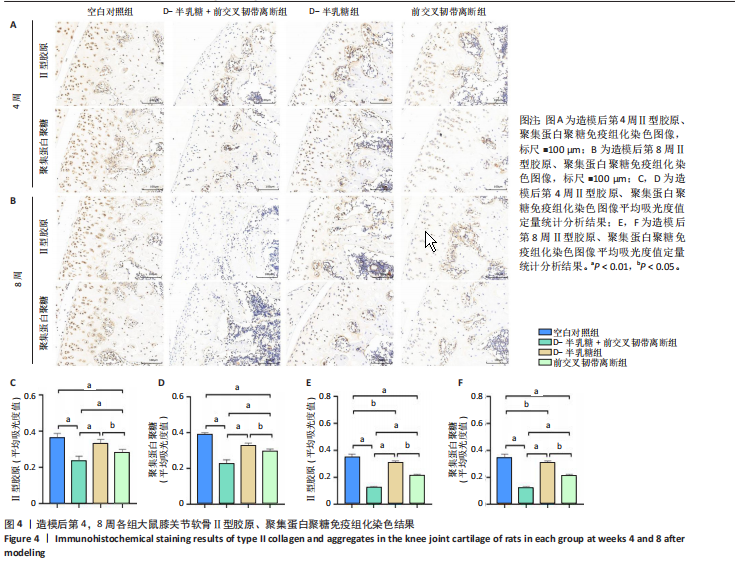
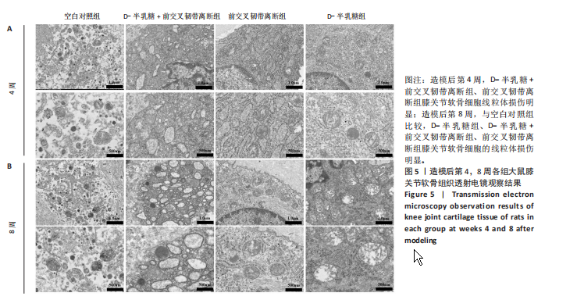
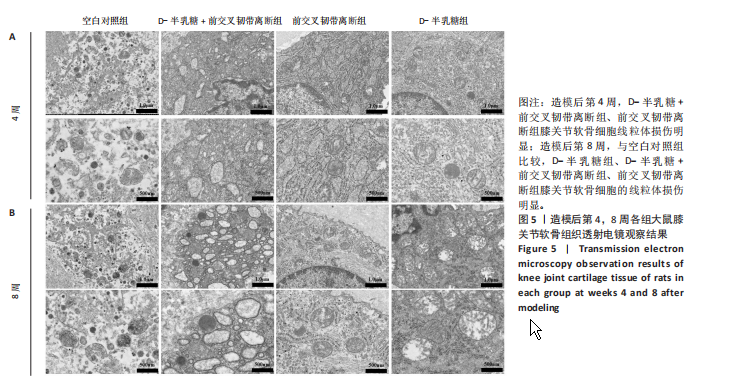
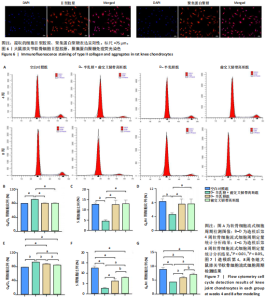
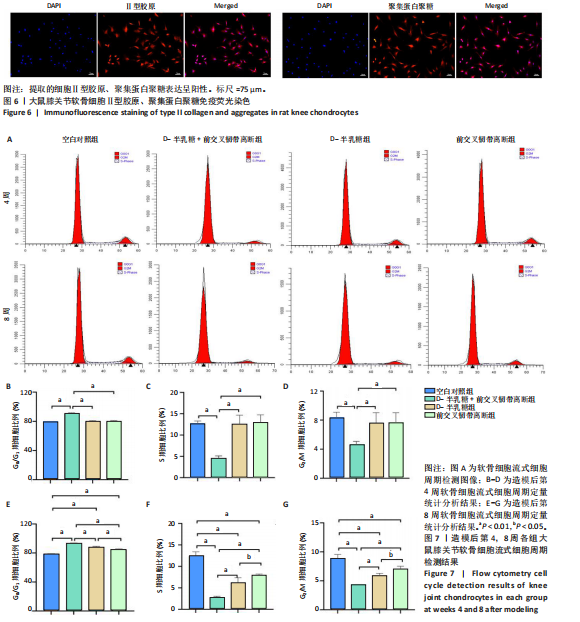
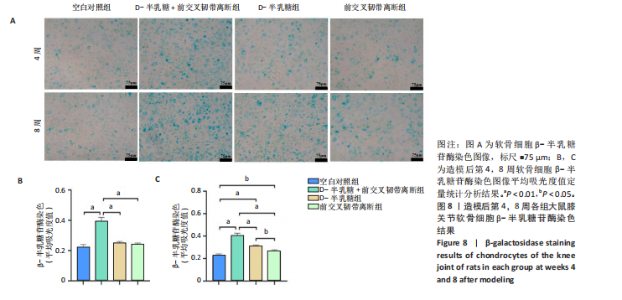
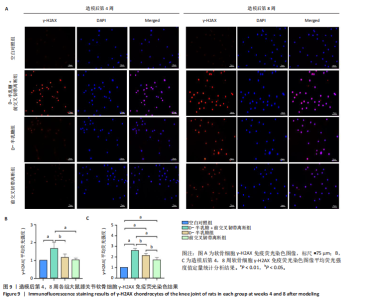
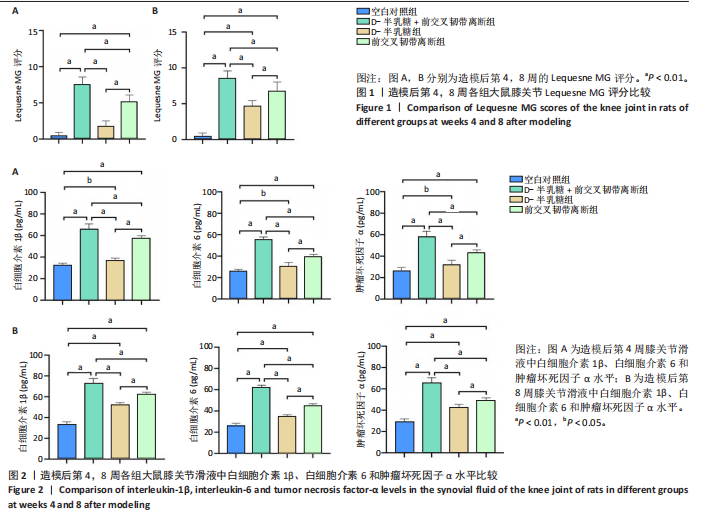
2.1 实验动物数量分析 40只SD大鼠全部进入结果分析。 2.2 各组大鼠Lequesne MG评分比较 造模后第4,8周,各组大鼠Lequesne MG评分比较,见图1。造模后第4周,D-半乳糖+前交叉韧带离断组、前交叉韧带离断组Lequesne MG评分高于空白对照组(P均< 0.05);D-半乳糖+前交叉韧带离断组的Lequesne MG评分高于前交叉韧带离断组(P < 0.05)。造模后第8周,D-半乳糖+前交叉韧带离断组、前交叉韧带离断组、D-半乳糖组Lequesne MG评分高于空白对照组(P均< 0.05),D-半乳糖+前交叉韧带离断组Lequesne MG评分高于前交叉韧带离断组(P < 0.05),前交叉韧带离断组Lequesne MG评分高于D-半乳糖组(P < 0.05)。 2.3 各组大鼠膝关节滑液中白细胞介素1β、白细胞介素6和肿瘤坏死因子α水平比较 造模后第4,8周,各组大鼠膝关节滑液中白细胞介素1β、白细胞介素6和肿瘤坏死因子α水平比较,见图2。造模后第4,8周,与空白对照组比较,D-半乳糖+前交叉韧带离断组、前交叉韧带离断组与D-半乳糖组白细胞介素1β、白细胞介素6、肿瘤坏死因子α水平升高(P < 0.05,P < 0.01);D-半乳糖+前交叉韧带离断组白细胞介素1β、白细胞介素6和肿瘤坏死因子α水平高于前交叉韧带离断组(P均< 0.01),前交叉韧带离断组白细胞介素1β、白细胞介素6和肿瘤坏死因子α水平高于D-半乳糖组(P均< 0.01)。 2.4 各组大鼠膝关节软骨组织苏木精-伊红、番红固绿染色结果 造模后第4,8周,各组大鼠膝关节软骨苏木精-伊红、番红固绿染色结果,如图3所示。造模后第4周,苏木精-伊红和番红固绿染色显示,空白对照组膝关节软骨表面光滑,浅层及深层均无明显破坏,软骨细胞形态正常、分布均匀且排列规则,软骨细胞数量无异常增多或减少,软骨基质染色均匀;D-半乳糖组膝关节软骨表面光滑,浅层及深层均无明显破坏,软骨细胞形态正常、分布基本均匀且排列基本规则,软骨细胞数量无异常增多或减少,软骨基质染色均匀;前交叉韧带离断组膝关节软骨表面不规则且存在裂隙,浅层有缺损,软骨基质染色较正常减少,软骨细胞分布异常,局部可见软骨细胞数量增多或减少;D-半乳糖+前交叉韧带离断组膝关节软骨表面毛糙,有明显裂隙,基质染色不均匀,潮线不清晰,软骨细胞分布异常,数量明显减少,软骨下骨可见硬化现象。造模后第8周,苏木精-伊红和番红固绿染色显示,空白对照组膝关节软骨表面光滑,浅层及深层均未见软骨破坏,软骨细胞形态正常、分布均匀且排列规则,软骨细胞数量无异常增多或减少,软骨基质染色均匀;D-半乳糖组膝关节软骨表面较光滑,浅层可见轻微的破坏,深层无明显破坏,软骨细胞形态较正常、分布稍均匀、排列稍规则,软骨细胞数量可见局部减少,软骨基质染色稍均匀;前交叉韧带离断组膝关节软骨表面不规则且存在明显裂隙,浅层及深层均可见局部缺损,软骨基质染色明显减少,软骨细胞分布异常,局部可见软骨细胞数量增多或减少;D-半乳糖+前交叉韧带离断组膝关节软骨表面明显毛糙,有明显裂隙,且裂隙到达深层,软骨基质染色不均匀,潮线不清晰,软骨细胞分布异常,数量显著减少,软骨下骨硬化。 造模后第4周,D-半乳糖+前交叉韧带离断组、前交叉韧带离断组Mankin评分高于空白对照组(P均< 0.01),D-半乳糖+前交叉韧带离断组Mankin评分高于前交叉韧带离断组(P < 0.05),前交叉韧带离断组Mankin评分明显高于D-半乳糖组(P < 0.01)。造模后第8周,D-半乳糖组、D-半乳糖+前交叉韧带离断组、前交叉韧带离断组Mankin评分高于空白对照组(P均< 0.01),D-半乳糖+前交叉韧带离断组Mankin评分高于前交叉韧带离断组(P < 0.05),前交叉韧带离断组Mankin评分明显高于D-半乳糖组(P < 0.01)。 2.5 各组大鼠膝关节软骨Ⅱ型胶原、聚集蛋白聚糖免疫组化染色结果 造模后第4,8周,各组大鼠膝关节软骨Ⅱ型胶原、聚集蛋白聚糖免疫组化染色结果,见图4。免疫组化染色显示,Ⅱ型胶原、聚集蛋白聚糖两种蛋白均表达于软骨细胞的细胞质中。