Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (25): 6669-6679.doi: 10.12307/2026.267
Causal relationship between immune cell-mediated circulating inflammatory proteins and rheumatoid arthritis
Wang Chun, Gan Lizhen, Ren He, Fang Yi, Gao Zishan, Wu Yunchuan
- School of Acupuncture-Moxibustion and Tuina, and School of Health Preservation and Rehabilitation, Nanjing University of Chinese Medicine, Nanjing 210046, Jiangsu Province, China
-
Received:2025-08-24Revised:2025-12-10Online:2026-09-08Published:2026-04-23 -
Contact:Gao Zishan, Associate professor, School of Acupuncture-Moxibustion and Tuina, and School of Health Preservation and Rehabilitation, Nanjing University of Chinese Medicine, Nanjing 210046, Jiangsu Province, China Co-corresponding author: Wu Yunchuan, Professor, School of Acupuncture-Moxibustion and Tuina, and School of Health Preservation and Rehabilitation, Nanjing University of Chinese Medicine, Nanjing 210046, Jiangsu Province, China -
About author:Wang Chun, School of Acupuncture-Moxibustion and Tuina, and School of Health Preservation and Rehabilitation, Nanjing University of Chinese Medicine, Nanjing 210046, Jiangsu Province, China Gan Lizhen, School of Acupuncture-Moxibustion and Tuina, and School of Health Preservation and Rehabilitation, Nanjing University of Chinese Medicine, Nanjing 210046, Jiangsu Province, China Wang Chun and Gan Lizhen contributed equally to this work. -
Supported by:the National Natural Science Foundation of China, No. 81973941 (to GZS); Health Commission of Jiangsu Province, No. LKM2023030 (to WYC)
CLC Number:
Cite this article
Wang Chun, Gan Lizhen, Ren He, Fang Yi, Gao Zishan, Wu Yunchuan. Causal relationship between immune cell-mediated circulating inflammatory proteins and rheumatoid arthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6669-6679.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
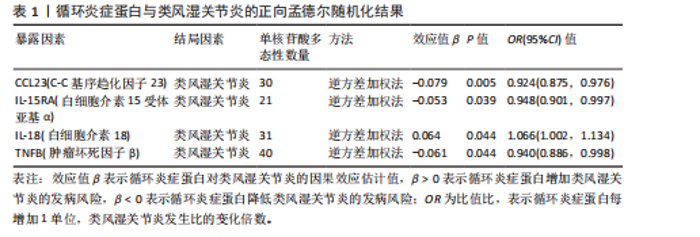
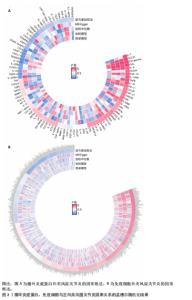
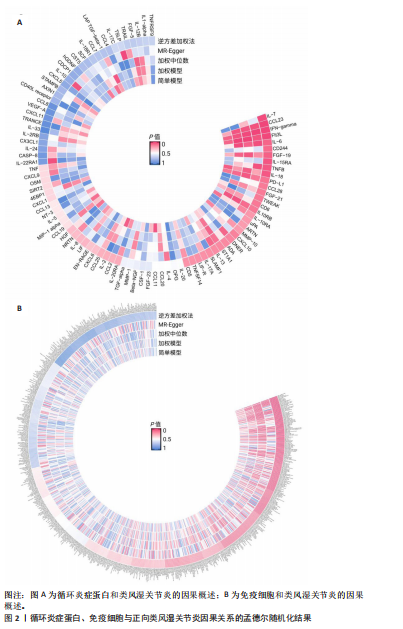
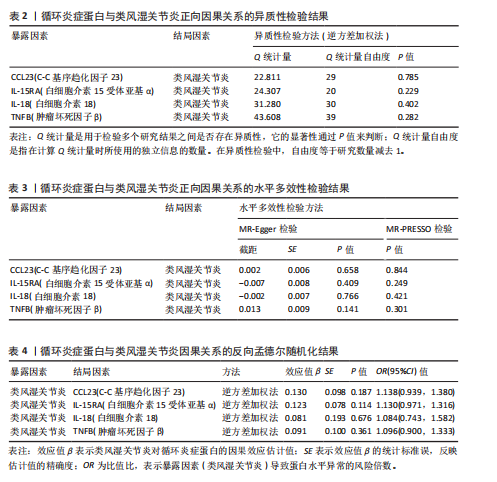
