Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (3): 464-470.doi: 10.3969/j.issn.2095-4344.0048
Previous Articles Next Articles
Particle-associated periprosthetic osteolysis after hip arthroplasty: the underlying biological mechanisms
Li Wen-bo, Song Ke-guan
- Third Department of Orthopedics, the First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang Province, China
-
Online:2018-01-28Published:2018-01-28 -
Contact:Song Ke-guan, M.D., Chief physician, Third Department of Orthopedics, the First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang Province, China -
About author:Li Wen-bo, Master, Third Department of Orthopedics, the First Affiliated Hospital of Harbin Medical University, Harbin 150001, Heilongjiang Province, China -
Supported by:the National Natural Science Foundation of China, No. 81272016
CLC Number:
Cite this article
Li Wen-bo, Song Ke-guan. Particle-associated periprosthetic osteolysis after hip arthroplasty: the underlying biological mechanisms[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(3): 464-470.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
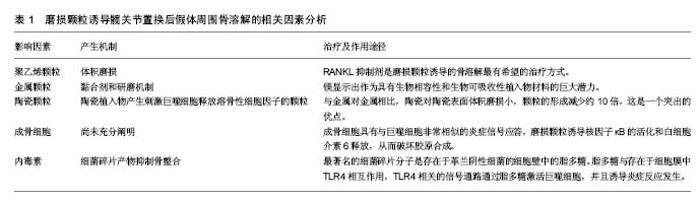
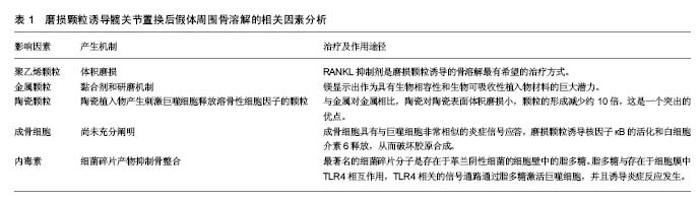
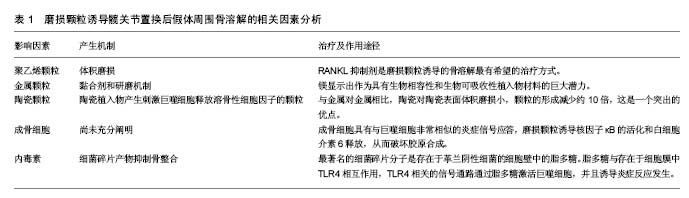
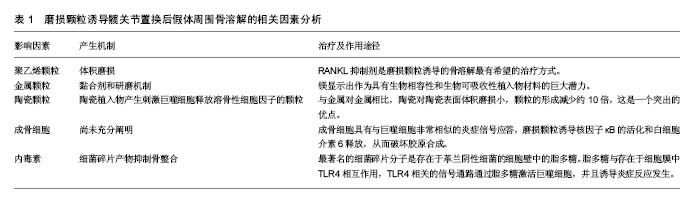
2.1 发病机制 任何材质及设计的功能性人工关节都会产生磨损颗粒。通常,植入物所产生的颗粒会被免疫细胞清除,并且磨损颗粒的产生和消除之间存在平衡,当颗粒的产出超过了免疫系统的消除能力,失代偿发生。据研究报告,年磨损率低于80 mm3的髋臼杯与骨溶解关联很小,年磨损率在80-140 mm3之间的髋臼杯都存在低到中度的骨溶解,而年磨损率达到140 mm3的髋臼杯骨溶解水平更显着增加[9]。 炎症反应与颗粒的类型无关,其目的是消除外来物质[10-11]。研究已经表明,这是以巨噬细胞为主,涉及成纤维细胞,淋巴细胞和破骨细胞的慢性炎性宿主反应。免疫细胞的失补偿表明它们是长期活化的,主要通过3种机制破坏骨组织形成和溶解之间的平衡。第一种机制涉及由活化的巨噬细胞诱导的过度炎症性破骨细胞,第二是假体周围骨形成的破坏,第三种机制是作为间充质骨祖细胞的细胞毒性反应增加的结果引起的骨再生的破坏[5]。这些过程在骨组织多细胞作用水平上将平衡从成骨转变为骨再吸收,导致植入物周围的肉眼可见的骨缺损[12]。骨缺损的程度是与影响修复再吸收部位的假体颗粒的数量,大小和来源相关[13]。 假体的磨损与腐蚀产生颗粒。磨损是在负载运动时相对滑动的两个表面的材料损失,磨损从表面释放颗 粒[14]。磨损颗粒可以是聚乙烯、骨、水泥、金属、金属腐蚀产物或羟基磷灰石颗粒[15],见表1。现今的关节成形术中,接触表面是金属、聚乙烯和陶瓷的双重组合。磨损颗粒的类型、尺寸和密度的不同组合决定了了宿主的免疫应答[14]。例如,从金属关节面-金属关节面(MoM)组合产生的颗粒将触发B和T淋巴细胞,以及细胞介导的延迟型超敏反应或浆细胞相关的4型超敏反应。 腐蚀是金属与其环境之间的化学相互作用及其随后的电化学溶解,因此,这个过程导致假体不断的钝化,并在界面处连续沉积金属氧化物颗粒[16-18]。在围绕腐蚀轴的假体周围组织反应中观察到的临床和组织学特征与磨损金属关节面-金属关节面和非金属关节面-金属关节面轴承中观察到的不良局部组织反应非常相似[19]。然而,有证据表明衍生的颗粒可能对软组织更具生物活性和破坏性[20]。各种局部因素,包括金属的溶解度、pH和有机组分的性质,引起各种腐蚀产物的出现,这些腐蚀产物可以是可溶性或不溶性盐,金属-蛋白质复合物和自由基。这些腐蚀产物可以使用包括原子吸收等离子体质谱的各种数字技术在体液中检测。由磨损或腐蚀产生的颗粒可以局部或全身传播[15,21]。 颗粒的特征,包括它们的组成、尺寸、形状和数量,特别是在生物活性强的亚微米尺寸范围内的颗粒,显著影响整个细胞和组织反应[20]。