Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (25): 6433-6445.doi: 10.12307/2026.455
10-Hydroxy-2-decenoic acid facilitates osteogenic differentiation via the enhancement of autophagy and antioxidant capacity
Hu Jie1, 2, He Hui1, 2, Ma Fengyu1, 2, Shen Xiaotian1, 2, Yuan Zhangqin1, 2, Liang Ting1, 2, Han Fengxuan1, 2
- 1Department of Orthopedic Surgery, First Affiliated Hospital, Soochow University, Suzhou 215000, Jiangsu Province, China; 2Orthopedic Institute, Suzhou Medical College, Soochow University, Suzhou 215000, Jiangsu Province, China
-
Received:2025-09-21Revised:2026-02-05Online:2026-09-08Published:2026-04-17 -
Contact:Han Fengxuan, PhD, Professor, Department of Orthopedic Surgery, First Affiliated Hospital, Soochow University, Suzhou 215000, Jiangsu Province, China; Orthopedic Institute, Suzhou Medical College, Soochow University, Suzhou 215000, Jiangsu Province, China -
About author:1Department of Orthopedic Surgery, First Affiliated Hospital, Soochow University, Suzhou 215000, Jiangsu Province, China; 2Orthopedic Institute, Suzhou Medical College, Soochow University, Suzhou 215000, Jiangsu Province, China Hu Jie, MS, Department of Orthopedic Surgery, First Affiliated Hospital, Soochow University, Suzhou 215000, Jiangsu Province, China; Orthopedic Institute, Suzhou Medical College, Soochow University, Suzhou 215000, Jiangsu Province, China He Hui, MS, Department of Orthopedic Surgery, First Affiliated Hospital, Soochow University, Suzhou 215000, Jiangsu Province, China; Orthopedic Institute, Suzhou Medical College, Soochow University, Suzhou 215000, Jiangsu Province, China Hu Jie and He Hui contributed equally to this work. -
Supported by:Jiangsu Provincial Natural Science Foundation for Basic Research - Outstanding Young Scientist Fund Project, No. BK20240020 (to HFX)
CLC Number:
Cite this article
Hu Jie, He Hui, Ma Fengyu, Shen Xiaotian, Yuan Zhangqin, Liang Ting, Han Fengxuan. 10-Hydroxy-2-decenoic acid facilitates osteogenic differentiation via the enhancement of autophagy and antioxidant capacity[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6433-6445.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
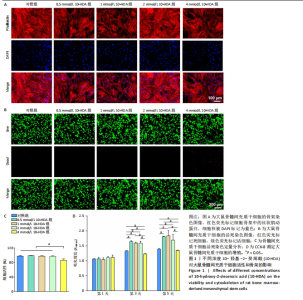
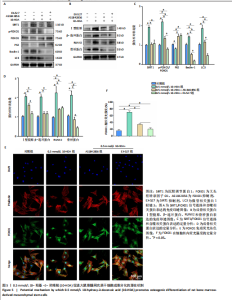
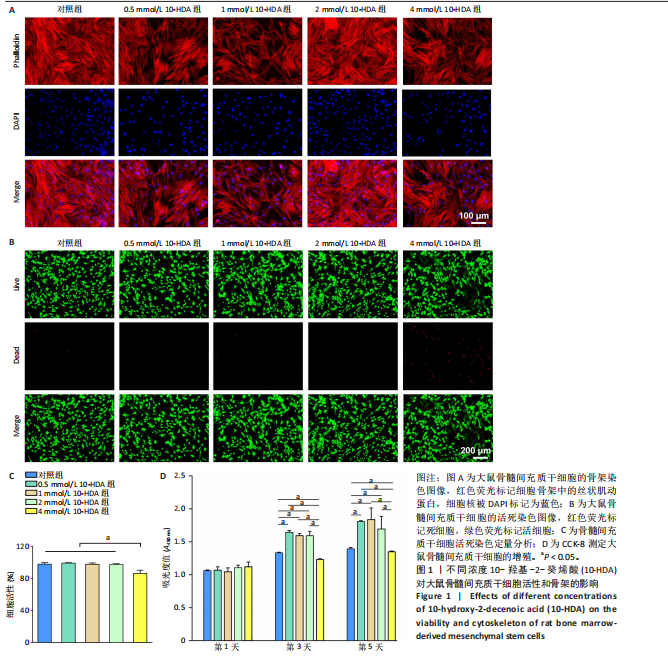
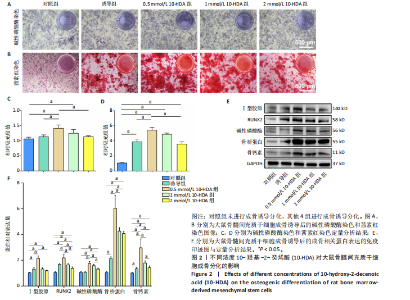
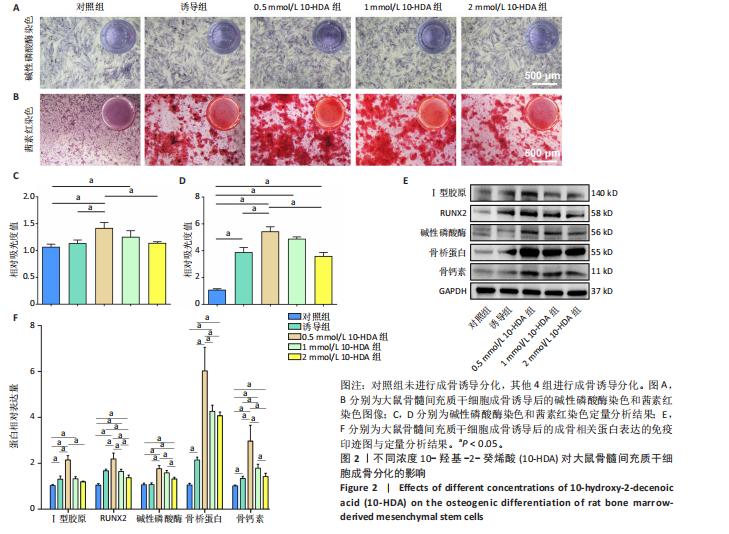
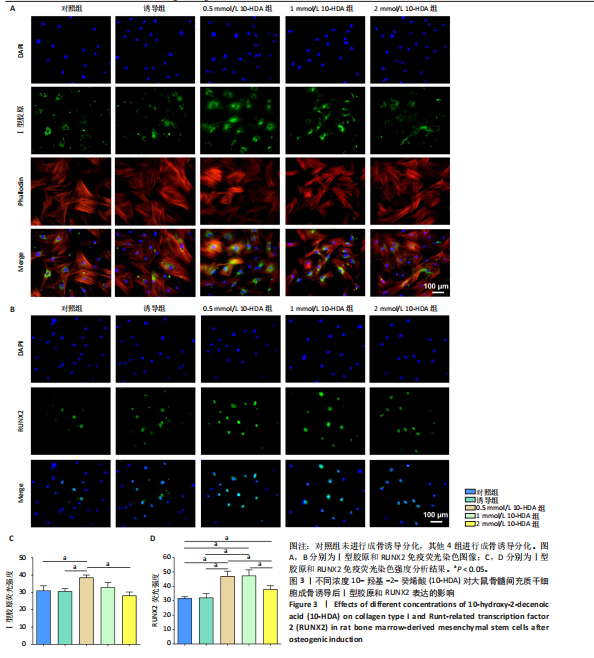
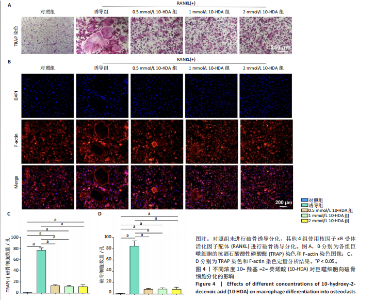
