Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (9): 1422-1429.doi: 10.3969/j.issn.2095-4344.2460
Previous Articles Next Articles
Risk factors for stress fracture of lower limbs and future research direction
Zhao Yanxu1, 2, Zhang Zhao2, Luo Dong2,
Li Manglai2, Wang Jing2, Fang Jie2, Jiang Wen2,
Feng Yong1, Peng Jiang2,
Xu Wenjing2, Wang Yu2, Wang Aiyuan2, Zhang
Minze1
- 1Department of Trauma and Repair Surgery, Affiliated Hospital of Yan’an University, Yan’an 716000, Shaanxi Province, China; 2Orthopedics Institute of Chinese PLA General Hospital, Beijing 100853, China
-
Received:2019-07-11Revised:2019-07-27Accepted:2019-09-02Online:2020-03-28Published:2020-02-13 -
Contact:Zhang Minze, Chief physician, Master’s supervisor, Department of Trauma and Repair Surgery, Affiliated Hospital of Yan’an University, Yan’an Shaanxi Province, China Wang Aiyuan, Master, Associate researcher, Professor, Master’s supervisor, Orthopedics Institute of Chinese PLA General Hospital, Beijing 100853, China -
About author:Zhao Yanxu, Master candidate, Department of Trauma and Repair Surgery, Affiliated Hospital of Yan’an University, Yan’an Shaanxi Province, China; Orthopedics Institute of Chinese PLA General Hospital, Beijing 100853, China -
Supported by:the Studying on Early Warning Evaluation System and Comprehensive Preventive Measures for Stressful Fractures in Military Training, No. 16CXZ044; the Researches on Key Technologies of Vital Tissue Warfare Trauma Tissue Engineering Regenerative Medicine, No. BWS11J025; the Diagnostic Criteria and Principles of Prevention and Treatment of Stress Fractures in Military Training, No. BWS17B053
CLC Number:
Cite this article
Zhao Yanxu, Zhang Zhao, Luo Dong, Li Manglai, Wang Jing, Fang Jie, Jiang Wen, Feng Yong, Peng Jiang, Xu Wenjing, Wang Yu, Wang Aiyuan, Zhang Minze. Risk factors for stress fracture of lower limbs and future research direction[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1422-1429.
share this article

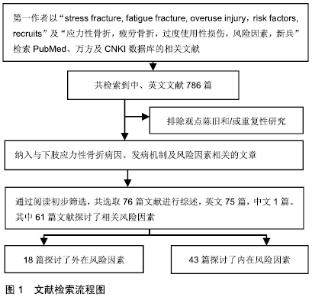
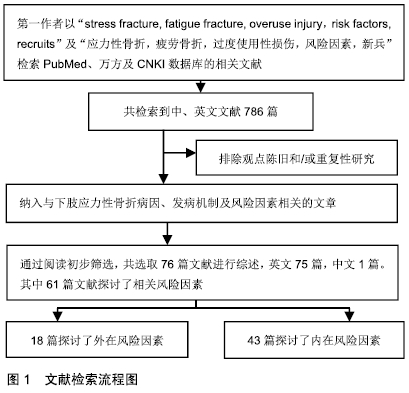
2.2 流行病学 应力性骨折常见于跑步人群,尤其是部队新兵、田径运动员以及舞蹈演员[6]。尽管应力性骨折在普通人群中的发病率低于1%,但在女运动员人群中,应力性骨折的发生率高达13%[7],新兵应力性骨折的发病率则为3%-9%[8]。然而在一项针对以色列男性军人的前瞻性研究中,应力性骨折发生率高达28.3%[9]。由此可见,应力性骨折的发病率差异很大,这可能与训练强度和训练项目的不同有关。 2.3 发病机制 应力性骨折是重复使用性损伤。在应力性骨折早期,反复的应力会导致骨重建,即出现新骨形成,但是持续或逐渐增强的应力将打破骨重建的平衡,当骨吸收速度快于骨形成速度时,引起骨疲劳,最终发展成为完全骨折[10]。导致应力性骨折发生的原因仍存在争议,目前普遍接受的有2种理论[11-12]:第1种理论认为,骨骼周围肌肉组织因长时间运动引起疲劳后,导致持续的应力经骨传递,从而引起骨骼的微骨折;第2种理论认为,在持续高强度运动的情况下,骨吸收速度快于骨形成速度,骨骼易发生微损伤,若得不到充分的休息,微损伤会逐渐积累,最终发展为完全骨折。 2.4 影像学诊断 X射线平片为应力性骨折首选的影像学检查方法,但是X射线平片检查对早期的应力性骨折缺乏敏感性,假阴性率较高。MRI对应力性骨折的诊断率较高,可作为诊断应力性骨折的金标准[13]。在MRI不能确定的病例中,CT可以作为一种选择的方法,但CT并不常规用于应力性骨折的诊断中[14]。此外,有学者认为,核素骨扫描可显示应力性骨折的代谢活跃区,是早期应力性骨折诊断的金标准[15]。 2.5 风险因素 在军事集训期间,应力性骨折的发生率与训练量成正比。预防下肢应力性骨折的关键是发现致伤因素,从源头干预,从而减少应力性骨折的发生。应力性骨折的发病机制非常复杂,其发生显然是多因素共同作用的结果,既包括外在因素又有内在因素,见表1。这些复杂的发病原因和发病机制可能有助于解释为什么在相同的运动条件下,给定的个体处于不同的风险中。 "


