Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (34): 8860-8867.doi: 10.12307/2026.846
Previous Articles Next Articles
Mechanism by which Hernandezine alleviates osteoporosis through macrophage polarization and osteoclast activation
Xia Wenyu, Zhang Wei, Li Wenhao, Jiang Kunlong, Wu Zebin, Yang Huilin
- Department of Orthopedics, First Affiliated Hospital of Suzhou University, Suzhou 215006, Jiangsu Province, China
-
Received:2025-10-04Revised:2026-01-21Online:2026-12-08Published:2026-04-11 -
Contact:Yang Huilin, Chief physician, Department of Orthopedics, First Affiliated Hospital of Suzhou University, Suzhou 215006, Jiangsu Province, China -
About author:Xia Wenyu, MS, Department of Orthopedics, First Affiliated Hospital of Suzhou University, Suzhou 215006, Jiangsu Province, China -
Supported by:2023 Interdisciplinary Innovation Project of Suzhou Medical College (to WZB)
CLC Number:
Cite this article
Xia Wenyu, Zhang Wei, Li Wenhao, Jiang Kunlong, Wu Zebin, Yang Huilin. Mechanism by which Hernandezine alleviates osteoporosis through macrophage polarization and osteoclast activation[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8860-8867.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
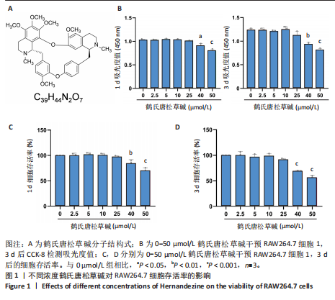
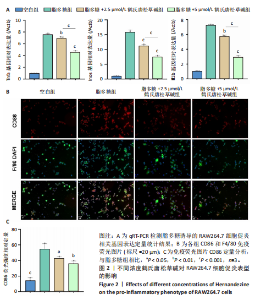
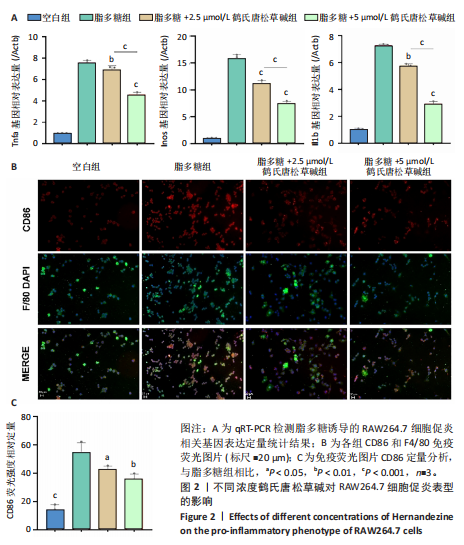
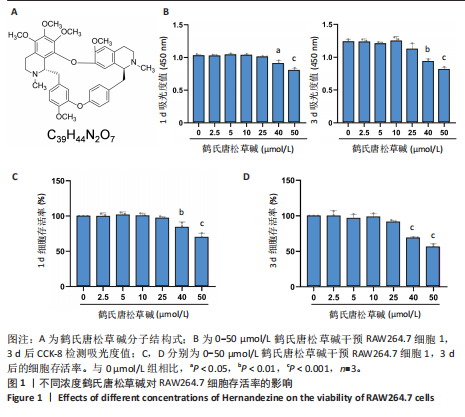
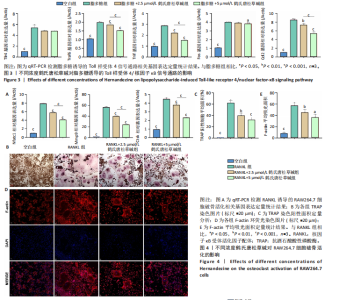
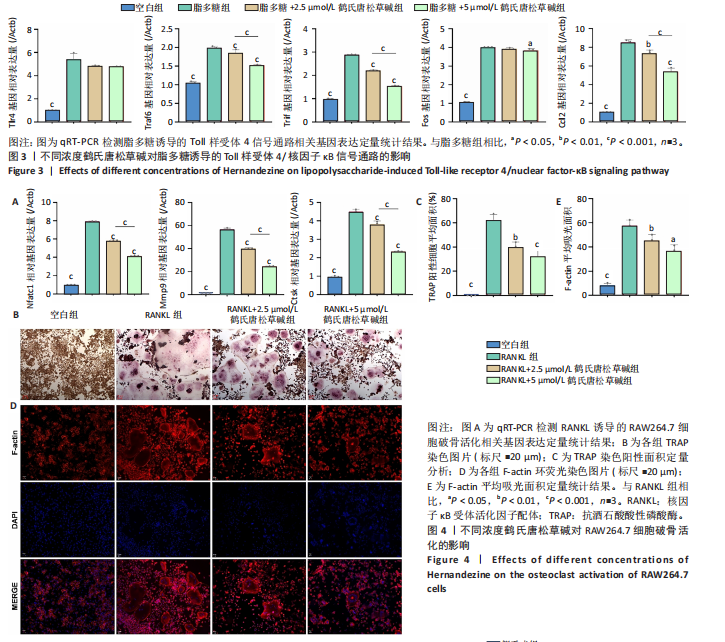
2.1 鹤氏唐松草碱对RAW264.7的细胞毒性 鹤氏唐松草碱是一种双苄基异喹啉生物碱,分子式为C39H44N2O7,分子质量为652.78 Da,图1A。将RAW264.7细胞分别暴露于0-50 μmol/L的鹤氏唐松草碱中进行1 d和3 d的干预,并使用CCK-8检测细胞毒性。结果显示,当暴露1 d在鹤氏唐松草碱25 μmol/L 及以下时,各组吸光度值均未有显著性差异;而当鹤氏唐松草碱浓度增加到40 μmol/L和50 μmol/L时,观察到吸光度值显著降低。此外,在3 d的暴露时间内也观察到相似的结果,见图1B。细胞存活率结果提示在鹤氏唐松草碱≤25 μmol/L时3 d内 RAW264.7细胞不会出现明显的生长抑制,见图1C;而40 μmol/L和50 μmol/L在3 d后对细胞的生长抑制率可达到31.04%和44.00%,见图1D。综合以上细胞毒性检测结果和已发表研究对鹤氏唐松草碱干预多种细胞实验结果[19],此次研究选择2.5 μmol/L和5 μmol/L为低剂量和高剂量的鹤氏唐松草碱干预细胞实验浓度。 2.2 鹤氏唐松草碱抑制脂多糖诱导的RAW264.7巨噬细胞极化 为明确鹤氏唐松草碱在RAW264.7中能否抑制因脂多糖诱导的巨噬细胞向促炎表型的M1极化。qRT-PCR检测在鹤氏唐松草碱干预经脂多糖诱导后的巨噬细胞促炎相关基因(肿瘤坏死因子α、Inos和Il1b)的转录水平变化[20]。结果发现,在脂多糖诱导下巨噬细胞促进炎症相关基因转录水平显著升高,而在鹤氏唐松草碱低浓度和高浓度干预下均发现肿瘤坏死因子α(Tnfα)、Inos和Il1b的转录水平显著降低,2.5 μmol/L和5 μmol/L鹤氏唐松草碱组间差异性提示鹤氏唐松草碱抑制炎症相关基因的表达呈现出浓度依赖性(图2A)。研究进一步通过对巨噬细胞促炎特异性蛋白CD86/F4/80进行免疫荧光染色,定量统计结果证明鹤氏唐松草碱2.5 μmol/L和5 μmol/L浓度干预下均可以降低CD86蛋白水平,抑制促炎相关蛋白的表达,见图2B,C。