Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6923-6929.doi: 10.12307/2026.828
Previous Articles Next Articles
Three-dimensional bioprinting and tendon repair: application advances and future directions
Liu Xuemiao1, 2, Zhang Yuchang1, 2, Zhang Weiguo1, Tian Kang1, Wang Xing2
- 1Department of Joint Surgery and Sports Medicine, The First Affiliated Hospital of Dalian Medical University, Dalian 116000, Liaoning Province, China; 2Institute of Chemistry, Chinese Academy of Sciences, Beijing 100190, China
-
Accepted:2026-01-01Online:2026-09-18Published:2026-03-16 -
Contact:Wang Xing, PhD, Associate researcher, Institute of Chemistry, Chinese Academy of Sciences, Beijing 100190, China -
About author:Liu Xuemiao, Doctoral candidate, Department of Joint Surgery and Sports Medicine, The First Affiliated Hospital of Dalian Medical University, Dalian 116000, Liaoning Province, China; Institute of Chemistry, Chinese Academy of Sciences, Beijing 100190, China -
Supported by:National Natural Science Foundation of China, No. 52373162 (to WX)
CLC Number:
Cite this article
Liu Xuemiao, Zhang Yuchang, Zhang Weiguo, Tian Kang, Wang Xing. Three-dimensional bioprinting and tendon repair: application advances and future directions [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6923-6929.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

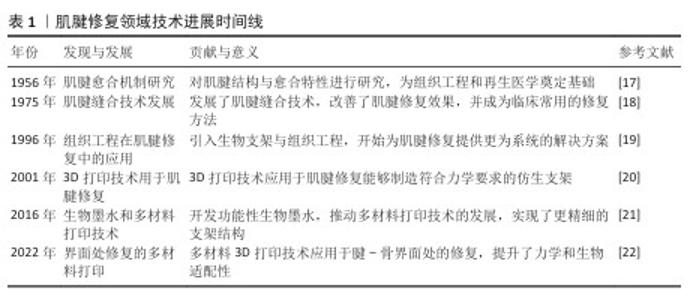
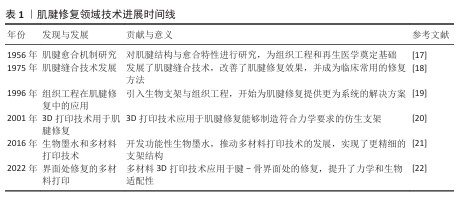
2.2 肌腱愈合基础 2.2.1 肌肉-肌腱-骨骼结构 见表2。 肌肉-肌腱-骨骼系统通过多层次结构与功能耦合实现运动力学传导[23]。肌肉-肌腱界面处作为跨尺度力学界面,肌动蛋白丝通过整合素-层粘连蛋白锚定于基底膜,微尺度应力由Ⅳ型胶原网络传递;而肌纤维束以30°-60°的扇形互嵌方式嵌入肌腱胶原,显著增加接触面积,同时弹性蛋白的富集赋予该区域动态的黏弹性[24-25]。肌腱本体由Ⅰ型胶原分级组装,从纳米到宏观层次形成高度定向结构,既提供了卓越的抗拉强度,又保持了良好的柔韧性[26]。此外,肌腱-骨骼界面处呈现四区渐变结构,通过胶原类型(Ⅰ型→Ⅱ型→Ⅹ型)与矿化程度(0%→60%羟基磷灰石)的连续梯度实现了软硬组织间的力学适配。然而,这些区域在损伤后的修复能力有限,常常形成纤维性瘢痕组织,显著影响功能恢复[27-29]。因此,深入理解这些结构特征及其修复机制,对提升运动损伤的修复策略具有重要意义。"


2.2.2 3D打印在肌腱愈合过程中的应用 肌腱愈合过程可分为炎症期、增殖期和重塑期3个阶段[30-31],而3D打印技术的介入为各阶段修复提供了精准调控手段。在炎症期(损伤后1-7 d),3D生物打印的仿生支架可快速植入损伤区域,支架的多孔结构既为血小板聚集和细胞因子释放提供物理支撑,又可负载抗炎药物调节局部免疫反应,促进坏死组织的高效清除[32-35]。 进入增殖期(第7天起持续数周),支架内部设计的定向微通道引导成纤维细胞有序迁移,结合缓释生长因子的协同作用加速Ⅲ型胶原合成与血管网络构建,同时通过仿生纤维拓扑结构引导细胞外基质的初步定向排列[36-37]。至重塑期(数周至数月),智能响应型打印材料可根据力学刺激动态调控支架降解速率,使Ⅲ型胶原向Ⅰ型胶原转化过程中纤维沿应力方向逐步重排[38-39]。通过整合生物活性调控、细胞定向引导及力学适配设计,3D打印构建的梯度化复合支架不仅显著提升修复组织的力学性能,还能优化胶原纤维的空间排列模式,从而降低愈合后期的再损伤风险[40]。这一技术实现了从微观分子调控到宏观力学支撑的全周期干预,为肌腱功能性修复提供了新的策略(表3)。 "

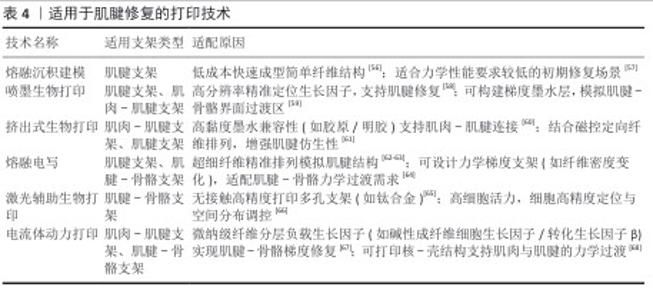
2.3 应用于肌腱修复的打印技术 在组织工程领域,肌腱修复因力学负荷的特殊性及再生微环境的复杂性,成为临床转化研究的难点之一[41]。肌肉-肌腱界面处、肌腱本体组织及肌腱-骨骼界面处在解剖结构、细胞外基质组成及生物力学性能上呈现显著异质性,这对支架的仿生设计与功能适配提出了多维度的挑战。例如,肌腱与骨骼的连接处需要高强度的支撑以承受较大的拉伸力,而肌腱与肌肉连接处则要求具有较好的延展性和弹性[42-43], 这样的需求使得传统的制造技术(如模具浇铸或注塑等)受到工艺精度和材料单一性限制,难以满足肌腱修复所需的力学梯度及生物活性因子时空控释等复杂要求。为了应对这些挑战,近年来3D生物打印技术取得了突破性进展,为构建解剖适配性支架提供了新的策略。3D打印技术的核心优势在于通过整合多尺度制造能力、智能材料设计以及细胞与材料的交互作用研究,可以实现细致入微的三维结构控制[44-45]。不同的打印技术各具优势,能够针对肌腱修复中的特定需求进行优化。例如电流体动力打印技术,通过微纳级纤维的分层沉积提供了精准的生物墨水加载与纤维取向控制能力[46]。电场的应用可以将生长因子和药物以精准的时空方式释放,同时促进细胞的迁移与增殖,极大地优化了细胞的生长环境,并在肌腱修复领域展示了巨大应用潜力[47]。与传统的打印技术相比,电流体动力打印技术能够在肌腱修复中实现更为精细的力学过渡层,特别是在肌肉-肌腱界面处的仿生结构构建中发挥了独特的作用[37,48]。 通过控制电场强度、喷射速度和生物墨水粘黏度等工艺参数,可以精确调节纤维的直径、排列密度及结构的力学性能。研究表明,电场强度的增加能够促进纤维的定向排列,但同时也可能加速材料的降解过程[49]。因此,在设计支架时必须平衡电场强度与材料降解之间的关系,以确保材料的力学稳定性和生物功能性。 生物墨水的选择对于3D打印技术的成功应用至关重要。生物墨水通常由天然和合成高分子材料构成[50]。天然材料如胶原蛋白、丝素蛋白、明胶和透明质酸,能够有效模拟肌腱的细胞外基质,提供良好的生物相容性和可生物降解性,特别是胶原蛋白能够促进细胞的附着和增殖,从而在肌腱修复中应用广泛[51-52]。合成材料如聚乳酸和聚己内酯则能通过调节其力学性能,特别是在力学过渡区域的设计中发挥关键作用。这些材料的选择不仅影响支架的物理性能,还决定了修复过程中的细胞行为。在肌腱修复中,支架需要模拟不同区域的力学性能,尤其是肌腱-肌肉和肌腱-骨骼的过渡层。通过控制生物墨水的交联度和黏度可以实现不同区域的力学梯度,交联度较高的材料可提供较强的机械强度,而低交联度材料则具有更好的柔韧性[53-54]。此外,生物墨水中生长因子和细胞因子的释放速率对修复效果至关重要,通过调节材料的交联度和结构设计可以实现生长因子与细胞因子的时空控释,促进细胞增殖和分化,从而优化肌腱修复过程[55]。 不同打印技术适用的支架类型与原因统计在表4中。 "
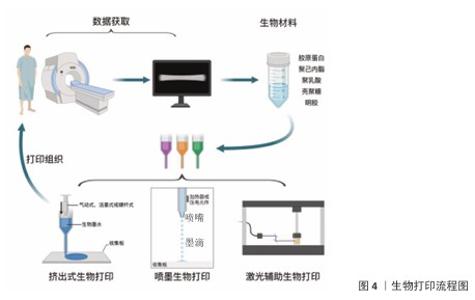
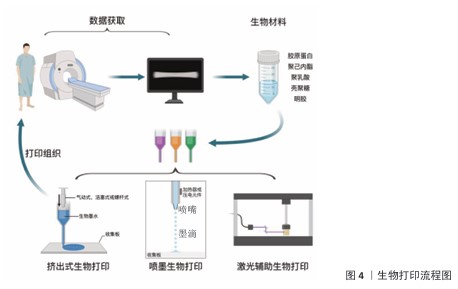
