Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (34): 8986-8993.doi: 10.12307/2026.892
Previous Articles Next Articles
Mitochondrial kinetic mechanism by which triptolide alleviates hydrogen peroxide-induced apoptosis in SH-SY5Y cells
Mu Bingtao1, Guo Minfang1, Hu Fenqi1, Liu Qiyuan1, Jia Hui1, Xu Mingyuan1, Chen Jiayuan2, Zhang Huiyu1, Meng Tao1, Yu Jiezhong1, 3#br#
- 1Institute of Brain Science/Key Laboratory of Molecular Cellular Immunology in Datong, Shanxi Datong University, Datong 037009, Shanxi Province, China; 2Shanxi University of Traditional Chinese Medicine, Jinzhong 030619, Shanxi Province, China; 3Datong Fifth People's Hospital, Shanxi Datong University, 037009, Shanxi Province, China
-
Received:2025-08-14Revised:2026-02-26Online:2026-12-08Published:2026-04-14 -
Contact:Wei Jiezhong, PhD, Professor, Doctoral supervisor, Institute of Brain Science/Key Laboratory of Molecular Cellular Immunology in Datong, Shanxi Datong University, Datong 037009, Shanxi Province, China; Datong Fifth People's Hospital, Shanxi Datong University, 037009, Shanxi Province, China -
About author:Mu Bingtao, MS, Associate professor, Institute of Brain Science/Key Laboratory of Molecular Cellular Immunology in Datong, Shanxi Datong University, Datong 037009, Shanxi Province, China -
Supported by:Research Project of Datong City Basic Research Program, No. 2024063 (to MBT); Research Project of Datong City Platform Research Program, No. 2024093 (to MT); Research Project of Shanxi Province Basic Research Program, No. 20210302123476 (to GMF); Research Project of Shanxi Province Basic Research Program, No. 20210302123478 (to ZHY); Project Unit of Key Research Laboratory of Traditional Chinese Medicine: Key Research Laboratory of Traditional Chinese Medicine for the Prevention and Treatment of Dementia, No. zyyyjs2024027 (to YJZ); Shanxi Province Traditional Chinese Medicine Research Project, No. 2023ZYYB2042 (to YJZ)
