Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7287-7296.doi: 10.12307/2026.822
Previous Articles Next Articles
Differential proteomic analysis of exercise-induced and pathological cardiac hypertrophy models in mice
Qin Di1, 2, Qin Xuelin2, 3, Li Zhu1, Ye Jiachi2, 3, Chen Gan4, Lin Yi5, Peng Yong2, 3
- 1School of Sports and Health, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China; 2Jiangsu Sports and Health Engineering Collaborative Innovation Center, Nanjing 210014, Jiangsu Province, China; 3Sports Science Research Institute, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China; 4School of Physical Education, Hunan Normal University, Changsha 410012, Hunan Province, China; 5State Owned Assets Management Office, Nanjing University of Chinese Medicine, Nanjing 210023, Jiangsu Province, China
-
Received:2025-08-14Revised:2025-12-08Online:2026-10-08Published:2026-02-10 -
Contact:Peng Yong, PhD, Senior experimentalist, Master’s supervisor, Jiangsu Sports and Health Engineering Collaborative Innovation Center, Nanjing 210014, Jiangsu Province, China; Sports Science Research Institute, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China -
About author:Qin Di, PhD, Associate professor, Master’s supervisor, School of Sports and Health, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China; Jiangsu Sports and Health Engineering Collaborative Innovation Center, Nanjing 210014, Jiangsu Province, China -
Supported by:Major Project of Basic Science (Natural Science) Research in Jiangsu Higher Education Institutions, No. 25KJA180002 (to QD); Major Project of Basic Science (Natural Science) Research in Jiangsu Higher Education Institutions, No. 25KJA180003 (to PY); Young and Middle-aged Academic Leaders of “Blue Project” in Jiangsu Province (to QD); National Level Project of Innovation and Entrepreneurship Training Program for College Students in Jiangsu Province, No. 202510330021 (to LZ); The "1+1" Excellent Academic Team Project of Nanjing Sport Institute, No. XSTD202406 (to YJC)
CLC Number:
Cite this article