2.4 生物打印过程 3D生物打印技术在肌腱修复中的核心优势在于其多尺度结构可控性与功能集成化设计能力,能够精准响应肌腱组织工程对解剖适配性、力学动态响应及生物微环境调控的复杂需求。现阶段的生物打印过程如图4所示。在建模与生物墨水设计阶段,研究者通过患者影像数据(如MRI或CT)重建损伤肌腱的三维解剖结构,并结合有限元分析优化支架的孔隙率(60%-80%)和纤维排列(间10-50 μm),以匹配天然肌腱的力学传导特性。例如,DU等[69]通过多细胞分层打印技术将肌腱干/祖细胞与骨髓间充质干细胞按梯度分布,成功模拟了肌腱-骨骼界面处的四层渐变结构(肌腱→非矿化纤维软骨→矿化纤维软骨→骨骼),显著提升了再生结构的应力缓冲能力,这种结构能有效分散应力/减少应力集中,力学性能达到天然肌腱的85%以上。 生物墨水的功能化创新是技术落地的关键,尤其在肌腱修复对高强度与动态愈合的需求下[69-71]。研究者开发了复合生物墨水体系,以增强材料的力学性能和生物功能。例如,聚乳酸-聚乙醇酸/聚己内酯复合支架通过调节二者的比例(75∶25)可将拉伸强度控制在40-60 MPa、屈服强度达13-16 MPa,同时实现降解速率的精准匹配(75∶25复合支架降解周期为四五个月,聚己内酯降解周期两三年)[72];此外,硅酸锰纳米颗粒的引入赋予支架免疫调节功能,通过释放锰离子刺激巨噬细胞分泌前列腺素E2,促进细胞分化和界面微结构再生[72]。跨物种共生支架(如海洋硅藻与哺乳动物细胞结合)则通过光合作用持续供氧,改善局部缺氧状态,加速运动功能恢复[73]。吡格列酮预处理干细胞可增强线粒体递送效率,实现受损组织的能量代谢重编程,进一步优化再生微环境[74-75]。 在打印工艺方面,多模态技术的协同应用成为趋势。挤出式打印凭借0.25-0.4 mm喷嘴直径、0.1-0.3 mm层厚控制、5-15 kPa挤出压力及30-80 mm/s的打印速度,实现定向纤维束的沉积,精确复现肌腱的层状排列[76]。光固化辅助技术通过局部激光固化(如405 nm波长)增强层间结合力,防止梯度界面剥离[77]。对于复杂解剖轮廓(如肩袖肌腱的螺旋形态),原位打印技术可实现术中精准填充(精度达±50 μm),减少二次损伤风险[78]。 后处理与功能激活环节的创新同样关键,研究者可通过动态力学加载(如循环拉伸应变5%-10%)模拟体内应力环境,促进细胞外基质的分泌与纤维重排[79-80];而生长因子梯度释放(如转化生长因子β3缓释21 d以上)通过协同作用诱导干细胞成腱分化,实现从静态支撑到动态调控的功能跃迁[81-82]。 综上所述,3D生物打印技术通过精准控制结构和功能化设计能够有效满足肌腱修复对解剖适配性、力学响应及生物微环境调控的需求,多细胞分层打印与功能化生物墨水的结合使得支架的力学性能和再生效果得到了显著优化,多模态技术的协同应用提高了打印精度与支架稳定性,后处理技术则进一步促进了细胞外基质的重排和成腱分化,为肌腱修复提供了具有潜力的解决方案。 "

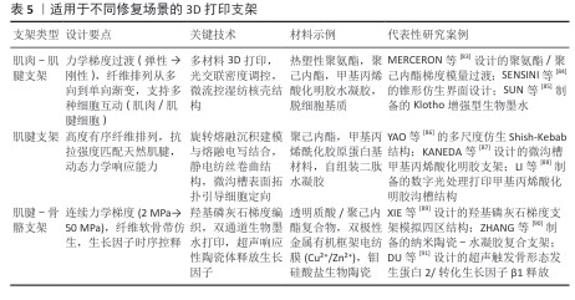
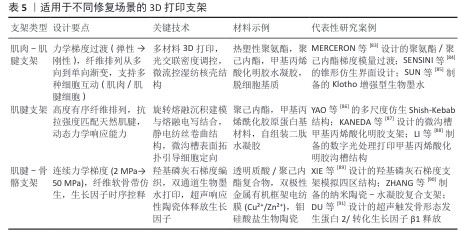
2.5.1 肌肉-肌腱界面修复 在肌肉-肌腱界面修复中,力学梯度的仿生构建是核心挑战。由于肌肉与肌腱的弹性模量差异可达2个数量级,传统均质材料难以匹配天然组织的力学特性[92-93]。研究者通过多材料复合策略突破这一瓶颈,例如MERCERON团队[83]将热塑性聚氨酯与聚己内酯结合,利用生物打印技术实现界面区模量梯度过渡,使应力集中降低60%,这一设计不仅模拟了天然组织的力学渐变特征,还通过孔隙结构的定向调控促进细胞迁移。进一步研究发现,数字光处理技术与人工智能逆向算法的结合可在单一材料体系中构建跨尺度刚度梯度(68-803 kPa),精确复现小鼠肌肉-肌腱界面的力学异质性,形态仿生设计为力学传导路径优化提供了新思路[94]。SENSINI团队[84]开发的锥形互锁结构通过纤维拓扑重构,将循环载荷下的应力峰值抑制至9%,其抗疲劳性能在10万次力学测试后仍保持稳定。生化微环境的同步调控同样关键,Klotho蛋白的引入可显著抑制氧化应激反应,使间充质干细胞存活率提升至92%并定向分化为功能性肌管与肌腱细胞,为界面再生提供了双重力学与生化保障[85]。 2.5.2 肌腱本体修复 肌腱本体的修复需兼顾结构有序性与动态响应能力。天然肌腱的高度定向胶原纤维(取向因子f > 0.9)赋予其优异的抗拉性能,而传统支架往往难以复现这种多层级结构[2,95]。旋转熔融沉积建模与熔融电写技术的集成创新为此提供突破,YAO团队通过该技术制备的波状纤维核心-多孔外壳支架,拉伸强度达6.94 MPa,其拓扑特征显著促进Ⅰ型胶原蛋白与双糖链蛋白聚糖的基因表达,力学性能与天然肌腱匹配度超过80%[96]。动态响应设计则拓展了支架的功能边界,壳聚糖压电凝胶不仅具备300%的可拉伸性,还能通过电阻变化实时监测肌腱活动状态,其内置的抗菌肽(LL-37)可有效抑制金黄色葡萄球菌生物膜形成,实现治疗-监测一体化功能。表面改性技术进一步提升了生物整合效率。微沟槽结构(宽10 μm)引导肌腱干细胞定向排列,使植入后胶原纤维直径增至100-200 nm,弹性模量提升166%,异位骨化面积减少70%[88]。这些技术的协同应用标志着肌腱修复从静态仿生向动态智能的范式转变。 2.5.3 肌腱-骨骼界面修复 腱骨界面修复因软-硬组织过渡特性成为最具挑战性的领域。力学与生化梯度的时空协调是该区域再生的关键,羟基磷灰石梯度支架(含量0%-15% )通过模量渐变(7.2-34.2 MPa)驱动细胞区域性分化,高羟基磷灰石区域促进成骨标志物表达,低羟基磷灰石区域则诱导肌腱特异性基因上调[89]。时序性因子控释技术为多阶段愈合提供精准调控,超声响应性陶瓷体可程序化释放骨形态发生蛋白2、转化生长因子β1与成纤维细胞生长因子2,在兔肩袖模型中使纤维软骨带厚度增加200%,力学强度恢复至天然组织的85%[91]。值得关注的是,神经血管网络的同步再生对功能恢复至关重要,血管内皮生长因子与神经生长因子的梯度递送可定向引导血管生成与神经轴突延伸,这种仿生策略为界面区建立生理传导功能奠定了基础[97-98]。 尽管上述研究显示出良好的动物实验效果,但临床转化仍面临多重瓶颈:术中快速成型技术的精度与效率不足,现有打印精度 (±0.3 mm)可能导致骨床微动风险;个性化支架设计周期较长(平均需72 h),限制急诊手术应用;长期生物相容性数据缺乏,如可降解材料的降解速率与组织再生的匹配性仍需验证。目前尚无基于增材制造的肌腱再生产品进入Ⅰ期临床研究,多数研究仍处于临床前评估阶段,因此,临床试验对于阐明新型生物工程肌腱样支架的适用性或缺点仍是必不可少的。 从技术演进角度看,肌腱修复支架设计正经历三大转变:从均质材料向多级梯度材料发展,从被动仿生向主动响应跨越,从单一结构修复向神经血管协同再生延伸。未来研究需进一步探索光-电-磁多模态调控机制,并推动实验室成果向临床转化。例如,可降解压电材料的开发有望实现无源植入式监测,而类器官3D打印技术或能构建具有代谢活性的微型肌腱单元,这些创新将推动组织工程从“结构替代”迈向“功能再生”的新阶段。 2.6 总结与展望 肌腱修复因多层级结构(纳米胶原纤维至宏观肌腱束)及界面区的特殊力学需求,传统方法难以复现梯度力学传导与细胞微环境,易致瘢痕愈合。尽管3D打印技术通过仿生结构设计提供了新路径,但其功能化与临床转化仍需智能材料与多模态技术的协同突破。环境响应型生物材料的开发是关键,其中,光热响应水凝胶可动态调控支架刚度以适配运动负荷;压电材料则通过力学-电信号耦合模拟内源性电微环境、调控细胞分化;酶敏感材料实现生长因子的时空可控释放;基因工程化细胞墨水则通过自分泌血管内皮生长因子等增强宿主整合[99-102]。此外,多模态技术的融合进一步拓展了应用维度:4D打印引入时间维度,使支架随pH值、温度等生理环境动态演变;类器官技术预构微型界面区生物活性单元,提升组织整合效率;多技术集成系统(熔融沉积建模宏观支撑+熔融电写纳米纤维排列+喷墨细胞定位)则同步满足宏观力学、微观结构与细胞分布的多尺度需求[103-105]。未来需系统验证智能材料的长期生物安全性(如压电效应的细胞毒性、光热材料的热损伤风险)及多模态技术的临床适配性(如类器官规模化制备、4D打印降解速率匹配),结合医学影像与AI构建个性化设计平台,推动技术从“结构仿生”向“功能再生”的临床转化。 "