CLC Number:
Cite this article
Mu Bingtao, Guo Minfang, Hu Fenqi, Liu Qiyuan, Jia Hui, Xu Mingyuan, Chen Jiayuan, Zhang Huiyu, Meng Tao, Yu Jiezhong. Mitochondrial kinetic mechanism by which triptolide alleviates hydrogen peroxide-induced apoptosis in SH-SY5Y cells[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8986-8993.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
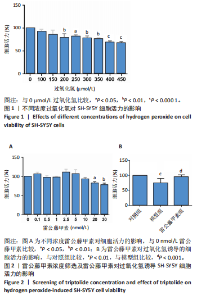
2.1 过氧化氢最佳浓度的确定 如图1所示,过氧化氢浓度在100,150 μmol/L时,细胞活力即有所下降,随着过氧化氢浓度增大,200 μmol/L过氧化氢干预后细胞活力明显下降(P < 0.01),当浓度增至400,450 μmol/L时,细胞活力更低(P < 0.000 1),参考文献[21-22],确定200 μmol/L为过氧化氢干预浓度。 2.2 雷公藤甲素药物浓度的确定 如图2A所示,雷公藤甲素浓度为2.5 nmol/L时细胞活力最高;与0 nmol/L雷公藤甲素相比,当雷公藤甲素浓度达到20 nmol/L时,细胞存活率开始下降,差异有显著性意义(P < 0.05),对细胞产生一定的毒性作用;如图2B"
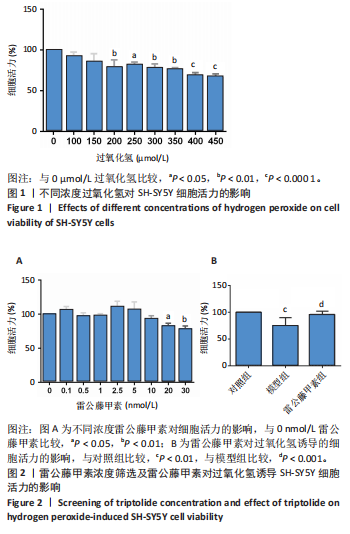
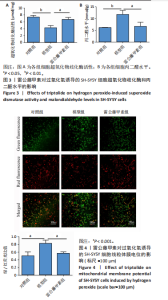
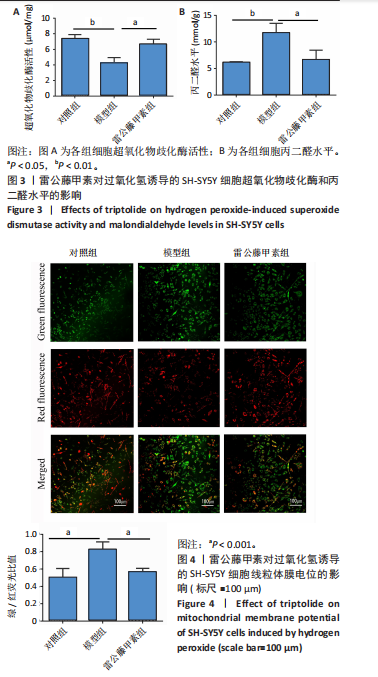
所示,用2.5 nmol/L雷公藤甲素干预过氧化氢诱导的SH-SY5Y细胞,细胞存活率较模型组显著升高(P < 0.001),可见雷公藤甲素对细胞有保护作用。后续实验均选用2.5 nmol/L雷公藤甲素干预SH-SY5Y细胞。 2.3 雷公藤甲素改善了过氧化氢诱导的氧化应激状态 超氧化物歧化酶活性间接反映机体清除活性氧的能力,如图3所示,模型组的超氧化物歧化酶活性较对照组显著下降(P < 0.01),雷公藤甲素组超氧化物歧化酶活性较模型组显著上升(P < 0.05);丙二醛水平可以直接提示氧化损伤程度,模型组丙二醛水平较对照组显著升高(P < 0.01);雷公藤甲素组丙二醛水平较模型组显著降低(P < 0.05)。 2.4 雷公藤甲素提高了过氧化氢诱导的线粒体膜电位水平 如图4所示,红色荧光提示线粒体膜电位正常,绿色荧光增强提示线粒体膜电位下降,细胞开始凋亡,常用绿/红荧光比值表示膜电位水平,比值越高,膜电位水平越低,呼吸链受损越重。与对照组相比,模型组的绿/红荧光比值明显升高(P < 0.001);与模型组相比,雷公藤甲素组的绿/红荧光比值明显下降(P < 0.001),说明雷公藤甲素改善了线粒体呼吸功能,减少细胞凋亡。 2.5 雷公藤甲素抑制了过氧化氢诱导的细胞凋亡 如图5所示,与对照组相比,模型组促凋亡蛋白Bax、Cleaved-caspase-3表达及细胞凋亡率都显著升高(P < 0.05,P < 0.05,P < 0.01),"
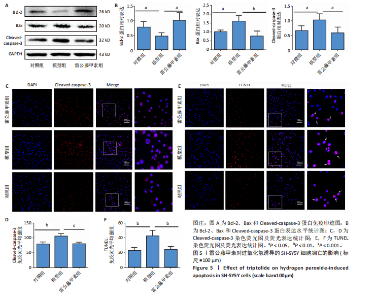
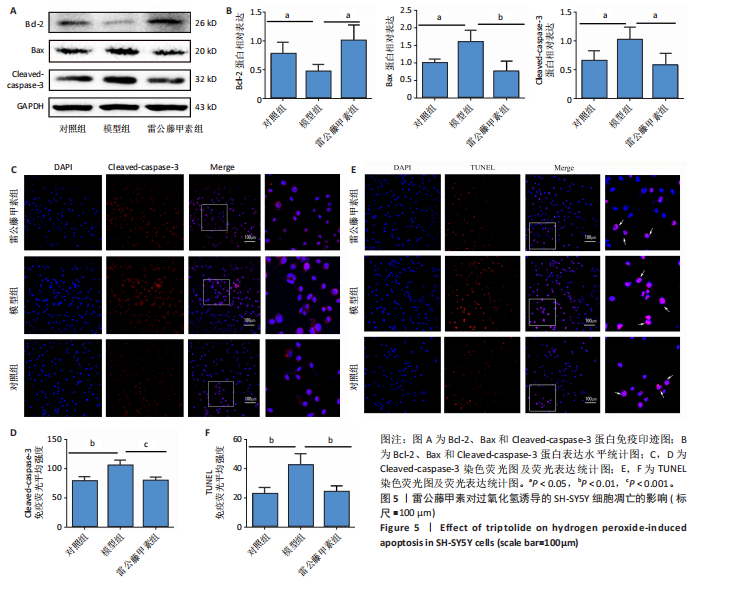
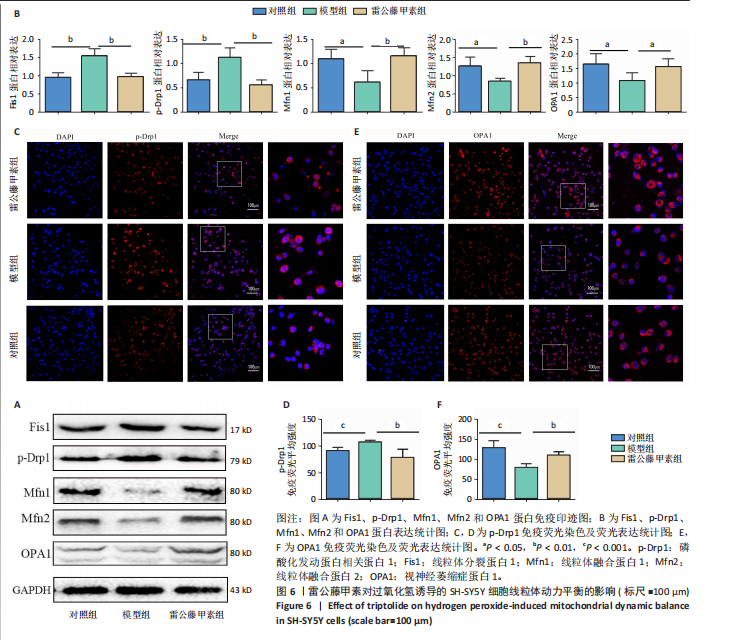
抗凋亡蛋白Bcl-2表达显著降低(P < 0.05);与模型组相比,雷公藤甲素组抗凋亡蛋白Bcl-2表达显著升高(P < 0.05),促凋亡蛋白Bax、cleaved-caspase-3表达及细胞凋亡率显著降低(P < 0.01,P < 0.05,P < 0.01)。 2.6 雷公藤甲素促进过氧化氢诱导的线粒体融合且抑制分裂 如图6所示,与对照组相比,模型组线粒体分裂蛋白1和磷酸化发动蛋白相关蛋白1表达显著升高(P < 0.01,P < 0.01);线粒体融合蛋白1、线粒体融合蛋白2和视神经萎缩症蛋白1表达显著降低(P < 0.05,P < 0.05,P < 0.05);与模型组相比,雷公藤甲素组线粒体分裂蛋白1和磷酸化发动蛋白相关蛋白1表达显著降低(P < 0.01,P < 0.01),线粒体融合蛋白1、线粒体融合蛋白2和视神经萎缩症蛋白1表达显著增高(P < 0.01,P < 0.01,P < 0.05)。上述结果表明雷公藤甲素通过促进过氧化氢诱导的线粒体融合,抑制分裂,调控线粒体动力平衡。"
