Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7396-7403.doi: 10.12307/2026.806
Previous Articles Next Articles
Research hotspots and trends of optogenetics in behavioral neuroscience
Liu Yan1, Zuo Qingchun1, Li Weiying1, Wu Xubo2
- 1School of Rehabilitation Medicine of Jiamusi University, Department of Child Health Care/Heilongjiang Provincial Key Laboratory of Pediatric Neurorehabilitation of Third Affiliated Hospital of Jiamusi University, Jiamusi 154002, Heilongjiang Province, China; 2Department of Rehabilitation Medicine, Pudong New Area People’s Hospital, Shanghai 200120, China
-
Received:2025-09-04Revised:2025-12-06Online:2026-10-08Published:2026-02-24 -
Contact:Wu Xubo, MD, Associate professor, Department of Rehabilitation Medicine, Pudong New Area People’s Hospital, Shanghai 200120, China -
About author:Liu Yan, MS, School of Rehabilitation Medicine of Jiamusi University, Department of Child Health Care/Heilongjiang Provincial Key Laboratory of Pediatric Neurorehabilitation of Third Affiliated Hospital of Jiamusi University, Jiamusi 154002, Heilongjiang Province, China Li Weiying, School of Rehabilitation Medicine of Jiamusi University, Department of Child Health Care/Heilongjiang Provincial Key Laboratory of Pediatric Neurorehabilitation of Third Affiliated Hospital of Jiamusi University, Jiamusi 154002, Heilongjiang Province, China Liu Yan and Li Weiying contributed equally to this article. -
Supported by:Investigator-Initiated Trial Program of Shanghai Pudong New Area Health Commission (the Medical and Industrial Integration Program), No. 2025-PWYC-11 (to WXB)
CLC Number:
Cite this article
Liu Yan, Zuo Qingchun, Li Weiying, Wu Xubo. Research hotspots and trends of optogenetics in behavioral neuroscience[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7396-7403.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
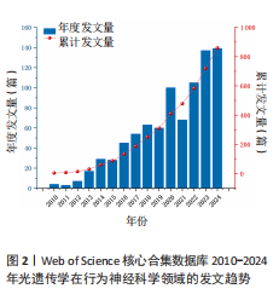
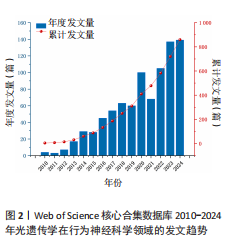
2.1 光遗传学在行为神经科学领域全球发文趋势 2010-2024年间,光遗传学在行为神经科学领域的研究热度持续上升。2010-2024年间,相关研究共发表文献859篇,年发文趋势如图2所示。整体可分为3个阶段:2010-2015年为初始阶段,年发文量较少,长期维持在个位数至30篇以内,尚处于起步探索阶段;2016-2019年进入增长阶段,发文数量稳步上升,从2016年的45篇增至2019年的60篇,光遗传学在神经环路调控与动物行为研究中的潜力逐渐引起重视;自2020年起,研究步入快速发展阶段,年发文量显著攀升,2020年首次突破百篇(n=100),2022年和2024年分别达105篇和139篇,光遗传学在行为神经科学中的应用日益广泛。"
