Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (34): 8852-8859.doi: 10.12307/2026.889
Previous Articles Next Articles
Role of myeloid-derived suppressor cells in osteoclast differentiation in primary osteoporosis
Cheng Xinyi, Chen Yida, Wang Yi, Liu Daihui, Zheng Yi, Shi Qin
- Department of Orthopedics, First Affiliated Hospital of Soochow University, Institute of Orthopedics of Soochow University, Suzhou 215006, Jiangsu Province, China
-
Received:2025-10-18Revised:2026-02-13Online:2026-12-08Published:2026-04-11 -
Contact:Shi Qin, PhD, Professor, Department of Orthopedics, First Affiliated Hospital of Soochow University, Institute of Orthopedics of Soochow University, Suzhou 215006, Jiangsu Province, China -
About author:Cheng Xinyi, MS, Department of Orthopedics, First Affiliated Hospital of Soochow University, Institute of Orthopedics of Soochow University, Suzhou 215006, Jiangsu Province, China -
Supported by:National Natural Science Foundation of China (General Program), No. 82172485 (to SQ)
CLC Number:
Cite this article
Cheng Xinyi, Chen Yida, Wang Yi, Liu Daihui, Zheng Yi, Shi Qin. Role of myeloid-derived suppressor cells in osteoclast differentiation in primary osteoporosis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8852-8859.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
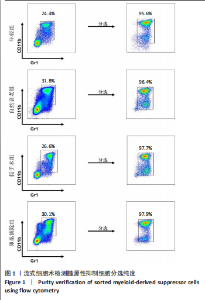
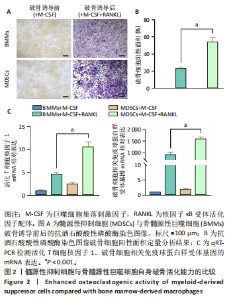
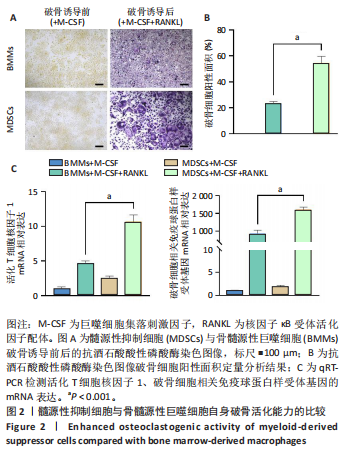
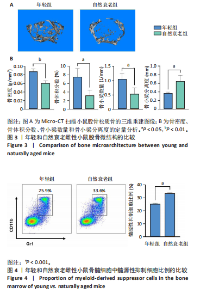
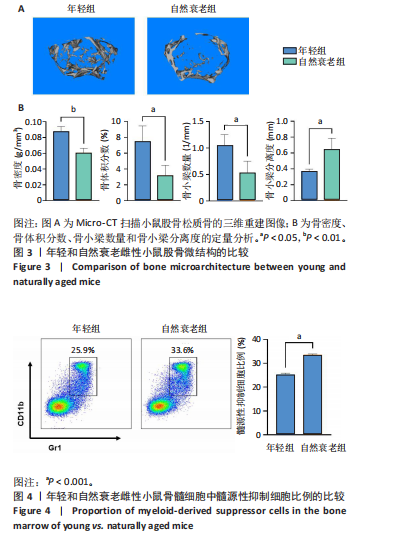
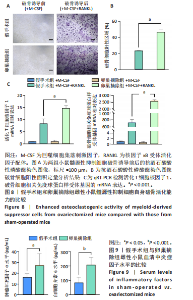
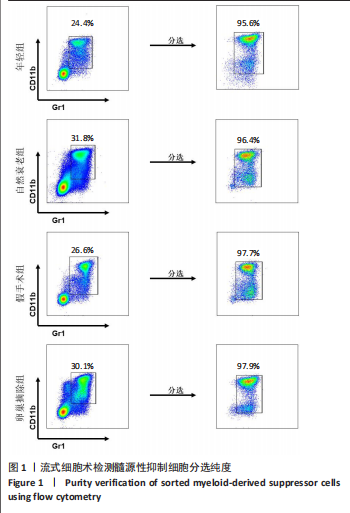
2.1 髓源性抑制细胞的分选纯度鉴定结果 流式细胞术检测结果显示,未分选前,年轻组、自然衰老组、假手术组、卵巢摘除组小鼠骨髓细胞中CD11b+Gr1+髓源性抑制细胞比例分别为24.4%,31.8%,26.6%,30.1%;经磁珠分选后,骨髓细胞中髓源性抑制细胞比例分别为95.6%,96.4%,97.7%,97.9%,见图1,说明分选后的髓源性抑制细胞纯度较高,可以进行后续实验。 2.2 髓源性抑制细胞与骨髓源性巨噬细胞自身破骨活化能力的比较 抗酒石酸酸性磷酸酶染色显示,破骨诱导分化后,髓源性抑制细胞组中多核破骨细胞(≥3个核)数量显著多于骨髓源性巨噬细胞组,并且髓源性抑制细胞组细胞体积更大、抗酒石酸酸性磷酸酶染色阳性区域比例更高,见图2A,B。qRT-PCR检测显示,破骨诱导分化后,髓源性抑制细胞组T细胞核因子1、破骨细胞相关免疫球蛋白样受体基因的mRNA表达高于骨髓源性巨噬细胞组(P < 0.001),见图2C。结果表明,相较于骨髓源性巨噬细胞,髓源性抑制细胞具有更强的活化为成熟破骨细胞的能力。 2.3 年轻和自然衰老雌性小鼠骨微结构的比较 年轻组与自然衰老组小鼠股骨远端骨微结构,如图3A所示,可见自然衰老组小鼠骨质流失严重。定量分析结果显示,与年轻组比较,自然衰老组小鼠骨密度、骨体积分数与骨小梁数量均降低(P < 0.05,P < 0.01),骨小梁分离度增加(P < 0.05),见图3B。 2.4 年轻和自然衰老雌性小鼠骨髓细胞中髓源性抑制细胞比例的比较 流式细胞术检测结果显示,自然衰老组小鼠骨髓细胞中CD11b+Gr1+髓源性抑制细胞比例(33.6±0.3)%高于年轻组(25.4±0.5)%(P < 0.001),见图4。"
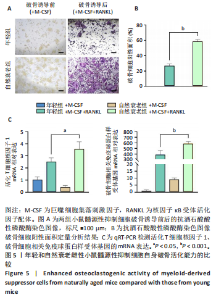
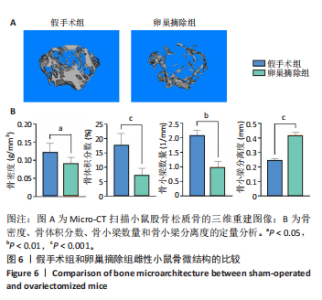
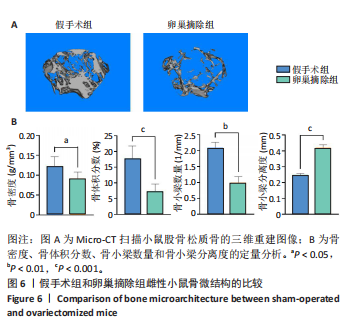
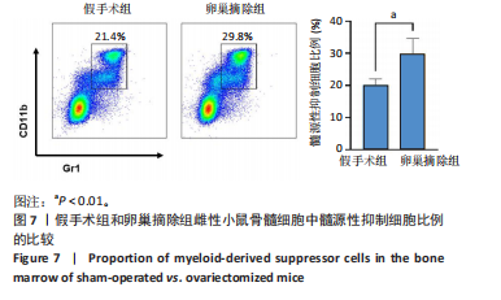
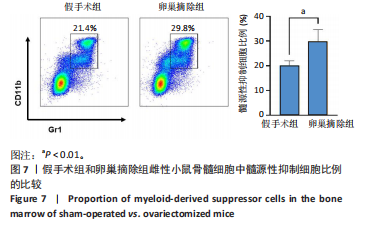
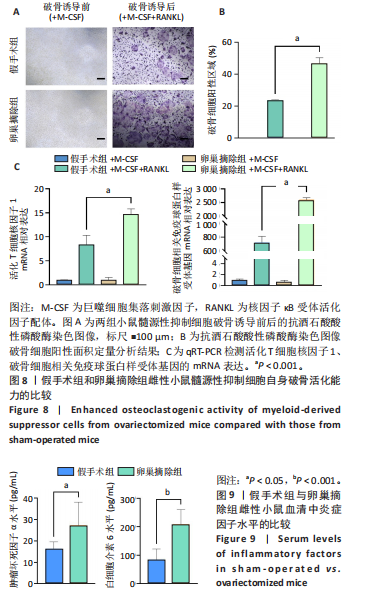