2.5.1 外在因素 (1)训练模式:军事研究表明,不同的军种和不同的训练模式可能使军事人员处于不同程度的骨折风险中。一项针对美国军人的流行病学研究表明,与其他军种相比,陆军和海军更容易发生下肢应力性骨折[16]。此外,每周跑步距离大于30 km(OR=3.28,95%CI:1.23-8.75)与训练伤的发生有显著相关性[17]。值得注意的是,每周进行较高的训练强度对运动员似乎具有保护作用[18],DAMSTED等[19]发现每周跑步少于15 km和/或跑步速度小于6 km/min的跑步者更容易受到伤害。但运动量的突然增加会提升跑步者应力性骨折的发生率,一项由214名跑步者参与的为期14周的前瞻性研究发现,与每周跑步距离增加不到20%的人相比,每周跑步距离增加20%-60%的人更容易受伤。 (2)入伍前体能训练与身体素质:在服兵役之前,定期和反复进行高强度体力活动可能是预防应力性骨折发生的重要预防措施[20],这可能与身体素质的提高有关。有研究发现在入伍/学前,每周锻炼时间少于7 h的男性学员发生应力性骨折的风险要比经常锻炼的学员高2.3倍[6]。一项针对美国陆军应力性骨折风险的分析发现,低体脂百分比的男性、女性新兵以及高体脂的男性新兵中,应力性骨折风险分别增加1.27,1.15和1.20 倍[21]。显然,肥胖和体能下降与下肢应力性骨折风险增加有关[22]。BECK等[23]的研究发现,与正常对照组相比,胫骨应力损伤患者的脂肪含量增加2.7%,肌肉含量减少2.6%,且差异有显著性意义。他们认为,增加肌肉力量和限制脂肪增加可以对应力性骨损伤起到一定的保护作用。近年来,随着青少年运动量的减少以及生活方式(久坐)的改变,军事新兵的身体素质有所下降,这些改变可能会降低训练效率并增加受伤风险。 (3)设备和训练环境因素:鞋的类型或矫形鞋垫与下肢应力性骨折的发生发展存在一定的关系,不同类型的跑鞋以及旧鞋与下肢应力性骨折的发生率有关[24]。佩戴更舒适的靴子或者足部矫形器有助于预防应力性骨折的发生[25-26],它们可能通过改变跑步者下肢的生物力学参数而起作用[27]。另外,有研究表明在地势平坦、表面坚硬的基地进行训练,并将累积行军距离减少1/3,可减少应力性骨折的发生率(OR=10.03,95%CI:3.5- 28.4)[26]。另外,也有人曾尝试研究紫外线指数低的地域环境是否与应力性骨折的发生率存在关系,但并没有发现有意义的结果[28]。目前,环境因素与应力性骨折关系的研究相对较少,缺乏足够的证据证实两者之间的关系。 (4)药物因素:口服避孕药的使用与骨折风险的降低显著相关(OR=0.81,95%CI:0.74-0.90)。在18-25岁、26-35岁年龄组以及口服避孕药使用持续时间超过1年的患者中,这种关联最强[29]。同样,口服避孕药的使用也显著降低了应力性骨折的发生率[30]。此外,由于非类固醇抗炎药在军队新兵中的使用比较广泛,一项针对非类固醇抗炎药与应力性骨折发生关系的研究发现,与不服用药物的士兵相比,服用非类固醇抗炎药和对乙酰氨基酚的士兵应力性骨折的风险分别增加3倍及2倍,尤其是在进行强化训练的时候,应力性骨折的风险增加了4倍[31]。原因在于非类固醇抗炎药可能通过抑制前列腺素的合成而增加应力性骨折的风险。 2.5.2 内在因素 (1)性别:性别和年龄是下肢应力性骨折发生的潜在风险因素。在集训期间,女性发生应力性骨折的可能性是男性的5倍[32]。女性有较高风险发生应力性骨折可能是多因素造成的,尤其是在伴有三联征(饮食紊乱、闭经和低骨密度)的女性人群中,应力性骨折风险增加15%-50%[6,32-34]。NOSE-OGURA等[35]对390名女运动员的研究中发现,三联征的每个组成部分仅是青少年运动员应力性骨折的危险因素,与成人应力性骨折无关,原因可能在于青春期是骨量积累的关键时期。其中,青少年女运动员继发性闭经、低骨密度(Z值<-1)或实际体质量与理想体质量的比例低(≤85%)分别使应力性骨折的风险增加了12.9倍、4.5倍和1.1倍。而且,相对于正常人,经常跑步的人更容易出现月经紊乱。PRATHER等[36]发现,不论年龄大小,几乎每5名参加高水平比赛的女子足球运动员中就有1人有月经功能障碍。因此,应重视对女性新兵或运动员的教育,尽量减少应力性骨折的风险。 (2)年龄:一项关于2 439名男性职业足球运动员的研究报道,年轻球员(22.1±2.1)岁应力性骨折发生率为年长球员(29.6±2.9岁)的11倍[37]。可以看出,应力性骨折的发病率与年龄成反比关系。一种可能的解释是年轻跑步者骨质积累峰值时间的相对滞后[38];另一种可能的解释是,与年轻跑步者相比,由于长期的训练,年长跑步者的肌肉骨骼结构已经习惯于承受更高的训练负荷,因此具有较少的过度使用性伤害。然而并不是所有的研究均得出类似的结论,正如KNAPIK等[32]的研究发现应力性骨折的发生风险随着年龄(17-35岁)的增加而呈现上升的趋势,该研究报道在男性和女性新兵中,应力性骨折人数每年分别增加2.2例/1 000名和3.9例/1 000名。建议未来的研究可以通过划分年龄组来确定两者之间的因果关系。 (3)种族/民族:种族和民族的不同在应力性骨折发生的风险因素中扮演一个重要的角色。一项对130万美国陆军士兵的队列研究报道,混合种族(5.4/1 000人年)以及白人士兵(4.7/1 000人年)患应力性骨折的风险最高[39]。然而,不同种族/民族导致应力性骨折风险差异的原因尚不完全清楚,可能是由于骨量、骨微结构和强度的差异造成的。与白人相比,黑人具有更大的骨密度、皮质面积以及更低的皮质孔隙率等有利的骨微结构,这可能有助于降低他们的骨折风险[40-41]。目前很少有研究在探讨其他危险因素(如体质量指数、年龄、教育程度)对应力性骨折影响的同时,也探讨种族/民族的作用。因此,白种人作为应力性骨折的潜在危险因素值得进一步研究。 (4)既往应力性骨折史:一项有关海军新兵胫骨内侧应力综合征风险因素的研究报道,有胫骨内侧应力综合征病史的新兵发生胫骨内侧应力综合征的风险是无胫骨内侧应力综合征病史新兵的18.28倍[42]。以往对应力性骨折的研究中也确定了既往应力性骨折病史为未来应力性骨折的风险因素[43]。一项针对156名男性运动员开展的为期7年的研究发现,先前的骨应力损伤评分每增加1分,应力性骨折风险将增加57%[44]。在中国人民解放军陆军新兵中,存在应力性骨折病史的新兵更容易遭受应力性骨折(P?=0.011,OR=1.77)[45]。有应力性骨折史的跑步者具有较低的骨强度以及较小的肌肉横截面积,这可能使该个体易发生应力性骨折[46]。既往骨折病史是未来应力性骨折的一个重要危险因素,但它是不可改变的。因此,此类新兵或运动员在集训期间应注意控制其他风险因素。 (5)骨密度:骨密度作为骨质量的一个重要标志,常用来预测骨折发生的风险[47]。TENFORDE等[48]发现,在28例男性应力性骨折患者中有43%的骨密度Z值< -1.0。DAVEY等[49]对皇家海军陆战队新兵的研究发现,腰椎骨密度低于正常值的新兵发生应力性骨折的可能性是骨密度正常或高于正常值的新兵的4倍以上(OR=4.3,95%CI:1.9-9.9)。然而,DUCKHAM等[50]并没有发现髋、腰椎及桡骨远端骨密度与应力性骨折之间存在相关性。先前,作者开展的一项对中国人民解放军某陆军中新兵的研究发现,应力性骨折的发生与跟骨骨密度存在显著的相关性。作者认为,将跟骨超声骨密度仪应用于对入伍新兵和运动员日常的体检中,具有较高的实用价值。另外,值得注意的是,虽然多数文献报道骨密度与应力性骨折之间存在相关性,但是单独使用骨密度预测应力性骨折发生风险的准确性有待进一步研究,未来在进行此类研究时应控制好其他混杂因素。 (6)钙和维生素D摄入不足:骨强度的降低会增加发生应力性骨折的风险[51]。维生素D以及钙是人体必需的营养物质,对维持骨强度和骨健康是必不可少的[52]。一项针对1 082名皇家海军男性新兵开展的为期32周的研究报道,在训练开始时维生素D水平较低即血清25-羟基维生素D(25(OH)D)浓度低于50 nmol/L的新兵发生应力性骨折的风险将增加(OR=1.6)[53]。NIEVES等[54]的研究是第一个证明钙和维生素D是降低女性应力性骨折发生的保护因素的前瞻性研究,他们发现每天多喝一杯脱脂牛奶,应力性骨折的发生率就会降低62%。一项对124例应力性骨折患者的研究发现,维生素D水平<40 μg/L的人员发生应力性骨折的风险较高[55]。