Qin Di, Qin Xuelin, Li Zhu, Ye Jiachi, Chen Gan, Lin Yi, Peng Yong. Differential proteomic analysis of exercise-induced and pathological cardiac hypertrophy models in mice[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7287-7296.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
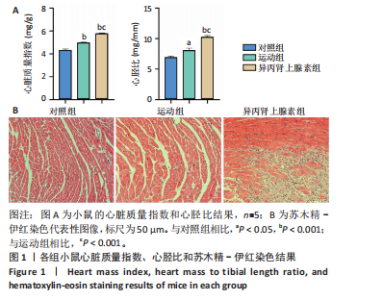
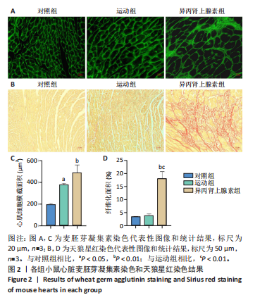
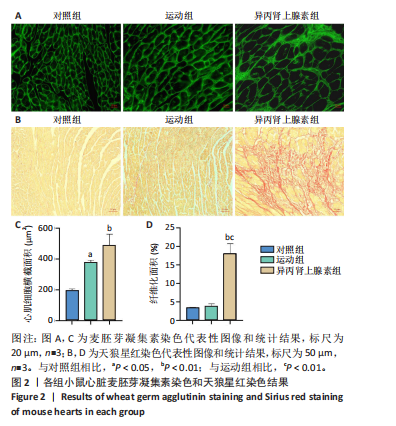
2.1 实验动物数量分析 纳入 21只小鼠,随机分为对照组、运动组、异丙肾上腺素组,每组7只。因为小鼠有个体差异,评估小鼠心脏质量指数和心胫比时,每组选择5只;另外每组小鼠中选取3只进行串联质量标签定量蛋白质组学检测之前,采用苏木精-伊红染色、麦胚芽凝集素染色和天狼星红染色确定造模成功。 2.2 运动性和病理性心脏肥大造模结果 2.2.1 小鼠心脏质量指数、心胫比和苏木精-伊红染色的比较 结果显示:运动组与对照组相比,心脏质量指数和心胫比均显著升高(P < 0.001,P < 0.05)。异丙肾上腺素组与对照组相比,心脏质量指数和心胫比也显著升高(P < 0.001)。异丙肾上腺素组与运动组相比,心脏质量指数和心胫比显著升高(P < 0.001)。苏木精-伊红染色结果显示,运动组与对照组的心肌细胞排列整齐有序,未发现明显的心肌细胞损伤。与对照组相比,异丙肾上腺素组小鼠心肌细胞排列紊乱,观察到大量炎症细胞浸润,心肌细胞损伤明显,见图1。上述结果提示,运动和异丙肾上腺素均可以诱导小鼠心脏肥大,异丙肾上腺素诱导的病理性心脏肥大显著大于运动诱导的生理性心脏肥大。运动组和异丙肾上腺素组小鼠在心脏组织学上存在显著差异,异丙肾上腺素组的心脏表现出明显的病理性损伤。 2.2.2 小鼠心脏麦胚芽凝集素染色和天狼星红染色的比较 麦胚芽凝集素染色结果显示,与对照组相比,运动组和异丙肾上腺"
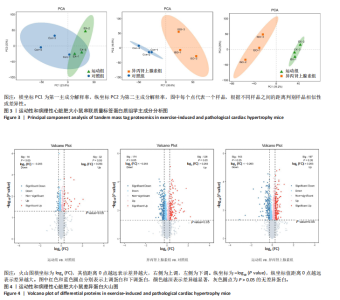
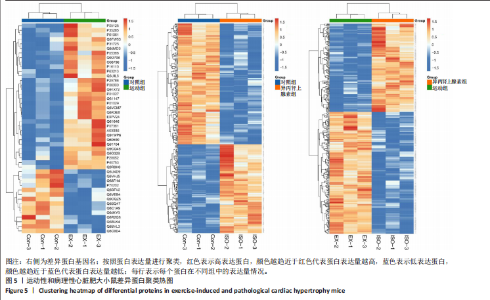
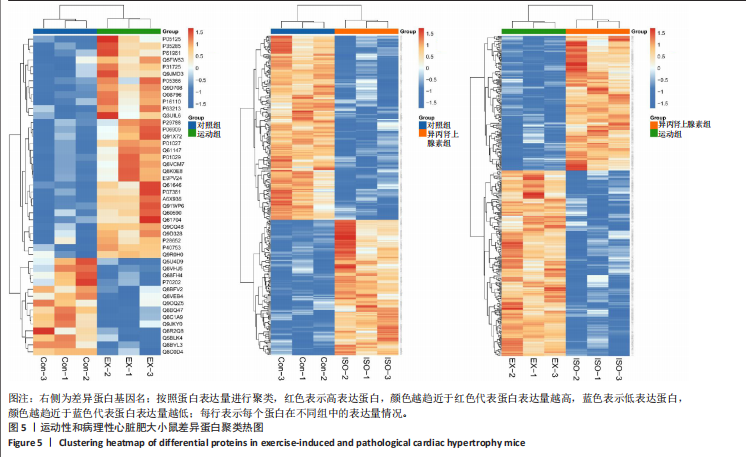
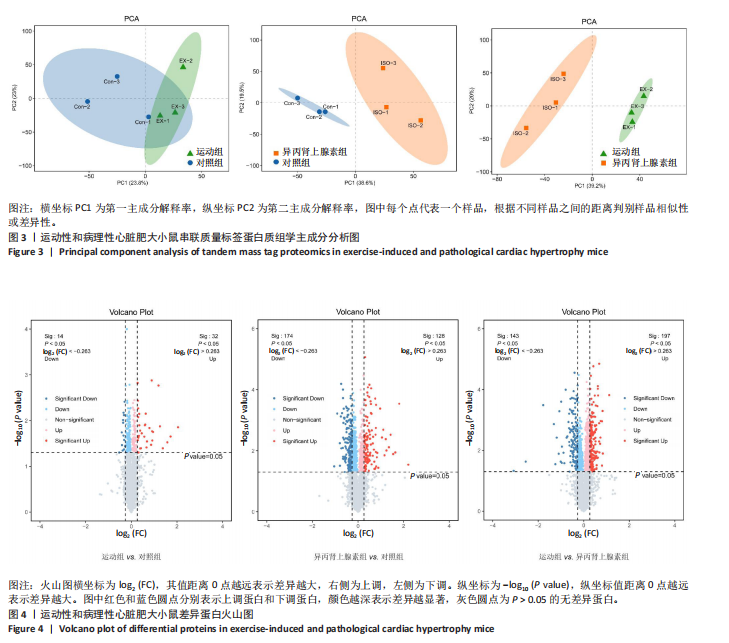
2.3 串联质量标签蛋白质组学分析结果 2.3.1 主成分分析 样品经液相色谱-串联质谱检测、搜库后,进行蛋白定性、定量分析,得到肽段总数为35 279个,鉴定到的蛋白总数为4 796个。利用数据库检索得到原始数据后,按照Score Sequest HT > 0和unique peptide≥1的标准筛选可信蛋白,去除空白值。利用可信蛋白的表达量进行主成分分析,从不同维度展现样品间的关系,每个点代表一个分组实验中的一次重复,不同颜色区分不同分组。从图中可以直观看出同一组样品之间差异很小,不同组样品之间差异显著,见图3。 2.3.2 差异表达蛋白的筛选和火山图 在可信蛋白基础上,以FC≥1.2或FC≤1/1.2且P < 0.05为标准筛选显著差异表达蛋白。运动组和对照组相比,有46个差异表达蛋白,其中,上调蛋白32个,下调蛋白14个;异丙肾上腺素组和对照组相比,有302个差异表达蛋白,其中,上调蛋白128个,下调蛋白174个;运动组和异丙肾上腺素组相比,有340个差异表达蛋白,其中,上调蛋白197个,下调蛋白143个。火山图展示了各组间的蛋白质显著性差异,横坐标为log2 (FC),纵坐标为-log10 (P value), 红色和蓝色圆点分别表示上调蛋白和下调蛋白,颜色越深表示差异越显著,灰色圆点为无差异蛋白,见图4。 2.3.3 差异表达蛋白的聚类分析 采用层次聚类对各组之间的差异表达蛋白质进行分组归类,以热图形式展示,横坐标为样品信息,纵坐标为显著性差异表达蛋白,红色代表显著性上调的蛋白质,蓝色代表显著性下调的蛋白质。从图中可以看出,显著差异蛋白质可以有效地将各个样品分开,同一组样品通过聚类出现在同一簇中,见图5。 2.3.4 差异表达蛋白的韦恩分析 韦恩图被用于分析各组中差异蛋白的特性和共性,发现运动组vs.对照组和异丙肾上腺素组vs.对照组两组差异表达蛋白中,交集的蛋白数有9个:过氧化物酶酰基辅酶A氧化酶1(peroxisomal acyl-coenzyme A oxidase 1,Acox1)、半乳糖凝集素3(galectin-3,Gal-3)、丝氨酸蛋白酶抑制剂A3N(serine protease inhibitor A3N,Serpina3n)、间-α-胰蛋白酶抑制因子重链3(inter-alpha-trypsin inhibitor heavy chain 3,Itih3)、Itih4、血红素结合蛋白(haptoglobin,Hp)、血清淀粉样蛋白A1(serum amyloid A-1 protein,Saa1)、骨髓基质抗原"