| [1] STAŃCZAK M, KACPRZAK B, GAWDA P. Tendon Cell Biology: Effect of Mechanical Loading. Cell Physiol Biochem. 2024;58(6):677-701. [2] DONG Z, PENG R, ZHANG Y, et al. Tendon Repair and Regeneration Using Bioinspired Fibrillation Engineering That Mimicked the Structure and Mechanics of Natural Tissue. ACS Nano. 2023; 17(18):17858-17872. [3] SCHULZE-TANZIL GG. Healing of Ligaments and Tendons: Tissue Engineering and Models. Int J Mol Sci. 2022;23(24):15503. [4] CHATTERJEE M, EVANS MK, BELL R, et al. Histological and Immunohistochemical Guide to Tendon Tissue. J Orthop Res. 2023;41(10):2114-2132. [5] ALTUNBEK M, AFGHAH F, CALISKAN OS, et al. Design and Bioprinting for Tissue Interfaces. Biofabrication. 2023;15(2). doi: 10.1088/1758-5090/acb73d. [6] XU Z, XU W, ZHANG T, et al. Mechanisms of Tendon-Bone Interface Healing: Biomechanics, Cell Mechanics, and Tissue Engineering Approaches. J Orthop Surg Res. 2024;19(1):817. [7] SHEN S, LIN Y, SUN J, et al. A New Tissue Engineering Strategy to Promote Tendon-bone Healing: Regulation of Osteogenic and Chondrogenic Differentiation of Tendon-derived Stem Cells. Orthop Surg. 2024;16(10):2311-2325. [8] KANCHANATHEPSAK T, WAIROJANAKUL W, SUPPAPHOL S, et al. Evaluation of Biomechanical Properties on Partial and Complete Epitendinous Suture in Human Cadaver Flexor Tendon Repair. J Orthop Surg Res. 2021;16(1):489. [9] DAI W, LENG X, WANG J, et al. Quadriceps Tendon Autograft Versus Bone-Patellar Tendon-Bone and Hamstring Tendon Autografts for Anterior Cruciate Ligament Reconstruction: A Systematic Review and Meta-analysis. Am J Sports Med. 2022;50(12):3425-3439. [10] BUTLER DL. Evolution of Functional Tissue Engineering for Tendon and Ligament Repair. J Tissue Eng Regen Med. 2022;16(12):1091-1108. [11] RUIZ-ALONSO S, LAFUENTE-MERCHAN M, CIRIZA J, et al. Tendon Tissue Engineering: Cells, Growth Factors, Scaffolds and Production Techniques. J Control Release. 2021;333:448-486. [12] JIANG X, KONG Y, KUSS M, et al. 3D Bioprinting of Multilayered Scaffolds with Spatially Differentiated Admscs for Rotator Cuff Tendon-to-Bone Interface Regeneration. Appl Mater Today. 2022;27:101510. [13] WANG Y, REN C, BI F, et al. The Hydroxyapatite Modified 3D Printed Poly L-Lactic Acid Porous Screw in Reconstruction of Anterior Cruciate Ligament of Rabbit Knee Joint: A Histological and Biomechanical Study. BMC Musculoskel Dis. 2023;24(1):151. [14] KLAK M, KOWALSKA P, DOBRZAŃSKI T, et al. Bionic Organs: Shear Forces Reduce Pancreatic Islet and Mammalian Cell Viability during the Process of 3D Bioprinting. Micromachines (Basel). 2021;12(3):304. [15] ZHANG X, LI K, WANG C, et al. Facile and Rapid Fabrication of A Novel 3D-Printable, Visible Light-Crosslinkable and Bioactive Polythiourethane for Large-to-Massive Rotator Cuff Tendon Repair. Bioact Mater. 2024;37:439-458. [16] MAKUKU R, WERTHEL JD, ZANJANI LO, et al. New Frontiers of Tendon Augmentation Technology in Tissue Engineering and Regenerative Medicine: A Concise Literature Review. J Int Med Res. 2022; 50(8):3000605221117212. [17] LI Y, WANG W, XU W. Mechanisms and New Advances in The Efficacy of Plant Active Ingredients in Tendon-Bone Healing. J Orthop Surg Res. 2025;20(1):106. [18] ZHOU H, CHEN Y, YAN W, et al. Advances and Challenges in Biomaterials for Tendon and Enthesis Repair. Bioact Mater. 2025;47:531-545. [19] BUTLER DL. Evolution of Functional Tissue Engineering for Tendon and Ligament Repair. J Tissue Eng Regen Med. 2022;16(12):1091-1108. [20] ALHASKAWI A, ZHOU H, DONG Y, et al. Advancements in 3D-Printed Artificial Tendon. J Biomed Mater Res B. 2024;112(2):e35364. [21] CHAE S, CHOI YJ, CHO DW. Mechanically and Biologically Promoted Cell-Laden Constructs Generated Using Tissue-Specific Bioinks for Tendon/Ligament Tissue Engineering Applications. Biofabrication. 