显然,血清25(OH)D与应力性骨折的发生有关联,但是目前的研究对25(OH)D临界值的报道尚不一致,尚需更加深入的研究进行确认。 (7)心理因素:心理压力就像病原体一样,会影响到身体的防御功能,破坏机体的平衡,可能会造成身体的某些疾病。MORAN等[56]的研究表明,患应力性骨折的新兵承受着更大的心理压力,他们建议开发一种基于身体表现和心理状态评估的新方法去干预以及调整训练模式,从而减少应力性骨折的发生。 (8)遗传易感性:近年来,应力性骨折的遗传易感性已经成为了一个热点话题,可能在应力性骨折的发生发展中起着不可或缺的作用。ZHAO等[45]评估了189例应力性骨折患者和1 209例健康对照人群的GDF-5rs143383单核苷酸多态性分布,结果发现携带GDF-5rs143383基因的人群应力性骨折的发病风险是非携带者的1.75倍。VARLEY等[57]在对精英运动员的11个遗传变异的研究中发现,SOST SNP rs1877632和VDR SNPs rs10735810和rs731236的变异等位基因与应力性骨折损伤相关。 (9)解剖学因素:下肢形态学和解剖学与应力性骨折之间的关系已有报道,但这些因素不容易被干预。最近的2项Meta分析指出,足内翻、高弓足和扁平足等足部形态的变异使得这些个体易于发生下肢损伤[58-59]。可能的原因是,不同的足部姿势在运动时显示出不同的足底压力特征,比如,扁平足即表现出前足外侧的峰值压力与压力-时间积分等异常的生物力学参数,这可能改变了人体的生物力学性能[60]。NUNNS等[61]发现较窄的双踝距离、较低的体质量指数、较高的足跟压力以及较低的胫骨旋转范围显著增加了新兵发生胫骨应力性骨折的风险。一项针对男性皇家海军陆战队新兵的研究也得出了类似的结论,他们发现胫骨距离越窄,胫骨力量指数降低,应力性骨折的发生风险越高[62]。此外,髋关节内旋范围也与应力性骨折的发生有关,幸运的是,这是一个可改变的因素,可通过监测和改善髋关节内旋范围以减少应力性骨折的发生。MEARDON等[63]的研究发现,骨的几何形状以及施加的载荷均可提高胫骨远端骨应力,这提示可以改变军事新兵的训练模式,增强骨骼的结构特征,促进骨骼的适应。另外,可通过调整新兵运动中的步态,抵抗外加的负荷,以减少运动过程中的应力,从而减少应力性骨折的发生[64]。 (10)生物力学因素:步态模式的异常可能会导致下肢过度使用性损伤,但其潜在的机制尚不清楚。一项前瞻性研究发现关节活动范围、肌肉力量、足底压力分布以及运动学因素在胫骨内侧应力综合征的发展中起了重要作用,其中站立时后脚外翻持续时间每增加1%,发生胫骨内侧应力综合征的可能性就增加1.38倍(P=0.015)[65]。一项关于胫骨内侧应力综合征生物力学因素的综述收集了以往大量的文献,报道胫骨内侧应力综合征的主要风险因素是体质量指数的增加、跖屈活动度和髋关节外旋活动度的改变[66]。尽管上述文献基本都是在研究胫骨内侧应力综合征,但是,下肢应力性骨折的发生发展与其有一定的相似性,这为研究下肢应力性骨折的主要风险因素提供了思路和方向。 足底压力分析是指在行走过程中对足底表面施加的力的大小和分布的测量,是研究足部姿势与下肢生物力学功能相互作用的一种技术[67]。足底压力分析系统在下肢过度使用性损伤以及其他运动伤的发生风险领域中已经有了广泛的研究[68-69]。DIXONA等[70]首次研究了跖骨应力性骨折与动态足底压力的关系,他们的数据表明,动态弓形指数每增加1%,发生第二跖骨应力性骨折的风险就会降低25%。与正常人员相比,第三跖骨应力性骨折患者表现出更大的足外展角度以及更高的第4跖骨区峰值压力。此外,第2跖骨峰值压力时间每增加1个单位,应力性骨折风险将增加19%。一项为期32周的军事训练的前瞻性研究发现第1跖骨区的高峰值压力和跑步期间内侧高度集中的压力与损伤风险增加有关[71]。改变的步态生物力学如垂直地面反作用力可能使个体易于发生应力性骨折[46]。还有研究发现应力性骨折患者与正常人的足底压力参数之间存在差异,遗憾的是他们并没有做前瞻性研究,其结果并不能充分说明两者间的相关性,但他们的研究结果指明了研究方向。同时,他们还指出下肢动力学和运动学差异可能能够反映应力性骨折的风险[72]。足底压力负荷的增加可能是应力性骨折发生发展的一个诱发因素。足底压力分布因足部姿势的不同而不同,足底压力参数与应力性骨折的联系尚不清楚,需要进一步的研究来确定足部姿势和足底压力对应力性骨折的影响。 此外,一项关于应力性骨折的有限元分析发现同时进行跑步和步行试验的参与者,两者的胫骨应力存在显著的差异[73]。跑步期间的胫骨应力峰值比行走期间的胫骨应力峰值大3倍以上。然而,令人惊讶的是,每个行走周期的累积应力(13.6 MPa·s)是跑步的累积应力的88%(15.2 MPa·s)。他们认为胫骨应力的一次性升高并不一定会增加应力性骨折的风险,因为机械负荷升高只是导致应力性骨损伤的一个风险因素。 2.5.3 其他因素 另外,吸烟、饮酒以及营养状况等诸多因素均可能会增加下肢应力性骨折的风险[31,74-75]。未来的研究可参考上述风险因素进行方案设计,并在军事集训期间跟踪随访,以确定新兵应力性骨折的各类风险因素,从而给予相应的预防措施。虽然性别、年龄、种族以及既往应力性骨折病史等风险因素不可改变,但可以针对诸如训练模式和心理状态等因素进行干预,还可以通过使用合适的矫形器调整新兵的异常生物力学参数,有效减少新兵应力性骨折的发病率。 "

| [1] MOREIRA CA, BILEZIKIAN JP. Stress fractures: concepts and therapeutics. J Clin Endocrinol Metab.2017;102(2): 525-534. [2] HUGHES JM, POPP KL, YANOVICH R, et al. The role of adaptive bone formation in the etiology of stress fracture. Exp Biol Med (Maywood). 2017; 242(9): 897-906. [3] WELCK MJ, HAYES T, PASTIDES P, et al. Stress fractures of the foot and ankle. Injury. 2017;48(8): 1722-1726. [4] MILLER TL, JAMIESON M, EVERSON S, et al. Expected time to return to athletic participation after stress fracture in division i collegiate athletes. Sports Health. 2018;10(4):340-344. [5] ENGEBRETSEN L, BAHR R, COOK JL, et al. The IOC Centres of Excellence bring prevention to sports medicine. Br J Sports Med. 2014;48(17):1270-1275. [6] COSMAN F, RUFFING J, ZION M, et al. Determinants of stress fracture risk in United States Military Academy cadets. Bone. 2013; 55(2): 359-366. [7] ABBOTT A, BIRD ML, WILD E, et al. Part I: epidemiology and risk factors for stress fractures in female athletes. Phys Sportsmed. 