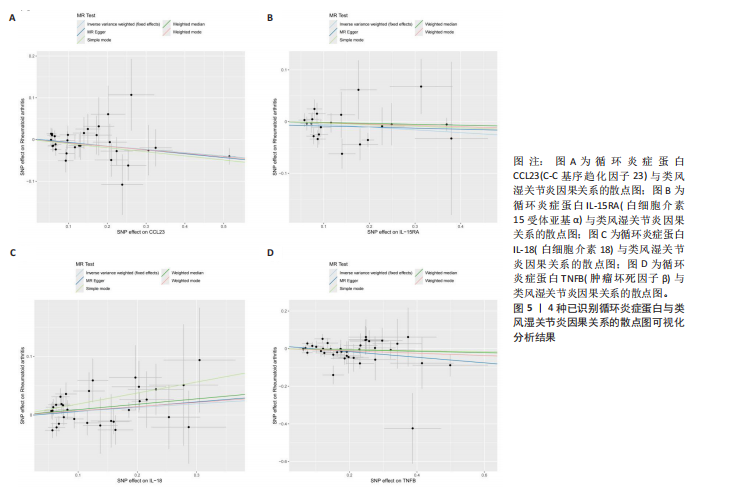
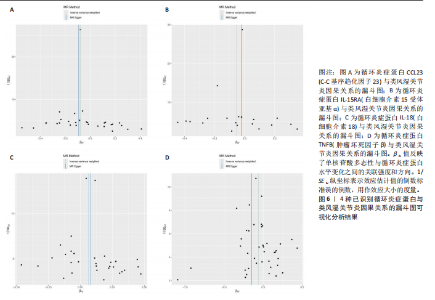
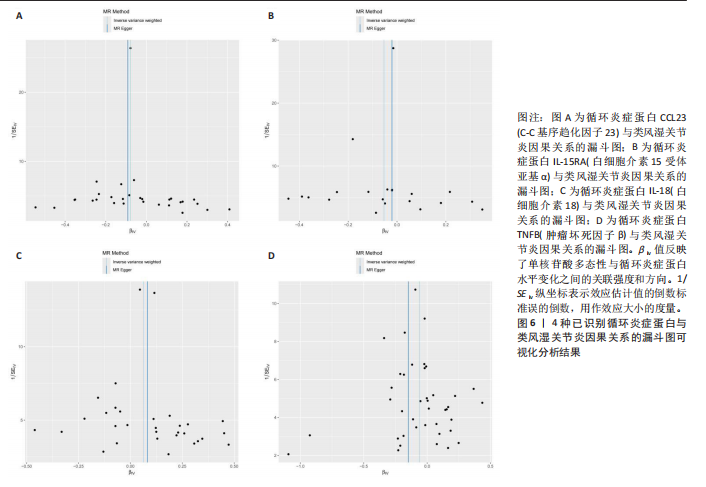
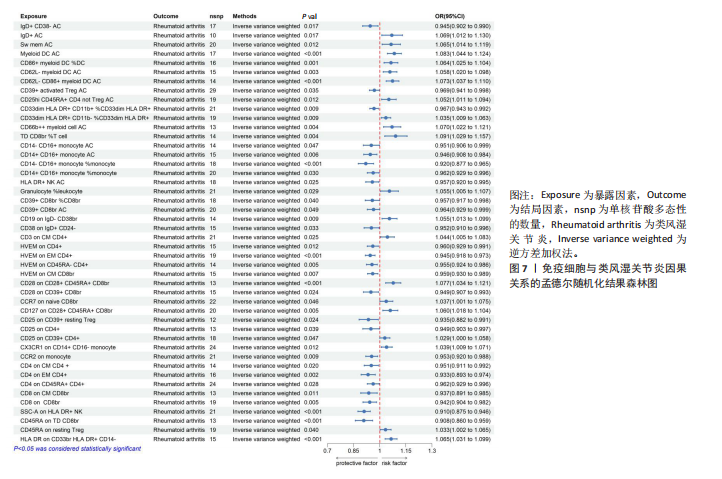
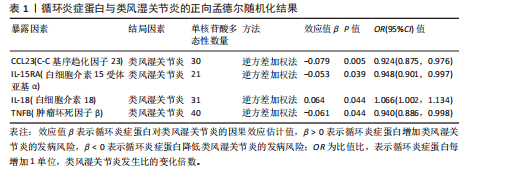
2.1 循环炎症蛋白与类风湿关节炎的正向孟德尔随机化分析结果 以逆方差加权法为主要分析方法,经过一系列严格筛选后,识别出4种循环炎症蛋白与类风湿关节炎存在显著因果关联,见表1、图2,3。IL-18(白细胞介素18)(β=0.064,OR=1.066,P=0.044)与类风湿关节炎风险呈正相关,CCL23(C-C基序趋化因子23)(β=-0.079,OR=0.924,P=0.005)、IL-15RA(白细胞介素15受体亚基α)(β=-0.053,OR=0.948,P=0.039)和TNFB(肿瘤坏死因子β)(β=-0.061,OR=0.940,P=0.044)与类风湿关节炎风险呈负相关。 Cochran’Q异质性检验结果显示P > 0.05,表明已识别的4种循环炎症蛋白相关工具变量在效应估计中未呈现显著异质性。MR-Egger回归的截距项检验与MR-PRESSO全局多效性检验均显示P > 0.05,表明工具变量集合中不存在具有统计学意义的水平多效性偏倚。留一法敏感性分析结果表明,在依次剔除每个单核苷酸多态性后,总体效应估计值变化不大,各个单核苷酸多态性的影响较为均匀,没有单个单核"
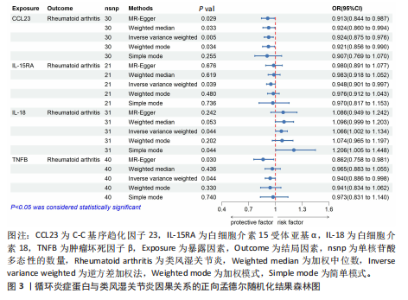
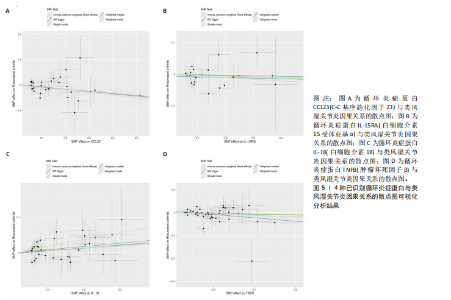
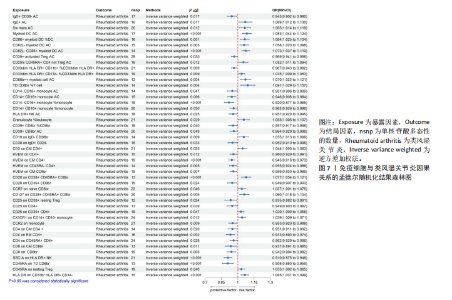
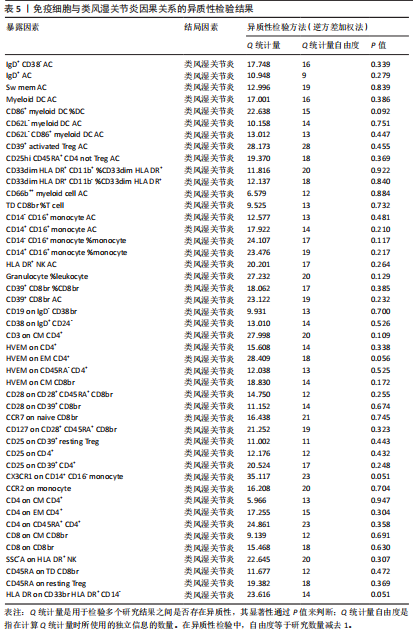
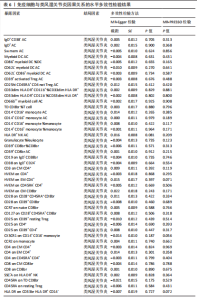
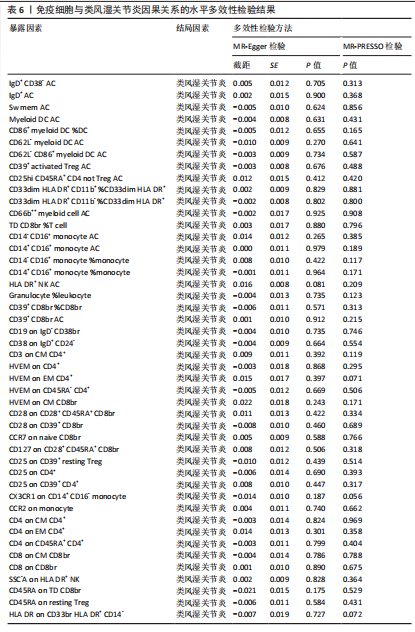
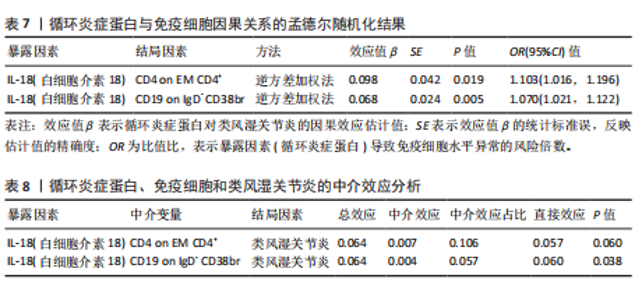
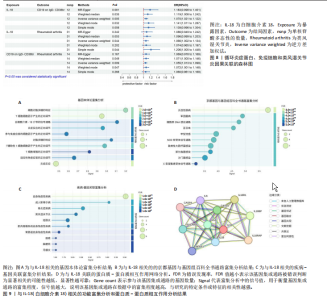
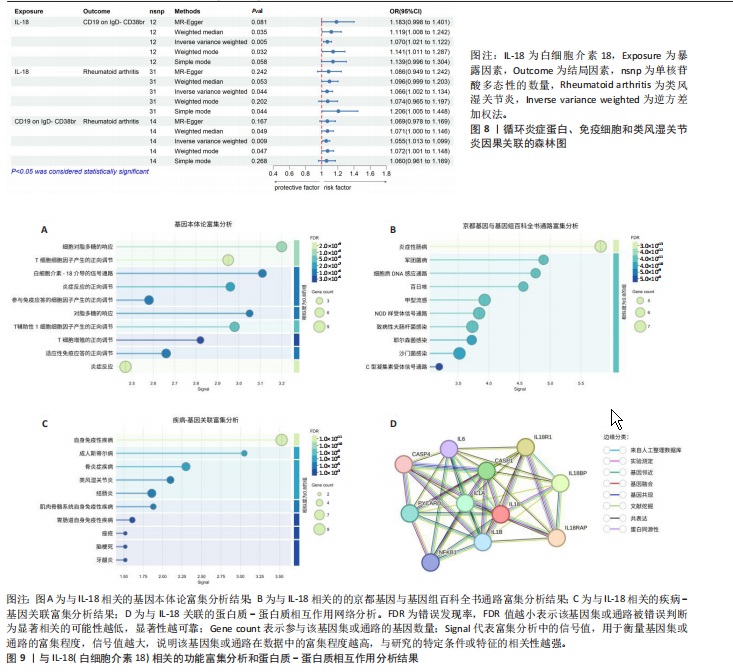
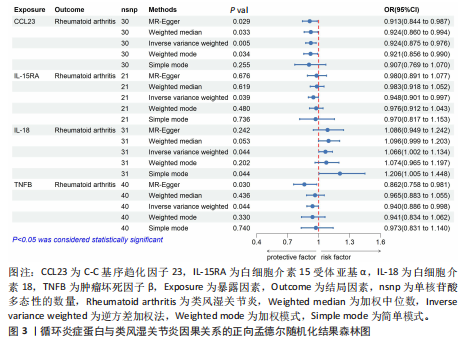
苷酸多态性对结果产生显著影响,分析结果具有较好的稳健性。可视化结果显示,散点图中5种分析方法的因果效应方向保持一致,未发现异常值。漏斗图中各单核苷酸多态性的效应值呈对称分布态势,数据点集中分布于基准线两侧,未观察到明显偏离正常分布范围的离群值。循环炎症蛋白与类风湿关节炎正向因果关系的异质性检验和水平多效性检验结果,见表2,3。循环炎症蛋白与类风湿关节炎正向因果关系的留一法敏感性分析结果见图4。循环炎症蛋白与类风湿关节炎正向因果关系的散点图结果,见图5。循环炎症蛋白与类风湿关节炎正向因果关系的漏斗图结果,见图6。 2.2 循环炎症蛋白与类风湿关节炎的反向孟德尔随机化分析结果 将类风湿关节炎作为暴露因素,已识别的4种循环炎症蛋白作为结局因素进行反向孟德尔随机化分析。以逆方差加权法作为主要分析方法,结果显示类风湿关节炎与CCL23(β=0.130,OR=1.138,P=0.187)、IL-15RA(β=0.123,OR=1.130,P=0.114)、IL-18(β=0.081,OR=1.084,P=0.676)、TNFB(β=0.091,OR=1.096,P=0.361)的P均>0.05,见表4,表明类风湿关节炎与4种循环炎症蛋白不存在反向因果关系,未观察到类风湿关节炎影响4种循环炎症蛋白的证据。 2.3 免疫细胞与类风湿关节炎因果关系的孟德尔随机化结果 将免疫细胞作为暴露因素,类风湿关节炎作为结局因素,以逆方差加权法为主要分析法,经过一系列筛选后,发现46种免疫细胞与类风湿关节炎存在显著相关性,见图2,图7。敏感性分析结果表明,46种免疫细胞与类风湿关节炎不存在异质性和水平多效性,并且因果效应方向显示一致性。异质性检验和水平多效性检验结果见表5,6。 2.4 中介分析结果 在上述研究的基础上,以免疫细胞作为中介,以确定免疫细胞是否充当从循环炎症蛋白到类风湿关节炎的介质,分析过程同上述过程一致,结果表明,炎症蛋白IL-18与免疫细胞CD4 on EM CD4+和CD19 on IgD- CD38br存在显著因果关系,见表7。