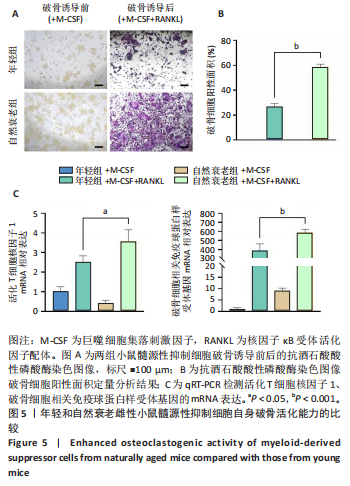
2.5 年轻和自然衰老雌性小鼠髓源性抑制细胞自身破骨活化能力的比较 抗酒石酸酸性磷酸酶染色显示,破骨诱导分化后,自然衰老组髓源性抑制细胞中多核破骨细胞(≥3个核)数量高于年轻组,并且自然衰老组髓源性抑制细胞细胞体积更大、抗酒石酸酸性磷酸酶染色阳性区域比例更高,见图5A,B。qRT-PCR检测显示,破骨诱导分化后,自然衰老组细胞中T细胞核因子1、破骨细胞相关免疫球蛋白样受体基因的mRNA表达高于年轻组(P < 0.05,P < 0.001),见图5C。结果表明,与年轻组髓源性抑制细胞相比,自然衰老组髓源性抑制细胞具有更强的活化为成熟破骨细胞的能力。 2.6 假手术组和卵巢摘除组小鼠骨微结构的比较 假手术组和卵巢摘除组小鼠股骨远端骨微结构,如图6A所示,可见卵巢摘除组小鼠骨质流失严重。定量分析结果显示,与假手术组比较,卵巢摘除组小鼠骨密度、骨体积分数与骨小梁数量均减少(P < 0.05,P < 0.001,P < 0.01),骨小梁分离度增加(P < 0.001),见图6B。 2.7 假手术组和卵巢摘除组小鼠骨髓细胞中髓源性抑制细胞比例的比较 流式细胞术检测结果显示,卵巢摘除组小鼠骨髓细胞中CD11b+Gr1+髓源性抑制细胞比例(29.8±4.7)%高于假手术组(20.1±1.9)%(P < 0.01),如图7所示。"
| [1] FANG H, DENG Z, LIU J, et al. The Mechanism of Bone Remodeling After Bone Aging. Clin Interv Aging. 2022;17:405-415. [2] GENANT HK, COOPER C, POOR G, et al. Interim report and recommendations of the World Health Organization Task-Force for Osteoporosis. Osteoporos Int. 1999;10(4):259-264. [3] PERIS P, MARTÍNEZ-FERRER A, MONEGAL A, et al. Aetiology and clinical characteristics of Male osteoporosis. Have they changed in the last few years? Clin Exp Rheumatol. 2008;26(4):582-588. [4] SØE K, DELAISSE JM, BORGGAARD XG. Osteoclast formation at the bone marrow/bone surface interface: Importance of structural elements, matrix, and intercellular communication. Semin Cell Dev Biol. 2021;112:8-15. [5] SONG S, GUO Y, YANG Y, et al. Advances in pathogenesis and therapeutic strategies for osteoporosis. Pharmacol Ther. 2022;237:108168. [6] UMUR E, BULUT SB, YIĞIT P, et al. Exploring the Role of Hormones and Cytokines in Osteoporosis Development. Biomedicines. 2024;12(8):1830. [7] PAROLINI C. Pathophysiology of bone remodelling cycle: role of immune system and lipids. Biochem Pharmacol. 2025;235:116844. [8] ZHAO Z, DU Y, YAN K, et al. Exercise and osteoimmunology in bone remodeling. FASEB J Off Publ Fed Am Soc Exp Biol. 2024;38(7):e23554. [9] GEUSENS P, LEMS WF. Osteoimmunology and osteoporosis. Arthritis Res Ther. 2011;13(5):242. [10] ZHANG W, GAO R, RONG X, et al. Immunoporosis: role of immune system in the pathophysiology of different types of osteoporosis. Front Endocrinol. 2022;13:965258. [11] PARK Y, KWOK SK. Recent Advances in Cell Therapeutics for Systemic Autoimmune Diseases. Immune Netw. 2022;22(1):e10. [12] FINN OJ. Immuno-oncology: understanding the function and dysfunction of the immune system in cancer. Ann Oncol. 2012;23 Suppl 8(Suppl 8):viii6-9. [13] ZHAO F, GONG W, SONG J, et al. The paradoxical role of MDSCs in inflammatory bowel diseases: From bench to bedside. Front Immunol. 