这些颗粒最初引起炎症,增加关节液中炎性细胞的数量,特别是巨噬细胞和成纤维细胞的数量。关节内压力也升高,并且来自关节表面的磨损颗粒可以从该区域迁移,即使当假体并没有松动时,它们也将最终扩散遍及关节腔[22-24]。 此外,压缩的关节液也可以直接诱导骨吸收[24]。许多研究已经证实,在同一患者不同解剖位置取得的组织样本的组织学切片之间有明显的相似性[15]。当关节液和磨损颗粒贯穿骨和假体之间时,在膨胀的区域中形成滑膜样膜。这种界面膜是包含丰富的成纤维细胞、巨噬细胞、内皮细胞和炎症递质的肉芽组织,它为细胞和酶反应提供场所,最终破坏邻近的骨[25]。上述过程被认为是骨溶解的病理机制[25-27],见表1。 2.2 聚乙烯颗粒 目前最常见的表面组合是高度交联的聚乙烯颗粒,置于金属髋臼内盖和金属股骨头上[25]。假体部件之间的相互作用不断形成高浓度聚乙烯颗粒磨屑,并在整个关节腔中扩散。确定所产生的聚乙烯颗粒数量最重要的因素是体积磨损,这与假体头部的尺寸相关联。例如,36 mm金属头引起体积磨损,并与聚乙烯颗粒的数量的增加相关。相反,当使用22 mm小金属头时,在线性帽中发生线性磨损,并且产生颗粒的数量比大的金属头减少。临床上松动倾向于作为骨缺失的结果,并且经常伴随着聚乙烯帽的磨损发生[28]。颗粒大小与其数量一样重要。聚乙烯颗粒的尺寸范围为0.1-10 μm,0.1-0.5 μm之间的颗粒大多与磨损有关[29]。此外,当考虑到磨屑的总体积时,则更大的颗粒也可存在[9,29]。介于0.3-1.0 μm之间的颗粒是体内单核吞噬细胞最有效的刺激物[17];如果颗粒小于0.3 μm,则被代偿,不引起严重的反应,并且大多通过胞饮作用而不是吞噬作用从该区域消除。 虽然这些小颗粒不是高反应性的,但它们可以累积并触发反应。被巨噬细胞吞噬的碎片材料大多由小于 7 μm的颗粒组成。较大的颗粒被多核巨细胞包围时被捕获[30]。虽然交联的聚乙烯产生的颗粒较小,但比常规聚乙烯颗粒更具反应性,因为刺激产生相同水平的肿瘤坏死因子α所需的常规聚乙烯颗粒的量大约是交联聚乙烯颗粒的量100倍。但是交联聚乙烯的使用更安全,更常应用于临床[29],因为交联聚乙烯内帽体积磨损小。如上所述,体积磨损是决定产生的聚乙烯颗粒数量的最重要因素;因此,当在体内使用交联聚乙烯颗粒时,引起炎症程度较低。交联聚乙烯颗粒(超高分子量聚乙烯颗粒)具有较轻的磨损,小而多的颗粒,导致磨损颗粒性疾病较少。 聚乙烯磨损颗粒被软骨组织中的破骨细胞、成纤维细胞和成骨细胞/基质细胞吞噬。聚乙烯颗粒引起的免疫反应是非特异性的,因此,参与宿主反应的淋巴细胞很少。通常材料在噬菌体吞噬后在吞噬体内迅速分解。但磨损颗粒对酶消化有抗性[30-31],这些颗粒产物激活炎症细胞,诱导促炎细胞因子和蛋白水解酶的释放[15]。促进释放肿瘤坏死因子α、白细胞介素1α、白细胞介素1β、白细胞介素6、白细胞介素8和前列腺素E2的信号被触发,并且这些促炎症递质增加破骨细胞形成。这些促炎递质具有自分泌或旁分泌效应,并且能够影响各种信号机制的功能。在这些递质中,肿瘤坏死因子α和白细胞介素1β在引起骨质溶解的级联反应中具有最重要的作用。吞噬并不是巨噬细胞活化所必需的。实际上,巨噬细胞和颗粒可以在巨噬细胞的外膜上相互作用,这可以类似地刺激促炎细胞因子的释放。这种相互作用可以通过单个受体或多个受体发生,例如分化簇(CD)11b,CD14和Toll样受体(TLR)家族[32-33]。 在含有颗粒的细胞中,核因子κB控制促炎细胞因子产生的几种基因的表达,包括白细胞介素1、白细胞介素6和肿瘤坏死因子α。核转录因子κB激活是启动宿主反应的关键步骤。最近,核因子κB配体(RANKL)途径的受体激活剂-骨保护素已被证明在溶骨性损伤的起始和进展中起主要作用[34-35]。释放到环境中的促炎细胞因子到达成骨细胞,从而增加RANKL的活化。RANKL是由骨髓相关基质细胞和成骨细胞产生的肿瘤坏死因子相关细胞因子。RANKL在成骨细胞表面形成,以溶解形式释放到该区域,并且通过结合核因子κB的受体活化剂(RANK)增加核因子κB基因表达(图1)。骨保护素是RANKL的竞争受体。它结合RANKL并阻止其与RANK的连接,从而减少破骨细胞形成[25]。最近的研究已经得出结论,全髋关节置换后出现无菌性松动的患者中的成骨细胞和基质细胞表现出比原发性全髋关节置换患者更高水平的RANKL表达以及在滑液中的更低的骨保护素/RANKL比率[36]。RANKL/骨保护素比率在破骨细胞形成的调节中起着关键作用,并且与多种骨疾病相关[37]。在炎症组织内的细胞中呼应假体颗粒的RANKL表达可以促进破骨细胞分化和活性,从而促进骨溶解。RANKL抑制剂是磨损颗粒诱导的骨溶解最有希望的治疗方式[38]。 2.3 金属颗粒 金属颗粒比聚乙烯颗粒小得多 (0.05 μm)。与硬-软材料组合,即金属-聚乙烯或陶瓷-聚乙烯相比,所有硬-硬材料组合,即金属对金属或陶瓷对聚乙烯产生更小的颗粒。在金属对金属表面之间产生的颗粒的总体积远大于其他关节[34]。参与由金属对金属表面产生的颗粒的免疫应答是非常强力的,这表明了材料本身或其形状对于随后的免疫应答的重要性。 黏合剂和研磨机制导致金属对金属表面的磨损。由于局部重复加载引起表层和深层的磨损或裂纹促进了磨屑形成。在非金属对金属植入物中,腐蚀产物也可以源自头颈或颈-杆接头。 实验应当使用人体细胞或细胞系及公认的标准试验进行体外试验,以确定合金元素释放的影响[35,39]。在过去几年研究中,镁(Mg)显示出作为具有生物相容性和生物可吸收性植入物材料的巨大潜力。Mg最具吸引力的物理特性是其高比强度和弹性模量,非常类似于人类骨骼[40]。