2(bone marrow stromal antigen 2,Bst2)、微管亲和力调节激酶1 (microtubule affinity-regulating kinase 1,Mark1),其中,半乳糖凝集素3、丝氨酸蛋白酶抑制剂A3N、间-α-胰蛋白酶抑制因子重链3、间-α-胰蛋白酶抑制因子重链4、血红素结合蛋白、血清淀粉样蛋白A1在两组差异表达蛋白中均显著上调,骨髓基质抗原2和微管亲和力调节激酶1在两组差异表达蛋白中均显著下调,而过氧化物酶酰基辅酶A氧化酶1在运动组vs.对照组差异表达蛋白中显著上调,在异丙肾上腺素组vs.对照组差异表达蛋白中显著下调。运动组vs.对照组、异丙肾上腺素组vs.对照组以及运动组vs.异丙肾上腺素组3组差异表达蛋白中,交集的蛋白数有2个即过氧化物酶酰基辅酶A氧化酶1和半乳糖凝集素3,半乳糖凝集素3在运动组vs.对照组中上调1.31倍,在异丙肾上腺素组vs.对照组中上调3.11倍,在运动组vs.异丙肾上腺素组中下调0.42倍,见图6。"

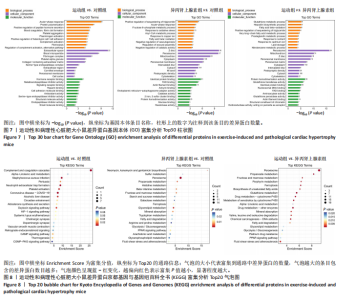
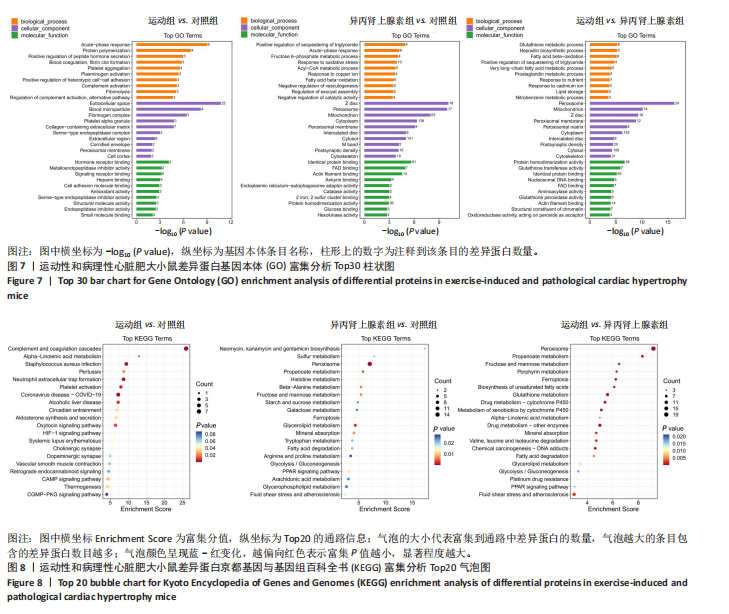
2.3.5 差异表达蛋白的基因本体富集分析 对差异表达蛋白进行基因本体富集分析,运动组vs.对照组差异蛋白富集结果显示,生物过程主要富集在急性期反应(acute-phase response)、蛋白质聚合(protein polymerization)和肽激素分泌的正向调节(positive regulation of peptide hormone secretion)等方面;细胞组分主要富集在细胞外间隙(extracellular space)、血液微粒(blood microparticle)和纤维蛋白原复合物(fibrinogen complex)等方面;分子功能主要富集在激素受体结合(hormone receptor binding)、金属内肽酶抑制剂活性(metalloendopeptidase inhibitor activity)和信号受体结合(signaling receptor binding)等方面。异丙肾上腺素组vs.对照组差异蛋白富集结果显示,在生物过程中,甘油三酯螯合的正向调节(positive regulation of sequestering of triglyceride)、急性期反应和果糖6-磷酸代谢过程(fructose 6-phosphate metabolic process)等功能类别富集度最高;在细胞组分中,Z盘(Z disc)、过氧化物酶体(peroxisome)和线粒体(mitochondrion)等功能类别富集度最高;在分子功能中,同种蛋白结合(identical protein binding)、黄素腺嘌呤二核苷酸结合(FAD binding)和肌动蛋白丝结合(actin filament binding)等功能类别富集度最高。运动组vs.异丙肾上腺素组差异蛋白富集结果显示,生物过程主要富集在谷胱甘肽代谢过程(glutathione metabolic process)、羟环氧二十碳三烯酸生物合成过程(hepoxilin biosynthetic process)和脂肪酸β-氧化(fatty acid beta-oxidation)等方面;细胞组分主要富集在过氧化物酶体、线粒体和Z盘等方面;分子功能主要富集在蛋白质同源二聚化活性(protein homodimerization activity)、谷胱甘肽转移酶活性(glutathione transferase activity)和同种蛋白结合等方面,见图7。上述结果提示,运动性心脏肥大可能涉及炎症或应激反应、激素调节,病理性心脏肥大可能涉及代谢异常、细胞结构改变,心脏两种肥大模型可能涉及抗氧化和能量代谢的差异。 2.3.6 差异表达蛋白的京都基因与基因组百科全书通路分析 对差异表达蛋白质进行京都基因与基因组百科全书通路富集分析,横坐标Enrichment Score为富集分值,纵坐标为top20的通路信息,气泡大小代表代谢通路上富集的差异蛋白数目,气泡颜色越红表示P值越小,对应代谢通路富集度的显著性水平越高。在运动组vs.对照组比较组中,补体和凝血级联反应(complement and coagulation cascades)通路富集最显著且富集的差异蛋白数目最多(7个),差异蛋白在中性粒细胞胞外陷阱形成(neutrophil extracellular trap formation)、血小板活化(platelet activation)、催产素信号通路(oxytocin signaling pathway)、环腺苷酸信号通路(cAMP signaling pathway)等通路中也显著富集。