2022;13:1021634. [14] ZOU L, JIANG W, WANG Z, et al. Effect of advanced oxidation protein products (AOPPs) and aging on the osteoclast differentiation of myeloid-derived suppressor cells (MDSCs) and its preliminary mechanism. Biochem Biophys Res Commun. 2022;636(Pt 2):87-96. [15] LI Z, ZHAO Y, CHEN Z, et al. Age-related expansion and increased osteoclastogenic potential of myeloid-derived suppressor cells. Mol Immunol. 2021;137:187-200. [16] ABDELMAGID SM, BARBE MF, SAFADI FF. Role of inflammation in the aging bones. Life Sci. 2015;123:25-34. [17] LI Z, XIA Q, HE Y, et al. MDSCs in bone metastasis: mechanisms and therapeutic potential. Cancer Lett. 2024;592:216906. [18] MONTERAN L, ERSHAID N, SCHARFF Y, et al. Combining TIGIT Blockade with MDSC Inhibition Hinders Breast Cancer Bone Metastasis by Activating Antitumor Immunity. Cancer Discov. 2024;14(7):1252-1275. [19] SAWANT A, DESHANE J, JULES J, et al. Myeloid-derived suppressor cells function as novel osteoclast progenitors enhancing bone loss in breast cancer. Cancer Res. 2013;73(2):672-682. [20] REN F, ZHENG S, LUO H, et al. Fibroblast derived C3 promotes the progression of experimental periodontitis through macrophage M1 polarization and osteoclast differentiation. Int J Oral Sci. 2025;17(1):30. [21] HU L, XIE X, XUE H, et al. MiR-1224-5p modulates osteogenesis by coordinating osteoblast/osteoclast differentiation via the Rap1 signaling target ADCY2. Exp Mol Med. 2022;54(7):961-972. [22] FUJIKAWA Y, SENDO S, DEL PERAL FANJUL A, et al. Myeloid-derived suppressor cell-derived osteoclasts with bone resorption capacity in the joints of arthritic SKG mice. Front Immunol. 2024;15:1168323. [23] SENDO S, SAEGUSA J, MORINOBU A. Myeloid-derived suppressor cells in non-neoplastic inflamed organs. Inflamm Regen. 2018;38:19. [24] KWACK KH, MAGLARAS V, THIYAGARAJAN R, et al. Myeloid-derived suppressor cells in obesity-associated periodontal disease: a conceptual model . Periodontol 2000. 2021;87(1):268-275. [25] FRANCESCHI C, CAMPISI J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J Gerontol A Biol Sci Med Sci. 2014;69 Suppl 1:S4-9. [26] FRANCESCHI C, GARAGNANI P, PARINI P, et al. Inflammaging: a new immune–metabolic viewpoint for age-related diseases. Nat Rev Endocrinol. 2018;14(10): 576-590. [27] YU K, YU C, JIAO L, et al. The Function and Therapeutic Implications of TNF Signaling in MDSCs. Biomolecules. 2022;12(11):1627. [28] WEBER R, GROTH C, LASSER S, et al. IL-6 as a major regulator of MDSC activity and possible target for cancer immunotherapy. Cell Immunol. 