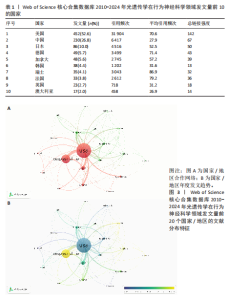
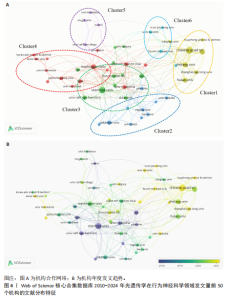
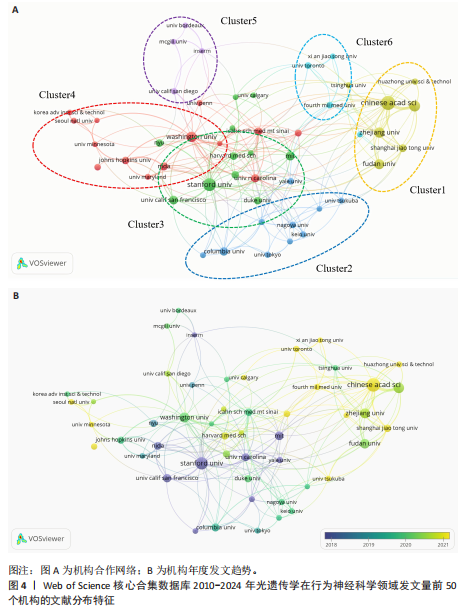
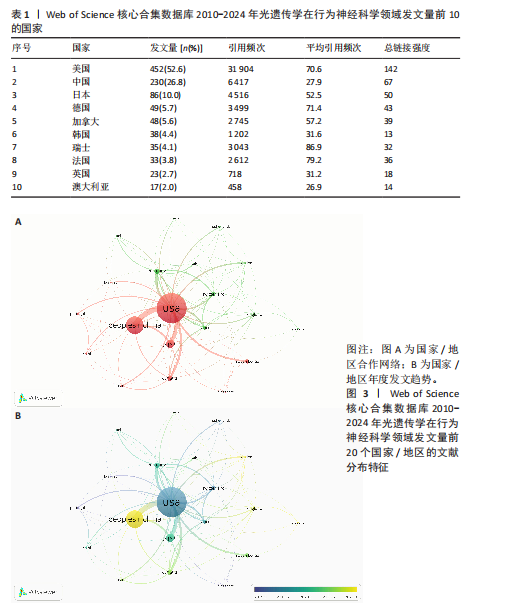
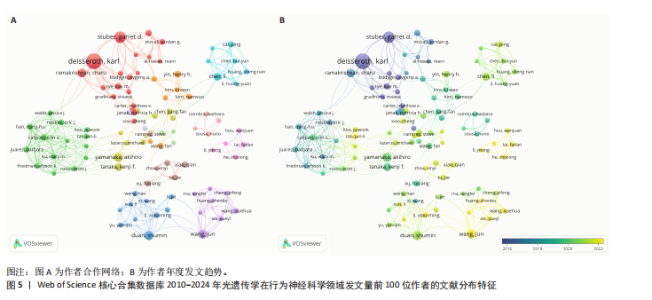
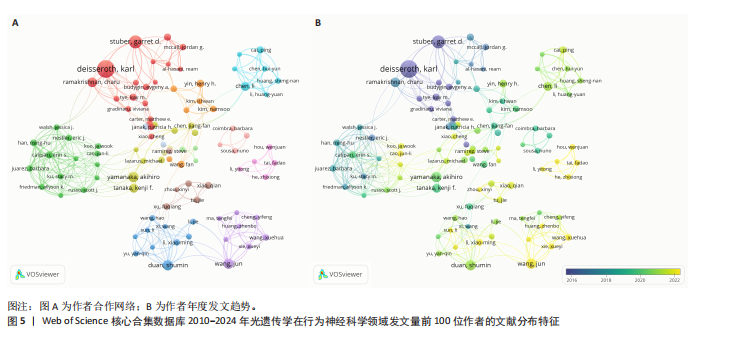
2.2 光遗传学在行为神经科学领域发文国家分析 全球共有46个国家在光遗传学调控行为领域发表了相关研究,美国在该领域的研究产出遥遥领先,共发表452篇文献,占总发文量的52.6%,数量超过其他所有国家的总和,具有显著的引领地位;中国和日本分别以230篇(26.8%)和86篇(10.0%)位列第二与第三,构成亚太地区的主要研究力量。值得注意的是,尽管瑞士、法国和德国的发文量均未超过50篇,但平均被引次数分别为86.9,79.2和71.4,位居全球前三,展现出较高的研究质量与国际影响力。光遗传学在行为神经科学领域发文量前10的国家,见表1。 在形成的6个国家群集中,前20个最多产出的国家之间构建了国际合作网络图(图3A),这些国家之间有着积极的合作,特别是美国和中国之间。从动态和趋势来看,美国、德国、瑞士早在2019年就开展了该领域的研究,2020年后,世界各地的许多其他研究人员开始更加关注这一领域(图3B)。 2.3 光遗传学在行为神经科学领域发文机构分析 在光遗传学与行为神经科学领域,共有834所高校及科研机构发表了相关研究成果。通过对机构间合作网络的可视化分析,共识别出6个主要聚类(图4A),其中,中国科学院(n=51,5.9%,Cluster1)、哥伦比亚大学(n=20,2.3%,Cluster2)、斯坦福大学(n=44,5.1%,Cluster3)、华盛顿大学(n=20,2.3%,Cluster4)、加利福尼亚大学圣迭戈分校(n=12,1.4%,Cluster5)以及第四军医大学(n=13,1.5%,Cluster6)分别为各自聚类中的主要节点。Cluster4为最大聚类,包含14个节点,而Cluster5与Cluster6为最小聚类,仅包含4个节点,其余聚类的节点数量分别为:Cluster3(11个节点)、Cluster2(10个节点)和Cluster1(7个节点)。此外,斯坦福大学、麻省理工学院、耶鲁大学和加利福尼亚大学旧金山分校等机构在2018年之前率先开展了光遗传学在行为神经科学中的研究,推动了该领域的早期发展。相比之下,近年来,中国科学院、中国科学技术大学、浙江大学和上海交通大学等机构在该领域取得了显著进展(图4B)。 2.4 光遗传学在行为神经科学领域发文作者分析 共有5 525位作者在光遗传学与行为神经科学领域发表了相关成果,前100名合作生产作者的网络地图和叠加可视化形成了几个集群(图5)。在这些作者中,有5个科研团队在全球相关文献中贡献较为显著,其中,Deisseroth Karl(29篇,占比3.4%)和Stuber Garret D(18篇,占比2.1%)是合作网络中最活跃的核心作者,他们分别隶属于斯坦福大学和华盛顿大学,形成了最大合作集群;段树明(12篇,占比1.4%)来自浙江大学脑科学与脑医学学院,构成了第三大集群的主要成员;王军(12篇,占比1.4%)与Yamanaka,Akihiro(12篇,占比1.4%)分别来自德克萨斯农工大学健康科学中心和中国脑科学研究院,共同构成了第四、五核心节点。此外,来自美国的Ramakrishnan,Charu迄今已发布了11篇文献(表2)。 2.5 光遗传学在行为神经科学领域发文期刊分析 在关于光遗传学调控行为研究的领域,共有125种期刊发表了相关文献,产出最多的期刊是《Journal of Neuroscience》(78篇,占比9.0%),而被引次数最多的期刊是《Neuron》(53篇,占比6.2%),共计被引6 433次;平均被引次数最高的期刊是《Nature》(9篇,占比1.0%),"
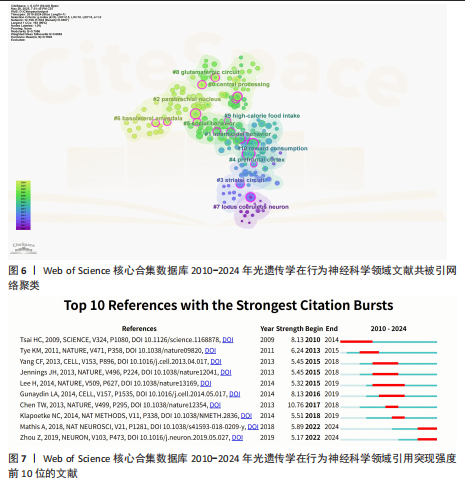
文献的平均被引次数为362.9次。表3列出了产出最高、被引最多以及平均被引次数最高的前10期刊。 2.6 光遗传学在行为神经科学领域发文共被引分析 在859篇论文中,共被引排名前10篇出版物列于表4。第一篇论文由CHEN 等[13]于2013年发表在《Nature》上,报告了一种新的GCaMP6荧光蛋白可超灵敏监测神经活动,引用次数为68次,远高于第二篇论文(53次引用)[14]。在这10篇文献中,有4篇发表在《Nature》杂志上,主题包括超敏感荧光蛋白(GCaMP6)在成像神经元活动中的应用[13]、调控下丘脑腹内侧核Esr1阳性神经元诱导爬跨和攻击行为[15]、调控中脑神经环路诱导防御行为以及调控杏仁核延伸部独立神经环路驱动截然不同的动机状态[16-17]。最新论文发表于2019年,被引用13次[18]。 使用CiteSpace对参考文献共被引网络进行可视化分析(图6)和文献突现(图7),共识别出11个共引聚类,代表了该领域内具有代表性的研究方向与热点主题。最大聚类为Cluster 0“central processing”,聚焦于中枢神经系统中的信息整合与调控机制。