2022;14(2):025013. [22] SNOW F, O’CONNELL C, YANG P, et al. Engineering Interfacial Tissues: The Myotendinous Junction. APL Bioeng. 2024;8(2):021505. [23] 王彦军,孔令同,许硕贵.组织工程技术在肌腱损伤修复中的研究进展[J].中国骨伤,2024, 37(11):1126-1131. [24] PASSERIEUX E, ROSSIGNOL R, LETELLIER T, et al. Physical Continuity of the Perimysium from Myofibers to Tendons: Involvement in Lateral Force Transmission in Skeletal Muscle. J Struct Biol. 2007;159(1):19-28. [25] GREGORY J, HAZEL AL, SHEARER T. A Microstructural Model of Tendon Failure. J Mech Behav Biomed Mater. 2021;122:104665. [26] TERZI A, GALLO N, SIBILLANO T, et al. Travelling Through the Natural Hierarchies of Type I Collagen with X-Rays: from Tendons of Cattle, Horses, Sheep and Pigs. Materials (Basel). 2023;16(13):4753. [27] WANG C, ZHANG X, WANG DM, et al. Optimized Design of an Enthesis-Mimicking Suture Anchor-Tendon Hybrid Graft for Mechanically Robust Bone-Tendon Repair. Acta Biomater. 2024;176: 277-292. [28] YU C, WANG T, DIAO H, et al. Photothermal-Triggered Structural Change of Nanofiber Scaffold Integrating with Graded Mineralization to Promote Tendon–Bone Healing. Adv Fiber Mater. 2022;4(4):908-922. [29] ZUO R, LIU J, ZHANG Y, et al. In Situ Regeneration of Bone-to-Tendon Structures: Comparisons Between Costal-Cartilage Derived Stem Cells and Bmscs in the Rat Model. Acta Biomater. 2022; 145:62-76. [30] DARRIEUTORT-LAFFITE C, BLANCHARD F, SOSLOWSKY LJ, et al. Biology and Physiology of Ttendon Healing. Joint Bone Spine. 2024;91(5): 105696. [31] OLIVA F, MARSILIO E, ASPARAGO G, et al. The Impact of Hyaluronic Acid on Tendon Physiology and Its Clinical Application in Tendinopathies. Cells. 2021;10(11):3081. [32] LI C, WANG J, YANG W, et al. 3D-Printed Hydrogel Particles Containing PRP Laden with Tdscs Promote Tendon Repair in A Rat Model of Tendinopathy. J Nanobiotechnology. 2023;21(1):177. [33] WANG T, YU Z, LIN S, et al. 3D-Printed Mg-Incorporated PCL-Based Scaffolds Improves Rotator Cuff Tendon-Bone Healing Through Regulating Macrophage Polarization. Front Bioeng Biotechnol. 2024;12:1407512. [34] 尹嘉鹏,印卫锋,李光辉,等.肌腱修复中的炎症反应及其治疗进展[J].骨科,2025,16(1):70-76. [35] 盛显浩,张文,宋首龙,等.镁合金通过调控PI3K/AKT信号通路促进巨噬细胞M2极化在肩袖损伤修复中肌腱-骨愈合作用的分子机制研究[J].中国修复重建外科杂志,2025,39(2):174-186. [36] CORTELLA G, LAMPARELLI EP, CIARDULLI MC, et al. Colma-Based Bioprinted 3D Scaffold Allowed to Study Tenogenic Events in Human Tendon Stem Cells Bioeng Transl Med. 2025;10(1):e10723. [37] WANG L, SHI Y, QIU Z, et al. Bioactive 3D Electrohydrodynamic Printed Lattice Architectures Augment Tenogenesis of Tendon Stem/Progenitor Cells. ACS Appl Mater Interfaces. 2024;16(15): 18574-18590. [38] TOURÉ ABR, MELE E, CHRISTIE JK. Multi-layer Scaffolds of Poly(caprolactone), Poly(glycerol sebacate) and Bioactive Glasses Manufactured by Combined 3D Printing and Electrospinning. Nanomaterials (Basel). 2020;10(4):626. [39] SILVA M, GOMES S, CORREIA C, et al. Biocompatible 3D-Printed Tendon/Ligament Scaffolds Based on Polylactic Acid/Graphite Nanoplatelet Composites. Nanomaterials (Basel). 2023; 13(18):2518. [40] ALHASKAWI A, ZHOU H, DONG Y, et al. Advancements in 3D-Printed Artificial Tendon. J Biomed Mater Res B Appl Biomater. 2024;112(2):e35364. [41] HE Z, ZENG S, QIN B, et al. Investigation on the Role of Icariin in Tendon Injury Repair: Focusing on Tendon Stem Cell Differentiation. J Orthop Surg Res. 2025;20(1):379. [42] ZHU C, QIU J, THOMOPOULOS S, et al. Augmenting Tendon-to-Bone Repair with Functionally Graded Scaffolds. Adv Healthc Mater. 2021;10(9):2002269. [43] KAMALITDINOV TB, FUJINO K, KEITH LANG S, et al. Targeting the Hedgehog Signaling Pathway to Improve Tendon-to-Bone Integration. Osteoarthr Cartilage. 