2.1 不同浓度10-HDA对大鼠骨髓间充质干细胞活性和骨架的影响 如图1A所示,低浓度(0.5,1,2 mmol/L)10-HDA组的大鼠骨髓间充质干细胞铺展良好,F-肌动蛋白结构完整且分布有序;在4 mmol/L 10-HDA处理后,部分细胞明显出现收缩,骨架结构紊乱,提示该浓度可能具有一定的细胞毒性作用。活死染色结果显示,4 mmol/L 10-HDA组红色荧光死细胞较多,0.5,1,2 mmol/L 10-HDA组红色荧光死细胞较少,如图1B,C所示,表明0.5-2 mmol/L 10-HDA具有良好的细胞相容性。CCK-8检测结果显示,培养第3,5天,0.5,1,2 mmol/L 10-HDA可促进大鼠骨髓间充质干细胞增殖,而4 mmol/L 10-HDA抑制大鼠骨髓间充质干细胞的增殖,见图1D。 综上所述,0.5,1,2 mmol/L 10-HDA具有良好的细胞相容性,可促进大鼠骨髓间充质干细胞的增殖。10-HDA作为一种不饱和脂肪酸衍生物,在高浓度下可能插入细胞膜或线粒体膜中,影响膜的流动性与完整性、破坏细胞器功能,尤其是线粒体膜电位,从而激活细胞凋亡通路[32],因此,后续实验选择0.5,1,2 mmol/L作为10-HDA适宜处理浓度,以兼顾细胞活性与实验安全性。 2.2 不同浓度10-HDA对大鼠骨髓间充质干细胞成骨分化的影响 为评估10-HDA的促成骨潜能,采用碱性磷酸酶染色和茜素红染色分别检测它对成骨早期活性和晚期基质矿化的影响。如图2A,B所示,对照组表现出较低的碱性磷酸酶活性和较弱的矿化结节形成,提示在未诱导条件下未启动成骨分化;相比之下,诱导组在成骨诱导培养基处理后碱性磷酸酶染色和钙沉积增强,证实成骨过程被成功激活。0.5,1 mmol/L 10-HDA组碱性磷酸酶活性高于诱导组(P < 0.05),2 mmol/L 10-HDA组的碱性磷酸酶活性与诱导组相似,见图2C,表明低浓度10-HDA可促进成早期阶段的骨分化。与碱性磷酸酶染色结果一致,茜素红染色显示,0.5,1 mmol/L 10-HDA组钙化结节数量多于诱导组(P < 0.05),如图2D所示,提示低浓度10-HDA对成骨晚期分化及钙沉积具有促进作用。 综上所述,10-HDA在适当浓度下能显著促进成骨分化,增强成骨早期活性和晚期矿化能力。 2.3 不同浓度10-HDA对大鼠骨髓间充质干细胞成骨相关蛋白表达的影响 为进一步探究10-HDA的促成骨作用,采用Western blot检测关键成骨标志蛋白表达,其中,Ⅰ型胶原是骨组织细胞外基质的主要成分,主要参与成骨早期的基质合成;RUNX2是成骨细胞谱系分化所必需的关键转录因子;碱性磷酸酶是成骨活性的早期标志物,而骨桥蛋白和骨钙素则在基质成熟和矿化过程中发挥重要作用。如图2E,F所示,0.5 mmol/L 10-HDA处理显著上调了成骨标志蛋白表达,然而,随着10-HDA浓度升高(1,2 mmol/L),这一促进作用逐渐减弱。 免疫荧光染色显示,与诱导组相比,0.5 mmol/L 10-HDA显著增强了Ⅰ型胶原和RUNX2蛋白表达,见图3,表明基质合成增加、成骨分化能力增强,进一步证实0.5 mmol/L 10-HDA可通过上调关键调控蛋白和结构蛋白促进早期成骨分化。 2.4 不同浓度10-HDA对破骨细胞生成的影响 抗酒石酸酸性磷酸酶是成熟破骨细胞表达的标志性酶,而F-actin环是破骨细胞形成骨吸收腔的关键结构。抗酒石酸酸性磷酸酶与细胞骨架染色显示,0.5 mmol/L 10-HDA处理后,抗酒石酸酸性磷酸酶阳性的多核细胞数量显著减少,并且F-actin环结构出现破坏或缺失,但当10-HDA浓度增加至1,2 mmol/L 时该抑制作用并未进一步增强,如图4所示,提示10-HDA的抗破骨分化作用中可能存在一个浓度阈值。 综上所述,10-HDA不仅能促进成骨细胞介导的骨形成,还能有效抑制破骨细胞生成,从而有助于构建有利于骨再生的微环境。"
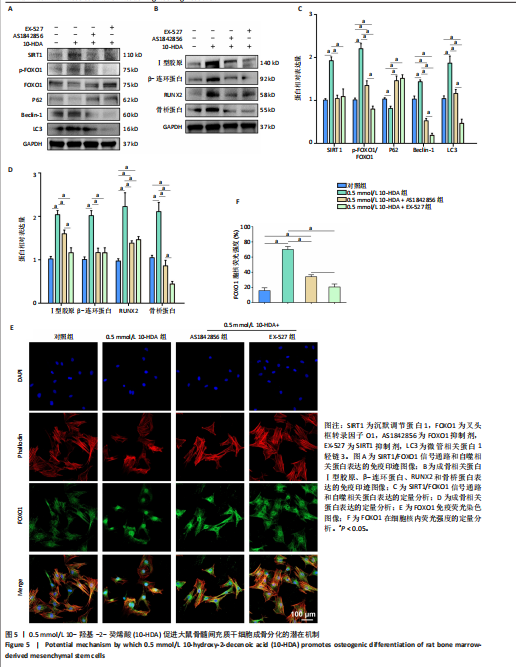
2.5 10-HDA促进成骨分化的潜在机制 自噬作为维持细胞内稳态的重要机制,在骨形成过程中也发挥着关键作用。已有研究表明,在成骨细胞中,自噬有助于清除受损的细胞器,同时提供能量和必要的代谢成分[33];此外,自噬还能通过调控多种信号通路促进成骨分化、细胞外基质合成及矿化过程[34]。因此,增强自噬活性被认为是一种有前景的策略,可用于促进骨再生并提高成骨效率。在众多调控自噬的信号通路中,SIRT1/FOXO1轴受到了广泛关注,SIRT1是一种烟酰胺腺嘌呤二核苷酸阳性依赖性的去乙酰化酶,可通过去乙酰化激活转录因子FOXO1,而被激活的FOXO1可促进Beclin-1和微管相关蛋白1轻链3等下游自噬相关基因的表达,从而增强自噬活性,并支持成骨细胞的功能维持与分化。此外,FOXO1还可直接调控RUNX2等成骨关键转录因子表达参与骨基质蛋白(如Osterix、碱性磷酸酶、骨钙素)的调控,从多个层面促进成骨分化过程[35]。因此,SIRT1/FOXO1信号通路不仅通过调控自噬维持细胞功能,还可能通过调节成骨相关基因表达直接参与骨髓间充质干细胞向成骨谱系的分化调控。 为探究10-HDA是否通过激活SIRT1/FOXO1信号通路并增强自噬来发挥促成骨作用,采用Western blot分析检测了该信号通路及自噬相关关键蛋白的表达,包括SIRT1、p-FOXO1、FOXO1、微管相关蛋白1轻链3、Beclin-1和P62。Western blot检测显示,与对照组比较,0.5 mmol/L 10-HDA组SIRT1蛋白表"
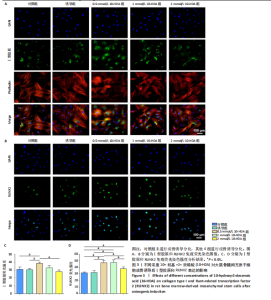
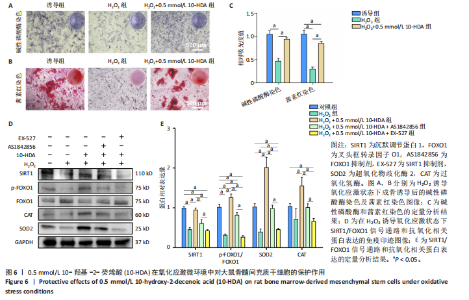
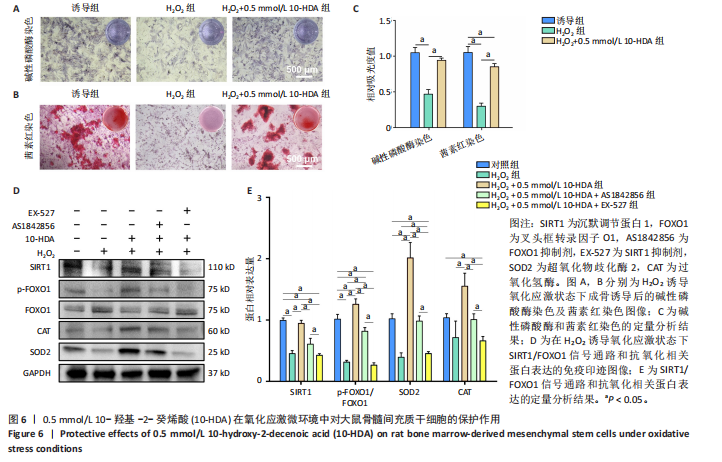