造模后第4周,D-半乳糖+前交叉韧带离断组、前交叉韧带离断组Ⅱ型胶原、聚集蛋白聚糖表达低于空白对照组(P均< 0.01),D-半乳糖+前交叉韧带离断组Ⅱ型胶原、聚集蛋白聚糖表达低于前交叉韧带离断组(P均< 0.01),前交叉韧带离断组Ⅱ型胶原、聚集蛋白聚糖表达低于D-半乳糖组(P均< 0.05)。造模后第8周,D-半乳糖组、D-半乳糖+前交叉韧带离断组、前交叉韧带离断组Ⅱ型胶原、聚集蛋白聚糖表达均低于空白对照组(P < 0.05,P < 0.01),D-半乳糖+前交叉韧带离断组Ⅱ型胶原、聚集蛋白聚糖表达低于前交叉韧带离断组(P均< 0.01),前交叉韧带离断组Ⅱ型胶原、聚集蛋白聚糖表达低于D-半乳糖组(P均< 0.05)。 2.6 各组大鼠膝关节软骨透射电镜观察结果 造模后第4,8周,各组大鼠膝关节软骨透射电镜观察结果,如图5所示。造模后第4周,与空白对照组比较,D-半乳糖+前交叉韧带离断组、前交叉韧带离断组膝关节软骨细胞线粒体损伤明显,D-半乳糖组膝关节软骨细胞线粒体无明显变化。造模后第8周,与空白对照组比较,D-半乳糖组、D-半乳糖+前交叉韧带离断组、前交叉韧带离断组膝关节软骨细胞的线粒体损伤明显。 2.7 软骨细胞鉴定结果 Ⅱ型胶原、聚集蛋白聚糖免疫荧光染色结果显示,软骨细胞的细胞质均着色并呈现红色荧光,细胞核呈现出蓝色,提取的细胞符合软骨细胞的特征,见图6。 2.8 各组软骨细胞流式细胞周期检测结果 造模后第4,8周,各组软骨细胞流式细胞周期结果,见图7。造模后第4周,D-半乳糖+前交叉韧带离断组G0/G1期细胞比例高于空白对照组,S期、G2/M期细胞比例低于空白对照组(P均< 0.01);D-半乳糖+前交叉韧带离断组G0/G1期细胞比例高于D-半乳糖组,S期细胞比例明于D-半乳糖组(P均< 0.01)。造模后第8周,D-半乳糖组、D-半乳糖+前交叉韧带离断组、前交叉韧带离断组G0/G1期细胞比例高于空白对照组(P均< 0.01),S期、G2/M期细胞比例低于空白对照组(P均< 0.01);D-半乳糖+前交叉韧带离断组G0/G1期细胞比例高于D-半乳糖组(P均< 0.01),S期、G2/M期细胞比例低于D-半乳糖组(P均< 0.01);D-半乳糖组G0/G1期细胞比例高于前交叉韧带离断组(P < 0.01),S期、G2/M期细胞比例低于前交叉韧带离断组(P均< 0.05)。"
| [1] KATZ JN, ARANT KR, LOESER RF. Diagnosis and Treatment of Hip and Knee Osteoarthritis: A Review. JAMA. 2021;325(6):568-578. [2] ARDEN NK, PERRY TA, BANNURU RR, et al. Non-surgical management of knee osteoarthritis: comparison of ESCEO and OARSI 2019 guidelines. Nat Rev Rheumatol. 2021;17(1):59-66. [3] FU B, SHEN J, ZOU X, et al. Matrix stiffening promotes chondrocyte senescence and the osteoarthritis development through downregulating HDAC3. Bone Res. 2024;12(1):32. [4] CAO Y, RUAN J, KANG J, et al. Extracellular Vesicles in Infrapatellar Fat Pad from Osteoarthritis Patients Impair Cartilage Metabolism and Induce Senescence. Adv Sci (Weinh). 2024;11(3):e2303614. [5] DREVET S, FAVIER B, BRUN E, et al. Mouse Models of Osteoarthritis: A Summary of Models and Outcomes Assessment. Comp Med. 2022;72(1):3-13. [6] CAI N, WU Y, HUANG Y. Induction of Accelerated Aging in a Mouse Model. Cells. 2022;11(9):1418. [7] 张伟波,吴丽洁,高原,等.自然衰老致增龄性骨骼肌萎缩SD大鼠模型评价[J].