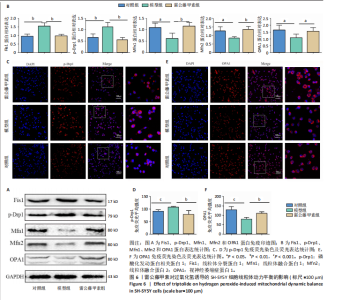
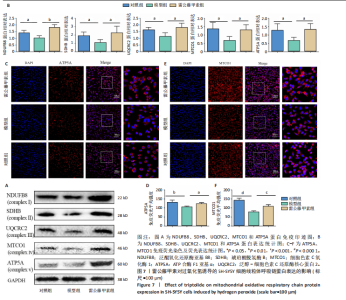
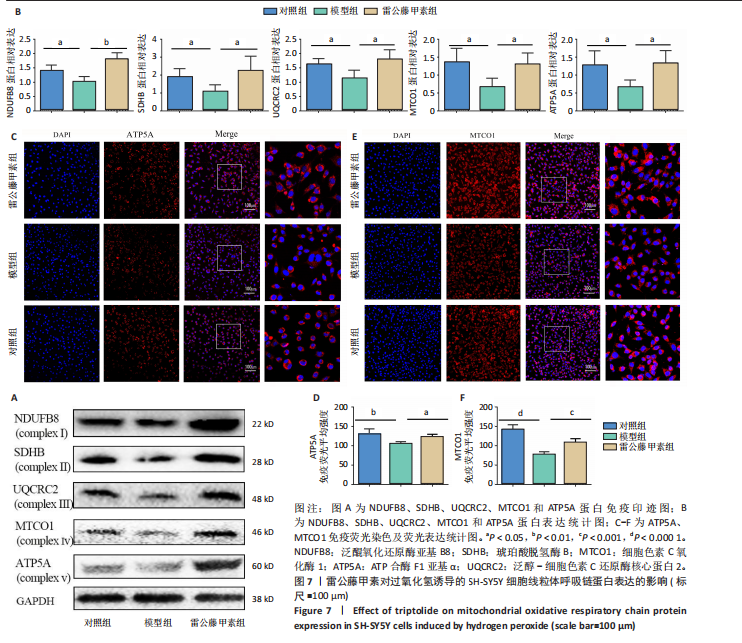
2.7 雷公藤甲素促进过氧化氢诱导的线粒体氧化呼吸链复合蛋白表达 如图7A,B所示,Western blot结果显示,与对照组相比,模型组泛醌氧化还原酶亚基B8、琥珀酸脱氢酶B、泛醇-细胞色素C还原酶核心蛋白2、细胞色素C氧化酶1和ATP合酶F1亚基α蛋白表达显著降低(P均< 0.05);与模型组相比,雷公藤甲素组泛醌氧化还原酶亚基B8、琥珀酸脱氢酶B、泛醇-细胞色素C还原酶核心蛋白2、细胞色素C氧化酶1和ATP合酶F1亚基α蛋白表达显著升高(P < 0.01,P < 0.05,P < 0.05,P < 0.05,P < 0.05)。如图7C-F所示,免疫荧光结果显示,与对照组相比,模型组ATP合酶F1亚基α和细胞色素C氧化酶1表达显著降低(P < 0.01,P < 0.000 1);与模型组相比,雷公藤甲素组ATP合酶F1亚基α和细胞色素C氧化酶1表达显著升高(P < 0.05,P < 0.001)。上述结果表明雷公藤甲素恢复了过氧化氢诱导的线粒体氧化磷酸化水平。"
| [1] ROSTAGNO AA. Pathogenesis of Alzheimer’s Disease. Int J Mol Sci. 2022;24(1): 107. [2] KNAPSKOG AB, ENGEDAL K, SELBÆK G, et al. Alzheimer’s disease – diagnosis and treatment. Tidsskr Nor Laegeforen. 2021;141(7). doi: 10.4045/tidsskr.20.0919. [3] XU X, PANG Y, FAN X. Mitochondria in oxidative stress, inflammation and aging: from mechanisms to therapeutic advances. Signal Transduct Target Ther. 2025; 10(1):190. [4] KLEMMENSEN MM, BORROWMAN SH, PEARCE C, et al. Mitochondrial dysfunction in neurodegenerative disorders. Neurotherapeutics. 2024;21(1):e00292. [5] ATTWELL D, LAUGHLIN SB. An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab. 2001;21(10):1133-1145. [6] COYLE JT, PUTTFARCKEN P. Oxidative stress, glutamate, and neurodegenerative disorders. Science. 1993;262(5134):689-695. [7] RAI SN, SINGH C, SINGH A, et al. Mitochondrial Dysfunction: a Potential Therapeutic Target to Treat Alzheimer’s Disease. Mol Neurobiol. 2020;57(7): 3075-3088. [8] MACDONALD R, BARNES K, HASTINGS C, et al. Mitochondrial abnormalities in Parkinson’s disease and Alzheimer’s disease: can mitochondria be targeted therapeutically? Biochem Soc Trans. 