达、p-FOXO1/FOXO1比值升高(P < 0.05),自噬相关蛋白微管相关蛋白1轻链3与Beclin-1表达升高(P < 0.05),P62蛋白表达降低(P < 0.05),提示自噬活性增强;成骨相关蛋白Ⅰ型胶原、β-连环蛋白、RUNX2、骨桥蛋白表达升高(P < 0.05),见图5A-D。与0.5 mmol/L 10-HDA组比较,0.5 mmol/L 10-HDA+AS1842856组、0.5 mmol/L 10-HDA+EX-527组SIRT1、微管相关蛋白1轻链3、Beclin-1、Ⅰ型胶原、β-连环蛋白、RUNX2、骨桥蛋白表达及p-FOXO1/FOXO1比值均降低(P < 0.05),P62蛋白表达升高(P < 0.05),见图5A-D。表明使用FOXO1抑制剂AS1842856处理后0.5 mmol/L 10-HDA的上述作用被部分逆转,提示自噬在10-HDA的成骨机制中起关键作用;使用SIRT1特异性抑制剂EX-527处理后,SIRT1蛋白表达被有效抑制,FOXO1激活程度及Beclin-1表达下降,P62出现积累;与此同时,10-HDA诱导的成骨标志物水平显著降低,说明SIRT1的抑制削弱了10-HDA增强自噬和促成骨的作用。 为进一步探究10-HDA是否促进FOXO1的激活,进行了免疫荧光染色以观察FOXO1的亚细胞定位。FOXO1进入细胞核是它转录活化的关键标志,只有转位至细胞核后才能调控参与自噬和成骨分化的下游基因表达[36]。如图5E,F所示,对照组中FOXO1主要分布于胞质中,激活水平较低;0.5 mmol/L 10-HDA处理后,细胞核中FOXO1荧光显著增强,提示FOXO1转录活性提高;联合FOXO1抑制剂AS1842856处理后FOXO1核定位部分减弱,而联合SIRT1抑制剂EX-527后FOXO1核定位显著减少。这些结果表明,10-HDA通过激活SIRT1促进FOXO1核转位,进而可能增强自噬及成骨基因的转录活性。 2.6 10-HDA在氧化应激微环境中对骨髓间充质干细胞的保护作用及机制探究 通过H2O2诱导骨髓间充质干细胞进入氧化应激状态,采用碱性磷酸酶和茜素红染色评估成骨分化,如图6A-C所示。H2O2处理显著抑制了骨髓间充质干细胞的成骨分化,表现为碱性磷酸酶染色强度和面积的显著下降以及钙结节形成减少;0.5 mmol/L 10-HDA处理显著恢复了骨髓间充质干细胞的成骨能力,表明0.5 mmol/L 10-HDA在氧化应激条件下具有保护作用并可促进成骨。 为探究SIRT1/FOXO1信号通路的功能,研究通过Western blot检测该通路相关蛋白及抗氧化蛋白超氧化物歧化酶2和过氧化氢酶的表达水平,如图6D,E所示。H2O2处理后,大鼠骨髓间充质干细胞内SIRT1蛋白表达显著下调,p-FOXO1/FOXO1比值降低,超氧化物歧化酶2和过氧化氢酶蛋白表达亦有所减少;0.5 mmol/L 10-HDA处理后可显著恢复上述蛋白表达,尤其是超氧化物歧化酶2蛋白表达明显上调;相反,在与AS1842856或EX-527联合处理的情况下,10-HDA的保护作用明显减弱。这些结果提示,10-HDA可能通过激活SIRT1/FOXO1信号通路来增强"
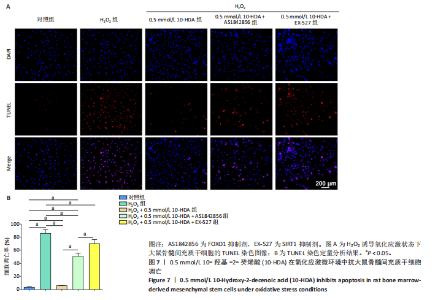
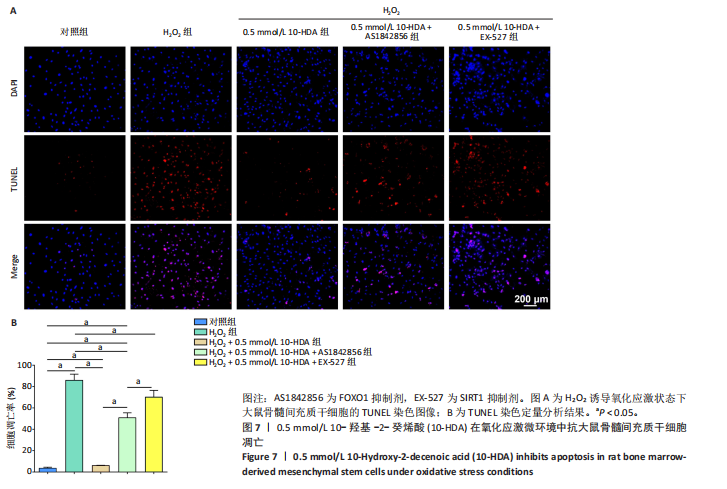
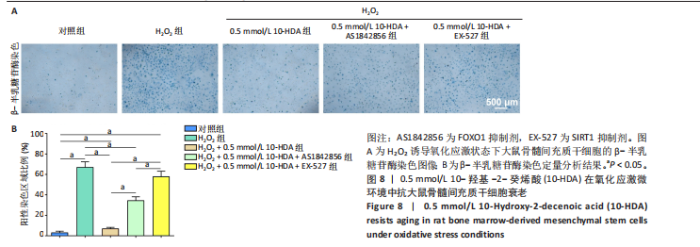
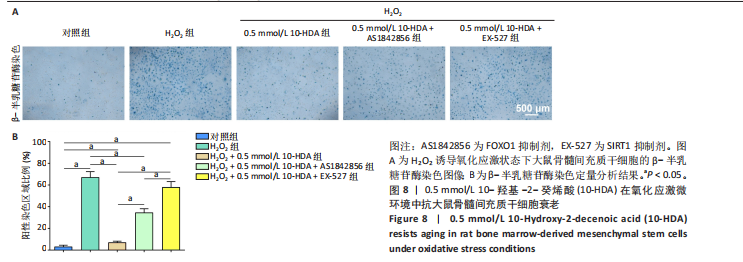
骨髓间充质干细胞的抗氧化能力并维持其正常功能。 此外,考虑到氧化应激常伴随细胞凋亡,通过TUNEL染色检测骨髓间充质干细胞的凋亡水平,如图7所示。H2O2处理后,骨髓间充质干细胞凋亡数量显著增加,而经0.5 mmol/L 10-HDA处理后细胞凋亡明显减少,提示10-HDA具有良好的抗凋亡保护作用;然而,当与AS1842856或EX-527联合处理时,10-HDA的抗凋亡保护作用被显著削弱,说明10-HDA的抗凋亡效果可能依赖于SIRT1/FOXO1信号通路的激活。 为进一步评估细胞衰老情况,采用β-半乳糖苷酶染色观察骨髓间充质干细胞的衰老状态,如图8所示。H2O2处理显著增加了骨髓间充质干细胞衰老的数量,而0.5 mmol/L 10-HDA干预有效减少了骨髓间充质干细胞的衰老;同样地,联合AS1842856或EX-527处理后,10-HDA的抗衰老作用明显减弱,进一步支持10-HDA通过SIRT1/FOXO1信号通路缓解氧化应激引发的细胞衰老。"

| [1] LANE JM, RUSSELL L, KHAN SN. Osteoporosis. Clin Orthop Relat Res. 2000;(372): 139-150. [2] YU B, WANG CY. Osteoporosis: The result of an ‘Aged’ bone microenvironment. Trends Mol Med. 2016;22(8):641-644. [3] GREWE JM, KNAPSTEIN PR, DONAT A, et al. The role of sphingosine-1-phosphate in bone remodeling and osteoporosis. Bone Res. 2022;10(1):34. [4] COMPSTON JE, MCCLUNG MR, LESLIE WD. Osteoporosis. Lancet. 2019; 393(10169):364-376. [5] SONG S, GUO Y, YANG Y, et al. Advances in pathogenesis and therapeutic strategies for osteoporosis. Pharmacol Therapeut. 