中国老年学杂志,2022,42(22):5570-5574. [8] SONG X, LIU Y, CHEN S, et al. Knee osteoarthritis: A review of animal models and intervention of traditional Chinese medicine. Animal Model Exp Med. 2024; 7(2):114-126. [9] ZAKI S, BLAKER CL, LITTLE CB. OA foundations - experimental models of osteoarthritis. Osteoarthritis Cartilage. 2022;30(3):357-380. [10] 王喆,李瑞生.衰老动物模型的研究进展[J].中国比较医学杂志,2013,23(3): 67-70. [11] TAORMINA G, FERRANTE F, VIENI S, et al. Longevity: Lesson from Model Organisms. Genes (Basel). 2019;10(7):518. [12] 尹丹阳,胡怡,史仍飞.动物衰老模型的研究进展[J].实验动物与比较医学, 2023,43(2):156-162. [13] 杨秋红,赵琛,吴焕淦,等.衰老相关动物模型的应用进展[J].吉林中医药, 2013,33(2):206-207. [14] HOCHHEISER K, KUEH AJ, GEBHARDT T, et al. CRISPR/Cas9: A tool for immunological research. Eur J Immunol. 2018;48(4):576-583. [15] MORIWAKI T, ABE S, OSHIMURA M, et al. Transchromosomic technology for genomically humanized animals. Exp Cell Res. 2020;390(2):111914. [16] LI JH, WEI TT, GUO L, et al. Curcumin protects thymus against D-galactose-induced senescence in mice. Naunyn Schmiedebergs Arch Pharmacol. 2021;394(2):411-420. [17] LI XL, XU M, YU F, et al. Effects of D-pinitol on myocardial apoptosis and fibrosis in streptozocin-induced aging-accelerated mice. J Food Biochem. 2021;45(4): e13669. [18] PENG XM, GAO L, HUO SX, et al. The Mechanism of Memory Enhancement of Acteoside (Verbascoside) in the Senescent Mouse Model Induced by a Combination of D-gal and AlCl3. Phytother Res. 2015;29(8):1137-1144. [19] ABBAS EY, EZZAT MI, RAMADAN NM, et al. Characterization and anti-aging effects of Opuntia ficus-indica (L.) Miller extracts in a D-galactose-induced skin aging model. Food Funct. 2023;14(7):3107-3125. [20] LI Y, LIN M, WANG G, et al. Atractylodes macrocephala polysaccharides shield a D-galactose-induced aging model via gut microbiota modulation. Int J Biol Macromol. 2024;281(Pt 1):136205. [21] 赵凡凡,周玉枝,高丽,等.D-半乳糖致衰老大鼠模型的研究进展[J].药学学报,2017,52(3):347-354. [22] ALI T, BADSHAH H, KIM TH, et al. Melatonin attenuates D-galactose-induced memory impairment, neuroinflammation and neurodegeneration via RAGE/NF-K B/JNK signaling pathway in aging mouse model. J Pineal Res. 2015;58(1):71-85. [23] 赵忠胜,陈振沅,黄云梅,等.