在异丙肾上腺素组vs.对照组比较组中,过氧化物酶体(peroxisome)通路富集最显著且富集的差异蛋白数目最多(14个),差异表达蛋白还涉及甘油酯代谢(glycerolipid metabolism)、丙酸代谢(propanoate metabolism)、β-丙氨酸代谢(beta-Alanine metabolism)、果糖和甘露糖代谢(fructose and mannose metabolism)、矿物吸收(mineral absorption)、过氧化物酶体增殖物激活受体信号通路[(peroxisome proliferator-activated receptor,PPAR) signaling pathway]、铁死亡(ferroptosis)等通路。在运动组vs.异丙肾上腺素组比较组中,过氧化物酶体通路富集最显著且富集的差异蛋白数目最多(19个),差异表达蛋白还涉及谷胱甘肽代谢(glutathione metabolism)、丙酸代谢、药物代谢-细胞色素P450(drug metabolism - cytochrome P450)、铁死亡、细胞色素P450对外源性物质代谢(metabolism of xenobiotics by cytochrome P450)、果糖和甘露糖代谢等通路,见图8。上述结果表明,运动组vs.对照组比较组中补体和凝血级联通路显著,可能与炎症和止血有关,异丙肾上腺素组vs.对照组比较组中过氧化物酶体通路突出,可能涉及脂代谢异常,运动组vs.异丙肾上腺素组比较组中过氧化物酶体通路依然显著,同时谷胱甘肽代谢和细胞色素P450通路出现,提示心脏两种肥大模型在代谢解毒和抗氧化方面的差异。"
| [1] AGABITI-ROSEI E, MUIESAN ML. Cardiac hypertrophy and hypertension. Curr Opin Nephrol Hypertens. 1998;7(2):211-216. [2] MARTIN TG, JUARROS MA, LEINWAND LA. Regression of cardiac hypertrophy in health and disease: mechanisms and therapeutic potential. Nat Rev Cardiol. 2023;20(5):347-363. [3] WEINER RB, BAGGISH AL. Exercise-induced cardiac remodeling. Prog Cardiovasc Dis. 2012;54(5):380-386. [4] HENNING RJ. The differentiation of the competitive athlete with physiologic cardiac remodeling from the athlete with cardiomyopathy. Curr Probl Cardiol. 2024;49(9):102473. [5] CARRENO JE, APABLAZA F, OCARANZA MP, et al. [Cardiac hypertrophy: molecular and cellular events]. Rev Esp Cardiol. 2006;59(5):473-486. [6] NAKAMURA M, SADOSHIMA J. Mechanisms of physiological and pathological cardiac hypertrophy. Nat Rev Cardiol. 2018;15(7):387-407. [7] BAI Y, ZHANG X, LI Y, et al. Protein Kinase A Is a Master Regulator of Physiological and Pathological Cardiac Hypertrophy. Circ Res. 2024;134(4):393-410. [8] LIU X, LI H, HASTINGS MH, et al. miR-222 inhibits pathological cardiac hypertrophy and heart failure. Cardiovasc Res. 2024;120(3):262-272. [9] SUOMINEN A, SUNI A, RUOHONEN S, et al. Melanocortin 1 Receptor Regulates Pathological and Physiological Cardiac Remodeling. J Am Heart Assoc. 