Cluster1 “infanticidal behavior”、Cluster5“social behavior”与Cluster6“basolateral amygdala”分别涉及攻击性行为、社交行为以及情绪调节相关神经环路的研究。Cluster2“parabrachial nucleus”、Cluster4“prefrontal cortex”与Cluster7“locus coeruleus neuron”则关注于感觉传导、认知调控和觉醒/应激调节机制。近年来,Cluster8“glutamatergic circuit”与Cluster9“high-calorie food intake”逐渐成为新兴热点,分别指向兴奋性神经环路及摄食行为调控的研究方向。Cluster10“reward consumption”则关注奖赏摄取过程中的神经机制,而Cluster3“striatal circuit”主要聚焦于运动控制与多巴胺系统,代表了该领域的早期研究主题。从时间演化趋势来看,早期研究多集中于“striatal "
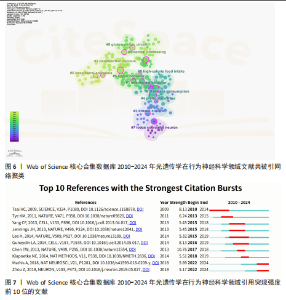
circuit”与“locus coeruleus neuron”等基础神经环路的功能探讨;近年来,研究重心逐步转向“glutamatergic circuit”“high-calorie food intake”“reward consumption”等与复杂行为调控及病理机制相关的方向,反映该领域正在从基础神经机制研究向行为调节与疾病模型转化的趋势。文献突现显示,光遗传学工具(爆发强度8.13)引发初始爆发[19],钙成像技术(爆发强度10.76)在4年延迟后成为最强爆发点[13],深度学习行为分析工具(爆发强度5.89)则推动当前多模态融合研究[20]。技术类文献占据突现强度前3名。自2022年起,该领域进入神经计算建模与行为量化分析的交叉创新阶段,AI驱动的高通量神经行为关联研究将成为未来核心方向。 2.7 光遗传学在行为神经科学领域文献关键词分析 在859篇文献中,共提取出3 089个关键词,出现频率前100的关键词构建关键词共现网络图谱,形成3个聚类(图8A)。“neurons” “prefrontal cortex”与“optogenetics”为网络中节点权重最大的关键词。图8B,C分别呈现了2019-2021年间前100个高频关键词的叠加可视化图及2010-2024年间前20个关键词爆发强度,揭示了近年来研究热点的时间演化趋势。 Cluster1以“neurons”为核心,代表光遗传学在调控特定脑区神经活动及情绪行为中的应用方向。以小鼠和大鼠等为动物模型,聚焦“amygdala hypothalamus”及“central amygdala”等情绪相关脑区,探讨它在恐惧“fear”“anxiety”“pain”等行为调控中的神经机制。Cluster2以“prefrontal cortex”核心,反映奖赏系统与神经递质调控机制的研究,关键词包括“dopamine”“reward”“activation”“ventral tegmental area”“nucleus accumbens”“glutamate”与“plasticity”等,旨在阐明中脑-边缘通路在奖赏学习、成瘾行为及动机调节中的作用,构建神经环路与行为功能之间的联系,为精神疾病机制研究提供基础。Cluster3以“optogenetics”为核心,聚焦前额皮质功能调控及其在神经精神障碍中的潜力,关键词包括“medial prefrontal cortex”“inhibitory neurons”“neuropathic pain”“sex differences”等,强调前额皮质与边缘系统之间的交互机制及其在情绪调节、慢"
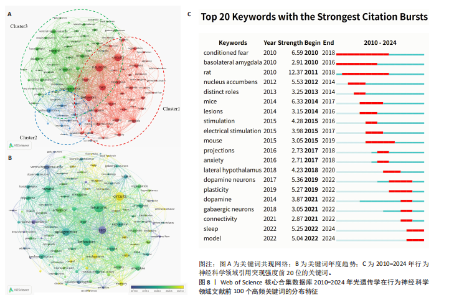
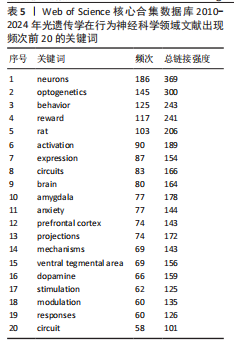
性疼痛、性别差异相关疾病模型中的关键作用。 时间维度上,早期研究多集中于“neurons”“reward”与“behavior”等基础主题;近年来,“circuits”“glutamatergic”“feeding behavior”与“plasticity”等关键词逐渐活跃,光遗传学正逐步融入认知调节、情绪障碍与疾病模型等研究领域。关键词突现显示,2010-2014年聚焦边缘系统(基底外侧杏仁核、伏隔核)与条件性恐惧机制,以大鼠模型为主导(强度12.37);2015-2018年依托电刺激技术深化神经投射与功能分化研究,小鼠模型广泛普及(突现持续5年),并拓展至焦虑及下丘脑调控;2019-2021年以多巴胺系统(神经元/递质)和神经可塑性为核心(强度> 5.0),结合γ-氨基丁酸能神经元等细胞特异性研究,揭示环路调控机制;2022年后突现睡眠神经机制(强度5.25)与计算模型构建(强度5.04),标志神经调控技术(光遗传学/化学遗传学)与计算神经科学的深度融合,共同推动脑功能-行为的系统性研究。 表5总结了共现频率排名前20的关键词及其总链接强度,出现频率较高的关键词如“neurons”“optogentics”“behavior”“reward”和“rat”,反映出该研究领域的主要热点集中在神经元功能、动物行为及奖赏机制等方向。"