荣筋拈痛方对膝关节软骨细胞外基质代谢的作用及机制[J].中国组织工程研究,2023,27(28):4448-4455. [24] GELBER AC. Knee Osteoarthritis. Ann Intern Med. 2024;177(9):ITC129-ITC144. [25] DU X, LIU ZY, TAO XX, et al. Research Progress on the Pathogenesis of Knee Osteoarthritis. Orthop Surg. 2023;15(9):2213-2224. [26] WANG S, YANG J, XIANG R, et al. Research and publication trends on knee osteoarthritis and cellular senescence: a bibliometric analysis. Front Physiol. 2023;14:1269338. [27] XIE J, WANG Y, LU L, et al. Cellular senescence in knee osteoarthritis: molecular mechanisms and therapeutic implications. Ageing Res Rev. 2021;70:101413. [28] RAHMATI M, NALESSO G, MOBASHERI A, et al. Aging and osteoarthritis: Central role of the extracellular matrix. Ageing Res Rev. 2017;40:20-30. [29] DIEKMAN BO, LOESER RF. Aging and the emerging role of cellular senescence in osteoarthritis. Osteoarthritis Cartilage. 2024;32(4):365-371. [30] MASTROGIOVANNI M, MARTÍNEZ-NAVARRO FJ, BOWMAN TV, et al. Inflammation in Development and Aging: Insights from the Zebrafish Model. Int J Mol Sci. 2024; 25(4):2145. [31] ZHANG S, LI F, ZHOU T, et al. Caenorhabditis elegans as a Useful Model for Studying Aging Mutations. Front Endocrinol (Lausanne). 2020;11:554994. [32] 刘传铃,王佳贺.衰老相关动物模型研究进展[J].实用老年医学,2018,32(12): 1103-1105,1120. [33] 陈强威,江涛,唐春萍.动物衰老模型建立与机制的研究进展[J].广东药科大学学报,2018,34(1):119-123. [34] AZMAN KF, ZAKARIA R. D-Galactose-induced accelerated aging model: an overview. Biogerontology. 2019;20(6):763-782. [35] 洪晶,张娅俐,闫莎莎,等.D-半乳糖诱导衰老小鼠模型研究进展[J].中国比较医学杂志,2023,33(3):136-142. [36] BAY-JENSEN AC, MOBASHERI A, THUDIUM CS, et al. Blood and urine biomarkers in osteoarthritis - an update on cartilage associated type II collagen and aggrecan markers. Curr Opin Rheumatol. 2022;34(1):54-60. [37] GUO Y, GUAN T, SHAFIQ K, et al. Mitochondrial dysfunction in aging. Ageing Res Rev. 2023;88:101955. [38] OGRODNIK M. Cellular aging beyond cellular senescence: Markers of senescence prior to cell cycle arrest in vitro and in vivo. Aging Cell. 2021;20(4):e13338. [39] ØVREBØ JI, MA Y, EDGAR BA. Cell growth and the cell cycle: New insights about persistent questions. Bioessays. 