2019. [8] CHANGSTROM BG, BROU L, KHODAEE M, et al. Epidemiology of stress fracture injuries among US high school athletes, 2005-2006 through 2012-2013. Am J Sports Med. 2015;43(1): 26-33. [9] SCHWARTZ O, MALKA I, OLSEN CH, et al. Overuse injuries among female combat warriors in the israeli defense forces: a cross-sectional study. Mil Med. 2018;183(11-12):e610-e616. [10] DAO D, SODHI S, TABASINEJAD R, et al. Serum 25-Hydroxyvitamin D Levels and Stress Fractures in Military Personnel: A Systematic Review and Meta-analysis. Am J Sports Med.2015;43(8):2064-2072. [11] SAUNIER J, CHAPURLAT R. Stress fracture in athletes. Joint Bone Spine. 2018; 85(3): 307-310. [12] GREASER MC. Foot and ankle stress fractures in athletes. Orthop Clin North Am. 2016;47(4): 809-822. [13] 高飞.应力性骨折早期诊断技术研究进展[J].中华灾害救援医学, 2017,5(1):51-54. [14] WRIGHT AA, HEGEDUS EJ, LENCHIK L, et al. Diagnostic accuracy of various imaging modalities for suspected lower extremity stress fractures: a systematic review with evidence-based recommendations for clinical practice. Am J Sports Med. 2016;44(1):255-263. [15] VASIKARAN S, COOPER C, EASTELL R, et al. International osteoporosis foundation and international federation of clinical chemistry and laboratory medicine position on bone marker standards in osteoporosis. Clin Chem Lab Med.2011;49(8): 1271-1274. [16] WATERMAN BR, GUN B, BADER JO, et al. Epidemiology of Lower Extremity Stress Fractures in the United States Military. Mil Med. 2016;181(10): 1308-1313. [17] VAN DER WORP MP, DE WIJER A, VAN CINGEL R, et al. The 5- or 10-km marikenloop run: a prospective study of the etiology of running-related injuries in women. J Orthop Sports Phys Ther. 2016;46(6):462-470. [18] VAN POPPEL D, SCHOLTEN-PEETERS GGM, VAN MIDDELKOOP M, et al. Risk models for lower extremity injuries among short- and long distance runners: A prospective cohort study. Musculoskelet Sci Pract. 2018;36:48-53. [19] DAMSTED C, PARNER ET, SORENSEN H, et al. ProjectRun21: Do running experience and running pace influence the risk of running injury-A 14-week prospective cohort study. J Sci Med Sport. 2019;22(3):281-287. [20] PIHLAJAMAKI H, PARVIAINEN M, KYROLAINEN H, et al. Regular physical exercise before entering military service may protect young adult men from fatigue fractures. BMC Musculoskelet Disord. 2019;20(1):126. [21] KNAPIK JJ, SHARP MA, MONTAIN SJ. Association between stress fracture incidence and predicted body fat in United States Army Basic Combat Training recruits. BMC Musculoskelet Disord. 2018;19(1):161. [22] KRAUSS MR, GARVIN NU, BOIVIN MR, et al. Excess stress fractures, musculoskeletal injuries, and health care utilization among unfit and overweight female army trainees. Am J Sports Med. 2017;45(2):311-316. [23] BECK BR, RUDOLPH K, MATHESON GO, et al. Risk factors for tibial stress injuries: a case-control study. Clin J Sport Med. 2015; 25(3):230-236. [24] GRIER T, CANHAM-CHERVAK M, BUSHMAN T, et al. Minimalist Running Shoes and Injury Risk Among United States Army Soldiers. Am J Sports Med. 2016;44(6):1439-1446. [25] BONANNO DR, LANDORF KB, MUNTEANU SE, et al. Effectiveness of foot orthoses and shock-absorbing insoles for the prevention of injury: a