2023;31(9):1202-1213. [44] Leaf-nosed bat. Encyclopædia Britannica. Encyclopedia Britannica Online. 2009. [45] 何树坤,秦廷武.肩袖损伤修复的界面组织工程研究进展[J].中国修复重建外科杂志,2021, 35(10):1341-1351. [46] WU Y. Electrohydrodynamic Jet 3D Printing in Biomedical Applications. Acta Biomater. 2021; 128:21-41. [47] REIZABAL A, TANDON B, LANCEROS-MÉNDEZ S, et al. Electrohydrodynamic 3D Printing of Aqueous Solutions. Small. 2023;19(7):2205255. [48] GU B, MA Q, LI J, et al. Multi-material Electrohydrodynamic Printing of Bioelectronics with Sub-Microscale 3D Gold Pillars for In Vitro Extra- and Intra-Cellular Electrophysiological Recordings. Adv Sci. 2025;12(9):2407969. [49] QIU Z, ZHU H, WANG Y, et al. Functionalized Alginate-Based Bioinks for Microscale Electrohydrodynamic Bioprinting of Living Tissue Constructs with Improved Cellular Spreading and Alignment. Bio-Des Manuf. 2023;6(2):136-149. [50] HASAN MM, AHMAD A, AKTER MZ, et al. Bioinks for Bioprinting Using Plant-Derived Biomaterials. Biofabrication. 2024;16(4):042004. [51] SOWBHAGYA R, MUKTHA H, RAMAKRISHNAIAH TN, et al. Collagen as the Extracellular Matrix Biomaterials in the Arena of Medical Sciences. Tissue Cell. 2024;90:102497. [52] JAFARI H, LISTA A, SIEKAPEN MM, et al. Fish Collagen: Extraction, Characterization, and Applications for Biomaterials Engineering. Polymers (Basel). 2020;12(10):2230. [53] SYNOFZIK J, HEENE S, JONCZYK R, et al. Ink-Structing the Future of Vascular Tissue Engineering: A Review of the Physiological Bioink Design. Bio-Des Manuf. 2024;7(2):181-205. [54] JAIN P, KATHURIA H, RAMAKRISHNA S, et al. In Situ Bioprinting: Process, Bioinks, and Applications. ACS Appl. Bio Mater. 2024;7(12):7987-8007. [55] WANG F, SONG P, WANG J, et al. Organoid Bioinks: Construction and Application. Biofabrication. 2024;16(3):032006. [56] TAN DK, MÜNZENRIEDER N, MANIRUZZAMAN M, et al. A Low-Cost Method to Prepare Biocompatible Filaments with Enhanced Physico-Mechanical Properties for FDM 3D Printing. Curr Drug Deliv. 2021;18(6):700-711. [57] MASZYBROCKA J, DWORAK M, NOWAKOWSKA G, et al. The Influence of the Gradient Infill of PLA Samples Produced with the FDM Technique on Their Mechanical Properties. Materials (Basel). 2022;15(4):1304. [58] KER ED, NAIN AS, WEISS LE, et al. Bioprinting of Growth Factors onto Aligned Sub-Micron Fibrous Scaffolds for Simultaneous Control of Cell Differentiation and Alignment. Biomaterials. 2011;32(32):8097-8107. [59] UEHLIN AF, VINES JB, FELDMAN DS, et al. Inkjet Printing of Nanohydroxyapatite Gradients on Fibrous Scaffold for Bone–Ligament Enthesis. JOM. 2022;74(9):3336-3348. [60] ZHOU K, SUN Y, YANG J, et al. Hydrogels for 3D Embedded Bioprinting: A Focused Review on Bioinks and Support Baths. J Mater Chem B. 2022; 10(12):1897-1907. [61] BU H, ZHAN X, HU Z, et al. Enhanced Interlayer Strength in 3D-Printed PA12 Composites via Electromagnetic Induction Post-Processing. Addit Manuf. 2024;92:104383. [62] CHEN Q, MEI X, SHEN Z, et al. Direct Write Micro/Nano Optical Fibers by Near-Field Melt Electrospinning. Opt Lett. 2017;42(24):5106-5109. [63] GRASA J, URBIOLA A, FLANDES-IPARRAGUIRRE M, et al. Modeling and Fabrication of MEW-3D Tubular Scaffolds with Tendon Mechanical Behavior for Tenocyte Differentiation. Acta Biomater. 2025;197:226-239. [64] MA S, ZHENG S, LI D, et al. Melt Electrowriting Combined with Fused Deposition Modeling Printing for the Fabrication of Three-Dimensional Biomimetic Scaffolds for Osteotendinous Junction Regeneration. Int J Nanomedicine. 2024;19:3275-3293. [65] ZHENG Y, HAN Q, LI D, et al. Promotion of Tendon Growth into Implant through Pore-Size Design of A Ti-6Al-4 V Porous Scaffold Prepared by 3D Printing. Mater Design. 