2022;237:108168. [6] EASTELL R, O’NEILL TW, HOFBAUER LC, et al. Postmenopausal osteoporosis. Nat Rev Dis Primers. 2016;2(1):16069. [7] MANOLAGAS SC. From Estrogen-Centric to Aging and Oxidative Stress: A Revised Perspective of the Pathogenesis of Osteoporosis. Endocr Rev. 2010;31(3):266-300. [8] ENSRUD KE. Bisphosphonates for Postmenopausal Osteoporosis. JAMA. 2021; 325(1):96. [9] BOLOGNESE MA. SERMs and SERMs with estrogen for postmenopausal osteoporosis. Rev Endocr Metab Dis. 2010;11(4):253-259. [10] RUSSELL RG, WATTS NB, EBETINO FH, et al. Mechanisms of action of bisphosphonates: similarities and differences and their potential influence on clinical efficacy. Osteoporos Int. 2008;19(6):733-759. [11] FUGGLE N, AL-DAGHRI N, BOCK O, et al. Novel formulations of oral bisphosphonates in the treatment of osteoporosis. Aging Clin Exp Res. 2022; 34(11):2625-2634. [12] WOTTON CJ, GREEN J, BROWN A, et al. Use of oral bisphosphonates and risk of hospital admission with osteonecrosis of the jaw: Large prospective cohort study in UK women. Bone. 2019;124:69-74. [13] OKUMURA N, ITO T, DEGAWA T, et al. Royal jelly protects against epidermal stress through upregulation of the NQO1 expression. Int J Mol Sci. 2021;22(23):12973. [14] WANG J, ZHANG W, ZOU H, et al. 10-Hydroxy-2-decenoic acid inhibiting the proliferation of fibroblast-like synoviocytes by PI3K-AKT pathway. Int Immunopharmacol. 2015;28(1):97-104. [15] YOU M, MIAO Z, TIAN J, et al. Trans-10-hydroxy-2-decenoic acid protects against LPS-induced neuroinflammation through FOXO1-mediated activation of autophagy. Eur J Nutr. 2020;59(7):2875-2892. [16] SHIRAKAWA T, MIYAWAKI A, MATSUBARA T, et al. Daily oral administration of protease-treated royal jelly protects against denervation-induced skeletal muscle atrophy. Nutrients. 2020;12(10):3089. [17] GAO K, SU B, DAI J, et al. Anti-biofilm and anti-hemolysis activities of 10-Hydroxy-2-decenoic acid against staphylococcus aureus. Molecules. 2022;27(5):1485. [18] RUOLAN W, LIANGJIAO C, LONGQUAN S. The mTOR/ULK1 signaling pathway mediates the autophagy-promoting and osteogenic effects of dicalcium silicate nanoparticles. J Nanobiotechnol. 2020;18(1):119. [19] WANG J, YI Z, JIN C, et al. The role of autophagy in bone metabolism and clinical significance. Autophagy. 2023;19(9):2409-2427. [20] AMBROGINI E, ALMEIDA M, MARTIN-MILLAN M, et al. FoxO-mediated defense against oxidative stress in osteoblasts is indispensable for skeletal homeostasis in mice. Cell Metab. 