这些结果提示鹤氏唐松草碱抑制脂多糖诱导的RAW264.7促炎相关基因的表达和向促炎表型转换,且呈现出浓度依赖性。 2.3 鹤氏唐松草碱抑制脂多糖诱导的Toll样受体4/核因子κB信号通路激活 为进一步探究鹤氏唐松草碱抑制脂多糖诱导的促炎表型信号机制,对Toll样受体4/核因子κB通路相关基因进行qRT-PCR检测,因Toll样受体4/核因子κB信号通路相关基因变化发生在脂多糖干预早期[21],检测时间点选择为脂多糖干预2 h后。结果显示,脂多糖干预可以显著增加Tlr4,Traf6,Trif,Fos和Ccl2等Toll样受体4/核因子κB信号通路相关基因转录水平。而鹤氏唐松草碱的干预可以降低Traf6,Trif,Fos和Ccl2的表达,5 μmol/L时抑制效果更显著。针对Toll样受体4/核因子κB信号通路相关基因转录检测说明鹤氏唐松草碱可能通过下调Toll样受体4/核因子κB信号通路激活发挥抑制巨噬细胞促炎表型转变的作用,见图3。 2.4 鹤氏唐松草碱抑制核因子κB受体活化因子配体诱导的RAW264.7破骨细胞活化 为探究鹤氏唐松草碱对巨噬细胞在破骨细胞分化中的作用,对核因子κB受体活化因子配体诱导的破骨分化相关关键基因Nfatc1,Mmp9和Ctsk进行qRT-PCR验证,以Actb作为内参基因。结果显示,与空白组相比,核因子κB受体活化因子配体诱导显著增加了破骨细胞活化相关基因的表达,表明破骨细胞诱导分化成功。2.5 μmol/L鹤氏唐松草碱干预时可显著下调Nfatc1,Mmp9和Ctsk的转录水平,当提高鹤氏唐松草碱浓度至5 μmol/L时,抑制破骨相关基因的作用更为显著,同时2.5 μmol/L和5 μmol/L组间存在显著性差异,见图4A;提示鹤氏唐松草碱抑制破骨细胞活化作用具有浓度依赖性。进一步通过抗酒石酸酸性磷酸酶染色实验观察破骨细胞前体融合和成熟情况,鹤氏唐松草碱2.5 μmol/L和5 μmol/L干预均显著抑制了成熟破骨细胞形成,抗酒石酸酸性磷酸酶阳性细胞显著减少,但是低浓度、高浓度组间没有显著性差异,见图4B,C;这可能是由于PCR实验检测时间点为核因子κB受体活化因子配体诱导早期,并不能完全反映破骨细胞活化基因全时间段抑制情况[22]。通过对成熟破骨细胞肌动蛋白环荧光染色间接评估破骨细胞的骨吸收能力,结果提示2.5,5 μmol/L鹤氏唐松草碱干预后,肌动蛋白环面积减少、荧光强度减弱,其中5 μmol/L鹤氏唐松草碱的抑制作用更为明显,见图4D,E。"
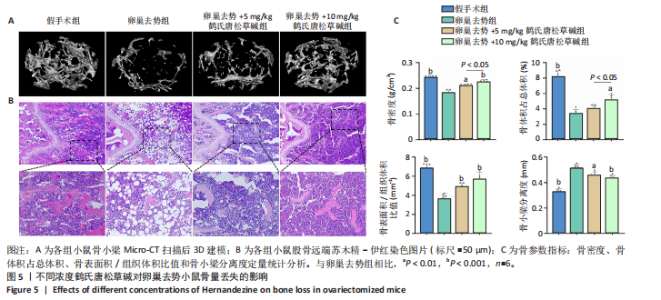
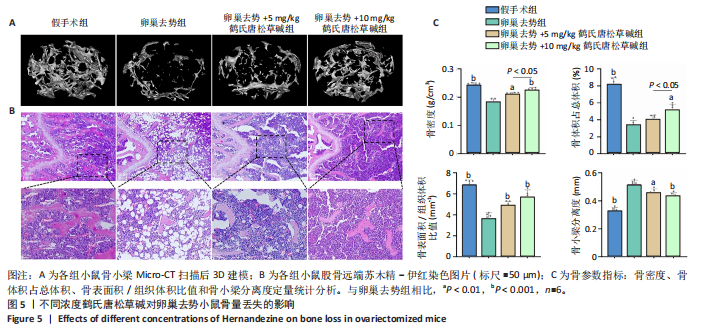
2.5 鹤氏唐松草碱减少雌激素缺乏引发的过量骨丢失 使用小鼠卵巢去势模拟骨质疏松状态下的过量骨丢失[23],应用鹤氏唐松草碱干预观察小鼠体内实验是否能得到与体外实验对应的结果。使用Micro-CT对股骨扫描进行二维和三维重建,以股骨远端生长板下方区域(100层×9 μm)作为感兴趣区域分析骨小梁结构破坏情况和骨参数分析。骨小梁区域三维重建结构显示,双侧卵巢去势成功诱导了小鼠因雌激素缺乏而导致的骨质疏松,与假手术组相比卵巢去势组小鼠骨小梁结构明显稀疏;而应用鹤氏唐松草碱干预明显观察到了骨小梁结构的部分恢复,见图5A。进一步通过骨组织切片和苏木精-伊红染色也观察到了骨小梁结构的增加,见图5B。围绕骨小梁区域为感兴趣区域(ROI)的骨参数分析发现,卵巢去势模型显著降低了感兴趣区域的骨密度、骨体积占比和骨表面积/组织体积比值,显著增加了骨小梁分离度,成功在体内模拟了骨质疏松症状态下的过量骨质丢失。通过应用不同浓度的鹤氏唐松草碱干预,发现鹤氏唐松草碱可部分恢复骨量,骨密度、骨体积占比和骨表面积/组织体积比值均有显著升高,而骨小梁分离度也显著减少,10 mg/kg剂量治疗效果更明显,其中骨密度较卵巢去势组小鼠提升22.7%,骨体积分数提升56.1%而骨小梁分离度降低15.3%(P < 0.001)。尽管仅在骨密度和骨体积占比中发现了5和10 mg/kg间的显著性差异,见图5C。以上结果证明,鹤氏唐松草碱在雌激素缺乏诱导的小鼠骨质疏松症模型中可以恢复骨量,减少骨质丢失。"
| [1] VILACA T, EASTELL R, SCHINI M. Osteoporosis in men. Lancet Diabetes Endocrinol. 2022;10(4): 273-283. [2] 中华医学会骨质疏松和骨矿盐疾病分会. 原发性骨质疏松症诊疗指南(2022)[J].中国全科医学,2023,26(14):1671-1691. [3] FUGGLE NR, BEAUDART C, BRUYÈRE O, et al. Evidence-Based Guideline for the management of osteoporosis in men. Nat Rev Rheumatol. 2024;20(4):241-251. [4] JEON HH, HUANG X, ROJAS CORTEZ L, et al. Inflammation and mechanical force-induced bone remodeling. Periodontol 2000. 