2025; 14(4):e037961. [10] BOOTH FW, ROBERTS CK, LAYE MJ. Lack of exercise is a major cause of chronic diseases. Compr Physiol. 2012;2(2):1143-1211. [11] MOTR PACSG, LEAD A. Temporal dynamics of the multi-omic response to endurance exercise training. Nature. 2024;629(8010):174-183. [12] MI S, JIANG H, ZHANG L, et al. Regulation of Cardiac-Specific Proteins Expression by Moderate-Intensity Aerobic Exercise Training in Mice With Myocardial Infarction Induced Heart Failure Using MS-Based Proteomics. Front Cardiovasc Med. 2021;8:732076. [13] 彭勇, 史绍蓉, 黄思敏, 等. 4周中等强度有氧运动诱导大鼠心房肌蛋白质组差异表达的研究[J]. 中国应用生理学杂志,2018,34(5):450-456+463. [14] 彭勇, 史绍蓉, 黄思敏, 等. 基于蛋白质组学的中等强度有氧运动对大鼠心房肌物质能量代谢蛋白差异表达的研究[J]. 中国康复医学杂志,2019,34(5): 521-526. [15] DAVIS RT, 3RD, SIMON JN, UTTER M, et al. Knockout of p21-activated kinase-1 attenuates exercise-induced cardiac remodelling through altered calcineurin signalling. Cardiovasc Res. 2015;108(3):335-347. [16] CHEN H, PEREZ JN, CONSTANTOPOULOS E, et al. A method to study the impact of chemically-induced ovarian failure on exercise capacity and cardiac adaptation in mice. J Vis Exp. 2014;86:51083. [17] 秦娣, 秦学林, 郑一苇, 等. 有氧运动和恩格列净抑制铁死亡减轻异丙肾上腺素诱导的心脏重构[J]. 中国实验动物学报,2024,32(10):1281-1294. [18] 强睿, 姚子昂, 韩东, 等. 基于TMT标记联合LC-MS/MS阐释茵芪三黄解毒汤抗肝纤维化的药效作用机制[J]. 中华中医药杂志,2023,38(12):5987-5992. [19] COHN JN, FERRARI R, SHARPE N. Cardiac remodeling--concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. Behalf of an International Forum on Cardiac Remodeling. J Am Coll Cardiol. 2000;35(3):569-582. [20] SHIMIZU I, MINAMINO T. Physiological and pathological cardiac hypertrophy. J Mol Cell Cardiol. 2016;97:245-262. [21] MARON BJ, PELLICCIA A. The heart of trained athletes: cardiac remodeling and the risks of sports, including sudden death. Circulation. 2006;114(15):1633-1644. [22] LIMYATI Y, LUCRETIA T, GUNADI JW, et al. Chronic moderate‑intensity exercise can induce physiological hypertrophy in aged cardiomyocytes through autophagy, with minimal Yap/Taz involvement. Biomed Rep. 2025;22(3):44. [23] VAN BILSEN M, VAN NIEUWENHOVEN FA, VAN DER VUSSE GJ. Metabolic remodelling of the failing heart: beneficial or detrimental? Cardiovasc Res. 2009; 81(3):420-428. [24] NEUBAUER S. The failing heart--an engine out of fuel. N Engl J Med. 2007; 356(11):1140-1151. [25] MORA C, PINTADO C, RUBIO B, et al. Central leptin regulates heart lipid content by selectively increasing PPAR beta/delta expression. J Endocrinol. 