2010;11(2):136-146. [21] TSAY J, YANG Z, ROSS FP, et al. Bone loss caused by iron overload in a murine model: importance of oxidative stress. Blood. 2010;116(14):2582-2589. [22] TIAN B, LI X, ZHANG J, et al. A 3D-printed molybdenum-containing scaffold exerts dual pro-osteogenic and anti-osteoclastogenic effects to facilitate alveolar bone repair. Int J Oral Sci. 2022;14(1):45. [23] XU C, WANG L, FOZOUNI P, et al. SIRT1 is downregulated by autophagy in senescence and ageing. Nat Cell Biol. 2020;22(10):1170-1179. [24] YANG K, VELAGAPUDI S, AKHMEDOV A, et al. Chronic SIRT1 supplementation in diabetic mice improves endothelial function by suppressing oxidative stress. Cardiovasc Res. 2023;119(12):2190-2201. [25] RACHED MT, KODE A, XU L, et al. FoxO1 is a positive regulator of bone formation by favoring protein synthesis and resistance to oxidative stress in osteoblasts. Cell Metab. 2010;11(2):147-160. [26] CHANG Y. The comparison study of using density gradient media in isolation of marrow derived human mesenchymal stem cells. Bone. 2009;44:S322-S323. [27] PURWANINGRUM M, JAMILAH NS, PURBANTORO SD, et al. Comparative characteristic study from bone marrow-derived mesenchymal stem cells. J Vet Sci. 2021;22(6):e74. [28] ZHANG X, GONCALVES R, MOSSER DM. The isolation and characterization of murine macrophages. Curr Protoc Immunol. 2008;83(1):14.1.1-14.1.14. [29] BOYLE WJ, SIMONET WS, LACEY DL. Osteoclast differentiation and activation. Nature. 2003; 423(6937):337-342. [30] LIANG C, XING H, WANG C, et al. Resveratrol protection against IL-1β-induced chondrocyte damage via the SIRT1/FOXO1 signaling pathway. J Orthop Surg Res. 2022;17(1):406. [31] ZOU J, CHEN H, FAN X, et al. Garcinol prevents oxidative stress-induced bone loss and dysfunction of BMSCs through NRF2-antioxidant signaling. Cell Death Discov. 2024;10(1):82. [32] LISTENBERGER LL, HAN X, LEWIS SE, et al. Triglyceride accumulation protects against fatty acid-induced lipotoxicity. Proc Natl Acad Sci. 2003;100(6):3077-3082. [33] YIN Y, TIAN BM, LI X, et al. Gold nanoparticles targeting the autophagy–lysosome system to combat the inflammation-compromised osteogenic potential of periodontal ligament stem cells: From mechanism to therapy. Biomaterials. 2022;288:121743. [34] HU R, LUO H, JI Y, et al. Activation of NLRP3 signaling contributes to cadmium-induced bone defects, associated with autophagic flux obstruction. Sci Total Environ. 