2024:10.1111/prd.12619. [5] KALYANARAMAN H, PAL CHINA S, CASTEEL DE, et al. Crosstalk between androgen receptor and protein kinase G signaling in bone: implications for osteoporosis therapy. Trends Pharmacol Sci. 2025;46(3):279-294. [6] CHOTIYARNWONG P, MCCLOSKEY EV. Pathogenesis of glucocorticoid-induced osteoporosis and options for treatment. Nat Rev Endocrinol. 2020;16(8):437-447. [7] ADAMOPOULOS IE, CHOI Y, TAKAYANAGI H. Novel insights and recent progress in osteoimmunology. Trends Immunol. 2025;46(3):192-194. [8] FUGGLE N, LASLOP A, RIZZOLI R, et al. Treatment of Osteoporosis and Osteoarthritis in the Oldest Old. Drugs. 2025;85(3):343-360. [9] DING X, YANG J, WEI Y, et al. The Nexus Between Traditional Chinese Medicine and Immunoporosis: Implications in the Treatment and Management of Osteoporosis. Phytother Res. 2025;39(4):1826-1846. [10] LI P, LI X, WU Y, et al. A novel AMPK activator hernandezine inhibits LPS-induced TNFα production. Oncotarget. 2017;8(40):67218-67226. [11] SONG CF, HU YH, MANG ZG, et al. Hernandezine induces autophagic cell death in human pancreatic cancer cells via activation of the ROS/AMPK signaling pathway. Acta Pharmacol Sin. 2023;44(4):865-876. [12] GARCIA D, SHAW RJ. AMPK: Mechanisms of Cellular Energy Sensing and Restoration of Metabolic Balance. Mol Cell. 2017;66(6):789-800. [13] LIU JY, LIU JX, LI R, et al. AMPK, a hub for the microenvironmental regulation of bone homeostasis and diseases. J Cell Physiol. 2024;239(11):31393. [14] WANG Y, XU Y, ZHANG P, et al.Smiglaside A ameliorates LPS-induced acute lung injury by modulating macrophage polarization via AMPK-PPARγ pathway. Biochem Pharmacol. 2018;156:385-395. [15] TANAKA M, INOUE H, TAKAHASHI N, et al. AMPK negatively regulates RANKL-induced osteoclast differentiation by controlling oxidative stress. Free Radic Biol Med. 2023;205:107-115. [16] LV JL, REN YS, TAN YJ, et al. Hernandezine acts as a CDK4 suppressor inhibiting tumor growth by the CDK4/PKM2/NRF2 axis in colon cancer. Phytomedicine. 2024;131:155775. [17] BAI J, ZHANG S, CAO J, et al. Hernandezine, a natural herbal alkaloid, ameliorates type 2 diabetes by activating AMPK in two mouse models. Phytomedicine. 2022; 105:154366. [18] DEMPSTER DW, COMPSTON JE, DREZNER MK, et al. Standardized nomenclature, symbols, and units for bone histomorphometry: a 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res. 2013;28(1):2-17. [19] WANG X, LI X, XIA Y, et al. Hernandezine Regulates Proliferation and Autophagy-Induced Apoptosis in Melanoma Cells. J Nat Prod. 2022;85(5):1351-1362. [20] LOCATI M, CURTALE G, MANTOVANI A. Diversity, Mechanisms, and Significance of Macrophage Plasticity. Annu Rev Pathol. 