将CD4 on EM CD4+输入到中介分析中评估IL-18与类风湿关节炎的相关性,结果显示P > 0.05,相关性不具有统计学意义。将CD19 on IgD- CD38br输入到中介分析中评估IL-18与类风湿关节炎的相关性,结果显示P < 0.05,相关性仍然具有统计学意义。具体而言,IL-18对类风湿关节炎的总效应值为0.064,表明IL-18与类风湿关节炎风险的总体效应呈正相关。进一步中介分析结果显示,CD19 on IgD- CD38br对类风湿关节炎的中介效应为0.004,中介占比为5.7%,直接效应为0.060,表明控制CD19 on IgD- CD38br的影响后IL-18与类风湿关节炎风险仍呈正相关关系,见表8,图8。综合以上结果表明,CD19 on IgD- CD38br可能介导循环炎症蛋白IL-18与类风湿关节炎风险的关联。 2.5 功能富集分析和蛋白质-蛋白质相互作用分析结果 多种功能富集分析显示,已识别的炎症蛋白IL-18在多个关键的生物学过程中表现出显著的参与性:在基因本体论富集层面,炎症蛋白IL-18显著参与细胞对脂多糖的响应、"

T细胞增殖正向调节及炎症反应调控等关键生物学过程;在京都基因与基因组百科全书信号通路层面,IL-18与炎症性肠病、军团菌病以及细胞质DNA感应通路存在强关联;在疾病-基因关联谱系层面,IL-18与成人斯蒂尔病、骨炎症疾病等慢性炎性疾病及类风湿关节炎等自身免疫性疾病存在显著富集。综合上述结果表明,IL-18与介导免疫细胞活化调控、病原相关分子模式识别信号转导及炎症因子网络平衡显著相关,可能在维系免疫稳态与推动病理炎症循环中发挥重要作用。针对此分子靶点,构建蛋白质-蛋白质相互作用网络分析,展示与其交互最强的前10位分子,结果显示IL-18RAP(白细胞介素18受体辅助蛋白)、IL-18R1(白细胞"

| [1] CUSH JJ. Rheumatoid Arthritis: Early Diagnosis and Treatment. Med Clin North Am. 2021;105(2): 355-365. [2] ALMUTAIRI K, NOSSENT J, PREEN D, et al. The global prevalence of rheumatoid arthritis: a meta-analysis based on a systematic review. Rheumatol Int. 2021;41(5):863-877. [3] MA Y, CHEN H, LV W, et al. Global, regional and national burden of rheumatoid arthritis from 1990 to 2021, with projections of incidence to 2050: a systematic and comprehensive analysis of the Global Burden of Disease study 2021. Biomark Res. 2025;13(1):47. [4] LIN YJ, ANZAGHE M, SCHÜLKE S. Update on the Pathomechanism, Diagnosis, and Treatment Options for Rheumatoid Arthritis. Cells. 2020; 9(4):880. [5] BOUZIT L, MALSPEIS S, SPARKS JA, et al. Assessing improved risk prediction of rheumatoid arthritis by environmental, genetic, and metabolomic factors. Semin Arthritis Rheum. 2021;51(5):1016-1022. [6] KERSCHBAUMER A, SEPRIANO A, BERGSTRA SA, et al. Efficacy of synthetic and biological DMARDs: a systematic literature review informing the 2022 update of the EULAR recommendations for the management of rheumatoid arthritis. Ann Rheum Dis. 2023;82(1):95-106. [7] GAUTAM S, WHITTAKER JE, VEKARIYA R, et al. The distinct transcriptomic signature of the resolution phase fibroblast-like