2018;46(4):891-909. [9] EBADPOUR N, MAHMOUDI M, KAMAL KHEDER R, et al. From mitochondrial dysfunction to neuroinflammation in Parkinson’s disease: Pathogenesis and mitochondrial therapeutic approaches. Int Immunopharmacol. 2024;142(Pt A): 13015. [10] EO H, YU SH, CHOI Y, et al. Mitochondrial transplantation exhibits neuroprotective effects and improves behavioral deficits in an animal model of Parkinson’s disease. Neurotherapeutics. 2024;21(4):e00355. [11] WEI YM, WANG YH, XUE HQ, et al. Triptolide, A Potential Autophagy Modulator. Chin J Integr Med. 2019;25(3):233-240. [12] JIA L, NIE XQ, JI HM, et al. Multiple-Coated PLGA Nanoparticles Loading Triptolide Attenuate Injury of a Cellular Model of Alzheimer’s Disease. Biomed Res Int. 2021;2021:8825640. [13] XU P, WU Z, PENG Y, et al. Neuroprotection of Triptolide against Amyloid-Beta1-42-induced toxicity via the Akt/mTOR/p70S6K-mediated Autophagy Pathway. An Acad Bras Cienc. 2022;94(2):e20210938. [14] XU P, WANG H, LI Z, et al. Triptolide attenuated injury via inhibiting oxidative stress in Amyloid-Beta25-35-treated differentiated PC12 cells. Life Sci. 2016;145:19-26. [15] HE Z, BOTCHWAY BOA, ZHANG Y, et al. Triptolide activates the Nrf2 signaling pathway and inhibits the NF-κB signaling pathway to improve Alzheimer disease. Metab Brain Dis. 2024;39(1):173-182. [16] ZHENG Y, ZHANG WJ, WANG XM. Triptolide with potential medicinal value for diseases of the central nervous system. CNS Neurosci Ther. 2013;19(2):76-82. [17] SONG W, LIU M, WU J, et al. Preclinical Pharmacokinetics of Triptolide: A Potential Antitumor Drug. Curr Drug Metab. 2019;20(2):147-154. [18] CHEN P, ZHONG X, SONG Y, et al. Triptolide induces apoptosis and cytoprotective autophagy by ROS accumulation via directly targeting peroxiredoxin 2 in gastric cancer cells. Cancer Lett. 2024;587:216622. [19] LI W, YANG Y, HU Z, et al. Neuroprotective effects of DAHP and Triptolide in focal cerebral ischemia via apoptosis inhibition and PI3K/Akt/mTOR pathway activation. Front Neuroanat. 