2020;15:123-147. [21] ZHAO X, HE M-J, ZHAO M, et al. Crude Polygalae Radix after boiling with licorice decoction alleviates intestinal mucosal barrier injury of rats by regulating TLR4/NF-κB signaling pathway. J Ethnopharmacol. 2025;346:119661. [22] YANG R, YU W, LIN L, et al. NAT10 promotes osteoclastogenesis in inflammatory bone loss by catalyzing Fos mRNA ac4C modification and upregulating MAPK signaling pathway. J Adv Res. 2025;72:303-317. [23] DONG Q, FU H, LI W, et al. Nuclear farnesoid X receptor protects against bone loss by driving osteoblast differentiation through stabilizing RUNX2. Bone Res. 2025;13(1):20. [24] BOYLE WJ, SIMONET WS, LACEY DL. Osteoclast differentiation and activation. Nature. 2003;423(6937):337-342. [25] YU B, WANG CY. Osteoporosis and periodontal diseases - An update on their association and mechanistic links. Periodontol 2000. 2022;89(1):99-113. [26] BILLINGTON E, AGHAJAFARI F, SKULSKY E, et al. Bisphosphonates. BMJ. 2024; 386:e076898. [27] CHANG JW, TANG CH. The role of macrophage polarization in rheumatoid arthritis and osteoarthritis: Pathogenesis and therapeutic strategies. Int Immunopharmacol. 2024;142(Pt A):113056. [28] XIANG C, LI H, TANG W. Targeting CSF-1R represents an effective strategy in modulating inflammatory diseases. Pharmacol Res. 2023;187:106566. [29] BURGAN J, RAHMATI M, LEE M, et al. Innate immune response to bone fracture healing. Bone. 2025;190:117327. [30] WANG H, LI Y, LI H, et al. T cell related osteoimmunology in fracture healing: Potential targets for augmenting bone regeneration. J Orthop Translat. 2025;51: 82-93. [31] KODAMA J, KAITO T. Osteoclast Multinucleation: Review of Current Literature. Int J Mol Sci. 2020;21(16):5680. [32] NAKASHIMA T, HAYASHI M, FUKUNAGA T, et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat Med. 2011;17(10):1231-1234. [33] COMPSTON JE, MCCLUNG MR, LESLIE WD. Osteoporosis. Lancet (London, England). 2019;393(10169):364-376. [34] AMBROSI TH, MARECIC O, MCARDLE A, et al. Aged skeletal stem cells generate an inflammatory degenerative niche. Nature. 