synoviocytes supports endothelial cell dysfunction. Commun Biol. 2025;8(1):837. [8] GAUTAM S, KUMAR R, KUMAR U, et al. Yoga maintains Th17/Treg cell homeostasis and reduces the rate of T cell aging in rheumatoid arthritis: a randomized controlled trial. Sci Rep. 2023;13(1):14924. [9] ZHENG Y, LIU Q, GORONZY JJ, et al. Immune aging - A mechanism in autoimmune disease. Semin Immunol. 2023;69:101814. [10] JANG S, KWON EJ, LEE JJ. Rheumatoid Arthritis: Pathogenic Roles of Diverse Immune Cells. Int J Mol Sci. 2022;23(2):905. [11] KOMATSU N, TAKAYANAGI H. Mechanisms of joint destruction in rheumatoid arthritis - immune cell-fibroblast-bone interactions. Nat Rev Rheumatol. 2022;18(7):415-429. [12] SKRIVANKOVA VW, RICHMOND RC, WOOLF BAR, et al. Strengthening the Reporting of Observational Studies in Epidemiology Using Mendelian Randomization: The STROBE-MR Statement. JAMA. 2021;326(16):1614-1621. [13] RICHMOND RC, DAVEY SMITH G. Mendelian Randomization: Concepts and Scope. Cold Spring Harb Perspect Med. 2022;12(1):a040501. [14] ZHAO JH, STACEY D, ERIKSSON N, et al. Genetics of circulating inflammatory proteins identifies drivers of immune-mediated disease risk and therapeutic targets. Nat Immunol. 2023;24(9):1540-1551. [15] ORRÙ V, STERI M, SIDORE C, et al. Complex genetic signatures in immune cells underlie autoimmunity and inform therapy. Nat Genet. 2020;52(10):1036-1045. [16] KURKI MI, KARJALAINEN J, PALTA P, et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature. 2023; 613(7944):508-518. [17] LIU QP, DU HC, XIE PJ, et al. Effect of the immune cells and plasma metabolites on rheumatoid arthritis: a mediated mendelian randomization study. Front Endocrinol (Lausanne). 2024;15:1438097. [18] XU J, SI S, HAN Y, et al. Genetic insight into dissecting the immunophenotypes and inflammatory profiles in the pathogenesis of Sjogren syndrome. J Transl Med. 2025;23(1): 56. [19] LI DH, WU Q, LAN JS, et al. Plasma metabolites and risk of myocardial infarction: a bidirectional Mendelian randomization study. J Geriatr Cardiol. 