2021;197:109219. [66] CHICHKOV B. Laser Printing: Trends and Perspectives. Appl Phys A Mater Sci Process. 2022;128(11):1015. [67] BAI L, XU M, MENG Z, et al. Coaxial Electrohydrodynamic Printing of Core–Shell Microfibrous Scaffolds with Layer-Specific Growth Factors Release for Enthesis Regeneration. Int J Extreme Manuf. 2024;6(5):055003. [68] LI M, WU Y, YUAN T, et al. Biofabrication of Composite Tendon Constructs with the Fibrous Arrangement, High Cell Density, and Enhanced Cell Alignment. ACS Appl Mater Interfaces. 2023; 15(41):47989-8000. [69] DU L, QIN C, ZHANG H, et al. Multicellular Bioprinting of Biomimetic Inks for Tendon-to-Bone Regeneration. Adv Sci. 2023;10(21):2301309. [70] PENICHE SILVA CJ, DOMINGUEZ R, BAKHT SM, et al. Mirna-Laden Magnetic-Responsive Bioink for Tendon and Enthesis Tissue Engineering. Orthop Proc. 2024;106-B(SUPP_2):74-74. [71] KESHARWANI P, KUMAR V, GOH KW, et al. Pegylated PLGA Nanoparticles: Unlocking Advanced Strategies for Cancer Therapy. Mol Cancer. 2025;24(1):205. [72] DU L, WU J, HAN Y, et al. Immunomodulatory Multicellular Scaffolds for Tendon-To-Bone Regeneration. Sci Adv. 2024;10(10):eadk6610. [73] HAN Y, DU L, WU J, et al. Diatomaceous Cross-Species Constructs for Tendon-to-Bone Regeneration. Mater Today. 2025;83:64-84. [74] KIM W, LEE SK, KWON YW, et al. Pioglitazone-Primed Mesenchymal Stem Cells Stimulate Cell Proliferation, Collagen Synthesis and Matrix Gene Expression in Tenocytes. Int J Mol Sci. 2019; 20(3):472. [75] WANG Y, ZHANG R, CHEN Q, et al. PPARγ Agonist Pioglitazone Prevents Hypoxia-induced Cardiac Dysfunction by Reprogramming Glucose Metabolism. Int J Biol Sci. 2024;20(11):4297-4313. [76] WU H, XU F, JIN H, et al. 3D Nanofiber-Assisted Embedded Extrusion Bioprinting for Oriented Cardiac Tissue Fabrication. ACS Biomater Sci Eng. 2024;10(11):7256-7265. [77] QUAN H, ZHANG T, XU H, et al. Photo-Curing 3D Printing Technique and Its Challenges. Bioact Mater. 2020;5(1):110-115. [78] KIM W, JANG CH, KIM G. Bioprinted Hasc-Laden Structures with Cell-Differentiation Niches for Muscle Regeneration. Chem Eng J. 2021;419: 129570. [79] GRIER WK, SUN HAN CHANG RA, RAMSEY MD, et al. The Influence of Cyclic Tensile Strain on Multi-Compartment Collagen-GAG Scaffolds for Tendon-Bone Junction Repair. Connect Tissue Res. 2019;60(6):530-543. [80] RAABE O, SHELL K, FIETZ D, et al. Tenogenic Differentiation of Equine Adipose-Tissue-Derived Stem Cells Under the Influence of Tensile Strain, Growth Differentiation Factors and Various Oxygen Tensions. Cell Tissue Res. 2013;352(3):509-521. [81] SONG Y, CHOE G, KWON SH, et al. Dual Growth Factor Delivery Using Photo-Cross-Linkable Gelatin Hydrogels for Effectively Reinforced Regeneration of the Rotator Cuff Tendon. ACS Appl Bio Mater. 2024;7(2):1146-1157. [82] LI Y, GE Z, LIU Z, et al. Integrating Electrospun Aligned Fiber Scaffolds with Bovine Serum Albumin-Basic Fibroblast Growth Factor Nanoparticles to Promote Tendon Regeneration. J Nanobiotechnology. 2024;22(1):799. [83] MERCERON TK, BURT M, SEOL YJ, et al. A 3D Bioprinted Complex Structure for Engineering the Muscle-Tendon Unit. Biofabrication. 2015; 7(3):035003. [84] SENSINI A, D’ANNIBALLE R, GOTTI C, et al. Bi-Material Nanofibrous Electrospun Junctions: A Versatile Tool to Mimic the Muscle–Tendon Interface. Mater Des. 2024;242:113015. [85] SUN Y, SHENG R, CAO Z, et al. Bioactive Fiber-Reinforced Hydrogel to Tailor Cell Microenvironment for Structural and Functional Regeneration of Myotendinous Junction. Sci Adv. 2024;10(17):eadm7164. [86] YAO K, LV S, ZHANG X, et al. 3D Printing of Multiscale Biomimetic Scaffold for Tendon Regeneration. Adv Funct Mater. 