最近的研究突出了Mg刺激骨生长和愈合的能力。其他实验已经证实,Mg能够增强骨细胞对氧化铝的黏附,并且成骨细胞和生长板软骨细胞耐受良好[41]。 2.3.1 超敏性 金属对金属表面由于金属[例如,钴(Co)铬(Cr),钛(Ti),镍(Ni)和钼(Mo)]腐蚀而产生金属颗粒和游离金属离子。金属颗粒的离子形式与体液和吞噬体中的阴离子和蛋白质相互作用,最终导致有机金属络合物,无机金属盐和氧化物的形成。这些离子化合物具有不同的尺寸,并且具有不同的稳定性,分辨率,生物利用度和生物活性电位。在围绕松动的植入物的组织中,通常存在包括血管炎和淋巴细胞积聚的Ⅳ型超敏反 应[37,42]。由于暴露在特定材料的免疫应答尚未在所有患者中得到证实,所以不同的机制被提出。在金属对金属接触表面上产生的金属离子激活内皮细胞,从而增加淋巴细胞运输和黏附。内皮细胞在拮抗金属磨损颗粒的组织反应中起着中心调节作用。在不存在Ⅳ型超敏反应的情况下,已经跨内膜迁移并累积在血管周区中的淋巴细胞引发针对金属离子的反应。 人体对不同金属离子的响应不同。在体外研究中,Posada等[42]观察到Co导致更高水平的肿瘤坏死因子α表达,并且具有更高的刺激效应。由于高水平的肿瘤坏死因子α刺激,相关报道表明Co甚至在较低浓度下比Cr更具毒性。Co和Cr离子引起剂量和时间依赖性的巨噬细胞凋亡[43]。此外,Ti通过诱导细胞凋亡减少成骨细胞的数量,聚甲基丙烯酸甲酯减少成骨细胞增殖[37]。 与正常人群相比,存在金属对金属植入物的人体血液中的金属离子浓度更高。这些金属离子和颗粒可以通过循环遍及全身并积聚在脑、肝和肾组织中,这些累积的物质具有包括毒性,致癌性和致畸性的不良反应。 2.3.2 钽颗粒 使用激光技术在体外分析用钽(Ta)覆盖的Ti植入物的摩擦学性能。Ta优选用于植入物,因为其具有高度耐腐蚀,并且它是具有低离子释放速率的生物相容性金属。该涂层增加了高碳CoCrMo生物医学合金的机械和摩擦学性质。研究表明,激光涂覆有Ta的表面能够减少早期阶段的骨植入物界面的磨损颗粒的发展。与Ti和不锈钢相比,Ta高度耐腐蚀和耐侵蚀,并且其重量或粗糙度方面没有任何显著变化。钽也是与成骨细胞最相容的金属[37]。 2.4 陶瓷颗粒 陶瓷植入物产生刺激巨噬细胞释放溶骨性细胞因子的颗粒。铝颗粒引起的刺激比聚乙烯更小,并且它们引起肿瘤坏死因子α和前列腺素E2释放也相对较少。与金属对金属相比,陶瓷对陶瓷表面体积磨损小,颗粒的形成减少约10倍,这是一个突出的优点,由颗粒介导的炎症反应和骨吸收低于由聚乙烯颗粒引起的炎症反应。最近的研究表明,氧化铝(Al2O3)陶瓷颗粒在体外比CoCr合金颗粒对人体细胞基因毒性更小。氧化铝颗粒无细胞毒性,但对人体细胞在体内是弱基因毒性的[43]。 2.5 成骨细胞和骨溶解 成骨细胞的作用尚未充分阐明。然而,可以通过它们分泌的细胞因子间接增加骨溶解,从而减少骨形成。磨损颗粒通过增加破骨细胞活性和抑制骨形成引起骨溶解[38],并抑制正常的成骨细胞活性。成骨细胞具有与巨噬细胞非常相似的炎症信号应答,磨损颗粒诱导核因子κB的活化和白细胞介素6释放,从而破坏胶原合成[44]。金属和聚合物颗粒减少成骨细胞Ⅰ型和Ⅲ型胶原的表达[37,45]。O’Neill等[44]研究表明显示,磨损颗粒对成骨细胞中的原胶原a1基因(Ⅰ)有拮抗作用。聚乙烯颗粒还能减少成骨细胞的基质产生。所有因素都对成骨细胞活性产生负面影响,并且被吸收的骨质不能修复。 2.6 内毒素和骨溶解 许多研究表明存在负面影响骨整合和导致局部骨吸收的因素。最近的研究也集中在细菌碎片产物[3,46-48]。现已在植入物上检测到高浓度的细菌碎片产物,并且最近已经确定它们抑制骨整合。这暗示亚临床感染可以作为导致无菌性松动的病因学因素。炎症性关节病和免疫受损患者的感染率更高。有研究的结果表明,在假体周围组织有更高浓度的细菌碎片产物存在[49]。 最著名的细菌碎片分子是存在于革兰阴性细菌的细胞壁中的脂多糖[48]。此外,来自革兰阳性细菌的脂磷壁酸和脂肽和肽聚糖也具有与脂多糖类似的作 用[50]。脂多糖对生物材料表面具有亲和力[48]。脂多糖与存在于细胞膜中TLR4相互作用,TLR4相关的信号通路通过脂多糖激活巨噬细胞,并且诱导炎症反应发生[51-52]。当脂多糖与TLR4相互作用时,白细胞介素受体相关激酶IRAK1,IRAK2,IRAK4和肿瘤坏死因子受体相关因子6等因子相互作用,引起核因子κB活化的连锁反应,导致促炎细胞因子如肿瘤坏死因子α、白细胞介素1b和白细胞介素6的释放[52]。 脂多糖诱导成骨细胞释放白细胞介素1、白细胞介素6、前列腺素E2和RANKL。这些介质负责破骨细胞的成熟和活化[53-54]。活化的破骨细胞启动局部骨吸收。脂多糖还抑制成骨细胞分化,从而防止新的骨形成。在骨溶解区域中检测到的这种细菌碎片产物来自3个已知来源。首先,尽管已经灭菌,脂多糖仍可以高浓度存在于植入物上,并且当植入物被植入时其被转移到体内[55-56]。严重的炎症反应和局部骨吸收却与无感染的任何临床症状皆可以通过这种机制来解释。体内的系统性内毒素可能提供细菌碎片的第二来源[43],这些内毒素来源于肠道菌群、轻微感染和牙科手术。虽然携带全身性内毒素的患者没有显示任何临床或微生物学症状,但植入物产生的磨损颗粒暴露于源于远端感染灶的脂多 糖[51]。细菌碎片的第3个来源是以亚临床浓度存在于植入物和磨损颗粒上的细菌[49]。"
