Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (3): 471-477.doi: 10.3969/j.issn.2095-4344.0049
Previous Articles Next Articles
Treatment strategies for infection after hip arthroplasty in the elderly patients
Shi Zhi-yong1, Zhang Xue-yun1, Chen Feng2, Wang Fei3, Guo Zhi3
- 1Department of Orthopedics, Yanjiao People’s Hospital, Sanhe 101600, Hebei Province, China; 2Department of Orthopedics, the Central Hospital of Wuhan, Wuhan 430014, Hubei Province, China; 3Department of Orthopedics, the General Hospital of Beijing Military Region of Chinese PLA, Beijing 100700, China
-
Online:2018-01-28Published:2018-01-28 -
About author:Shi Zhi-yong, Associate chief physician, Department of Orthopedics, Yanjiao People’s Hospital, Sanhe 101600, Hebei Province, China -
Supported by:the Key Technology Research & Development Project of Langfang City, No. 2016013065
CLC Number:
Cite this article
Shi Zhi-yong, Zhang Xue-yun, Chen Feng, Wang Fei, Guo Zhi. Treatment strategies for infection after hip arthroplasty in the elderly patients[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(3): 471-477.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
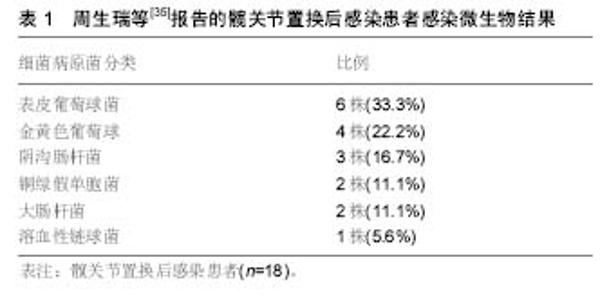
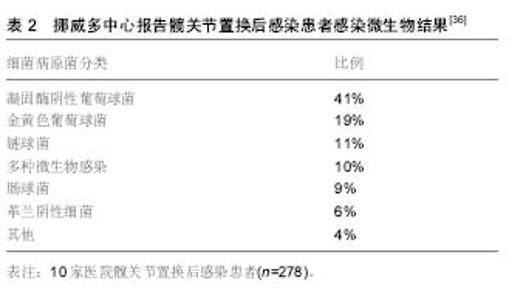
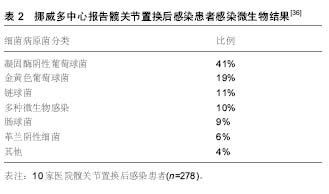
2.1 单纯抗生素治疗 一般而言,对于具有髋关节翻修术禁忌证的患者,可以考虑使用单纯抗生素治疗,通常是由于合并严重并发症或多种合并症患者或者用于姑息性治疗,单纯抗生素治疗较少用于髋关节置换术后感染,是否能彻底清除细菌尚值得怀疑,Milosev等[3]报告一组24例髋关节置换术后感染的患者,这些患者中17例是用单一抗生素治疗,7例为联合抗生素治疗,随访3年没有复发,因考虑入选病例数较少,作者没有得出明确结论。吕亮等[4]收集21例髋关节置换后感染的患者人工髋关节置换后感染率为8.86%,检出病原菌前3位为金黄色葡萄球菌、大肠埃希菌及铜绿假单胞菌,均给予单纯抗生素治疗,药敏结果显示,革兰阳性菌对替考拉宁和利奈唑胺的耐药率较低,革兰阴性菌对妥布霉素、阿米卡星、亚胺培南和美罗培南的耐药率较低。 2.2 清创和植入物保留 在早期髋关节置换术后感染的患者中可以考虑手术清创,抗生素治疗和植入物保留,可考虑开放的方法彻底清创、灌洗。Byren等[5]对52例髋关节置换后感染患者进行回顾性研究,清创手术前 48 h停止使用抗生素,在手术中患者开始使用万古霉素和美罗培南,当明确感染微生物后再做调整,如果没有培养出革兰阴性杆菌则停用美罗培南,全组病例中金黄色葡萄球菌感染发生率最高,占42%的病例,耐甲氧西林金黄色葡萄球菌仅占8%。术后感染防控需要长时间使用抗生素,平均治疗疗程达到1.5年,1,2和3年的成功救治率分别为89%、81%和78%,金黄色葡萄球菌感染是抗感染治疗失败的独立风险,作者的分析表明停止抗生素治疗后失败率会增加,这点支持了长期使用抗生素的相关理论。 Estes等[6]回顾性分析了20例髋关节置换后感染患者,他认为如果患者明确诊断为髋关节置换术后感染,应在诊断后28 d内进行清创手术并且在手术时确定假体稳定,其中2例患者应用清创术和放置含有庆大霉素或妥布霉素的抗生素骨水泥,18例患者放置含有万古霉素和头孢唑啉抗生素骨水泥,在第7天进行第二次清创,静脉抗生素共持续6周,再继续口服抗生素,时间范围从6周到终生,患者清创术后应该至少抗生素治疗1年,其中有18例患者1年内没有表现出感染复发的证据,最终全组有8例患者接受持续的抗生素治疗。 2.3 一期翻修 Winkler等[7]分析37例髋关节置换术后感染患者的结果,采用一期非骨水泥假体植入术治疗。在这些患者中有12例为早期感染,9例迟发感染和16例急性血源性感染,使用了混合了万古霉素或万古霉素加妥布霉素的松质骨。随访时间平均为4.4年,无感染率为92%,其中有5例耐甲氧西林金黄色葡萄球菌成功治疗。