2023;893:164787. [35] YANG Q, ZHOU Y, WANG T, et al. MiRNA-1271-5p regulates osteogenic differentiation of human bone marrow-derived mesenchymal stem cells by targeting forkhead box O1 (FOXO1). Cell Biol Int. 2021;45(7):1468-1476. [36] FENG YL, JIANG XT, MA FF, et al. Resveratrol prevents osteoporosis by upregulating FoxO1 transcriptional activity. Int J Mol Med. 2018;41(1):202-212. [37] LIU J, BAO X, HUANG J, et al. TMEM135 maintains the equilibrium of osteogenesis and adipogenesis by regulating mitochondrial dynamics. Metabolism. 2024;152: 155767. [38] BAI J, WANG H, CHEN H, et al. Biomimetic osteogenic peptide with mussel adhesion and osteoimmunomodulatory functions to ameliorate interfacial osseointegration under chronic inflammation. Biomaterials. 2020;255:120197. [39] CHURCHMAN SM, BOXALL SA, MCGONAGLE D, et al. A4.08 Predicting senescence-related loss of MSC osteogenic capacity irrespective of donor age. Ann Rheum Dis. 2016;75:A40. [40] YU Y, SHEN X, LUO Z, et al. Osteogenesis potential of different titania nanotubes in oxidative stress microenvironment. Biomaterials. 2018;167:44-57. [41] CHEN Y, ZHOU F, LIU H, et al. SIRT1, a promising regulator of bone homeostasis. Life Sci. 2021;269:119041. [42] YU X, CHEN X, WU W, et al. Zinc Alleviates diabetic muscle atrophy via modulation of the SIRT1/FoxO1 autophagy pathway through GPR39. J Cachexia Sarcopeni. 2025;16(2):e13771. [43] RUAN Y, XUE Y, ZHANG P, et al. Acetylation of FOXO1 is involved in cadmium-induced rat kidney injury via mediating autophagosome-lysosome fusion blockade and autophagy inhibition. Ecotox Environ Safe. 2024;287:117253. [44] LI H, DANHUI L, ZHENGMIN M, et al. Defective autophagy in osteoblasts induces endoplasmic reticulum stress and causes remarkable bone loss. Autophagy. 2018;14(10):1726-1741. [45] YOSHIDA G, TSUYOSHI K, HYOTA T, et al. Degradation of the NOTCH intracellular domain by elevated autophagy in osteoblasts promotes osteoblast differentiation and alleviates osteoporosis. Autophagy. 2022;18(10):2323-2332. [46] ZHANG Y, LIU W, YUAN W, et al. Impairment of APPL1/Myoferlin facilitates adipogenic differentiation of mesenchymal stem cells by blocking autophagy flux in osteoporosis. Cell Mol Life Sci. 2022;79(9):488. [47] ZHAN W, DENG M, HUANG X, et al. Pueraria lobata-derived exosome-like nanovesicles alleviate osteoporosis by enhacning autophagy. J Control Release. 