2018;236(1): 43-56. [26] ZHANG H, SHEN Y, KIM IM, et al. The Impaired Bioenergetics of Diabetic Cardiac Microvascular Endothelial Cells. Front Endocrinol (Lausanne). 2021;12:642857. [27] GUTIERREZ-CUEVAS J, SANDOVAL-RODRIGUEZ A, MONROY-RAMIREZ HC, et al. Prolonged-release pirfenidone prevents obesity-induced cardiac steatosis and fibrosis in a mouse NASH model. Cardiovasc Drugs Ther. 2021;35(5):927-938. [28] WANG Q, WANG X. The Effects of a Low Linoleic Acid/alpha-Linolenic Acid Ratio on Lipid Metabolism and Endogenous Fatty Acid Distribution in Obese Mice. Int J Mol Sci. 2023;24(15):12117. [29] LIU L, NING X, WEI L, et al. Twist1 downregulation of PGC-1alpha decreases fatty acid oxidation in tubular epithelial cells, leading to kidney fibrosis. Theranostics. 2022;12(8):3758-3775. [30] HU H, WANG J, PENG Z, et al. Dapagliflozin attenuates diabetes-induced podocyte lipotoxicity via ERRalpha-Mediated lipid metabolism. Free Radic Biol Med. 2025; 234:178-191. [31] VERGARO G, PRUD’HOMME M, FAZAL L, et al. Inhibition of Galectin-3 Pathway Prevents Isoproterenol-Induced Left Ventricular Dysfunction and Fibrosis in Mice. Hypertension. 2016;67(3):606-612. [32] XU GR, ZHANG C, YANG HX, et al. Modified citrus pectin ameliorates myocardial fibrosis and inflammation via suppressing galectin-3 and TLR4/MyD88/NF-kappaB signaling pathway. Biomed Pharmacother. 2020;126:110071. [33] SUN JH, YANG HX, YAO TT, et al. Gentianella acuta prevents acute myocardial infarction induced by isoproterenol in rats via inhibition of galectin-3/TLR4/MyD88/NF-small ka, CyrillicB inflammatory signalling. Inflammopharmacology. 2021;29(1):205-219. [34] XING Z, YANG C, FENG Y, et al. Understanding aconite’s anti-fibrotic effects in cardiac fibrosis. Phytomedicine. 