2024;21(2):219-231. [20] 容向宾,郑海波,莫学燊,等.血浆代谢物、免疫细胞与髋骨关节炎的因果推断:GWAS数据欧洲群体资料分析[J].中国组织工程研究, 2026,30(4):1028-1035. [21] JIN C, LU Z, CHEN Y, et al. Identification of biomarkers for chronic lymphocytic leukemia risk: a proteome-wide Mendelian randomization study. Discov Oncol. 2025;16(1):2. [22] MA H, CHEN Y. Examining the causal relationship between sex hormone-binding globulin (SHBG) and infertility: A Mendelian randomization study. PLoS One. 2024;19(6):e0304216. [23] GAO B, WANG Z, WANG K, et al. Relationships among gut microbiota, plasma metabolites, and juvenile idiopathic arthritis: a mediation Mendelian randomization study. Front Microbiol. 2024;15:1363776. [24] HUANG M, XING F, HU Y, et al. Causal inference study of plasma proteins and blood metabolites mediating the effect of obesity-related indicators on osteoporosis. Front Endocrinol (Lausanne). 2025;16:1435295. [25] YE CJ, LIU D, CHEN ML, et al. Mendelian randomization evidence for the causal effect of mental well-being on healthy aging. Nat Hum Behav. 2024;8(9):1798-1809. [26] CHEN JY, WANG JF, HU Y, et al. Evaluating the advancements in protein language models for encoding strategies in protein function prediction: a comprehensive review. Front Bioeng Biotechnol. 2025;13:1506508. [27] DONCHEVA NT, MORRIS JH, HOLZE H, et al. Cytoscape stringApp 2.0: Analysis and Visualization of Heterogeneous Biological Networks. J Proteome Res. 2023;22(2):637-646. [28] SZKLARCZYK D, GABLE AL, LYON D, et al. STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019;47(D1):D607-D613. [29] IHIM SA, ABUBAKAR SD, ZIAN Z, et al. Interleukin-18 cytokine in immunity, inflammation, and autoimmunity: Biological role in induction, regulation, and treatment. Front Immunol. 2022; 13:919973. [30] WANG Y, XU D, LONG L, et al. Correlation between plasma, synovial fluid and articular cartilage Interleukin-18 with radiographic severity in 33 patients with osteoarthritis of the knee. Clin Exp Med. 2014;14(3):297-304. [31] 郑宝林,李婷,刘奔流,等.通痹1号方对实验性类风湿关节炎大鼠血清IL-18与VEGF水平的影响[J].