2022;44(11):e2200150. [40] LOZANO-TORRES B, BLANDEZ JF, SANCENÓN F, et al. Chromo-fluorogenic probes for β-galactosidase detection. Anal Bioanal Chem. 2021;413(9):2361-2388. [41] ITAHANA K, CAMPISI J, DIMRI GP. Methods to detect biomarkers of cellular senescence: the senescence-associated beta-galactosidase assay. Methods Mol Biol. 2007;371:21-31. [42] YOU L, NEPOVIMOVA E, VALKO M, et al. Mycotoxins and cellular senescence: the impact of oxidative stress, hypoxia, and immunosuppression. Arch Toxicol. 2023;97(2):393-404. |
| [1] | Li Sa, Sun Ning, Sun Zhaozhong, Feng Zhimeng, Li Xuedong. Evaluation parameters and specific region of C6 nerve oppression by uncinate process degeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2294-2302. |
| [2] | Zhu Xiaolong, Zhang Wei, Yang Yang. Visualization analysis of research hotspots and cutting-edge information in the field of intervertebral disc regeneration and repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2391-2402. |
| [3] | Wen Fayan, Li Yan, Qiang Tianming, Yang Chen, Shen Linming, Li Yadong, Liu Yongming. Unilateral biportal endoscopic technology for treatment of lumbar degenerative diseases: global research status and changing trends [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2380-2390. |
| [4] | Zhao Jiaqing, Liu Dong, Lu Suni, Wang Dawei, Geng Xiaopeng, Ning Huaxiu. One-hole split endoscope surgery with the aid of digital 3D technology for highly migrated lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2278-2285. |
| [5] | Zhang Xianxu, Ma Zhong, Liu Xin, Huang Lei, Shen Wenxiang, Luo Zhiqiang . Lumbar fusion combined with unilateral fixation for lumbar degenerative diseases: biomechanics, technical evolution, and clinical applications [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2334-2342. |
| [6] | Sun Lei, Zhang Qi, Zhang Yu. Pro-osteoblastic effect of chlorogenic acid protein microsphere/polycaprolactone electrospinning membrane [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1877-1884. |