Rudelli等[8]报告了32例接受一期翻修和松质骨移植治疗的患者结果,12例为早期感染,17例迟发感染和3例急性血源性感染,15例使用钛网杯来支持假体,移植骨中不加入抗生素,但植入骨水泥假体时使用了抗生素骨水泥,他们报告平均随访103个月中的无感染率为93.7%。 较多的报道使用多种技术成功治愈髋关节置换后感染,外科医生的临床培训和经验应该是选择重建方面工作,重点强调细致的清创手术,需要清除死腔和残留的细菌,选择特异性抗生素成功地浸渍到骨移植物中,骨水泥中的抗生素浓度会受到限制,因为高浓度可能会降低组织的完整性,一期翻修的优点仍值得进一步研究[9-10]。 2.4 二期翻修 分期翻修通常被认为是治疗髋关节置换后感染的金标准。Chen等[11]对155例髋关节置换术后感染的患者进行二期髋关节翻修术,翻修间隔时间为平均17.8周,患者平均Harris评分28.3分改善到85.7分。郭国栋等[12]报告23 例髋关节置换后感染病例,一期彻底清创加含抗生素骨水泥间隔置入,二期行关节重建,随访后Harris评分显著提高且无感染复发。分期翻修的原则是去除所有可能感染的组织和骨头,然后使用混合抗生素的骨水泥占位器局部释放抗生素,联合静脉系统性使用抗生素,根据患者临床体征症状的消退以及血化验指标如C-反应蛋白和红细胞沉降率等恢复正常,然后进行第二阶段,在重新植入之前进行重复冷冻切片,如果感染仍存在可继续应用当前抗生素,大多数中心规定了约6周的静脉用抗生素治疗及再继续口服抗生素[13-14]。 抗生素骨水泥占位器可以大致分为静态或关节占位器,静态占位器由留在关节腔内的抗生素骨水泥块组成,关节占位器可以用金属架支撑的抗生素骨水泥制成,这类关节占位器有现成的产品,也可以由外科医生使用金属杆、骨水泥通过模具制造[15],成分都是高浓度的抗生素骨水泥。占位器的使用具有几个优点,它们允许以高浓度局部递送抗生素,关节间隔器有助于保持正确的肢体长度和阶段之间的软组织张力。这提高了患者生活质量,保留骨骼储备,防止软组织挛缩和有利于再次植入假体。然而如果感染微生物未知,则间隔区可能含有敏感性不合适的抗生素,因此常规都推荐使用万古霉素和碳青霉烯抗生素[16-17]。 Disch等[18]提出髋关节置换术后感染仅用清创根治术和全身抗生素治疗,报告病例中平均随访41个月,30例患者保持无感染复发,但9例患者需要重复进行清创术,但其它使用占位器的患者均无感染复发,证明了使用占位器的有较好的预后结果。Cabrita等[19]提出了用二期翻修治疗的68例髋关节置换术后感染患者,30个没有占位器(对照组)和38个万古霉素负载占位器(研究组)。平均随访7年(5-11.5年)时,对照组和研究组无感染率分别为66.7%和89.1%,Harris髋关节评分对照组和研究组分别为69分和75分,显示了占位器治疗具有更好的临床结果。Hsieh等[20]比较了将负载抗生素的骨水泥珠用于预制占位器,无感染率为95.3%(122/128)。 2.5 抗生素骨水泥使用 Enayatollahi等[21]进行了文献回顾分析髋关节置换术后抗感染治疗相关因素,共评估了12项研究,总共对1 299例髋关节置换术后感染患者进行了随访,虽然在99%的病例中使用了抗生素骨水泥,但抗生素方面的选择、给药和持续时间方面存在很大差异。平均随访时间为4.8年(0.1-17.1年),总体治愈率为83%。成功控制感染的关键因素包括:①初始髋关节置换术后感染没有其他并发症;②患者一般情况较好,无严重基础疾病;③药敏结果提示微生物为敏感的葡萄球菌或链球菌属;④微生物对骨水泥中的抗生素敏感。预后不良的相关因素为:①多重微生物感染;②革兰阴性杆菌感染,特别是假单胞菌;③发生耐甲氧西林金黄色葡萄球菌和D组链球菌感染。 在抗生素骨水泥中,为了保持骨水泥的机械完整性,抗生素与骨水泥的比率不应超过所用骨水泥的10%,即40 g骨水泥中的4 g总抗生素。最常见的抗生素包括妥布霉素,庆大霉素和万古霉素,庆大霉素现在只有液体制 剂[22],Gulotta等[23]报道,液体庆大霉素用于骨水泥仍然能有效杀菌,该研究显示,当将480 mg庆大霉素加入40 g骨水泥中时,极限压缩强度和拉伸强度都下降了近50%。Hsieh等[24]分析42例髋关节置换术后感染接受二期翻修的患者,并得出结论液体庆大霉素掺入骨水泥垫片有效且安全。 目前,髋关节置换术后革兰阴性细菌感染比例在逐渐增多,因此碳青霉烯使用的越来越多。Baleani等[22]认为40 g骨水泥中加入0.5 g美罗培南和0.5 g万古霉素可行,但是如果单纯把万古霉素剂量增加至1 g反而不可接受。Andollina等[25]进行了一个与抗生素骨水泥有关的体外研究,结果表明,单纯用1 g万古霉素对肠球菌作用是缓慢的,如果分别用0.5 g万古霉素和0.5 g美罗培南来消除假单胞菌,同时显示了对抗肠球菌的快速反应,也达到了清除假单胞菌作用,目前没有万古霉素、美罗培南的最佳浓度比的相关研究,不管怎样,维持抗生素骨水泥的完整性,需要同时提供足够的抗生素来覆盖抗生素骨水泥。 2.6 二期翻修后的抗生素治疗 在二期翻修后进行抗生素治疗的持续时间无相关共识,文献提出了很多种方案,从术后无抗生素到长期静脉内抗生素。大多数中心考虑为4-6周的静脉内抗生素,有或无口服抗生素。减少不必要的抗生素防止出现细菌耐药,降低并发症的风险,并大大降低治疗成本[26-29]。李超等[30]将初次髋关节置换的患者随机分为试验组和对照组各150例,住院期间内所有患者静脉应用头孢类抗生素,出院后试验组口服利福平和左氧氟沙星,疗程6周,结果显示试验组和对照组在末次随访时各有4例、13例患者发生了关节假体周围感染,两组感染率比较差异有显著性意义,说明髋关节置换后长期口服利福平和左氧氟沙星可有效预防髋关节置换术后感染的复发。 