| [1] Greidanus NV, Peterson RC, Masri BA, et al. Quality of life outcomes in revision versus primary total knee arthroplasty. J Arthroplasty. 2011; 26(4): 615-620. [2] Harris WH. Osteolysis and particle disease in hip replacement. A review. Acta Orthop Scand. 1994; 65(1): 113-123.[3] Lewallen EA, Riester SM, Bonin CA, et al. Biological strategies for improved osseointegration and osteoinduction of porous metal orthopedic implants. Tissue Eng Part B Rev. 2014;21(2): 218-230.[4] Gallo J, Raska M, Mrázek F, et al. Bone remodeling, particle disease and individual susceptibility to periprosthetic osteolysis. Physiol Res. 2008; 57(3): 339-349. [5] Omran MTA, Miley DD, McLeod DE, et al. Retrospective assessment of survival rate for short endosseous dental implants. Implant Dentistry. 2015;24(2):185-191. [6] Pedersen AB, Mehnert F, Odgaard A, et al. Existing data sources for clinical epidemiology: The Danish Knee Arthroplasty Register. Clin Epidemiol 2012; 4: 125-35. [7] Paxton EW, Furnes O, Namba RS, et al. Comparison of the Norwegian knee arthroplasty register and a United States arthroplasty registry. J Bone Joint Surg Am 2011; 93(Suppl. 3): 20-30. [8] Illingworth KD, Wachter N, Maloney WJ, et al. Advances in acetabular osteolysis: biomarkers, imaging, and pharmacologic management. Instr Course Lect.2014; 63: 177-186.[9] Glyn-Jones S, Thomas GER, Garfjeld-Roberts P, et al. The John Charnley Award: highly crosslinked polyethylene in total hip arthroplasty decreases long-term wear: a double-blind randomized trial. Clin Orthop Relat Res.2015;473(2): 432-438. [10] Bitar D, Parvizi J. Biological response to prosthetic debris. World J Orthop. 2015; 6(2): 172-189. [11] Rujitanaroj PO, Jao B, Yang J, et al. Controlling fibrous capsule formation through long-term down-regulation of collagen type I (COL1A1) expression by nanofiber-mediated siRNA gene silencing. Acta Biomater. 2013;9(1):4513-4524.[12] Messier SP, Gutekunst DJ, Davis C, et al. Weight loss reduces knee-joint loads in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005; 52(7): 2026-2032.[13] Gallo J, Goodman SB, Konttinen YT, et al. Particle disease: biologic mechanisms of periprosthetic osteolysis in total hip arthroplasty. Innate Immun. 2013;19(2): 213-224. [14] Davies AP, Willert HG, Campbell PA, et al. An unusual lymphocytic perivascular infiltration in tissues around contemporary metal-on-metal joint replacements. J Bone Joint Surg Am.2005; 87(1): 18-27.