2021;597(7875):256-262. [35] HU K, SHANG Z, YANG X, et al. Macrophage Polarization and the Regulation of Bone Immunity in Bone Homeostasis. J Inflamm Res. 2023;16:3563-3580. [36] LOI F, CÓRDOVA LA, PAJARINEN J, et al. Inflammation, fracture and bone repair. Bone. 2016;86:119-130. [37] IBÁÑEZ L, ABOU-EZZI G, CIUCCI T, et al. Inflammatory Osteoclasts Prime TNFα-Producing CD4+ T Cells and Express CX3 CR1. J Bone Miner Res. 2016;31(10): 1899-908. [38] PANEZ-TORO I, HEYMANN D, GOUIN F, et al. Roles of inflammatory cell infiltrate in periprosthetic osteolysis. Front Immunol. 2023;14:1310262. [39] TAO H, LI W, ZHANG W, et al. Urolithin A suppresses RANKL-induced osteoclastogenesis and postmenopausal osteoporosis by, suppresses inflammation and downstream NF-κB activated pyroptosis pathways. Pharmacol Res. 2021;174:105967. [40] WINTER E, DEKKERS O, ANDREASEN C, et al. Expert consensus recommendations for the diagnosis and treatment of chronic non-bacterial osteitis (CNO) in adults.Ann Rheum Dis. 2025;84(2):169-187. [41] GAO Y, XU X, ZHANG X. Targeting different phenotypes of macrophages: A potential strategy for natural products to treat inflammatory bone and joint diseases. Phytomedicine. 2023;118:154952. [42] BOTT KN, FELDMAN E, DE SOUZA RJ, et al. Lipopolysaccharide-Induced Bone Loss in Rodent Models: A Systematic Review and Meta-Analysis. J Bone Miner Res. 2023;38(1):198-213. [43] HSIAO SH, LU YJ, YANG CC, et al. Hernandezine, a Bisbenzylisoquinoline Alkaloid with Selective Inhibitory Activity against Multidrug-Resistance-Linked ATP-Binding Cassette Drug Transporter ABCB1. J Nat Prod. 2016;79(8):2135-2142. [44] LOW A M, BERDIK M, SORMAZ L, et al. Plant alkaloids, tetrandrine and hernandezine, inhibit calcium-depletion stimulated calcium entry in human and bovine endothelial cells. Life Sci. 1996;58(25):2327-2335. [45] TONG X, GANTA RR, LIU Z. AMP-activated protein kinase (AMPK) regulates autophagy, inflammation and immunity and contributes to osteoclast differentiation and functionabs. Biol Cell. 2020;112(9):251-264. [46] JIANG L, HE H, TANG Y, et al. Activation of BK channels prevents diabetes-induced osteopenia by regulating mitochondrial Ca2+ and SLC25A5/ANT2-PINK1-PRKN-mediated mitophagy. Autophagy. 2024;20(11):2388-2404. [47] AHSAN M, GARNEAU L, AGUER C. The bidirectional relationship between AMPK pathway activation and myokine secretion in skeletal muscle: How it affects energy metabolism]. Front Physiol. 