Stockley等[31]报告了114例髋关节置换后感染患者,进行根治性清创和二期翻修后,术后3种抗生素一起使用,在平均随访74个月时报告了87.7%的无感染率。Whittaker等[32]报告了43例患者二期翻修后,接受系统性万古霉素2周治疗,平均随访49个月时,无感染率为92.7%。McKenna等[33]回顾性分析了30例迟发感染患者,在二期翻修后,使用抗生素骨水泥占位器与5 d静脉抗生素治疗,再口服即利福平或利奈唑胺,在平均随访35个月,成功根除所有患者的感染。使用静脉注射抗生素简化过程的原因是抗生素从骨水泥间隔器有效和持续地洗脱到局部组织中。许多研究已经证明,在植入后几个月内,抗生素水平维持在常见病原体的最小抑制浓度以上[34],短期抗生素的基本原理是消除任何从手术区域排出的细菌。 2.7 髋关节置换后感染致病菌分析 周生瑞等[35]报告18例髋关节置换后感染患者,培养病原菌如下:表皮葡萄球菌 6例,金黄色葡萄球菌4例,阴沟肠杆菌3例,铜绿假单胞菌2例,大肠杆菌 2 例,溶血性链球菌 1 例,平均随访25(12-48)个月,6例清创、5例一期翻修、7例使用抗生素骨水泥二期翻修,随访均未见感染复发,髋关节 Harris评分由43.2分提高到86.5分,感染微生物情况见表1。"
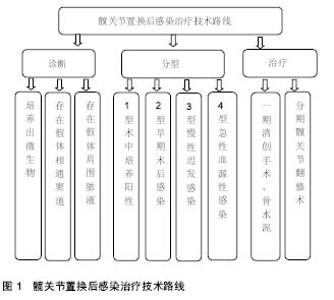
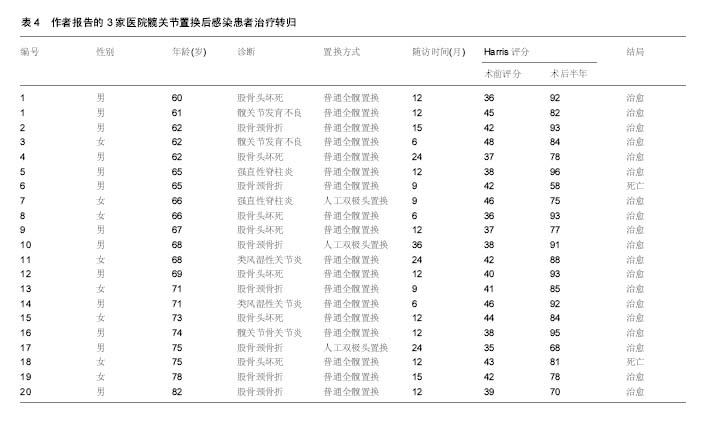
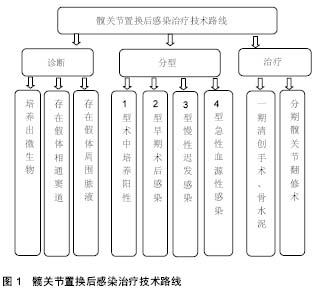
2.8.2 髋关节置换后感染治疗策略 清创方法包括清除炎性肉芽组织,去掉硬化的骨组织,在股骨侧清创时还要取出髓腔内的人工假体,还需去除假体和骨水泥再进行清创。早期保留假体的伤口清创,持续创腔灌洗时间两三周,灌洗液为庆大霉素盐水,后期根据培养结果为敏感抗生素盐水,灌洗1周后每3 d取灌洗引流液培养,连续2次以上培养阴性先拔除灌洗引流入管,3 d后如无大量引流液拔除引流出管,进行清创之后采用过氧化氢溶液、聚维碘酮溶液浸泡,再用大量生理盐水冲洗。 本组患者中5例行一期翻修术,15例行二期翻修,一期手术取出假体、清创、抗生素骨水泥占位器植入。术后3-6个月局部情况稳定,红细胞沉降率、C-反应蛋白正常后再行二期关节翻修术,翻修手术时尽量使用非骨水泥假体以提高远期疗效,部分骨缺损严重患者翻修时行异体骨植入。翻修过程中要彻底清除感染组织和失活组织,使感染创面变为一个接近无菌的新鲜创面,创面的局部血液循环得到改善,彻底的清创是翻修手术成功关键,二期翻修手术中还需使用含抗生素的骨水泥间隔器来减少关节内的瘢痕形成,通过间隔器持续释放高浓度的抗生素,术后再联合应用对细菌药敏敏感的抗生素。髋关节置换后感染治疗技术路线见图1。"
| [1] Dirk Jan F Moojen, Jasper H Zwiers, Vanessa AB Scholte,et al. Similar success rates for single and multiple debridement surgery for acute hip arthroplasty infection.Acta Orthop.2014;85(4): 383-388.[2] Ong T, Anand V, Tan W, et al.Patient characteristics and outcomes of a hip fracture and concomitant fracture compared with hip fracture alone: results from a United Kingdom teaching hospital. Arch Orthop Trauma Surg.2016;136(4):463-467.[3] Milosev I, Trebse R, Kovac S, et al. Survivorship and retrieval analysis of Sikomet metal-on-metal total hip replacements at a mean of seven years.J Bone Joint Surg Am.2006;88(6): 1173-1182.[4] 吕亮,王福生,卢敏,等. 人工髋关节置换术后感染的危险因素分析及预防[J].中华医院感染学杂志,2013,23(6):1334-1336.[5] Byren I, Bejon P, Atkins BL, et al. One hundred and twelve infected arthroplasties treated with 'DAIR' (debridement, antibiotics and implant retention): antibiotic duration and outcome. J Antimicrob Chemother.2009;63(6):1264-1271.[6] Estes CS, Beauchamp CP, Clarke HD, et al. A two-stage retention débridement protocol for acute periprosthetic joint infections.Clin Orthop Relat Res. 2010;468(8): 2029-2038.[7] Winkler H, Stoiber A, Kaudela K, et al. One stage uncemented revision of infected total hip replacement using cancellous allograft bone impregnated with antibiotics. J Bone Joint Surg Br.2008;90(12):1580-1584.