systematic review and meta-analysis. Br J Sports Med. 2017;51(2):86-96. [26] MILGROM C, FINESTONE AS. The effect of stress fracture interventions in a single elite infantry training unit (1983-2015). Bone.2017;103:125-130. [27] JAFARNEZHADGERO A, ALAVI-MEHR SM, GRANACHER U. Effects of anti-pronation shoes on lower limb kinematics and kinetics in female runners with pronated feet: The role of physical fatigue. PLoS One.2019;14(5):e0216818. [28] MONTAIN SJ, MCGRAW SM, ELY MR, et al. A retrospective cohort study on the influence of UV index and race/ethnicity on risk of stress and lower limb fractures. BMC Musculoskelet Disord. 2013;14:135. [29] DOMBROWSKI S, JACOB L, HADJI P, et al. Oral contraceptive use and fracture risk-a retrospective study of 12,970 women in the UK. Osteoporos Int. 2017;28(8):2349-2355. [30] HULME A, NIELSEN RO, TIMPKA T, et al. Risk and Protective Factors for Middle- and Long-Distance Running-Related Injury. Sports Med. 2017;47(5):869-886. [31] HUGHES JM, MCKINNON CJ, TAYLOR KM, et al. Nonsteroidal anti-inflammatory drug prescriptions are associated with increased stress fracture diagnosis in the us army population. J Bone Miner Res. 2019;34(3):429-436. [32] KNAPIK J, MONTAIN SJ, MCGRAW S, et al. Stress fracture risk factors in basic combat training. Int J Sports Med. 2012;33(11): 940-946. [33] BARRACK MT, GIBBS JC, DE SOUZA MJ, et al. Higher incidence of bone stress injuries with increasing female athlete triad-related risk factors: a prospective multisite study of exercising girls and women. Am J Sports Med. 2014;42(4):949-958. [34] TENFORDE AS, CARLSON JL, CHANG A, et al. Association of the female athlete triad risk assessment stratification to the development of bone stress injuries in collegiate athletes. Am J Sports Med. 2017;45(2):302-310. [35] NOSE-OGURA S, YOSHINO O, DOHI M, et al. Risk factors of stress fractures due to the female athlete triad: Differences in teens and twenties. Scand J Med Sci Sports. 2019. [36] PRATHER H, HUNT D, MCKEON K, et al. Are elite female soccer athletes at risk for disordered eating attitudes, menstrual dysfunction, and stress fractures? PM R. 2016; 8(3):208-213. [37] LARSSON D, EKSTRAND J, KARLSSON MK. Fracture epidemiology in male elite football players from 2001 to 2013: 'How long will this fracture keep me out? Br J Sports Med. 2016; 50(12):759-763. [38] BAXTER-JONES AD, FAULKNER RA, FORWOOD MR, et al. Bone mineral accrual from 8 to 30 years of age: an estimation of peak bone mass. J Bone Miner Res.2011;26(8):1729-1739. [39] BULATHSINHALA L, HUGHES JM, MCKINNON CJ, et al. Risk of stress fracture varies by race/ethnic origin in a cohort study of 1.3 million us army soldiers. J Bone Miner Res. 2017;32(7): 1546-1553. [40] POPP KL, HUGHES JM, MARTINEZ-BETANCOURT A, et al. Bone mass, microarchitecture and strength are influenced by race/ethnicity in young adult men and women. Bone. 2017;103: 200-208. [41] POPP KL, XU C, YUAN A, et al. Trabecular microstructure is influenced by race and sex in Black and White young adults. Osteoporos Int. 2019;30(1):201-209. [42] GARNOCK C, WITCHALLS J, NEWMAN P. Predicting individual risk for medial tibial stress syndrome in navy recruits. J Sci Med Sport. 