2025;35(4):2413970. [87] KANEDA G, CHAN JL, CASTANEDA CM, et al. Ipsc-Derived Tenocytes Seeded on Microgrooved 3D Printed Scaffolds for Achilles Tendon Regeneration. J Orthop Res. 2023;41(10):2205-2220. [88] LI S, SUN Y, CHEN Y, et al. Sandwich Biomimetic Scaffold Based Tendon Stem/Progenitor Cell Alignment in a 3D Microenvironment for Functional Tendon Regeneration. ACS Appl Mater Interfaces. 2023;15(3):4652-4567. [89] XIE X, CAI J, YAO Y, et al. A Woven Scaffold with Continuous Mineral Gradients for Tendon-to-Bone Tissue Engineering. Compos Part B-Eng. 2021;212:108679. [90] ZHANG X, SONG W, HAN K, et al. Three-Dimensional Bioprinting of a Structure-, Composition-, and Mechanics-Graded Biomimetic Scaffold Coated with Specific Decellularized Extracellular Matrix to Improve the Tendon-to-Bone Healing. ACS Appl Mater Interfaces. 2023; 15(24):28964-28980. [91] DU C, WU R, YAN W, et al. Ultrasound-Controlled Delivery of Growth Factor-Loaded Cerasomes Combined with Polycaprolactone Scaffolds Seeded with Bone Marrow Mesenchymal Stem Cells for Biomimetic Tendon-to-Bone Interface Engineering. ACS Appl Mater Interfaces. 2024;16(1):292-304. [92] IWASAKI N, MORRISON B, KARALI A, et al. Measuring Full-Field Strain of the Muscle-Tendon Junction Using Confocal Microscopy Combined with Digital Volume Correlation. J Mech Behav Biomed Mater. 2025;164:106925. [93] SNOW F, O’CONNELL C, YANG P, et al. Engineering Interfacial Tissues: The Myotendinous Junction. APL Bioeng. 2024;8(2):021505. [94] KIRATITANAPORN W, GUAN J, TANG M, et al. 3D Printing of A Biomimetic Myotendinous Junction Assisted by Artificial Intelligence. Biomater Sci. 2024;12(23):6047-6062. [95] GUNER MB, DALGIC AD, TEZCANER A, et al. A Dual-Phase Scaffold Produced by Rotary Jet Spinning and Electrospinning for Tendon Tissue Engineering. Biomed Mater. 2020;15(6):065014. [96] XIONG Z, LIN B, HUANG C, et al. Biocompatible and Stretchable Chitosan Piezoelectric Gel with Antibacterial Capability and Motion Monitoring Function for Achilles Tendon Rupture Treatment. Carbohydr Polym. 2025;352:123149. [97] MONTEIRO RF, BAKHT SM, GOMEZ-FLORIT M, et al. 3D Writing of Multicellular Tendon-on-Cnc-Chip Models. Orthop Proc. 2024;106-B(SUPP_2):15. [98] ANDREANI L, VOZZI G, PETRINI M, et al. Smart Bioactive Personalized and Implantable 3d-Printed Scaffold for Tendon Regeneration: Preliminary Results of Tritone Project. Orthop Proc. 2023; 105-B(SUPP_9):13. [99] NIU C, LIU X, WANG Y, et al. Photothermal-Modulated Drug Release from A Composite Hydrogel Based on Silk Fibroin and Sodium Alginate. Eur Polym J. 2021;146:110267. [100] Extracellular Piezoelectric Nanostickers Promote Neuronal Differentiation. Nat Mater. 2025;24(7): 1003-1004. [101] SCHNEIDER MC, CHU S, RANDOLPH MA, et al. An in Vitro and in Vivo Comparison of Cartilage Growth in Chondrocyte-Laden Matrix Metalloproteinase-Sensitive Poly(Ethylene Glycol) Hydrogels with Localized Transforming Growth Factor bei’taβ3. Acta Biomater. 2019;93:97-110. [102] ALMEIDA-PINTO J, MOURA BS, GASPAR VM, et al. Advances in Cell-Rich Inks for Biofabricating Living Architectures. Adv Mater. 2024;36(27):e2313776. [103] KIM YB, SONG H, KIM S, et al. 4D Printing of Magneto-Responsive Shape Memory Nano-Composite for Stents. Smart Mater Struct. 2024; 33(10):105004. [104] ZHAO Z, CHEN X, DOWBAJ AM, et al. Organoids. Nat Rev Method Prime. 2022;2(1):94. [105] NAIM G, MAGDASSI S, MANDLER D. Continuous Material Deposition on Filaments in Fused Deposition Modeling. Polymers (Basel). 