2023;364:644-653. [48] ABDULLAH KS, ALHAMDANY DA. AB0811 Total antioxidant status and oxidative stress in patients with osteopenia and osteoporosis. Ann Rheum Dis. 2014;73:1071. [49] PENG J, LIU R, XU J, et al. Acid-responsive aggregated carrot-derived nanoantioxidants alleviate oxidative stress and restore osteoblast activity. J Nanobiotechnol. 2025;23(1):206. [50] COMAS F, LATORRE J, ORTEGA F, et al. Permanent cystathionine-β-Synthase gene knockdown promotes inflammation and oxidative stress in immortalized human adipose-derived mesenchymal stem cells, enhancing their adipogenic capacity. Redox Biology. 2021;42:101668. [51] KANZAKI H, SHINOHARA F, KANAKO I, et al. Molecular regulatory mechanisms of osteoclastogenesis through cytoprotective enzymes. Redox Biol. 2016;8: 186-191. [52] EFFRAIMIDIS G, WATT T, FELDT-RASMUSSEN U. Levothyroxine therapy in elderly patients with hypothyroidism. Front Endocrinol. 2021;12:641560. [53] MEUNIER PJ, ROUX C, SEEMAN E, et al. The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. New Engl J Med. 2004;350(5):459-468. [54] LIU F, WANG X, HE Y, et al. Jaw osteoporosis: Challenges to oral health and emerging perspectives of treatment. Biomed Pharmacother. 2024;177:116995. [55] MURPHY SJ, MCCULLOUGH LD, LITTLETON-KEARNEY MT, et al. Estrogen and selective estrogen receptor modulators. Endocrine. 2003;21(1):17-26. [56] FUGGLE NR, COOPER C, HARVEY NC, et al. Assessment of cardiovascular safety of anti-osteoporosis drugs. Drugs. 2020;80(15):1537-1352. |
| [1] | Wu Yanting, Li Yu, Liao Jinfeng. Magnesium oxide nanoparticles regulate osteogenesis- and angiogenesis-related gene expressions to promote bone defect healing [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1885-1895. |
| [2] | Li Zhenyu, Zhang Siming, Bai Jiaxiang, Zhu Chen. Osthole improves osteogenic differentiation function of bone marrow mesenchymal stem cells under high-glucose conditions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1641-1648. |
| [3] | Jin Dongsheng, Zhao Zhanghong, Zhu Ziyin, Zhang Sen, Sun Zuyan, Deng Jiang. Effects of icariin-loaded microsphere-three-dimensional scaffold on osteogenic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1658-1668. |
| [4] | Zou Yulian, Chen Chaopei, Huang Haixia, Lan Yuyan, Liu Min, Huang Ting. Resveratrol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1669-1678. |
| [5] | Zhang Haiwen, Zhang Xian, Xu Taichuan, Li Chao. Bibliometric and visual analysis of the research status and trends of senescence in osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1580-1591. |