[8] Rudelli S,Uip D,Honda E,et al.One-stage revision of infected total hip arthroplasty with bone graft. J Arthroplasty.2008;23(8):1165-1177.[9] Matthias Wolf, Heimo Clar, Jörg Friesenbichler,et al. Prosthetic joint infection following total hip replacement: results of one-stage versus two-stage exchange. Int Orthop.2014;38(7):1363-1368.[10] Ke Liu,Jia Zheng,Yi Jin,et al.Application of temporarily functional antibiotic-containing bone cement prosthesis in revision hip arthroplasty.Eur J Orthop Surg Traumatol.2014;24(1): 51-55.[11] Chen SY, Hu CC, Chen CC,et al. Two-stage revision arthroplasty for periprosthetic hip infection: mean follow-up of ten years.Biomed Res Int.2015;2015:345475.[12] 郭国栋,樊根涛,郭亭,等.人工髋关节置换后感染二期翻修中的彻底清创[J].中国组织工程研究,2015,19(48):7741-7746.[13] 张理昂,周一新,李玉军,等.髋关节置换术后假体周围感染的治疗[J].中华骨科杂志,2009,29(10):924-928.[14] Kim BH, Lee S, Yoo B, et al. Risk factors associated with outcomes of hip fracture surgery in elderly patients. Korean J Anesthesiol.2015; 68(6):561-567.[15] Moerman S, Vochteloo AJ, Tuinebreijer WE,et al. Factors associated with the course of health-related quality of life after a hip fracture.Arch Orthop Trauma Surg.2016;136(7): 935-943.[16] Radtke K, Tetzlaff T, Vaske B,et al.Arthroplasty-center related retrospective analysis of risk factors for Periprosthetic Joint Infection after primary and after revision Total Hip Arthroplasty. Technol Health Care.2016;24(5):721-728.[17] Janz V,Bartek B,Wassilew GI,et al.Validation of synovial aspiration in girdlestone hips for detection of infection persistence in patients undergoing 2-stage revision total hip arthroplasty. J Arthroplasty.2016;31(3):684-687.[18] Disch AC, Matziolis G,Perka C.Two-stage operative strategy without local antibiotic treatment for infected hip arthroplasty: clinical and radiological outcome. Arch Orthop Trauma Surg. 2007;127(8):691-697.[19] Cabrita HB, Croci AT, Camargo OP,et al. Prospective study of the treatment of infected hip arthroplasties with or without the use of an antibiotic-loaded cement spacer. Clinics(Sao Paulo). 2007;62(2):99-108.[20] Hsieh PH, Chang Y, Chen DW,et al. Pain distribution and response to total hip arthroplasty: a prospective observational study in 113 patients with end-stage hip disease. J Orthop Sci. 2012;17(3):213-218.