2015;9:48. [20] NIE J, ZHOU M, LÜ C, et al. Effects of triptolide on the synaptophysin expression of hippocampal neurons in the AD cellular model. Int Immunopharmacol. 2012;13(2):175-180. [21] 宋哲,薛超,张小曼,等.脂联素通过激活PP2A减轻H2O2诱导的SH-SY5Y细胞损伤及tau蛋白过度磷酸化[J].中国病理生理杂志,2015,31(2):207-212. [22] 于婧文,郭敏芳,李苏垚,等.黄芪甲苷通过调控线粒体功能抑制H2O2诱导的SH-SY5Y细胞凋亡[J].中国病理生理杂志,2022,38(9):1553-1560. [23] ASHLEIGH T, SWERDLOW RH, BEAL MF. The role of mitochondrial dysfunction in Alzheimer’s disease pathogenesis. Alzheimers Dement. 2023;19(1):333-342. [24] YAO MF, DANG T, WANG HJ, et al. Mitochondrial homeostasis regulation: A promising therapeutic target for Parkinson’s disease. Behav Brain Res. 2024; 459:114811. [25] ELFAWY HA, DAS B. Crosstalk between mitochondrial dysfunction, oxidative stress, and age related neurodegenerative disease: Etiologies and therapeutic strategies. Life Sci. 2019;218:165-184. [26] CAO H, ZUO C, GU Z, et al. High frequency repetitive transcranial magnetic stimulation alleviates cognitive deficits in 3xTg-AD mice by modulating the PI3K/Akt/GLT-1 axis. Redox Biol. 2022;54:102354. [27] PERLUIGI M, DI DOMENICO F, BUTTERFIELD DA. Oxidative damage in neurodegeneration: roles in the pathogenesis and progression of Alzheimer disease. Physiol Rev. 2024;104(1):103-197. [28] YULUG B, ALTAY O, LI X, et al. Multi-omics characterization of improved cognitive functions in Parkinson’s disease patients after the combined metabolic activator treatment: a randomized, double-blinded, placebo-controlled phase II trial. Brain Commun. 2025;7(1):fcae478. [29] 于海杰,关启刚,郭亮,等.雷公藤内酯醇保护心肌H/R损伤的相关分子机制[J].贵州医科大学学报,2019,44(2):190-194. [30] LV C, CHENG T, ZHANG B, et al. Triptolide protects against podocyte injury in diabetic nephropathy by activating the Nrf2/HO-1 pathway and inhibiting the NLRP3 inflammasome pathway. Ren Fail. 2023;45(1):2165103. [31] ALQAHTANI T, DEORE SL, KIDE AA, et al. Mitochondrial dysfunction and oxidative stress in Alzheimer’s disease, and Parkinson’s disease, Huntington’s disease and Amyotrophic Lateral Sclerosis -An updated review. Mitochondrion. 