2022;13:1040809. [48] HSU SH, CHEN LR, CHEN KH. Primary Osteoporosis Induced by Androgen and Estrogen Deficiency: The Molecular and Cellular Perspective on Pathophysiological Mechanisms and Treatments. Int J Mol Sci. 2024;25(22):12139. [49] MA N, MA D, LIU X, et al. Bisphenol P exposure in C57BL/6 mice caused gut microbiota dysbiosis and induced intestinal barrier disruption via LPS/TLR4/NF-κB signaling pathway. Environ Int. 2023;175:107949. [50] WU X, QIAO S, WANG W, et al. Melatonin prevents peri‑implantitis via suppression of TLR4/NF-κB. Acta Biomater. 2021;134:325-336. [51] LI R, FENG J, LI L, et al. Recombinant fibroblast growth factor 4 ameliorates axonal regeneration and functional recovery in acute spinal cord injury through altering microglia/macrophage phenotype. Int Immunopharmacol. 2024;134:112188. |
| [1] | Cai Ziming, Yu Qinghe, Ma Pengfei, Zhang Xin, Zhou Longqian, Zhang Chongyang, Lin Wenping. Heme oxygenase-1 alleviates lipopolysaccharide-induced inflammatory response in nucleus pulposus mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1624-1631. |
| [2] | He Jiale, Huang Xi, Dong Hongfei, Chen Lang, Zhong Fangyu, Li Xianhui. Acellular dermal matrix combined with adipose-derived stem cell exosomes promotes burn wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1699-1710. |
| [3] | Xia Linfeng, Wang Lu, Long Qianfa, Tang Rongwu, Luo Haodong, Tang Yi, Zhong Jun, Liu Yang. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate blood-brain barrier damage in mice with septic encephalopathy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1711-1719. |
| [4] | Cui Lianxu, Li Haomin, Xu Junrong, Tan Baodong, Lu Dahong, Peng Siwei, Wang Jinhui. Effect of umbilical cord mesenchymal stem cell conditioned medium on tissue repair after traumatic craniocerebral injury in miniature pigs [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1730-1735. |
| [5] | Cao Yong, Teng Hongliang, Tai Pengfei, Li Junda, Zhu Tengqi, Li Zhaojin. Interactions between cytokines and satellite cells in muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1808-1817. |
| [6] | Pan Hongfei, Zhuang Zhenbing, Xu Baiyun, Yang Zhangyang, Lin Kairui, Zhan Bingqing, Lan Jinghan, Gao Heng, Zhang Nanbo, Lin Jiayu. Inhibitory effects of different concentrations of auranofin on M1 macrophage function and its therapeutic potential in diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1390-1397. |