海南医学,2018,29(3):297-299. [32] AMIN MA, RABQUER BJ, MANSFIELD PJ, et al. Interleukin 18 induces angiogenesis in vitro and in vivo via Src and Jnk kinases. Ann Rheum Dis. 2010;69(12):2204-2212. [33] SHEN J, ZHANG Y, TANG W, et al. Short IL-18 generated by caspase-3 cleavage mobilizes NK cells to suppress tumor growth. Nat Immunol. 2025;26(3):416-428. [34] MA H, HU T, TAO W, et al. A lncRNA from an inflammatory bowel disease risk locus maintains intestinal host-commensal homeostasis. Cell Res. 2023;33(5):372-388. [35] JIANG Q, WANG X, XU X, et al. Inflammasomes in rheumatoid arthritis: a pilot study. BMC Rheumatol. 2023;7(1):39. [36] 陈泽豪,王景,高翔宇,等.外周血单个核细胞AIM2炎症小体激活释放白细胞介素-18介导带状疱疹的临床研究[J].中国疼痛医学杂志, 2024,30(5):348-355. [37] HONG J, LUO F, DU X, et al. The immune cells in modulating osteoclast formation and bone metabolism. Int Immunopharmacol. 2024;133: 112151. [38] LI T, JIANG G, HU X, et al. Punicalin Attenuates Breast Cancer-Associated Osteolysis by Inhibiting the NF-κB Signaling Pathway of Osteoclasts. Front Pharmacol. 2021;12:789552. [39] WEI L, CHEN W, HUANG L, et al. Alpinetin ameliorates bone loss in LPS-induced inflammation osteolysis via ROS mediated P38/PI3K signaling pathway. Pharmacol Res. 2022;184:106400. [40] LIU C, ZUO M, ZHAO J, et al. DPHB inhibits osteoclastogenesis by suppressing NF-κB and MAPK signaling and alleviates inflammatory bone destruction. Int Immunopharmacol. 2025; 152: 114377. [41] XU S, WANG Y, LU J, et al. Osteoprotegerin and RANKL in the pathogenesis of rheumatoid arthritis-induced osteoporosis. Rheumatol Int. 2012;32(11):3397-3403. [42] QUARESMA TO, DE ALMEIDA SCL, DA SILVA TA, et al. Comparative study of the synovial levels of RANKL and OPG in rheumatoid arthritis, spondyloarthritis and osteoarthritis. Adv Rheumatol. 2023;63(1):13. [43] MCGRATH S, GRIMSTAD K, THORARINSDOTTIR K, et al. Correlation of Professional Antigen-Presenting Tbet(+)CD11c(+) B Cells With Bone Destruction in Untreated Rheumatoid Arthritis. Arthritis Rheumatol. 2024;76(8):1263-1277. [44] WANG X, GAO H, ZENG Y, et al. A Mendelian analysis of the relationships between immune cells and breast cancer. Front Oncol. 2024;14:1341292. |