2023;71:83-92. [32] HASHIMI H, GAHURA O, PÁNEK T. Bringing together but staying apart: decisive differences in animal and fungal mitochondrial inner membrane fusion. Biol Rev Camb Philos Soc. 2025;100(2):920-935. [33] PIRES M, REGO AC. Apoe4 and Alzheimer’s Disease Pathogenesis-Mitochondrial Deregulation and Targeted Therapeutic Strategies. Int J Mol Sci. 2023;24(1):778. [34] GREL H, WOZNICA D, RATAJCZAK K, et al. Mitochondrial Dynamics in Neurodegenerative Diseases: Unraveling the Role of Fusion and Fission Processes. Int J Mol Sci. 2023;24(17):13033. [35] BUCK SA, SANDERS LH. LRRK2-mediated mitochondrial dysfunction in Parkinson’s disease. Biochem J. 2025;482(11):721-739. [36] 姚美桦,黄浩.线粒体呼吸链超级复合物研究进展[J].生物学教学,2024, 49(8):2-5. [37] LÉVY E, EL BANNA N, BAÏLLE D, et al. Causative Links between Protein Aggregation and Oxidative Stress: A Review. Int J Mol Sci. 2019;20(16):3896. [38] LIU G, YANG C, WANG X, et al. Oxygen metabolism abnormality and Alzheimer’s disease: An update. Redox Biol. 2023;68:102955. [39] SONG T, SONG X, ZHU C, et al. Mitochondrial dysfunction, oxidative stress, neuroinflammation, and metabolic alterations in the progression of Alzheimer’s disease: A meta-analysis of in vivo magnetic resonance spectroscopy studies. Ageing Res Rev. 2021;72:101503. |
| [1] | Yang Xuetao, Zhu Menghan, Zhang Chenxi, Sun Yimin, Ye Ling. Applications and limitations of antioxidant nanomaterials in oral cavity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2044-2053. |
| [2] | Wang Zheng, Cheng Ji, Yu Jinlong, Liu Wenhong, Wang Zhaohong, Zhou Luxing. Progress and future perspectives on the application of hydrogel materials in stroke therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2081-2090. |
| [3] |
Dong Chunyang, Zhou Tianen, Mo Mengxue, Lyu Wenquan, Gao Ming, Zhu Ruikai, Gao Zhiwei.