2021;359:104254. [29] NEO SY, TONG L, CHONG J, et al. Tumor-associated NK cells drive MDSC-mediated tumor immune tolerance through the IL-6/STAT3 axis. Sci Transl Med. 2024;16(747):eadi2952. [30] GABRILOVICH DI, NAGARAJ S. Myeloid-derived-suppressor cells as regulators of the immune system. Nat Rev Immunol. 2009;9(3):162-174. [31] TAKAGI R, SAKAMOTO E, KIDO JI, et al. S100A9 Increases IL-6 and RANKL Expressions through MAPKs and STAT3 Signaling Pathways in Osteocyte-Like Cells. Biomed Res Int. 2020;2020:7149408. [32] ZHANG W, FANG X, GAO C, et al. MDSCs in sepsis-induced immunosuppression and its potential therapeutic targets. Cytokine Growth Factor Rev. 2023;69:90-103. [33] YAN L, LIANG M, YANG T, et al. The immunoregulatory role of myeloid-derived suppressor cells in the pathogenesis of rheumatoid arthritis. Front Immunol. 2020;11:568362. [34] REN Y, BÄCKER H, MÜLLER M, et al. The role of myeloid derived suppressor cells in musculoskeletal disorders. Front Immunol. 2023;14:1139683. [35] HEGDE S, LEADER AM, MERAD M. MDSC: Markers, development, states, and unaddressed complexity. Immunity. 2021;54(5):875-884. [36] ZHUANG J, ZHANG J, LWIN ST, et al. Osteoclasts in multiple myeloma are derived from gr-1+CD11b+myeloid-derived suppressor cells. PLoS One. 2012;7(11):e48871. |
| [1] | Zhang Haiwen, Zhang Xian, Xu Taichuan, Li Chao. Bibliometric and visual analysis of the research status and trends of senescence in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1580-1591. |
| [2] | Yang Jing, Wang Houmei, Wang Yi, Song Min, Ren Jie, Dai Lujun, Xiao Ziwen. Constructing a rat animal model of pelvic organ prolapse: a comparison of three modeling methods [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 864-872. |
| [3] | Xia Wenyu, Zhang Wei, Li Wenhao, Jiang Kunlong, Wu Zebin, Yang Huilin. Mechanism by which Hernandezine alleviates osteoporosis through macrophage polarization and osteoclast activation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8860-8867. |
| [4] | Li Yiwei, Luo Zongming, Rong Yifa, Jiang Kai, Zhang Jiahao, Lu Bowen, Li Gang. Druggable gene and single cell analyses reveal potential therapeutic targets for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6654-6660. |
| [5] | Hu Jie, He Hui, Ma Fengyu, Shen Xiaotian, Yuan Zhangqin, Liang Ting, Han Fengxuan. 10-Hydroxy-2-decenoic acid facilitates osteogenic differentiation via the enhancement of autophagy and antioxidant capacity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6433-6445. |