[21] Enayatollahi MA, Parvizi J. Diagnosis of infected total hip arthroplasty.Hip Int.2015;25(4):294-300.[22] Baleani M, Persson C, Zolezzi C,et al. Biological and biomechanical effects of vancomycin and meropenem in acrylic bone cement.J Arthroplasty.2008;23(8):1232-1238.[23] Gulotta LV, Padgett DE, Sculco TP, et al. Fast track THR: one hospital's experience with a 2-day length of stay protocol for total hip replacement.HSS J.2011;7(3):223-228.[24] Hsieh PH, Huang KC, Lee PC,et al. Two-stage revision of infected hip arthroplasty using an antibiotic-loaded spacer: retrospective comparison between short-term and prolonged antibiotic therapy. J Antimicrob Chemother.2009;64(2): 392-397.[25] Andollina A, Bertoni G,Zolezzi C,et al. Vancomycin and meropenem in acrylic cement: elution kinetics of in vitro bactericidal action. Chir Organi Mov.2008;91(3):153-158.[26] 陆文良,Dewan Sheilesh KUMAR,林伟龙,等.老年髋部骨折围术期风险及术后1年效果评估[J].中华老年多器官疾病杂志, 2014, 13(6):440-444.[27] Takada R, Jinno T, Koga D, et al. Is drain tip culture prognostic of surgical site infection? Results of 1380 drain tip cultures in total hip arthroplasty.J Arthroplasty.2015; 30(8): 1407-1409.[28] Westberg M, Frihagen F, Brun OC, et al. Effectiveness of gentamicin-containing collagen sponges for prevention of surgical site infection after hip arthroplasty: a multicenter randomized trial. Clin Infect Dis.2015;60(12):1752-1759.[29] 孙长鲛,周勇刚,柴伟,等. 髋关节置换术后感染的微生物特点及药敏分析[J]. 中华医学杂志,2014, 94(21):1657-1660.[30] 李超,尚希福,曹晓芳,等. 利福平联合左氧氟沙星预防全髋关节置换后感染[J].中国组织工程研究,2014,18(48):7714-7718.[31] Stockley I, Mockford BJ, Hoad-Reddick A, et al. The use of two-stage exchange arthroplasty with depot antibiotics in the absence of long-term antibiotic therapy in infected total hip replacement. J Bone Joint Surg Br.2008;90(2):145-148.[32] Whittaker JP, Warren RE, Jones RS, et al. Is prolonged systemic antibiotic treatment essential in two-stage revision hip replacement for chronic Gram-positive infection? .J Bone Joint Surg Br.2009;91(1):44-51.[33] McKenna PB,O'Shea K,Masterson EL. Two-stage revision of infected hip arthroplasty using a shortened post-operative course of antibiotics.Arch Orthop Trauma Surg.2009;129(4): 489-494.[34] Masri BA, Panagiotopoulos KP, Greidanus NV, et al. Cementless two-stage exchange arthroplasty for infection after total hip arthroplasty.J Arthroplasty.2007;22(1):72-78.[35] 周生瑞,苗胜,沙广钊. 人工髋关节置换后感染的细菌培养及治疗对策文献复习[J].中国组织工程研究, 2012,16(13): 2459-2462.