| [1] EMILIANI V, ENTCHEVA E, HEDRICH R, et al. Optogenetics for light control of biological systems. Nat Rev Methods Primers. 2022;2:55. [2] JOURJINE N, HOEKSTRA HE. Expanding evolutionary neuroscience: insights from comparing variation in behavior. Neuron. 2021; 109(7):1084-1099. [3] HARRIOTT AM, CHUNG DY, UNER A, et al. Optogenetic spreading depression elicits trigeminal pain and anxiety behavior. Ann Neurol. 2021;89(1):99-110. [4] THOMAS CS, MOHAMMADKHANI A, RANA M, et al. Optogenetic stimulation of lateral hypothalamic orexin/dynorphin inputs in the ventral tegmental area potentiates mesolimbic dopamine neurotransmission and promotes reward-seeking behaviours. Neuropsychopharmacology. 2022; 47(3):728-740. [5] HU RK, ZUO Y, LY T, et al. An amygdala-to-hypothalamus circuit for social reward. Nat Neurosci. 2021;24(6):831-842. [6] WATANABE H, SANO H, CHIKEN S, et al. Forelimb movements evoked by optogenetic stimulation of the macaque motor cortex. Nat Commun. 2020;11(1):3253. [7] SPREEN A, ALKHOURY D, WALTER H, et al. Optogenetic behavioral studies in depression research: a systematic review. Iscience. 2024; 27(5):109776. [8] JIANG CC, LIN LS, LONG S, et al. Signalling pathways in autism spectrum disorder: mechanisms and therapeutic implications. Signal Transduct Target Ther. 2022;7(1):229. [9] YU C, CASSAR IR, SAMBANGI J, et al. Frequency-specific optogenetic deep brain stimulation of subthalamic nucleus improves parkinsonian motor behaviors. J Neurosci. 2020;40(22):4323-4334. [10] 陈悦,陈超美,刘则渊,等.CiteSpace 知识图谱的方法论功能[J].科学学研究,2015,33(2): 2422-253. [11] CHEN C. CiteSpace II: detecting and visualizing emerging trends and transient patterns in scientific literature. J Am Soc Inf Sci Technol. 2006;57(3):359-377. [12] VAN ECK NJ, WALTMAN L. Citation-based clustering of publications using CitNetExplorer and VOSviewer. Scientometrics. 2017;111(2):1053-1070. [13] CHEN TW, WARDILL TJ, SUN Y, et al. Ultra-sensitive fluorescent proteins for imaging neuronal activity. Nature. 2013;499(7458):295-300. [14] TYE KM, PRAKASH R, KIM SY, et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature. 2011;471(7338):358-362. [15] LEE H, KIM DW, REMEDIOS R, et al. Scalable control of mounting and attack by ESR1+ neurons in the ventromedial hypothalamus. Nature. 