Action mechanism of metformin combined with Eomecon chionantha Hance dressing in treatment of deep second-degree burn wounds#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2001-2013.
|
| [4] | Liu Anting, Lu Jiangtao, Zhang Wenjie, He Ling, Tang Zongsheng, Chen Xiaoling. Regulation of AMP-activated protein kinase by platelet lysate inhibits cadmium-induced neuronal apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1800-1807. |
| [5] | Yuan Xiaoshuang, Yang Xu, Yang Bo, Chen Xiaoxu, Tian Ting, Wang Feiqing, Li Yanju, Liu Yang, Yang Wenxiu. Effect of conditioned medium of diffuse large B-cell lymphoma cells on proliferation and apoptosis of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1632-1640. |
| [6] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [7] | Jia Jinwen, Airefate·Ainiwaer, Zhang Juan. Effects of EP300 on autophagy and apoptosis related to allergic rhinitis in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1439-1449. |
| [8] | Zhang Di, Zhao Jun, Ma Guangyue, Sun Hui, Jiang Rong. Mechanism of depression-like behavior in chronic social defeat stress mice based on high-throughput sequencing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1139-1146. |
| [9] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [10] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Advance in the mechanisms underlying miRNAs in steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1207-1214. |
| [11] | Zhang Jiuxuan, Zhang Jinnan, Sui Xiaofan, Pei Xiaxia, Wei Jianhong, Su Qiang, Li Tian. Effects of ammonia poisoning on cognitive behavior and hippocampal synaptic damage in mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1122-1128. |
| [12] | Bao Zhuoma, Hou Ziming, Jiang Lu, Li Weiyi, Zhang Zongxing, Liu Daozhong, Yuan Lin. Effect and mechanism by which Pterocarya hupehensis skan total flavonoids regulates the proliferation, migration and apoptosis of fibroblast-like synoviocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 816-823. |
| [13] | Ma Hong, Ding Xueling, Wang Qi, Lyu Hui, Asya Albusm, Cheng Xinyi, Ma Xiang. Expression and significance of tumor necrosis factor alpha, nuclear factor kappaB and ionized calcium binding adaptor molecule-1 in the hippocampus of mice with aortic dissection [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 858-863. |
| [14] | Yang Xiao, Bai Yuehui, Zhao Tiantian, Wang Donghao, Zhao Chen, Yuan Shuo. Cartilage degeneration in temporomandibular joint osteoarthritis: mechanisms and regenerative challenges [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 926-935. |
| [15] | Ma Runqiu, Yang Huixia, Li Xuer, Bai Zhigang, Li Guizhong, Hao Yinju, Ma Shengchao, Jiang Yideng. Mechanism of glucocorticoid-induced mitochondrial dysfunction in osteoblasts in steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8845-8851. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||