[15] Liu X, Qu X, Wu C, et al. The effect of enoxacin on osteoclastogenesis and reduction of titanium particle-induced osteolysis via suppression of JNK signaling pathway. Biomaterials. 2014;35(22): 5721-5730. [16] Duisabeau L, Combrade P, Forest B. Environmental effect on fretting of metallic materials for orthopaedic implants. Wear. 2004; 256: 805-816.[17] Schramm M, Wirtz DC, Holzwarth U, et al. The Morse taper junction in modular revision hip replacement--a biomechanical and retrieval analysis. Biomed Tech (Berl). 2000; 45(4): 105-109. [18] Jauch SY, Huber G, Sellenschloh K, et al. Micromotions at the taper interface between stem and neck adapter of a bimodular hip prosthesis during activities of daily living. J Orthop Res. 2013; 31(8): 1165-1171. [19] Mistry JB, Chughtai M, Elmallah RK, et al. Trunnionosis in total hip arthroplasty: a review. J Orthop Traumatol. 2016; 17(1): 1-6. [20] Pastides PS, Dodd M, Sarraf KM, et al. Trunnionosis: A pain in the neck. World J Orthop. 2013; 4(4): 161-166. [21] Morawietz L, Classen RA, Schröder JH, et al. Proposal for a histopathological consensus classification of the periprosthetic interface membrane. J Clin Pathol. 2006; 59(6): 591-597.[22] Mert M, Oztürkmen Y, Ünkar EA, et al. Sciatic nerve compression by an extrapelvic cyst secondary to wear debris after a cementless total hip arthroplasty: A case report and literature review. Int J Surg Case Rep. 2013; 4(10): 805-808. [23] Berend ME, Ritter MA, Hyldahl HC, et al. Implant migration and failure in total knee arthroplasty is related to body mass index and tibial component size. J Arthroplasty. 2008; 23(6) (Suppl. 1): 104-109. [24] Wong J, Steklov N, Patil S, et al. Predicting the effect of tray malalignment on risk for bone damage and implant subsidence after total knee arthroplasty. J Orthop Res. 2011; 29(3): 347-353. [25] Pap G, Machner A, Rinnert T, et al. Development and characteristics of a synovial-like interface membrane around cemented tibial hemiarthroplasties in a novel rat model of aseptic prosthesis loosening. Arthritis Rheum. 2001; 44(4): 956-963.