2014; 509(7502):627-632. [16] TOVOTE P, ESPOSITO MS, BOTTA P, et al. Midbrain circuits for defensive behaviour. Nature. 2016; 534(7606):206-212. [17] JENNINGS JH, SPARTA R, STAMATAKIS AM, et al. Distinct extended amygdala circuits for divergent motivational states. Nature. 2013;496(7444):224-228. [18] ZHOU Z, LIU X, CHEN S, et al. A VTA GABAergic neural circuit mediates visually evoked innate defensive responses. Neuron. 2019;103(3):473-488.e6. [19] TSAI HC, ZHANG F, ADAMANTIDIS A, et al. Phasic firing in dopaminergic neurons is sufficient for behavioral conditioning. Science (new York, N.Y.). 2009;324(5930):1080-1084. [20] MATHIS A, MAMIDANNA P, CURY KM, et al. DeepLabCut: markerless pose estimation of user-defined body parts with deep learning. Nat Neurosci. 2018;21(9):1281-1289. [21] DONTHU N, KUMAR S, MUKHERJEE D, et al. How to conduct a bibliometric analysis: an overview and guidelines. J Bus Res. 2021;133:285-296. [22] GRILLNER S, IP N, KOCH C, et al. Worldwide initiatives to advance brain research. Nat Neurosci. 2016;19(9):1118-1122. [23] KLAPOETKE NC, MURATA Y, KIM SS, et al. Independent Optical Excitation of Distinct Neural Populations. Nat Methods. 2014;11(3):338-346. [24] TERVO DGR, HWANG BY, VISWANATHAN S, et al. A Designer AAV Variant Permits Efficient Retrograde Access to Projection Neurons. Neuron. 2016;92(2):372-382. [25] DEISSEROTH K. Optogenetics: 10 years of microbial opsins in neuroscience. Nat Neurosci. 2015;18(9):1213-1225. [26] TYE KM, PRAKASH R, KIM SY, et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature. 2011;471(7338):358-362. [27] GUNAYDIN LA, GROSENICK L, FINKELSTEIN JC, et al. Natural neural projection dynamics underlying social behavior. Cell. 2014;157(7):1535-1551. [28] TOVOTE P, FADOK JP, LÜTHI A. Neuronal circuits for fear and anxiety. Nat Rev Neurosci. 2015;16(6):317-331. [29] JENNINGS JH, UNG RL, RESENDEZ SL, et al. Visualizing hypothalamic network dynamics for appetitive and consummatory behaviors. Cell. 2015;160(3):516-527. [30] ZONG W, OBENHAUS HA, SKYTØEN ER, et al. Large-scale two-photon calcium imaging in freely moving mice. Cell. 2022;185(7):1240-1256.e30. [31] VAN BEEST EH, BIMBARD C, FABRE JM J, et al. Tracking neurons across days with high-density probes. Nat Methods. 