| [1] | Wu Zhilin, , He Qin, Wang Pingxi, Shi Xian, Yuan Song, Zhang Jun, Wang Hao . DYRK2: a novel therapeutic target for rheumatoid arthritis combined with osteoporosis based on East Asian and European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1569-1579. |
| [2] | Liu Hongtao, Wu Xin, Jiang Xinyu, Sha Fei, An Qi, Li Gaobiao. Causal relationship between age-related macular degeneration and deep vein thrombosis: analysis based on genome-wide association study data [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1602-1608. |
| [3] | Guo Ying, Tian Feng, Wang Chunfang. Potential drug targets for the treatment of rheumatoid arthritis: large sample analysis from European databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1549-1557. |
| [4] | Zheng Yin, Wu Zhenhua, Zhang Cheng, Ruan Kexin, Gang Xiaolin, Ji Hong. Safety and efficacy of immunoadsorption therapy for rheumatoid arthritis: a network meta-analysis and systematic review [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1260-1268. |
| [5] | Gao Zengjie, , Pu Xiang, Li Lailai, Chai Yihui, Huang Hua, Qin Yu. Increased risk of osteoporotic pathological fractures associated with sterol esters: evidence from IEU-GWAS and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1302-1310. |
| [6] | Liu Fengzhi, Dong Yuna, Tian Wenyi, Wang Chunlei, Liang Xiaodong, Bao Lin. Gene-predicted associations between 731 immune cell phenotypes and rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1311-1319. |
| [7] | Zhang Cuicui, Chen Huanyu, Yu Qiao, Huang Yuxuan, Yao Gengzhen, Zou Xu. Relationship between plasma proteins and pulmonary arterial hypertension and potential therapeutic targets [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1331-1340. |