2018;21(6):586-590. [43] DUARTE SOSA D, FINK ERIKSEN E. Women with previous stress fractures show reduced bone material strength. Acta Orthop. 2016;87(6):626-631. [44] KRAUS E, TENFORDE AS, NATTIV A, et al. Bone stress injuries in male distance runners: higher modified female athlete triad cumulative risk assessment scores predict increased rates of injury. Br J Sports Med. 2019;53(4):237-242. [45] ZHAO L, CHANG Q, HUANG T, et al. Prospective cohort study of the risk factors for stress fractures in Chinese male infantry recruits. J Int Med Res. 2016;44(4):787-795. [46] POPP KL, MCDERMOTT W, HUGHES JM, et al. Bone strength estimates relative to vertical ground reaction force discriminates women runners with stress fracture history. Bone. 2017;94: 22-28. [47] CURTIS EM, MOON RJ, HARVEY NC, et al. The impact of fragility fracture and approaches to osteoporosis risk assessment worldwide. Bone. 2017;104:29-38. [48] TENFORDE AS, PARZIALE AL, POPP KL, et al. Low bone mineral density in male athletes is associated with bone stress injuries at anatomic sites with greater trabecular composition. Am J Sports Med. 2018;46(1):30-36. [49] DAVEY T, LANHAM-NEW SA, SHAW AM, et al. Fundamental differences in axial and appendicular bone density in stress fractured and uninjured Royal Marine recruits--a matched case-control study. Bone. 2015;73:120-126. [50] DUCKHAM RL, BROOKE-WAVELL K, SUMMERS GD, et al. Stress fracture injury in female endurance athletes in the United Kingdom: A 12-month prospective study. Scand J Med Sci Sports. 2015;25(6):854-859. [51] NAKAYAMA AT, LUTZ LJ, HRUBY A, et al. A dietary pattern rich in calcium, potassium, and protein is associated with tibia bone mineral content and strength in young adults entering initial military training. Am J Clin Nutr. 2019;109(1):186-196. [52] GAFFNEY-STOMBERG E, NAKAYAMA AT, GUERRIERE KI, et al. Calcium and vitamin D supplementation and bone health in Marine recruits: Effect of season. Bone. 2019;123:224-233. [53] DAVEY T, LANHAM-NEW SA, SHAW AM, et al. Low serum 25-hydroxyvitamin D is associated with increased risk of stress fracture during Royal Marine recruit training. Osteoporos Int. 2016;27(1):171-179. [54] NIEVES JW, MELSOP K, CURTIS M, et al. Nutritional factors that influence change in bone density and stress fracture risk among young female cross-country runners. PM R. 2010;2(8):740-750; quiz 794. [55] MILLER JR, DUNN KW, CILIBERTI LJ JR, et al. Association of vitamin d with stress fractures: a retrospective cohort study. J Foot Ankle Surg. 2016;55(1):117-120. [56] MORAN DS, EVANS R, ARBEL Y, et al. Physical and psychological stressors linked with stress fractures in recruit training. Scand J Med Sci Sports. 2013;23(4):443-450. [57] VARLEY I, HUGHES DC, GREEVES JP, et al. The association of novel polymorphisms with stress fracture injury in Elite Athletes: Further insights from the SFEA cohort. J Sci Med Sport. 2018; 21(6):564-568. [58] TONG JW, KONG PW. Association between foot type and lower extremity injuries: systematic literature review with meta-analysis. J Orthop Sports Phys Ther. 2013;43(10):700-714. [59] NEAL BS, GRIFFITHS IB, DOWLING GJ, et al. Foot posture as a risk factor for lower limb overuse injury: a systematic review and meta-analysis. J Foot Ankle Res. 2014;7(1):55. [60] BULDT AK, FORGHANY S, LANDORF KB, et al. Foot posture is associated with plantar pressure during gait: A comparison of normal, planus and cavus feet. Gait Posture. 