[26] Gouron R, Petit L, Boudot C, et al. Osteoclasts and their precursors are present in the induced-membrane during bone reconstruction using the Masquelet technique. J Tissue Eng Regen Med. 2017;11(2):382-389. [27] Windisch C, Windisch B, Kolb W, et al. Osteodensitometry measurements of periprosthetic bone using dual energy X-ray absorptiometry following total knee arthroplasty. Arch Orthop Trauma Surg. 2012; 132(11): 1595-1601. [28] Banerjee S, Pivec R, Issa K, et al. Large-diameter femoral heads in total hip arthroplasty: an evidence-based review. Am J Orthop. 2014; 43(11): 506-12. [29] Musib MK. Response to ultra-high molecular weight polyethylene particles. Am J Biomed Eng. 2011; 1(1): 7-12.[30] Jiang Y, Jia T, Wooley PH, et al. Current research in the pathogenesis of aseptic implant loosening associated with particulate wear debris. Acta Orthop Belg. 2013;79(1): 1-9.[31] Hodrick JT, Severson EP, McAlister DS, et al. Highly crosslinked polyethylene is safe for use in total knee arthroplasty. Clin Orthop Relat Res. 2008; 466(11): 2806-2812. [32] Liu A, Ingham E, Fisher J, et al. Generation of a large volume of clinically relevant nanometre-sized ultra-high-molecular-weight polyethylene wear particles for cell culture studies. Proc Inst Mech Eng H. 2014;228(4):418-426. [33] Carossino AM, Carulli C, Ciuffi S, et al. Hypersensitivity reactions to metal implants: laboratory options. BMC Musculoskelet Disord. 2016;17(1):486. [34] Archibeck MJ, Jacobs JJ, Roebuck KA, et al. The basic science of periprosthetic osteolysis. Instr Course Lect. 2001;50:185-195.[35] Goodman SB, Gibon E, Yao Z. The basic science of periprosthetic osteolysis. Instr Course Lect. 2013; 62: 201-206.[36] Wang CT, Lin YT, Chiang BL, et al. Over-expression of receptor activator of nuclear factor-kappaB ligand (RANKL), inflammatory cytokines, and chemokines in periprosthetic osteolysis of loosened total hip arthroplasty. Biomaterials. 2010; 31(1): 77-82. [37] Jiang X, Sato T, Yao Z, et al. Local delivery of mutant CCL2 protein-reduced orthopaedic implant wear particle-induced osteolysis and inflammation in vivo. J Orthop Res. 2016;34(1): 58-64. [38] Atkins GJ, Haynes DR, Howie DW, et al. Role of polyethylene particles in peri-prosthetic osteolysis: a review. World J Orthop. 2011;2(10): 93-101.[39] Witte F, Hort N, Vogt C, et al. Degradable biomaterials based on magnesium corrosion. Curr Opin Solid State Mater Sci. 2008; 12(5-6): 63-72. [40] Feyerabend F, Fischer J, Holtz J, et al. Evaluation of short-term effects of rare earth and other elements used in magnesium alloys on primary cells and cell lines. Acta Biomater. 