2025;22(4):778-787. [32] TYE KM. Neural circuit motifs in valence processing. Neuron. 2018;100(2):436-452. [33] ZHANG Q, LI T, XU M, et al. Application of optogenetics in neurodegenerative diseases. Cell Mol Neurobiol. 2024;44(1):57. [34] LI H, ZHAO Z, JIANG S, et al. Brain circuits that regulate social behavior. Mol Psychiatry. 2025; 30(7):3240-3256. [35] SUK K. Uncovering optogenetic and chemogenetic induction of cognitive deficits: efficient techniques for manipulating and observing specific neural activities. Neural Regen Res. 2026;21(1):304-305. [36] DENG S, SHU S, ZHAI L, et al. Optogenetic stimulation of mPFC alleviates white matter injury‐related cognitive decline after chronic ischemia through adaptive myelination. Adv Sci. 2023;10(5):2202976. [37] DAUTAN D, MONAI A, MALTESE F, et al. Cortico-cortical transfer of socially derived information gates emotion recognition. Nat Neurosci. 2024; 27(7):1318-1332. |
| [1] | Zhu Xiaolong, Zhang Wei, Yang Yang. Visualization analysis of research hotspots and cutting-edge information in the field of intervertebral disc regeneration and repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2391-2402. |
| [2] | Wen Fayan, Li Yan, Qiang Tianming, Yang Chen, Shen Linming, Li Yadong, Liu Yongming. Unilateral biportal endoscopic technology for treatment of lumbar degenerative diseases: global research status and changing trends [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(9): 2380-2390. |
| [3] | Yang Zeyu, Zhi Liang, Wang Jia, Zhang Jingyi, Zhang Qingfang, Wang Yulong, Long Jianjun. A visualized analysis of research hotspots in high-frequency repetitive transcranial magnetic stimulation from the macroscopic perspective [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1320-1330. |
| [4] | Zhang Jiuxuan, Zhang Jinnan, Sui Xiaofan, Pei Xiaxia, Wei Jianhong, Su Qiang, Li Tian. Effects of ammonia poisoning on cognitive behavior and hippocampal synaptic damage in mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1122-1128. |
| [5] | Zhang Jingyi, Zhi Liang, Yang Zeyu, Li Yaning, Hu Jia, Wang Jia, Wang Yulong, Long Jianjun. Extracorporeal shock wave therapy: current research status, hotspots, and trends [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7410-7417. |