| [6] | Jia Jinwen, Airefate·Ainiwaer, Zhang Juan. Effects of EP300 on autophagy and apoptosis related to allergic rhinitis in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1439-1449. |
| [7] | You Huijuan, Wu Shuzhen, Rong Rong, Chen Liyuan, Zhao Yuqing, Wang Qinglu, Ou Xiaowei, Yang Fengying. Macrophage autophagy in lung diseases: two-sided effects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1516-1526. |
| [8] | Liu Kexin, , Hao Kaimin, Zhuang Wenyue, , Li Zhengyi. Autophagy-related gene expression in pulmonary fibrosis models: bioinformatic analysis and experimental validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1129-1138. |
| [9] | Hu Jing, Zhu Ling, Xie Juan, Kong Deying, Liu Doudou. Autophagy regulates early embryonic development in mice via affecting H3K4me3 modification [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1147-1155. |
| [10] | Ma Runqiu, Yang Huixia, Li Xuer, Bai Zhigang, Li Guizhong, Hao Yinju, Ma Shengchao, Jiang Yideng. Mechanism of glucocorticoid-induced mitochondrial dysfunction in osteoblasts in steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8845-8851. |
| [11] | Cheng Xinyi, Chen Yida, Wang Yi, Liu Daihui, Zheng Yi, Shi Qin. Role of myeloid-derived suppressor cells in osteoclast differentiation in primary osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8852-8859. |
| [12] | Yang Huaqun, Abudouainijiang·Abulimiti, Wang Fazheng, Maimaitishawutiaji·Maimaiti, Li Simi, Muhetaer·Maimaitirexiati. Weighted gene co-expression network analysis combined with machine learning identifies autophagy and senescence signature genes in osteoarthritis chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8889-8898. |
| [13] | Yang Yanjun, Zhu Lin, Gu Yongchun, Yan Zhanjun. Mechanism by which magnesium implant-activated integrin α10β1 promotes osteogenic differentiation of periosteal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8319-8326. |
| [14] | Zhou Zixiang, Zhao Baoxiang. Research progress in the relationship between nontraumatic necrosis of the femoral head and lipid metabolism and its treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 680-690. |
| [15] | Tian Tan, Bao Shanjun. Hedgehog signaling pathway and diabetic osteoporosis: a potential target for specific drug therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7340-7346. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||