| [8] | Bao Zhuoma, Hou Ziming, Jiang Lu, Li Weiyi, Zhang Zongxing, Liu Daozhong, Yuan Lin. Effect and mechanism by which Pterocarya hupehensis skan total flavonoids regulates the proliferation, migration and apoptosis of fibroblast-like synoviocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 816-823. |
| [9] | Zeng Hao, Sun Pengcheng, Chai Yuan, Huang Yourong, Zhang Chi, Zhang Xiaoyun. Association between thyroid function and osteoporosis: genome-wide data analysis of European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1019-1027. |
| [10] | Rong Xiangbin, , Zheng Haibo, Mo Xueshen, Hou Kun, Zeng Ping, . Plasma metabolites, immune cells, and hip osteoarthritis: causal inference based on GWAS data from European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1028-1035. |
| [11] | He Qiwang, , , Chen Bo, Liang Fuchao, Kang Zewei, Zhou Yuan, Ji Anxu, Tang Xialin, . Relationship between Alzheimer’s disease and sarcopenia and body mass index: analysis of GWAS datasets for European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1036-1046. |
| [12] | Ding Yu, Chen Jingwen, Chen Xiuyan, Shi Huimin, Yang Yudie, Zhou Meiqi, Cui Shuai, . Circulating inflammatory proteins and myocardial hypertrophy: large sample analysis of European populations from GWAS Catalog and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1047-1057. |
| [13] | Chen Cai, Hong Zhongyuan, Deng Huaidong, Zeng Qin, Chen Jiancong. Therapeutic targets for knee osteoarthritis: identification via a bioinformatics approach [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 8878-8888. |
| [14] | Zhao Feifan, Cao Yujing. An artificial neural network model of ankylosing spondylitis and psoriasis shared genes and machine learning-based mining and validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 770-784. |
| [15] | Liu Chu, Qiu Boyuan, Tong Siwen, He Linyuwei, Chen Haobo, Ou Zhixue. A genetic perspective reveals the relationship between blood metabolites and osteonecrosis: an analysis of information from the FinnGen database in Finland [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 785-794. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||