| [6] | Wang Yan, Lyu Hao, Hu Zhimu, Zhou Yao, Liu Qiang, Yang Yuxiang, Yi Hairu, Wang Jiuxiang, Jiang Ting. Intervention with Compound Kidney-Invigorating Granules in a mouse model of osteoporosis: role of the TRIB3/beta-catenin axis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6142-6149. |
| [7] | Du Xingbin, Jiang Fugao, Kong Jianda. Traditional Chinese sports in the treatment of osteoporosis: potential biological mechanisms and clinical application progress [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5943-5953. |
| [8] | Wu Lingjie, Zheng Kaiyuan, Wang Guangrong, Yin Chong . Strategies for the application of miRNA-targeted therapy in the treatment of osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5792-5803. |
| [9] | Wang Siwei, Yao Xiaosheng, Qi Xiaonan, Wang Yu, Cui Haijian, Zhao Jiaxuan. Matrix metalloproteinase 9 mediates mitophagy to regulate osteogenesis and myogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4557-4567. |
| [10] | Fu Jingyue, Zhou Qinfeng, Li Muzhe, Ma Yong, Pan Yalan, Sun Jie, Huang Xiangyang, Guo Yang. Preparation and evaluation of an animal model of osteoporosis and osteoarthritis comorbidity in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4299-4308. |
| [11] | Wang Wentao, Hou Zhenyang, Wang Yijun, Xu Yaozeng. Apelin-13 alleviates systemic inflammatory bone loss by inhibiting macrophage M1 polarization [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1548-1555. |
| [12] | Yang Peng, Zhang Wei, Li Wenming, Li Wenhao, Wu Zebin, Zhou Jun, Geng Dechun. Linagliptin alleviates wear particle-induced inflammatory osteolysis by regulating macrophage polarization and osteoclast formation [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(12): 2421-2428. |
| [13] | Yang Cheng, Li Yusheng, Jiao Hongzhuo, Shang Man, Liu Qi, Li Linzhen, Fan Fangyang, Zhang Chenglong, Zhang Xiaoyu, Zhang Juntao. Establishment and validation of the Sprague-Dawley rat model of osteoarthritis with kidney deficiency and blood stagnation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(27): 4273-4280. |
| [14] | Zhang Shudong, Huang Yilin, Yao Qi. Punicalagin treats postmenopausal osteoporosis by promoting osteogenesis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4101-4105. |
| [15] | Yang Shanshan, Ouyang Renjun, Tian Jia, Linghu Min, Wang Zhen, Yang Xiaohong. Detection of immune-related cytokines of bone marrow mesenchymal stem cells in postmenopausal osteoporosis mice by antibody chip and analysis of key differential genes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(25): 3947-3954. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||