2018; 62: 235-240. [61] NUNNS M, HOUSE C, RICE H, et al. Four biomechanical and anthropometric measures predict tibial stress fracture: a prospective study of 1065 Royal Marines. Br J Sports Med. 2016;50(19):1206-1210. [62] SAITA Y, NAGAO M, KAWASAKI T, et al. Range limitation in hip internal rotation and fifth metatarsal stress fractures (Jones fracture) in professional football players. Knee Surg Sports Traumatol Arthrosc. 2018;26(7):1943-1949. [63] MEARDON SA, WILLSON JD, GRIES SR, et al. Bone stress in runners with tibial stress fracture. Clin Biomech (Bristol, Avon). 2015;30(9):895-902. [64] HONDA A, MATSUMOTO M, KATO T, et al. Exercise characteristics influence femoral cross-sectional geometry: a magnetic resonance imaging study in elite female athletes. Osteoporos Int. 2015;26(3):1093-1098. [65] BECKER J, NAKAJIMA M, WU WFW. Factors Contributing to Medial Tibial Stress Syndrome in Runners: A Prospective Study. Med Sci Sports Exerc. 2018;50(10):2092-2100. [66] HAMSTRA-WRIGHT KL, BLIVEN KC, BAY C. Risk factors for medial tibial stress syndrome in physically active individuals such as runners and military personnel: a systematic review and meta-analysis. Br J Sports Med. 2015;49(6):362-369. [67] BULDT AK, ALLAN JJ, LANDORF KB, et al. The relationship between foot posture and plantar pressure during walking in adults: A systematic review. Gait Posture. 2018;62:56-67. [68] LAREAU CR, HSU AR, ANDERSON RB. Return to Play in National Football League Players After Operative Jones Fracture Treatment. Foot Ankle Int. 2016;37(1):8-16. [69] AZEVEDO RR, DA ROCHA ES, FRANCO PS, et al. Plantar pressure asymmetry and risk of stress injuries in the foot of young soccer players. Phys Ther Sport. 2017;24:39-43. [70] DIXON S, NUNNS M, HOUSE C, et al. Prospective study of biomechanical risk factors for second and third metatarsal stress fractures in military recruits. J Sci Med Sport. 2019;22(2): 135-139. [71] RICE H, NUNNS M, HOUSE C, et al. High medial plantar pressures during barefoot running are associated with increased risk of ankle inversion injury in Royal Marine recruits. Gait Posture. 2013;38(4):614-618. [72] QUEEN RM, ABBEY AN, CHUCKPAIWONG B, et al. Plantar loading comparisons between women with a history of second metatarsal stress fractures and normal controls. Am J Sports Med. 2009;37(2):390-395. [73] XU C, SILDER A, ZHANG J, et al. A cross-sectional study of the effects of load carriage on running characteristics and tibial mechanical stress: implications for stress-fracture injuries in women. BMC Musculoskelet Disord. 2017;18(1):125. [74] BARRACK MT, FREDERICSON M, TENFORDE AS, et al. Evidence of a cumulative effect for risk factors predicting low bone mass among male adolescent athletes. Br J Sports Med. 2017; 51(3):200-205. [75] HERBERT A J, WILLIAMS AG, HENNIS PJ, et al. The interactions of physical activity, exercise and genetics and their associations with bone mineral density: implications for injury risk in elite athletes. Eur J Appl Physiol. 2019;119(1):29-47. [76] BONANNO DR, MURLEY GS, MUNTEANU SE, et al. Effectiveness of foot orthoses for the prevention of lower limb overuse injuries in naval recruits: a randomised controlled trial. Br J Sports Med. 2018;52(5):298-302. |