| [6] | Guo Jun, Lu Zheng, Yu Jinling, Hao Yuanyuan, Liu Kaishun, Liu Xuexia, Huang Yourong. Oxidative stress and osteoporosis: a bibliometric analysis of literature from SCI core database [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7428-7436. |
| [7] | Wang Feifei, Wang Zhennan. Scientometric deconstruction of developmental dynamics in upper-limb rehabilitation robotics: evidence network analysis via CiteSpace [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6400-6409. |
| [8] | Wang Fei, Wang Hangping, Dai Rongjuan, Gong Lingxuan. Visual analysis of the research literature on plantar fasciitis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6410-6420. |
| [9] | Shi Gaolong, Ge Caijun, Chen Jianpeng, Wang Yuanbin, Fan Zelin, Yan Jun, Wang Qianliang. Mechanism by which the paraventricular nucleus of the hypothalamus is involved in chronic pain and anxiety in mice with lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5707-5715. |
| [10] | Pei Xiaxia, Li Tian, Zhang Yanli, Gao Yanping, Su Qiang. A novel treadmill-based method for assessing learning and memory in mice [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4694-4701. |
| [11] | Yang Jiangxi, Li Huangyan, Zhang Yeting, Yu Zuoyin . Research hotspots and thematic evolution in the field of exercise interventions for multiple sclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4771-4781. |
| [12] | Li Kanglin, Jiang Yongdong, Wu Yufeng. Visualization analysis of piriformis syndrome: research trends and hotspots [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2886-2895. |
| [13] | Wang Jiaying, Xu Chun, Mayila · Abudukelimu. Global research status, trends and hotspots of anxiety/depression in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(11): 2920-2932. |
| [14] | Jiao Jingya, Zhang Yeting. Analysis of thematic evolution pathways in the field of physical activity and neurogenesis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(10): 2653-2661. |
| [15] | Wang Yida, Liu Jun, Wang Xiaoling, Wang Liyan, Yang Chengru, Zhang Xuexiao. Effects of wearable electronic device-based interventions on physical activity and sedentary behavior in healthy adolescents: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1693-1704. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||