2024;122:155112. [35] YAKAR TULUCE S, TULUCE K, CIL Z, et al. Galectin-3 levels in patients with hypertrophic cardiomyopathy and its relationship with left ventricular mass index and function. Anatol J Cardiol. 2016;16(5):344-348. [36] LI Y, ZHOU WW, SUN JH, et al. Modified citrus pectin prevents isoproterenol-induced cardiac hypertrophy associated with p38 signalling and TLR4/JAK/STAT3 pathway. Biomed Pharmacother. 2021;143:112178. [37] KRUGER DN, BOSMAN M, VAN ASSCHE CXL, et al. Characterization of systolic and diastolic function, alongside proteomic profiling, in doxorubicin-induced cardiovascular toxicity in mice. Cardiooncology. 2024;10(1):40. [38] WANG L, ZHANG Y, YU M, et al. Identification of Hub Genes in the Remodeling of Non-Infarcted Myocardium Following Acute Myocardial Infarction. J Cardiovasc Dev Dis. 2022;9(12):409. [39] GAO J, LI Y, WANG T, et al. Analyzing gene expression profiles with preliminary validations in cardiac hypertrophy induced by pressure overload. Can J Physiol Pharmacol. 2018;96(8):701-709. [40] CHEN H, TESIC M, NIKOLIC VN, et al. Systemic Biomarkers and Unique Pathways in Different Phenotypes of Heart Failure with Preserved Ejection Fraction. Biomolecules. 2022;12(10):1419. [41] XU H, SHANG Q, CHEN H, et al. ITIH4: A New Potential Biomarker of “Toxin Syndrome” in Coronary Heart Disease Patient Identified with Proteomic Method. Evid Based Complement Alternat Med. 2013;2013:360149. [42] SAMPIETRO T, NEGLIA D, BIONDA A, et al. Inflammatory markers and serum lipids in idiopathic dilated cardiomyopathy. Am J Cardiol. 2005;96(12):1718-1720. [43] ZHOU LQ, LIU C, ZOU Y, et al. Development and verification of the nomogram for dilated cardiomyopathy gene diagnosis. Sci Rep. 2022;12(1):8908. [44] XIAO Y, NI L, SHI H, et al. SAA1 deficiency alleviates cardiac remodeling by inhibiting NF-kappaB/p38/JNK and TGFbeta/Smad pathways. FASEB J. 2023; 37(5):e22911. [45] WEN J, LI H, ZHOU Y, et al. Immunoglobin attenuates fulminant myocarditis by inhibiting overactivated innate immune response. Br J Pharmacol. 2025; 182(20):4984-5003. |