| [1] | Xu Feng, Kang Hui, Wei Tanjun, Xi Jintao. Biomechanical analysis of different fixation methods of pedicle screws for thoracolumbar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1313-1317. |
| [2] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [3] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [4] | Chen Xinmin, Li Wenbiao, Xiong Kaikai, Xiong Xiaoyan, Zheng Liqin, Li Musheng, Zheng Yongze, Lin Ziling. Type A3.3 femoral intertrochanteric fracture with augmented proximal femoral nail anti-rotation in the elderly: finite element analysis of the optimal amount of bone cement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1404-1409. |
| [5] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [6] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [7] | Song Chengjie, Chang Hengrui, Shi Mingxin, Meng Xianzhong. Research progress in biomechanical stability of lateral lumbar interbody fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 923-928. |
| [8] | Xu Yulin, Shen Shi, Zhuo Naiqiang, Yang Huilin, Yang Chao, Li Yang, Zhao Heng, Zhao Lu. Biomechanical comparison of three different plate fixation methods for acetabular posterior column fractures in standing and sitting positions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 826-830. |
| [9] | Cai Qunbin, Zou Xia, Hu Jiantao, Chen Xinmin, Zheng Liqin, Huang Peizhen, Lin Ziling, Jiang Ziwei. Relationship between tip-apex distance and stability of intertrochanteric femoral fractures with proximal femoral anti-rotation nail: a finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 831-836. |
| [10] | Xie Chongxin, Zhang Lei. Comparison of knee degeneration after anterior cruciate ligament reconstruction with or without remnant preservation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 735-740. |
| [11] | Nie Shaobo, Li Jiantao, Sun Jien, Zhao Zhe, Zhao Yanpeng, Zhang Licheng, Tang Peifu. Mechanical stability of medial support nail in treatment of severe osteoporotic intertrochanteric fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 329-333. |
| [12] | Tan Jiachang, Yuan Zhenchao, Wu Zhenjie, Liu Bin, Zhao Jinmin. Biomechanical analysis of elastic nail combined with end caps and wire fixation for long oblique femoral shaft fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 334-338. |
| [13] | Chen Lu, Zhang Jianguang, Deng Changgong, Yan Caiping, Zhang Wei, Zhang Yuan. Finite element analysis of locking screw assisted acetabular cup fixation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 356-361. |
| [14] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Comparison of the advantages and disadvantages of multiple implants in treatment of traumatic dislocation of sternoclavicular joint [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 443-448. |
| [15] | Li Kun, Li Zhijun, Zhang Shaojie, Gao Shang, Sun Hao, Yang Xi, Wang Xing, Dai Lina . A 4-year-old child model of occipito-atlanto-axial joints established by finite element dynamic simulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3773-3778. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||