[36] Langvatn H,Lutro O,Dale H et al. Bacterial and hematological findings in infected total hip arthroplasties in norway assessment of 278 revisions due to infection in the Norwegian arthroplasty register. Open Orthop J.2015;9: 445-449.[37] 张明德,朱俊峰. 全髋关节置换术后感染的危险因素分析及早期诊断[J].重庆医学,2013,42(33):3999-4002.[38] Yi PH, Cross MB, Moric M,et al. The 2013 Frank Stinchfield Award: diagnosis of infection in the early postoperative period after total hip arthroplasty. Clin Orthop Relat Res.2014;472(2): 424-429. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [3] | Li Dadi, Zhu Liang, Zheng Li, Zhao Fengchao. Correlation of total knee arthroplasty efficacy with satisfaction and personality characteristics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1346-1350. |
| [4] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [5] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [6] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [7] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [8] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [9] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [10] | Chen Junming, Yue Chen, He Peilin, Zhang Juntao, Sun Moyuan, Liu Youwen. Hip arthroplasty versus proximal femoral nail antirotation for intertrochanteric fractures in older adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1452-1457. |
| [11] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [12] | Yuan Jun, Yang Jiafu. Hemostatic effect of topical tranexamic acid infiltration in cementless total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 873-877. |
| [13] | Liu Lihua, Sun Wei, Wang Yunting, Gao Fuqiang, Cheng Liming, Li Zirong, Wang Jiangning. Type L1 steroid-induced osteonecrosis of the femoral head through femoral head and neck junction decompression by fenestration: a single-center prospective clinical study [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 906-911. |
| [14] | Li Yan, Wang Pei, Deng Donghuan, Yan Wei, Li Lei, Jiang Hongjiang. Electroacupuncture for pain control after total knee arthroplasty: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 957-963. |
| [15] | Hua Haotian, Zhao Wenyu, Zhang Lei, Bai Wenbo, Wang Xinwei. Meta-analysis of clinical efficacy and safety of antibiotic artificial bone in the treatment of chronic osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 970-976. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||