| [7] | Chen Haojie, Wang Dai, Shen Shan. Immune inflammatory microenvironment mechanisms in peri-implantitis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2054-2062. |
| [8] | Yang Qiongqiong, Liu Wei. Comparison of performance and clinical effects of zirconia and titanium implants [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2063-2071. |
| [9] | Wang Zheng, Cheng Ji, Yu Jinlong, Liu Wenhong, Wang Zhaohong, Zhou Luxing. Progress and future perspectives on the application of hydrogel materials in stroke therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2081-2090. |
| [10] | Fu Lyupeng, Yu Peng, Liang Guoyan, Chang Yunbing. Electroactive materials applied in spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2113-2123. |
| [11] | Lai Yu, Chen Yueping, Zhang Xiaoyun. Research hotspots and frontier trends of bioactive materials in treating bone infections [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2132-2144. |
| [12] | Yang Lixia, Diao Liqin, Li Hua, Feng Yachan, Liu Xin, Yu Yuexin, Dou Xixi, Gu Huifeng, Xu Lanju. Regulatory mechanism of recombinant type III humanized collagen protein improving photoaging skin in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1988-2000. |
| [13] | Tan Jing, Li Li, Wang Liangliang, Qin Xiangyu. Bionic functional coating improves the integration of titanium implants and skin tissue interface [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2014-2022. |
| [14] | Wang Qisa, Lu Yuzheng, Han Xiufeng, Zhao Wenling, Shi Haitao, Xu Zhe. Cytocompatibility of 3D printed methyl acrylated hyaluronic acid/decellularized skin hydrogel scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1912-1920. |
| [15] | Gao Yanguo, Guo Xu, Li Xiaohan, Chen Shiqi, Zhu Haitao, Huang Liangyong, Ye Fang, Lu Wei Wang Qibin, Zheng Tao, Chen Li. Optimization of prescription ratio of “Honghuangbai” gel by orthogonal test in diabetic skin wound mouse models [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1921-1928. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||