2010; 6(5): 1834-1842. [41] Pichler K, Kraus T, Martinelli E, et al. Cellular reactions to biodegradable magnesium alloys on human growth plate chondrocytes and osteoblasts. Int Orthop. 2014; 38(4): 881-889. [42] Posada OM, Tate RJ, Meek RM, et al. In vitro analyses of the toxicity, immunological, and gene expression effects of cobalt-chromium alloy wear debris and Co ions derived from metal-on-metal hip implants. Lubricants. 2015;(3): 539-568.[43] Tsaousi A, Jones E, Case CP. The in vitro genotoxicity of orthopaedic ceramic (Al2 O3) and metal (CoCr alloy) particles. Mutat Res. 2010;697(1-2):1-9. [44] O’Neill SC, Queally JM, Devitt BM, et al. The role of osteoblasts in peri-prosthetic osteolysis. Bone Joint J. 2013; 95(8): 1022-1026.[45] Shi Y, Xie Y, Pan H, et al. Plasma-sprayed titanium patterns for enhancing early cell responses. J Ther Spray Technol. 2016;25(5): 946-958. [46] Syggelos SA, Aletras AJ, Smirlaki I, et al. Extracellular matrix degradation and tissue remodeling in periprosthetic loosening and osteolysis: focus on matrix metalloproteinases, their endogenous tissue inhibitors, and the proteasome. Biomed Res Int. 2013;2013:230805.[47] Bonsignore LA, Anderson JR, Lee Z, et al. Adherent lipopolysaccharide inhibits the osseointegration of orthopedic implants by impairing osteoblast differentiation. Bone. 2013; 52(1): 93-101. [48] Nalepka JL, Lee MJ, Kraay MJ, et al. Lipopolysaccharide found in aseptic loosening of patients with inflammatory arthritis. Clin Orthop Relat Res. 2006;451: 229-235. [49] Morra M, Cassinelli C, Bollati D, et al. Adherent endotoxin on dental implant surfaces: a reappraisal. J Oral Implantol. 2015;41(1): 10-16. [50] Bi Y, Seabold JM, Kaar SG, et al. Adherent endotoxin on orthopedic wear particles stimulates cytokine production and osteoclast differentiation. J Bone Miner Res. 2001; 16(11): 2082-2091. [51] Hirayama T, Tamaki Y, Takakubo Y, et al. Toll-like receptors and their adaptors are regulated in macrophages after phagocytosis of lipopolysaccharide-coated titanium particles. J Orthop Res. 2011;29(7): 984-992.[52] Sims NA, Martin TJ. Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. Bonekey Rep. 2014;3:481.[53] Rajamannan NM, Cicek M, Hawse JR, et al. In vitro cell culture model of calcification: molecular regulation of myofibroblast differentiation to an osteoblast phenotype //Molecular biology of valvular heart disease. Springer London, 2014: 13-20. [54] Morra M, Cassinelli C, Bollati D, et al. Adherent endotoxin on dental implant surfaces: a reappraisal. J Oral Implantol. 