| [1] | Chen Xiaoqing, Bian Luyao, Lu Xingyu, Yang Tao, Li Xiang Hai. Thread embedding pretreatment at Xinshu (BL 15) improves cardiac function of acute myocardial ischemia rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 882-891. |
| [2] | Bu Xianzhong, Bu Baoxian, Xu Wei, Zhang Chi, Zhang Yisheng, Zhong Yuanming, Li Zhifei, Tang Fubo, Mai Wei, Zhou Jinyan. Analysis of serum differential proteomics in patients with acute cervical spondylotic radiculopathy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 535-541. |
| [3] | Yin Gonghua, Xu Ruoyao, Zhang Lijuan, Zhang Yifan, Qi Jie, Zhang Jun. Regulation of N6-methyladenosine on non-coding RNAs in pathological cardiac remodeling [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3252-2358. |
| [4] | Huang Huimin, Xie Bingying, Huang Jingwen, Huang Xiaobin, Xie Lihua, Li Shengqiang, Ge Jirong. Protective mechanism of alendronate granule in a rat osteoporosis model based on TMT proteomic analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2505-2511. |
| [5] | Zhuang Zhikun, He Mincong, Lin Tianye, Wu Rongkai, Guo Jinhua, Wu Zhaoke, Wei Qiushi. Exploration and clinical validation of the repair mode of the sclerotic zone of steroid-induced osteonecrosis of the femoral head based on Tandem Mass Tags technology [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2191-2196. |
| [6] | Xiong Tinglin, Ying Menghui, Zhang Lisha, Zhang Xiaogang, Yang Yan. Electrophysiological characteristics of cardiomyocytes differentiated from induced pluripotent stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1063-1067. |
| [7] | Xu Wenshan, Jiang Pingli, Liu Yulu, Ding Yanyi, Yu Yan, Yang Minguang, Liu Weilin, Chen Lidian. Salvianolic acid A effects on hippocampal protein expression in ischemic stroke rats: a tandem mass tag-based proteomic analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3714-3720. |
| [8] | Tang Yue-peng, Huang Gui-lin, Yao Li, Zhang Ni-ni, Yi Jie. Isoproterenol influence on stem/progenitor cells of submandibular glands: Proliferative number or capability? [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(40): 7084-7089. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||