2024; 16(20):2904. [106] GRGIĆ I, WERTHEIMER V, KARAKAŠIĆ M, et al. 3D Printed Clamps for In Vitro Tensile Tests of Human Gracilis and the Superficial Third of Quadriceps Tendons. Appl Sci. 2021;11(6):2563. [107] XU T, RAO J, MO Y, et al. 3D Printing in Musculoskeletal Interface Engineering: Current Progress and Future Directions. Adv Drug Deliv Rev. 2025;219:115552. [108] NI Y, TIAN B, LV J, et al. 3D-Printed PCL Scaffolds Loaded with bFGF and BMSCs Enhance Tendon-Bone Healing in Rat Rotator Cuff Tears by Immunomodulation and Osteogenesis Promotion. ACS Biomater Sci Eng. 2025;11(2):1123-1139. [109] FERNANDEZ-YAGUE MA, PALMA M, TOFAIL SAM, et al. A Tympanic Piezo-Bioreactor Modulates Ion Channel-Associated Mechanosignaling to Stabilize Phenotype and Promote Tenogenesis in Human Tendon-Derived Cells. Adv Sci (Weinh). 2024;11(45):e2405711. |
| [1] | Wang Qisa, Lu Yuzheng, Han Xiufeng, Zhao Wenling, Shi Haitao, Xu Zhe. Cytocompatibility of 3D printed methyl acrylated hyaluronic acid/decellularized skin hydrogel scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1912-1920. |
| [2] | Zhou Hongli, Wang Xiaolong, Guo Rui, Yao Xuanxuan, Guo Ru, Zhou Xiongtao, He Xiangyi. Fabrication and characterization of nanohydroxyapatite/sodium alginate/polycaprolactone/alendronate scaffold [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1962-1970. |
| [3] | Yang Guang, Yin Zhitao, Xu Yan. Three-dimensional printed isoniazid liposome photothermal composite scaffolds and their performance evaluation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6701-6709. |
| [4] | Liu Weiwei, Gou Yuanbin, Cui Xiaoxue, Li Xin, Liu Dawei, Shi Mengrou, Chen Bin, Li Zhifeng. Comparison of different 3D-printed allogeneic bone and artificial polymer composite porous scaffold materials for repairing cranial bone defects in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6744-6751. |
| [5] | Li Yujin, Ni Guansen, Mao Weiqing, Tang Jiayu, Li Xueqing. Biocompatibility and preclinical experiments of a Chinese-made 3D-printed minimally invasive tungsten alloy needle electrodes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6859-6867. |
| [6] | Tang Hao, Zhong Qian, Wu Honghan, Wu Hengpeng, Wu Xingkai, Wa Qingde. 3D-printed biodegradable polyester-based scaffolds in bone regeneration therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5304-5311. |
| [7] | Wang Zitong, Wu Zijian, Yang Aofei, Mao Tian, Fang Nan, Wang Zhigang. Biomaterials regulate microenvironment imbalance for treating spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5321-5330. |
| [8] | Liu Shuting, Qiu Muen, Li Wei. Application and development trend of hydrogels in ophthalmic diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5349-5360. |
| [9] | Yang Fengli, Zhou Chao, Xiong Wei, Zhou Yuxiang, Li Dengshun, Wang Xin, Li Zhanzhen. 3D printed poly-L-lactic acid bone scaffolds in repair of bone defects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 507-515. |
| [10] | Li Liang, Yang Han, Suo Hairui, Guan Lu, Wang Zhenlin. 3D printed methacrylated gelatin/chitosan scaffolds: evaluation of antibacterial, mechanical properties and cytocompatibility [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(14): 3636-3642. |
| [11] | Xiong Jiaying, Shen Jieyi, Lyu Jiahong. Characteristics and strategies of 3D-printed biomimetic bioceramic scaffolds for repairing jaw defects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(14): 3709-3716. |
| [12] | Shi Xiaonan, Wu Xuan, Zhang Daxu, Hu Jingjing, Zheng Yazhe, Liu Yutong, Zhao Shuo, Li Weilong, Ye Shujun, Wang Jingyi, Yan Li. Preparation and characterization of 3D printed microstructured silk fibroin scaffold for liver injury repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(14): 3618-3625. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||