2015;41(1): 10-16. [55] US Food and Drug Administration. Guideline on validation of the Limulus amebocyte lysate test as an end-product endotoxin test for human and animal parenteral drugs, biological products and medical devices 1987 [displayed 29 March 2010].[56] Sukur E, Akman YE, Ozturkmen Y, et al. Particle disease: a current review of the biological mechanisms in periprosthetic osteolysis after hip arthroplasty. Open Orthop J. 2016;10: 241. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [3] | Li Dadi, Zhu Liang, Zheng Li, Zhao Fengchao. Correlation of total knee arthroplasty efficacy with satisfaction and personality characteristics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1346-1350. |
| [4] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [5] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [6] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [7] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [8] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [9] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [10] | Chen Junming, Yue Chen, He Peilin, Zhang Juntao, Sun Moyuan, Liu Youwen. Hip arthroplasty versus proximal femoral nail antirotation for intertrochanteric fractures in older adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1452-1457. |
| [11] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [12] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [13] | Zhong Hehe, Sun Pengpeng, Sang Peng, Wu Shuhong, Liu Yi. Evaluation of knee stability after simulated reconstruction of the core ligament of the posterolateral complex [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 821-825. |
| [14] | Zhao Zhongyi, Li Yongzhen, Chen Feng, Ji Aiyu. Comparison of total knee arthroplasty and unicompartmental knee arthroplasty in treatment of traumatic osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 854-859. |
| [15] | Liu Shaohua, Zhou Guanming, Chen Xicong, Xiao Keming, Cai Jian, Liu Xiaofang. Influence of anterior cruciate ligament defect on the mid-term outcome of fixed-bearing unicompartmental knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 860-865. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||