| [7] | Hou Chaowen, Li Zhaojin, Kong Jianda, Zhang Shuli. Main physiological changes in skeletal muscle aging and the multimechanism regulatory role of exercise [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1464-1475. |
| [8] | You Huijuan, Wu Shuzhen, Rong Rong, Chen Liyuan, Zhao Yuqing, Wang Qinglu, Ou Xiaowei, Yang Fengying. Macrophage autophagy in lung diseases: two-sided effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1516-1526. |
| [9] | Wen Guangwei, Zhen Yinghao, Zheng Taikeng, Zhou Shuyi, Mo Guoye, Zhou Tengpeng, Li Haishan, Lai Yiyi. Effects and mechanisms of isoginkgetin on osteoclastogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1348-1358. |
| [10] | Zhang Di, Zhao Jun, Ma Guangyue, Sun Hui, Jiang Rong. Mechanism of depression-like behavior in chronic social defeat stress mice based on high-throughput sequencing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1139-1146. |
| [11] | Li Haojing, Wang Xin, Song Chenglin, Zhang Shengnan, Chen Yunxin. Therapeutic efficacy of extracorporeal shock wave therapy in the upper trapezius muscle area combined with exercise control training in patients with chronic non-specific neck pain [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1162-1170. |
| [12] | Liu Yu, Lei Senlin, Zhou Jintao, Liu Hui, Li Xianhui. Mechanisms by which aerobic and resistance exercises improve obesity-related cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1171-1183. |
| [13] | Yu Huifen, Mo Licun, Cheng Leping. The position and role of 5-hydroxytryptamine in the repair of tissue injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1196-1206. |
| [14] | Fu Zhenyi, Li Junhao, Zhang Yating, He Yunkai, Liu Junyu, Wei Yunhao, Liu Jiaxin. Schwann cells promote peripheral nerve regeneration: retrospect and prospect [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1236-1246. |
| [15] | Wen Xiaolong, Weng Xiquan, Feng Yao, Cao Wenyan, Liu Yuqian, Wang Haitao. Effects of inflammation on serum hepcidin and iron metabolism related parameters in patients with type 2 diabetes mellitus: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1294-1301. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||